Introduction
Obstructive sleep apnea (OSA), a common sleep disorder, affects a large proportion of the adult population [1, 2]. Intermittent hypoxia (IH) known as the primary characteristic of OSA is a potential key factor leading to the pathogenesis of OSA-related comorbidities, including cardiovascular disease [3], insulin resistance [4] and Alzheimer’s disease [5]. Over the past few decades, an increased risk factor for cardiovascular morbidities has been consistently observed among OSA patients [6, 7]. Furthermore, increasing evidence indicates that patients with OSA often show endothelial dysfunction, which is an early event in the process of cardiovascular disease [8, 9]. In animal OSA models, IH exposures and long-term sleep fragmentation could lead to endothelial dysfunction [10], thereby supporting a potential causal relationship between OSA and endothelial dysfunction. In two previous studies, they clearly indicated that the impairment of endothelial function was restored with improvement of IH exposures [11, 12]. However, the potential mechanisms involved in the occurrence of OSA-induced endothelial dysfunction are still poorly understood.
MiRNAs are a class of small, noncoding RNAs, with the length of 20-26 nucleotides. They regulate gene expression by binding to the 3′ untranslated region (3′-UTR) of target genes, which leads to the reduction of the corresponding genes by degradation of mRNA or inhibition of mRNA translation [13]. Increasing evidence indicates that miRNAs could regulate various physiological and pathological processes, including cell viability, apoptosis, autophagy, and differentiation [14]. Meanwhile, a number of miRNAs are involved and functional in cardiovascular disease, including acute myocardial infarction (AMI) [15], atherosclerosis [16], atrial fibrillation [17] and cardiac hypertrophy [18]. For instance, miRNA-214 was highly expressed in elderly AMI patients, which may regulate myocardial cell apoptosis via inhibiting miR-214 target genes expression [15]. Recently, miR-193a-3p has been verified as a key regulator in the development of numerous cancers such as non-small cell lung cancer [19], colorectal cancer [20] and bladder cancer [21]. However, the effects and modulatory mechanism of miR-193a-3p in protecting human umbilical vein endothelial cells (HUVECs) from IH-induced apoptosis have not been studied.
In the present study, we first used an in vitro model of endothelial injury induced by IH to investigate the role of and interaction between miR-193a-3p and Fas apoptotic inhibitory molecule 2 (FAIM2) in regulating IH-induced endothelial damage. We found that intermittent hypoxia induced endothelial injury in vitro, which was accompanied by the upregulation of miR-193a-3p. Inhibition of miR-193a-3p attenuated intermittent hypoxia-induced endothelial injury by regulating apoptosis via down-regulating FAIM2 expression. Our novel insights into miRNA functions will elaborate the effects of miR-193a-3p in preventing IH-mediated endothelial injury by negatively regulating FAIM2, with the goal of providing new treatments for OSA-related cardiovascular diseases.
Results
IH-induced endothelial damage in HUVECs
To evaluate the role of IH conditions for endothelial function, cell viability was detected exposure to normoxia or IH conditions. The results showed that IH treatment significantly decreased cell viability in HUVECs (Figure 1A). Meanwhile, western blot analysis showed that the activities of caspase-3 and the pro-apoptotic protein Bax expression were significantly increased, whereas markedly decreased anti-apoptotic Bcl-2 expression when compared to the normoxia group (Figure 1B and 1C).
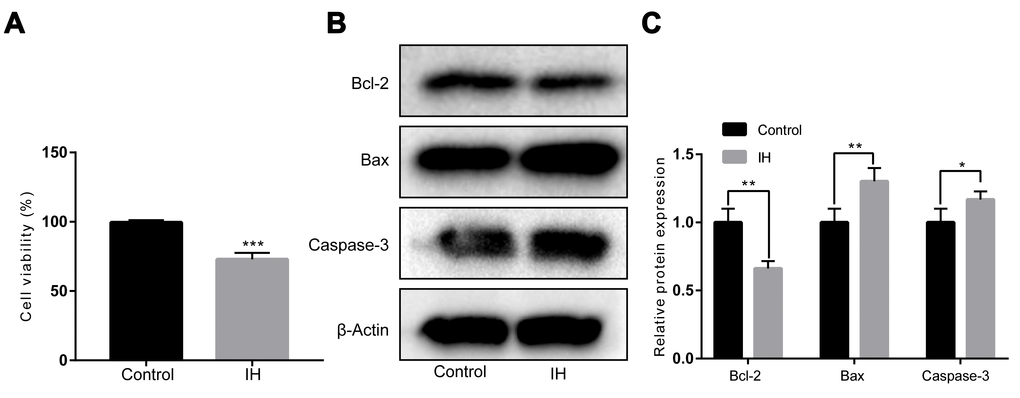
Figure 1. IH inhibits cell viability in HUVECs. (A) Cell viability by a Cell Counting Kit-8. (B, C) Western blotting assays for Bcl-2, Bax, and Caspase-3 protein levels. β-Actin was served as internal control. IH: intermittent hypoxia; n = 3. (Data are presented as the mean ± SD of three independent experiments. *P < 0.05, **P < 0.01, and ***P < 0.001).
miR-193a-3p was upregulated in HUVECs exposed to IH
To assess the effect of miR-193a-3p in endothelial function, we first measured the expression levels of miR-193a-3p in IH-mediated HUVECs by RT-qPCR. As shown in Figure 2A, miR-193a-3p was significantly up-regulated by IH compared to the control group (P < 0.001). Next, to investigate the roles of miR-193a-3p, transfection of HUVECs with the miR-193a-3p inhibitor, or negative control was further performed. After transfection, the expression of miR-193a-3p was determined by RT-qPCR. As expected, miR-193a-3p had a remarkable reduction after transfecting with miR-193a-3p inhibitor when compared to the negative control group (P < 0.0001; Figure 2B). These outcomes demonstrated that the transfection was efficient.
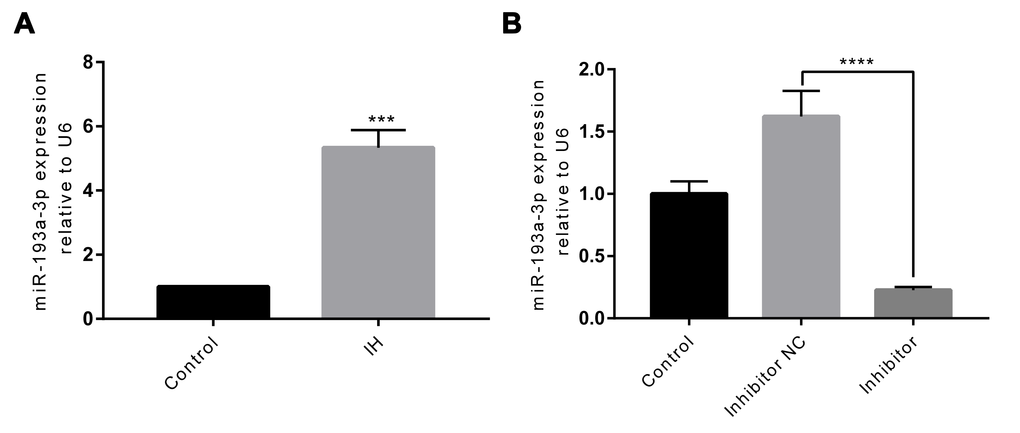
Figure 2. IH induces upregulation of miR-193a-3p, and miR-193a-3p is inhibited in HUVECs after transfection. (A) miR-193a-3p expression was measured by RT-qPCR. (B) Cells were transfected with miR-193a-3p inhibitor, and negative control. Relative miR-193a-3p expression was normalized to U6. IH: intermittent hypoxia; n = 3. (Data are presented as the mean ± SD of three independent experiments. ***P < 0.001, and ****P < 0.0001).
miR-193a-3p inhibition alleviated IH-induced endothelial injury
To validate if miR-193a-3p inhibitor can protect HUVECs from IH-induced injury, we carried out miR-193a-3p knockdown experiments. As shown in Figure 3A, results from CCK-8 assay indicated that the cell viability of HUVECs was notably higher than that in the control group after transfecting with miR-193a-3p inhibitor (P < 0.05). Additionally, the apoptosis-associated proteins Bcl-2, Bax and Caspase-3 were measured by western blotting. It showed that inhibition of miR-193a-3p significantly increased the expression of Bcl-2, whereas markedly decreased Bax and Caspase-3 expression in HUVECs exposure to IH (Figure 3B and 3C).
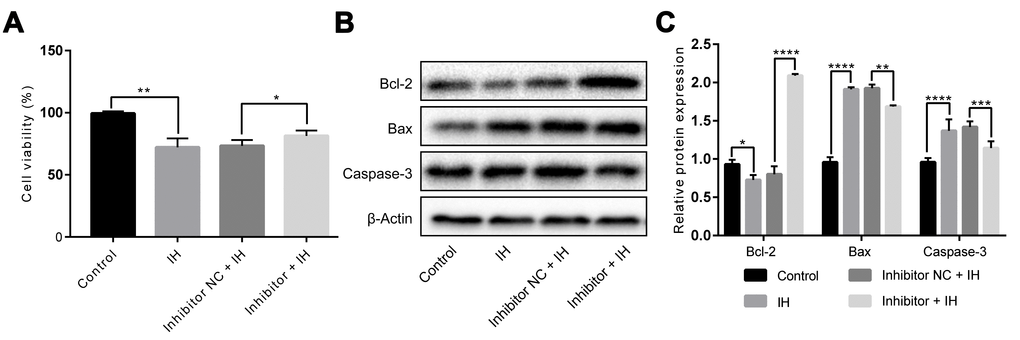
Figure 3. miR-193a-3p silence alleviates IH-induced injury in HUVECs. Cells were transfected with miR-193a-3p inhibitor, and negative control. Cells with normoxia treatment were acted as control. (A) Cell viability. (B, C) Expression levels of apoptosis-related proteins. β-Actin was served as internal control. IH: intermittent hypoxia; n = 3. (Data are presented as the mean ± SD of three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001).
miR-193a-3p directly targeted FAIM2, and inhibited FAIM2 expression
We carried out bioinformatic analysis to explore the potential mechanism underlying miR-193a-3p inhibition suppressed IH-induced endothelial injury. Using miRbase, starBase, and TargetScan, FAIM2 was predicted as a new target of miR-193a-3p. The binding site between FAIM2 3′UTR and miR-193a-3p is shown in Figure 4A. Next, we performed a dual-luciferase reporter assay to confirm whether miR-193a-3p directly targeted to the 3′UTR of FAIM2. As shown in Figure 4B, the results demonstrated that luciferase activity was significantly decreased in HUVECs co-transfected with miR-193a-3p mimics and FAIM2-WT compared to that of co-transfection with mimics control and FAIM2-WT. Additionally, the results also revealed that expressions of FAIM2 at mRNA and protein levels were markedly increased by knockdown of miR-193a-3p compared to the control group(Figure 4C to 4E). Collectively, these results identified that FAIM2 is a novel direct target of miR-193a-3p.
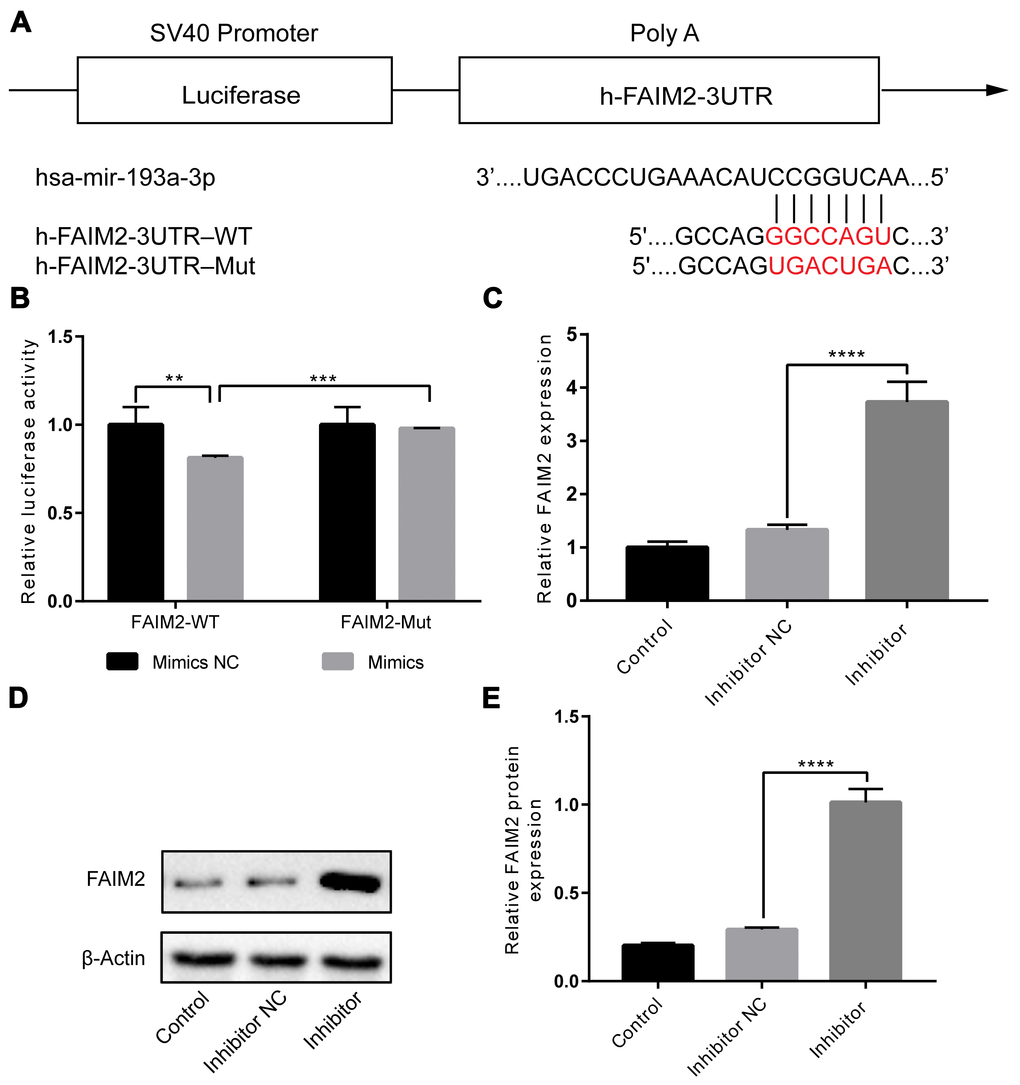
Figure 4. FAIM2 is a target of miR-193a-3p, and FAIM2 could be inhibited by miR-193a-3p in HUVECs. (A) The presumptive binding site of miR-193a-3p in the 3′-UTR of FAIM2. (B) Luciferase reporter assay. We cotransfected HUVECs with wild-type or mutant FAIM2 3′-UTR reporters and miR-193a-3p mimics or corresponding control. (C–E) HUVECs were transfected with miR-193a-3p mimics or corresponding control. mRNA and protein expressions of FAIM2 were determined by western blot. n = 3. (Data are presented as the mean ± SD of three independent experiments. **P < 0.01, ***P < 0.001, and ****P < 0.0001).
Knockdown of FAIM2 eliminated the protective effects of miR-193a-3p inhibition against IH-induced injury in HUVECs
Finally, we try to validate whether FAIM2 is linked to the effects of miR-193a-3p on IH-induced injury. HUVECs were transfected with si-FAIM2, miR-193a-3p inhibitor, or corresponding negative control. As shown in Figure 5A to 5C, the effectiveness of miR-193a-3p inhibition on cell viability, and the expression of apoptosis-related proteins were all reversed by knockdown of FAIM2 compared to the control group under IH condition. Therefore, we come up with the conclusion that miR-193a-3p silence may ameliorate IH-mediated endothelial injury through up-regulating FAIM2.
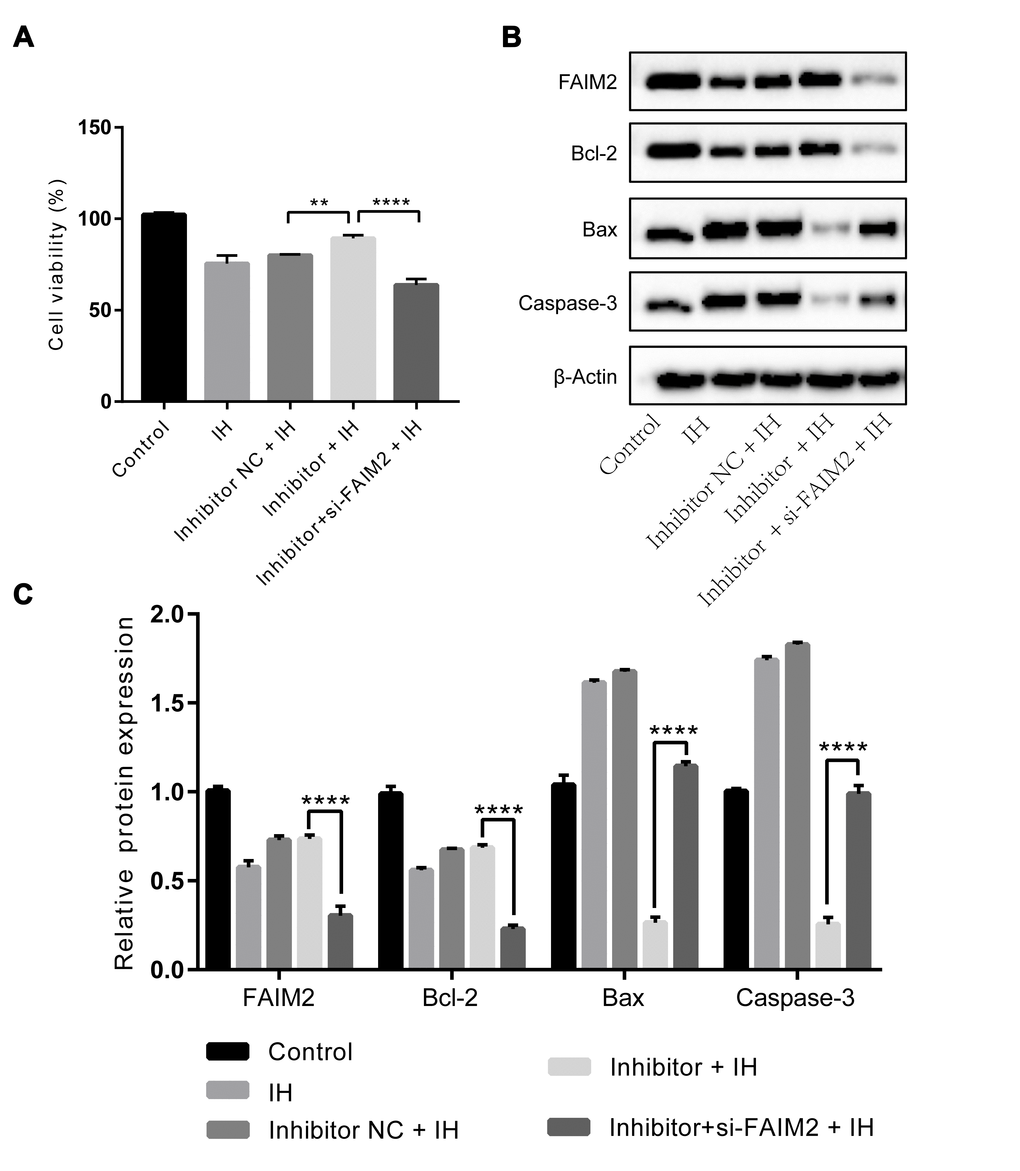
Figure 5. Effects of miR-193a-3p inhibition in HUVECs under IH condition are reversed by knockdown of FAIM2.miR-193a-3p inhibitor, si-FAIM2, and corresponding scrambled control were transfected into HUVECs. Cells without transfection were served as control. (A) Cell viability. (B, C) Western blot assays of FAIM2, Bcl-2, Bax, and Caspase-3 protein. β-Actin was served as internal control. IH: intermittent hypoxia; n = 3. (Data are presented as the mean ± SD of three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001).
Discussion
In the present study, our data indicated that IH could induced injury in HUVECs and miR-193a-3p was remarkably up-regulated under IH condition. However, miR-193a-3p inhibitor could protect HUVECs against IH-induced damage, as evidenced by the improvement of cell viability, the down-regulation of Bax, Caspase-3 and the up-regulation of Bcl-2. After that, miR-193a-3p was validated to inhibit FAIM2 and FAIM2 was further identified as a novel direct target of miR-193a-3p by luciferase reporter assay. Finally, effects of miR-193a-3p suppression on HUVECs could be relieved by knockdown of FAIM2. To our knowledge, the current study first revealed that inhibition of miR-193a-3p could protect HUVECs against IH-induced injury by targeting FIAM2.
OSA, characterized by intermittent hypoxia, is considered as an independent risk factor for a variety of cardiovascular diseases, including myocardial ischemia, hypertension, atherosclerosis and heart failure [8, 22]. Multiple contributing factors supporting the potential association between OSA and cardiovascular diseases have been suggested, including intermittent hypoxia, oxidative stress, increased sympathetic activity, and systemic inflammation, all of which may be linked to endothelial dysfunction [8, 23, 24]. Endothelial dysfunction is an important onset in the pathogenesis of atherosclerosis and other cardiovascular disease [25, 26]. Studies have confirmed the association between OSA or IH and endothelial dysfunction [27–29]. IH during OSA leads to several pathological responses including oxidative stress and inflammation, which is suggested to account for endothelial dysfunction [30, 31]. In a word, consistent evidence shows that OSA may cause endothelial dysfunction. As yet, little is known about the processes leading from endothelial dysfunction to pathological changes of cardiovascular consequences in OSA. In our study, IH stimulation significantly reduced cell viability and promoted cell apoptosis in endothelial cells. Therefore, how to relieve IH-related endothelial injury arouses more and more concern.
In the past decade, a number of microRNAs have already been demonstrated to play crucial roles in the biological functions of endothelial cells (ECs), such as cell proliferation, migration, apoptosis, and differentiation [30–34]. Liu et al. disclosed that miR-495 regulated the proliferation and apoptosis of HUVECs by directly targeting CCL2 [35]. Similarly, miR-497 was identified to play an important role in the development of atherosclerosis by inducing apoptosis and suppressing the proliferation of HUVECs [36]. These all highlighted the critical role miRNAs involved in the apoptosis of ECs. Furthermore, miRNAs are also involved in the regulation of initiation and development of cardiovascular disease [37, 38]. For example, miR-208 and miR-1 are identified as novel biomarkers for cardiovascular diseases, including coronary artery disease, essential hypertension and heart failure [39]. Our results showed that miR-193a-3p was markedly upregulated after IH stimulation, implying the crucial function of miR-193a-3p in the progression of IH-induced endothelial injury. Therefore, we chose miR-193a-3p to explore the relationship between miRNAs and endothelial injury induced by IH in the present study.
To the best of our knowledge, our study is the first report about the function of miR-193a-3p in HUVECs under IH condition. It has now been revealed that miR-193a-3p plays a vital role in multiple diseases, such as acute myeloid leukemia [40], osteosarcoma cells [41] and colorectal cancer [42]. Recent studies have declared that miR-193a-3p participated in various biological processes, such as proliferation, migration, and apoptosis [43]. For example, miR-193a-3p overexpression can promote apoptosis and inhibit proliferation in H295R cells by targeting CYP11B2 [44]. Our study showed that miR-193a-3p inhibitor could reverse IH-induced apoptosis in HUVECs. Taken together, we identified that miR-193a-3p could mediate IH-induced endothelial injury in HUVECs.
To further clarify the mechanism of miR-193a-3p in HUVECs proliferation and apoptosis, we performed bioinformatic analysis and dual-luciferase reporter assay to find its target gene. By using miRbase, starBase, and TargetScan software, we found that 3′-UTR of FAIM2 contained the putative binding sites for miR-193a-3p. FAIM2, also called Lifeguard (LFG) or neural membrane protein 35 (NMP35), is an anti-apoptotic protein known as a distinct gene of the LFG family [45]. FAIM2 also takes part in other apoptotic-independent processes, such as axonal growth, neuronal differentiation, and neuroplasticity [46, 47]. Next, we demonstrated that overexpression of miR-193a-3p resulted in suppression of luciferase activity. In addition, we observed that miR193a-3p downregulation significantly increased mRNA and protein expression of FAIM2 in HUVECs under IH condition. Based on the above data, we indicated that FAIM2 is an important direct target of miR-193a-3p in HUVECs during IH. Finally, FAIM2 suppression could abolish the inhibitory effect of miR-193a-3p inhibitor on HUVECs proliferation and apoptosis under IH. In brief, our study first demonstrated that downregulation of miR-193a-3p attenuated IH-induced HUVECs injury by targeting FAIM2.
The goal of our study was only to assess the effect and potential mechanism of miR-193a-3p inhibition in vitro experiments just as a preliminary exploration. However, we must acknowledge that our study presents some limitations. Firstly, different stimulation times of IH is likely to show different effects on HUVECs, which requires to be further verified. Secondly, we did not investigate the morphological change of apoptosis. Thirdly, this study was conducted in vitro, therefore, more in vivo experiments are still needed to confirm the present observations in the future. Fourthly, we did not perform these experiments using a second human endothelial cell line or primary cells, which had to be considered as a disadvantage. Finally, other miRNAs and genes are likely to play critical roles in IH-induced endothelial injury. A single miRNA could regulate various target genes, and vice versa. Therefore, we will focus our attention on roles of other miRNAs and target genes on IH-induced endothelial injury in future studies.
Taken together, we confirmed that miR-193a-3p was increased in HUVECs under IH condition and miR-193a-3p inhibition could protect HUVECs from IH-induced injury. In addition, we first identified that miR-193a-3p down-regulation mediated IH-induced endothelial injury by regulating FAIM2 expression. Our findings will provide a novel understanding of the mechanism of IH-induced endothelial injury and thus serve as a potential therapeutic target for treating OSA-associated cardiac diseases.
Materials and Methods
Cell culture
Human umbilical vein endothelial cells (HUVECs) was purchased from the Cell Bank of the Chinese Academy of Sciences(Shanghai, China). Cells were cultured in Dulbecco’s modified Eagle’s medium (HyClone) containing 10% fetal bovine serum (Gibco) and 1% penicillin/streptomycin, in a cell incubator with 5% CO2 at 37°C (Thermo, Waltham, MA, USA). HUVECs were found to be negative for mycoplasma by PCR to exclude the possibility of cryptic contamination.
Establishment of IH model
When HUVECs were propagated to 70-80% confluence, the method of IH stimulation was carried out as previously described [48], with slight modifications. In brief, cells were maintained under hypoxia condition induced by flushing a mixed air of 1% O2 and 5% CO2 balanced with N2 for 35 min, and then normoxia condition (21% O2 with 5% CO2 balanced with N2 for 25 min). Repeated IH exposure was performed for 6 times.
MiRNA target prediction
To predict the potential target genes of miR-193a-3p, three different miRNA target prediction algorithms: TargetScan7.2 (http://www.targetscan.org/), starBase (http://starbase.sysu.edu.cn/) and miRbase (http://www.mirbase.org/) were employed. Considering the high false positive rates of prediction, the three prediction tools were combined used to improve the quality of miRNA target prediction.
Real-time quantitative PCR (RT-qPCR)
After intervention, mRNA of HUVECs was isolated using Trizol reagent (Takara) according to manufacturer’s protocol. To analyze the expression of miR-193a-3p, the RevertAidTM First Strand cDNA Synthesis Kit (#K1622; Thermo) with a special stem-loop primer and SYBR Green PCR Master Mix (#K0223; Thermo) were used to reverse transcription and quantitative PCR. To detect the expression level of FAIM2, the One Step SYBR® PrimeScript® PLUS RT-RNA PCR Kit (Takara) was applied. U6 and Actin were used as an internal control. The RT-qPCR was performed on ABI 7500 thermocycler (Applied Biosystems, Foster City, CA, USA). Each sample was measured in triplicate. Relevant primers were listed in the Table 1. The relative expression of qPCR results was calculated by the 2−ΔΔCT method.
Table 1. Primers used for RT-qPCR.
| ID | Sequence (5′-3′) |
| miR-193a-3p | Sense: ACACTCCAGCTGGGTGGGTCTTTGCGGGCG |
| Antisense: TGGTGTCGTGGAGTCG | |
| miR-193a-3p inhibitor | ACUGGGACUUUGUAGGCCAGUU |
| Inhibitor control | CAGUACUUUUGUGUAGUACAA |
| FAIM2 | Sense: AGTTCGTCGAGTCTTTGTCAGA |
| Antisense: GGGTCCAGAACAGCAAGC | |
| si-FAIM2 | Sense: GCGGGUGUAUUUACAUUGUTT |
| Antisense: ACAAUGUAAAUACACCCGCTT | |
| U6 | Sense: CTCGCTTCGGCAGCACA |
| Antisense: AACGCTTCACGAATTTGCGT | |
| β-Actin | Sense: TGGACTTCGAGCAAGAGATG |
| Antisense: TGTTGGCGTACAGGTCTTTG |
Cell transfection
miR-193a-3p inhibitor, small interfering RNA targeting FAIM2 (si-FAIM2), and corresponding scrambled control were chemically synthesized by Sangon Biotech Co.(Shanghai, China). When HUVECs in 6-well plates grew to about 80% confluence, we replaced the medium with serum-free medium. The cells were then transfected with miR-193a-3p inhibitor, si-FAIM2 and corresponding scrambled control using Lipofectamine 3000 (Invitrogen, USA) following manufacturer’s instructions. Cells were then exposed to IH.
CCK-8 assay
The cell viability was detected by CCK-8 assay (TransGen Biotech, Beijing, China) following the manufacturer’s instructions. HUVECs were plated in 96-well (5 × 103 cells/well). After IH stimulation, 10ul/well of CCK-8 was added into each well. Next, the mixture of 96-well plates was maintained at cell incubator for additional 2h. Finally, the absorbance was measured at 450nm with the use of a Multiskan GO Spectrophotometer (Thermo Fisher Scientific, USA).
Western blot analysis
Proteins were extracted by using Mammalian Protein Extract on Reagent (CWBIO, Beijing, China) supplemented with protease inhibitors. Subsequently, BCA Protein Assay Kit (CWBIO, Beijing, China) was performed to determine protein concentrations. Equal amounts of protein were then separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride (PVDF) membranes. Next, the membranes were blocked in 5% non-fat dry milk for 1 h, and then followed by incubation with primary antibodies at 4°C overnight. After washes, relevant secondary antibodies were applied at room temperature for 1 h. Finally, the membranes were washed and developed using standard chemiluminescence and the Bio-Rad ChemiDoc™ XRS+System. The intensity of bands was analyzed with Image-Pro Plus 6.0 software (Media Cybernetics, Rockville, MD, USA) and normalized to β-Actin.
Dual-luciferase reporter assay
The fragment from FAIM2 3′-untranslated region (3′UTR), containing the predicted miR-193a-3p binding sequence, was amplified by PCR. To amplify the sequence for the mutation within the miR-193a-3p binding sites, we applied the point mutation method by using the KOD-Plus mutagenesis kit (Toyobo, Osaka, Japan). For dual-luciferase reporter experiments, the pSI-Check2 luciferase reporter vector containing the binding sites of 3′-UTR of FAIM2 mRNA or mutant 3′-UTR of FAIM2 was cotransfected with miR-193a-3p mimics or negative controls into HUVECs using LipofectamineTM 3000. After 48 h, we measured the firefly luciferase and renilla luciferase activity by a fluorescence detector (Promega). Renilla luciferase activities were normalized as control for each transfected well. Each experiment was replicated in triplicate.
Statistics and data analysis
All statistical analyses were performed with SPSS 22.0 software. All data are presented as mean ± SD. Differences were compared by one-way analysis of variance, followed by a modified Student’s t test. Differences were considered statistically significant if p < 0.05. All experiments were repeated at least three times.
Author Contributions
Qingshi Chen, Guofu Lin and Jianchai Huang: conceived of the study, carried out the molecular studies. Lida Chen, Yibin Liu and Jiefeng Huang: participated in the design of the study and performed the statistical analysis. Shuyi Zhang: carried out the molecular studies. Qichang Lin: conceived of the study.
Acknowledgments
We would like to appreciate the reviewers for their valuable comments on this article.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study was funded by the Chinese National Natural Science Foundation [grant number: 81870074]; Science and Technology Projects of Quanzhou [grant numbers: 2018N007S, and Z (2014) 0127]; Science and Technology Project of Fujian Education Department [grant number: JT180199] and Startup Fund for Scientific Research, Fujian Medical University [grant number: 2017XQ1102, and 2018QH1057].
References
- 1. Heinzer R, Vat S, Marques-Vidal P, Marti-Soler H, Andries D, Tobback N, Mooser V, Preisig M, Malhotra A, Waeber G, Vollenweider P, Tafti M, Haba-Rubio J. Prevalence of sleep-disordered breathing in the general population: the HypnoLaus study. Lancet Respir Med. 2015; 3:310–18. https://doi.org/10.1016/S2213-2600(15)00043-0 [PubMed]
- 2. Peppard PE, Young T, Barnet JH, Palta M, Hagen EW, Hla KM. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol. 2013; 177:1006–14. https://doi.org/10.1093/aje/kws342 [PubMed]
- 3. Gautier-Veyret E, Pépin JL, Stanke-Labesque F. Which place of pharmacological approaches beyond continuous positive airway pressure to treat vascular disease related to obstructive sleep apnea? Pharmacol Ther. 2018; 186:45–59. https://doi.org/10.1016/j.pharmthera.2017.12.006 [PubMed]
- 4. Murphy AM, Thomas A, Crinion SJ, Kent BD, Tambuwala MM, Fabre A, Pepin JL, Roche HM, Arnaud C, Ryan S. Intermittent hypoxia in obstructive sleep apnoea mediates insulin resistance through adipose tissue inflammation. Eur Respir J. 2017; 49:1601731. https://doi.org/10.1183/13993003.01731-2016 [PubMed]
- 5. Andrade AG, Bubu OM, Varga AW, Osorio RS. The Relationship between Obstructive Sleep Apnea and Alzheimer’s Disease. J Alzheimers Dis. 2018; 64:S255–70. https://doi.org/10.3233/JAD-179936 [PubMed]
- 6. Marin JM, Agusti A, Villar I, Forner M, Nieto D, Carrizo SJ, Barbé F, Vicente E, Wei Y, Nieto FJ, Jelic S. Association between treated and untreated obstructive sleep apnea and risk of hypertension. JAMA. 2012; 307:2169–76. https://doi.org/10.1001/jama.2012.3418 [PubMed]
- 7. Kohler M, Craig S, Nicoll D, Leeson P, Davies RJ, Stradling JR. Endothelial function and arterial stiffness in minimally symptomatic obstructive sleep apnea. Am J Respir Crit Care Med. 2008; 178:984–88. https://doi.org/10.1164/rccm.200805-717OC [PubMed]
- 8. Hoyos CM, Melehan KL, Liu PY, Grunstein RR, Phillips CL. Does obstructive sleep apnea cause endothelial dysfunction? A critical review of the literature. Sleep Med Rev. 2015; 20:15–26. https://doi.org/10.1016/j.smrv.2014.06.003 [PubMed]
- 9. Schwarz EI, Puhan MA, Schlatzer C, Stradling JR, Kohler M. Effect of CPAP therapy on endothelial function in obstructive sleep apnoea: A systematic review and meta-analysis. Respirology. 2015; 20:889–95. https://doi.org/10.1111/resp.12573 [PubMed]
- 10. Carreras A, Zhang SX, Peris E, Qiao Z, Gileles-Hillel A, Li RC, Wang Y, Gozal D. Chronic sleep fragmentation induces endothelial dysfunction and structural vascular changes in mice. Sleep . 2014; 37:1817–24. https://doi.org/10.5665/sleep.4178 [PubMed]
- 11. Champod AS, Eskes GA, Foster GE, Hanly PJ, Pialoux V, Beaudin AE, Poulin MJ. Effects of acute intermittent hypoxia on working memory in young healthy adults. Am J Respir Crit Care Med. 2013; 187:1148–50. https://doi.org/10.1164/rccm.201209-1742LE [PubMed]
- 12. Beaudin AE, Pun M, Yang C, Nicholl DD, Steinback CD, Slater DM, Wynne-Edwards KE, Hanly PJ, Ahmed SB, Poulin MJ. Cyclooxygenases 1 and 2 differentially regulate blood pressure and cerebrovascular responses to acute and chronic intermittent hypoxia: implications for sleep apnea. J Am Heart Assoc. 2014; 3:e000875. https://doi.org/10.1161/JAHA.114.000875 [PubMed]
- 13. Liu Q, Wu DH, Han L, Deng JW, Zhou L, He R, Lu CJ, Mi QS. Roles of microRNAs in psoriasis: immunological functions and potential biomarkers. Exp Dermatol. 2017; 26:359–67. https://doi.org/10.1111/exd.13249 [PubMed]
- 14. Javadian M, Gharibi T, Shekari N, Abdollahpour-Alitappeh M, Mohammadi A, Hossieni A, Mohammadi H, Kazemi T. The role of microRNAs regulating the expression of matrix metalloproteinases (MMPs) in breast cancer development, progression, and metastasis. J Cell Physiol. 2019; 234:5399–412. https://doi.org/10.1002/jcp.27445 [PubMed]
- 15. Yin Y, Lv L, Wang W. Expression of miRNA-214 in the sera of elderly patients with acute myocardial infarction and its effect on cardiomyocyte apoptosis. Exp Ther Med. 2019; 17:4657–62. https://doi.org/10.3892/etm.2019.7464 [PubMed]
- 16. Guan Y, Song X, Sun W, Wang Y, Liu B. Effect of Hypoxia-Induced MicroRNA-210 Expression on Cardiovascular Disease and the Underlying Mechanism. Oxid Med Cell Longev. 2019; 2019:4727283. https://doi.org/10.1155/2019/4727283 [PubMed]
- 17. Zhang X, Xiao XP, Ren XA, Cui T. Plasma miRNA-155 Levels Predict Atrial Fibrillation Recurrence after Cardioversion. Heart Surg Forum. 2019; 22:E140–48. https://doi.org/10.1532/hsf.2281 [PubMed]
- 18. Heymans S, Corsten MF, Verhesen W, Carai P, van Leeuwen RE, Custers K, Peters T, Hazebroek M, Stöger L, Wijnands E, Janssen BJ, Creemers EE, Pinto YM, et al. Macrophage microRNA-155 promotes cardiac hypertrophy and failure. Circulation. 2013; 128:1420–32. https://doi.org/10.1161/CIRCULATIONAHA.112.001357 [PubMed]
- 19. Liu X, Min S, Wu N, Liu H, Wang T, Li W, Shen Y, Zhao C, Wang H, Qian Z, Xu H, Chen Y, Wang X. miR-193a-3p inhibition of the Slug activator PAK4 suppresses non-small cell lung cancer aggressiveness via the p53/Slug/L1CAM pathway. Cancer Lett. 2019; 447:56–65. https://doi.org/10.1016/j.canlet.2019.01.027 [PubMed]
- 20. Pekow J, Meckel K, Dougherty U, Huang Y, Chen X, Almoghrabi A, Mustafi R, Ayaloglu-Butun F, Deng Z, Haider HI, Hart J, Rubin DT, Kwon JH, Bissonnette M. miR-193a-3p is a Key Tumor Suppressor in Ulcerative Colitis-Associated Colon Cancer and Promotes Carcinogenesis through Upregulation of IL17RD. Clin Cancer Res. 2017; 23:5281–91. https://doi.org/10.1158/1078-0432.CCR-17-0171 [PubMed]
- 21. Deng H, Lv L, Li Y, Zhang C, Meng F, Pu Y, Xiao J, Qian L, Zhao W, Liu Q, Zhang D, Wang Y, Zhang H, et al. miR-193a-3p regulates the multi-drug resistance of bladder cancer by targeting the LOXL4 gene and the oxidative stress pathway. Mol Cancer. 2014; 13:234. https://doi.org/10.1186/1476-4598-13-234 [PubMed]
- 22. Schillaci G, Battista F, Fiorenzano G, Basili MC, Crapa M, Alrashdi Y, Pucci G. Obstructive Sleep Apnea and Cardiovascular Disease - A New Target for Treatment. Curr Pharm Des. 2015; 21:3496–504. https://doi.org/10.2174/1381612821666150706110544 [PubMed]
- 23. Eisele HJ, Markart P, Schulz R. Obstructive Sleep Apnea, Oxidative Stress, and Cardiovascular Disease: Evidence from Human Studies. Oxid Med Cell Longev. 2015; 2015:608438. https://doi.org/10.1155/2015/608438 [PubMed]
- 24. Baguet JP, Barone-Rochette G, Tamisier R, Levy P, Pépin JL. Mechanisms of cardiac dysfunction in obstructive sleep apnea. Nat Rev Cardiol. 2012; 9:679–88. https://doi.org/10.1038/nrcardio.2012.141 [PubMed]
- 25. Gutiérrez E, Flammer AJ, Lerman LO, Elízaga J, Lerman A, Fernández-Avilés F. Endothelial dysfunction over the course of coronary artery disease. Eur Heart J. 2013; 34:3175–81. https://doi.org/10.1093/eurheartj/eht351 [PubMed]
- 26. Endemann DH, Schiffrin EL. Endothelial dysfunction. J Am Soc Nephrol. 2004; 15:1983–92. https://doi.org/10.1097/01.ASN.0000132474.50966.DA [PubMed]
- 27. Lavie L. Oxidative stress in obstructive sleep apnea and intermittent hypoxia—revisited—the bad ugly and good: implications to the heart and brain. Sleep Med Rev. 2015; 20:27–45. https://doi.org/10.1016/j.smrv.2014.07.003 [PubMed]
- 28. Lui MM, Lam DC, Ip MS. Significance of endothelial dysfunction in sleep-related breathing disorder. Respirology. 2013; 18:39–46. https://doi.org/10.1111/j.1440-1843.2012.02212.x [PubMed]
- 29. Lee MY, Wang Y, Mak JC, Ip MS. Intermittent hypoxia induces NF-κB-dependent endothelial activation via adipocyte-derived mediators. Am J Physiol Cell Physiol. 2016; 310:C446–55. https://doi.org/10.1152/ajpcell.00240.2015 [PubMed]
- 30. Golbidi S, Badran M, Ayas N, Laher I. Cardiovascular consequences of sleep apnea. Lung. 2012; 190:113–32. https://doi.org/10.1007/s00408-011-9340-1 [PubMed]
- 31. Badran M, Golbidi S, Devlin A, Ayas N, Laher I. Chronic intermittent hypoxia causes endothelial dysfunction in a mouse model of diet-induced obesity. Sleep Med. 2014; 15:596–602. https://doi.org/10.1016/j.sleep.2014.01.013 [PubMed]
- 32. Qu K, Wang Z, Lin XL, Zhang K, He XL, Zhang H. MicroRNAs: key regulators of endothelial progenitor cell functions. Clin Chim Acta. 2015; 448:65–73. https://doi.org/10.1016/j.cca.2015.06.017 [PubMed]
- 33. Santulli G. MicroRNAs and Endothelial (Dys) Function. J Cell Physiol. 2016; 231:1638–44. https://doi.org/10.1002/jcp.25276 [PubMed]
- 34. Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009; 136:215–33. https://doi.org/10.1016/j.cell.2009.01.002 [PubMed]
- 35. Liu D, Zhang XL, Yan CH, Li Y, Tian XX, Zhu N, Rong JJ, Peng CF, Han YL. MicroRNA-495 regulates the proliferation and apoptosis of human umbilical vein endothelial cells by targeting chemokine CCL2. Thromb Res. 2015; 135:146–54. https://doi.org/10.1016/j.thromres.2014.10.027 [PubMed]
- 36. Wu R, Tang S, Wang M, Xu X, Yao C, Wang S. MicroRNA-497 Induces Apoptosis and Suppresses Proliferation via the Bcl-2/Bax-Caspase9-Caspase3 Pathway and Cyclin D2 Protein in HUVECs. PLoS One. 2016; 11:e0167052. https://doi.org/10.1371/journal.pone.0167052 [PubMed]
- 37. Bao MH, Feng X, Zhang YW, Lou XY, Cheng Y, Zhou HH. Let-7 in cardiovascular diseases, heart development and cardiovascular differentiation from stem cells. Int J Mol Sci. 2013; 14:23086–102. https://doi.org/10.3390/ijms141123086 [PubMed]
- 38. Roncarati R, Viviani Anselmi C, Losi MA, Papa L, Cavarretta E, Da Costa Martins P, Contaldi C, Saccani Jotti G, Franzone A, Galastri L, Latronico MV, Imbriaco M, Esposito G, et al. Circulating miR-29a, among other up-regulated microRNAs, is the only biomarker for both hypertrophy and fibrosis in patients with hypertrophic cardiomyopathy. J Am Coll Cardiol. 2014; 63:920–27. https://doi.org/10.1016/j.jacc.2013.09.041 [PubMed]
- 39. Xu J, Zhao J, Evan G, Xiao C, Cheng Y, Xiao J. Circulating microRNAs: novel biomarkers for cardiovascular diseases. J Mol Med (Berl). 2012; 90:865–75. https://doi.org/10.1007/s00109-011-0840-5 [PubMed]
- 40. Fan Q, Hu X, Zhang H, Wang S, Zhang H, You C, Zhang CY, Liang H, Chen X, Ba Y. MiR-193a-3p is an Important Tumour Suppressor in Lung Cancer and Directly Targets KRAS. Cell Physiol Biochem. 2017; 44:1311–24. https://doi.org/10.1159/000485491 [PubMed]
- 41. Baumhoer D, Zillmer S, Unger K, Rosemann M, Atkinson MJ, Irmler M, Beckers J, Siggelkow H, von Luettichau I, Jundt G, Smida J, Nathrath M. MicroRNA profiling with correlation to gene expression revealed the oncogenic miR-17-92 cluster to be up-regulated in osteosarcoma. Cancer Genet. 2012; 205:212–19. https://doi.org/10.1016/j.cancergen.2012.03.001 [PubMed]
- 42. Lin M, Duan B, Hu J, Yu H, Sheng H, Gao H, Huang J. Decreased expression of miR-193a-3p is associated with poor prognosis in colorectal cancer. Oncol Lett. 2017; 14:1061–67. https://doi.org/10.3892/ol.2017.6266 [PubMed]
- 43. Seviour EG, Sehgal V, Mishra D, Rupaimoole R, Rodriguez-Aguayo C, Lopez-Berestein G, Lee JS, Sood AK, Kim MP, Mills GB, Ram PT. Targeting KRas-dependent tumour growth, circulating tumour cells and metastasis in vivo by clinically significant miR-193a-3p. Oncogene. 2017; 36:1339–50. https://doi.org/10.1038/onc.2016.308 [PubMed]
- 44. Zhang G, Zou X, Liu Q, Xie T, Huang R, Kang H, Lai C, Zhu J. MiR-193a-3p functions as a tumour suppressor in human aldosterone-producing adrenocortical adenoma by down-regulating CYP11B2. Int J Exp Pathol. 2018; 99:77–86. https://doi.org/10.1111/iep.12267 [PubMed]
- 45. Hu L, Smith TF, Goldberger G. LFG: a candidate apoptosis regulatory gene family. Apoptosis. 2009; 14:1255–65. https://doi.org/10.1007/s10495-009-0402-2 [PubMed]
- 46. Merianda TT, Vuppalanchi D, Yoo S, Blesch A, Twiss JL. Axonal transport of neural membrane protein 35 mRNA increases axon growth. J Cell Sci. 2013; 126:90–102. https://doi.org/10.1242/jcs.107268 [PubMed]
- 47. Tauber SC, Harms K, Falkenburger B, Weis J, Sellhaus B, Nau R, Schulz JB, Reich A. Modulation of hippocampal neuroplasticity by Fas/CD95 regulatory protein 2 (Faim2) in the course of bacterial meningitis. J Neuropathol Exp Neurol. 2014; 73:2–13. https://doi.org/10.1097/NEN.0000000000000020 [PubMed]
- 48. Xie S, Deng Y, Pan YY, Ren J, Jin M, Wang Y, Wang ZH, Zhu D, Guo XL, Yuan X, Shang J, Liu HG. Chronic intermittent hypoxia induces cardiac hypertrophy by impairing autophagy through the adenosine 5′-monophosphate-activated protein kinase pathway. Arch Biochem Biophys. 2016; 606:41–52. https://doi.org/10.1016/j.abb.2016.07.006 [PubMed]


