Introduction
Aging is characterized by a progressive decline in physiological function [1], which is controlled by a complex interaction between environmental and genetic factors [2]. While understanding the biological mechanisms of aging that result from these interactions is an area of intensive investigation, building evidence suggests that alterations in mitochondrial function plays a central role in coordinating the aging process [3]. Mitochondrial bioenergetics of skeletal muscle decline with age, and this decrement may pre-dispose to certain age-related diseases [4–6]. However, the mitochondrial biology of aging theory extends beyond that of altered intrinsic efficiencies in energy production to include a control over inflammatory responses, proteostasis, oxidative balance, stem cell function and initiation of adaptive stress responses [3, 7–9]. Coordination of such a complex array of cellular processes requires diverse mitochondrial initiated pathways of intra- and extra-cellular communication [10–12].
The mammalian mitochondrial proteome consists of >1000 proteins, of which 99% are encoded by the nuclear genome. All 13 proteins traditionally recognized as being encoded by mitochondrial DNA (mtDNA) form essential components of the oxidative phosphorylation (OXPHOS) complexes, with no known nuclear or mitochondrial-independent functions [13]. However, short open reading frames (sORFs) within the mitochondrial genome have recently been identified as harboring sequences for small regulatory peptides [14–17]. These mitochondrial-derived peptides (MDP), or mitokines, appear to form a critical retrograde communication pathway between the mitochondria and the wider cell, and may have an endocrine cytoprotective role [18–21].
MOTS-c, an MDP with its sequence located within the coding region for mitochondrial 12S rRNA gene [14], has recently been shown to be induced by metabolic perturbation and translocate to the nucleus where it is involved in regulating nuclear gene expression, including those with antioxidant response elements (ARE) to protect against metabolic stress [22, 23]. A stress mediated mitonuclear communication role of MOTS-c may partly explain why exogenous MOTS-c is capable of preventing diet, aging and menopause associated metabolic discourse and insulin resistance, but has limited impact on the resting metabolism of healthy young mice [14, 24–27]. Insulin sensitive tissues, such as skeletal muscle and fat, appear to be key target sites of MOTS-c, and levels of MOTS-c in skeletal muscle and plasma of aged mice are reduced [14, 26–28]. This has led to speculation that MOTS-c is an age related mitokine [29, 30], however whether human plasma and muscle MOTS-c levels are influenced by healthy aging is unclear. Therefore, we investigated plasma and vastus lateralis muscle MOTS-c levels in human males.
We report that while plasma MOTS-c levels are reduced with age, muscle levels are increased and this may be associated with aging-related fast-to-slow fiber type transition and the reduced ability of MOTS-c to leave the cell. Furthermore, we provide evidence that MOTS-c transcription is regulated independent of 12S rRNA, and MOTS-c protein or transcript expression does not correlate well with ARE-related skeletal muscle gene expression in resting skeletal muscle during aging.
Results
Plasma MOTS-c levels are reduced with aging while muscle levels are increased
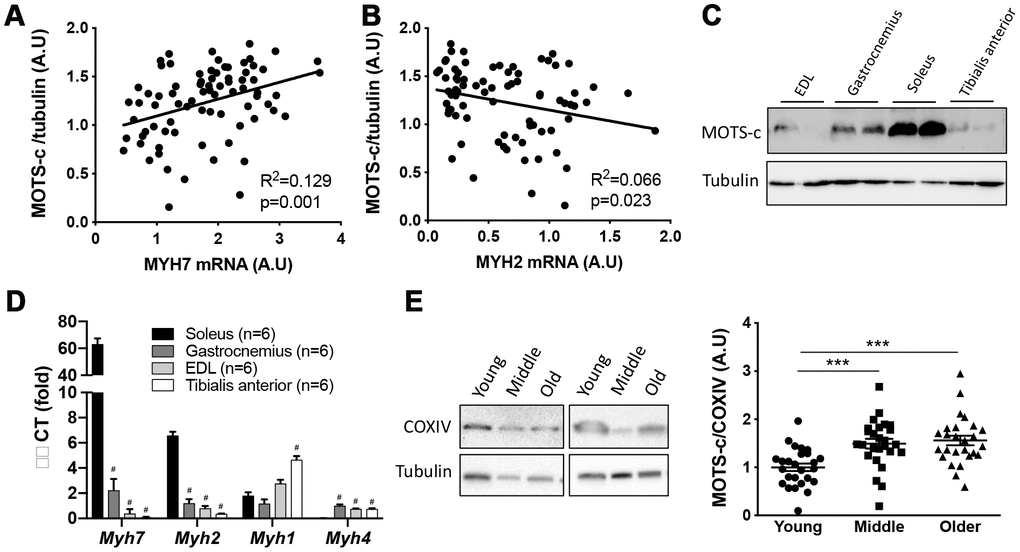
Plasma MOTS-c levels were measured in young (18-30 y), middle-aged (45-55 y) and older (70-81 y) males that were free from any overt disease following an overnight fast (Figure 1). Characteristics of participants are shown in Table 1, while all age groups had similar HOMA-IR, plasma LDL and HDL, the older-aged groups had higher fat mass and lower lean mass, and the middle-aged group high plasma triglycerides. Consistent with murine data [14], both the middle and older groups had lower circulating MOTS-c (by 11 and 21%, respectively) than the young group. Since exogenous MOTS-c treatment of aged or high fat diet challenged mice improves insulin sensitivity and alters body composition [14] we correlated plasma MOTS-c levels with HOMA-IR, and relative fat and lean mass (Figure 1B–1D). A weak association was observed with relative lean mass, but not fat mass or HOMA-IR (Figure 1B, 1C). To determine if differences between the groups could be explained by differences in clinical blood parameter or body composition, ANCOVA analysis was undertaken with % fat, % lean mass, HOMA-IR and plasma triglycerides as covariates (both independently and combined; Supplementary Table 1). With these covariates included the main effect for age on plasma MOTS-c remained significant (p<0.001; Supplementary Table 1) indicating that this is likely an age-dependent effect. To determine if skeletal muscle MOTS-c levels were similarly reduced with aging, MOTS-c protein expression was measured in vastus lateralis muscle samples at rest. In contrast to both plasma data, and MOTS-c muscle levels reported in aged mice [14], both the older and middle-aged groups had higher levels of muscle MOTS-c than the young group (Figure 1E and Supplementary Table 2). Plasma and muscle MOTS-c positively correlated in young, but not middle-aged or combined groups (Figure 1F–1H). The muscle and plasma samples for the older group were from different participants and therefore this comparison could not be made for the older men.
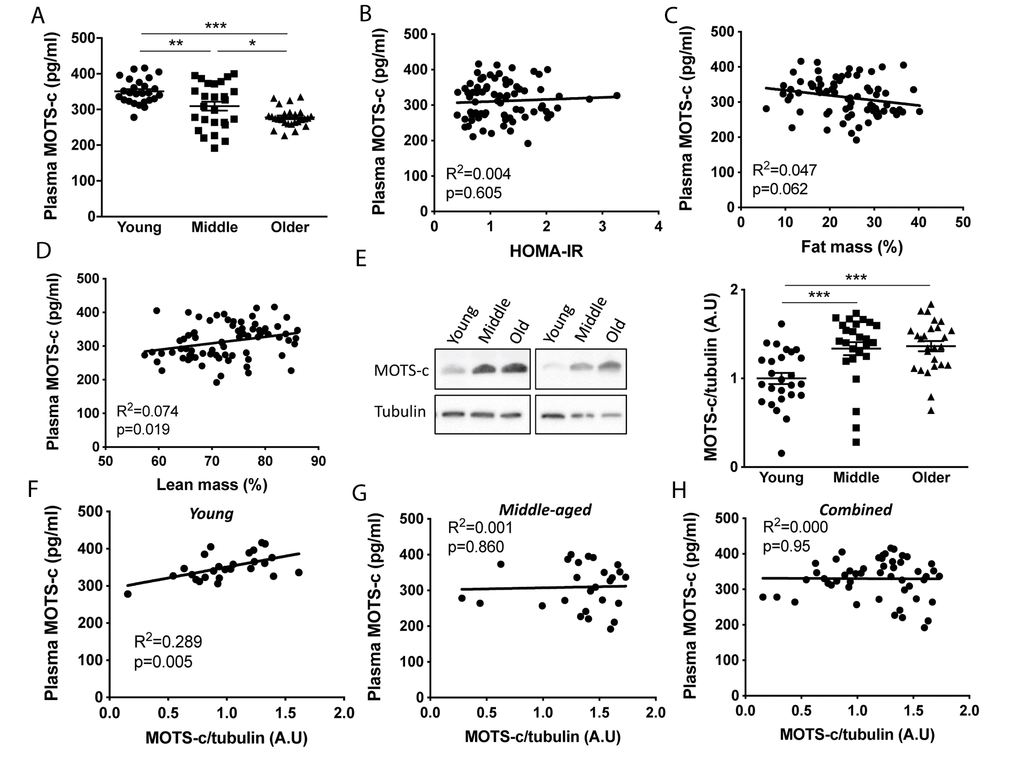
Figure 1. Plasma MOTS-c levels decrease and skeletal muscle levels increase with aging. Plasma MOTS-c (A), correlated with HOMA-IR (B), fat mass (C), lean mass (D), and muscle MOTS-c expression (E) in young, middle-aged and older males. Representative blots are independent and from different participants. Correlation between plasma MOTS-c and muscle MOTS-c expression in young (F), middle (G), and combined (H). Significance was determined using linear regression or one-way ANOVA. Data is presented as means ± SE, n=26 per group except for body composition measurements where data was not available for a young (n=25) and middle-aged (n=25) participant. *p<0.05, **p<0.01, ***p<0.001.
Table 1. Participant characteristics.
| Young | Middle-aged | Older 1 | Older 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (y) | 22 ± 3 | 50 ± 2** | 70 ± 4**,†† | 74 ± 3**,††,‡‡ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Weight (kg) | 77.8 ± 10.8 | 83.9 ± 9.8 | 88.5 ± 13.7* | 85.7 ± 11.7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 24 ± 3 | 26 ± 38 | 28 ± 4** | 28 ± 4** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fat mass (%) | 18.8 ± 7.8 | 23.9 ± 6.1* | NC | 28.4 ± 4.8**,† | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lean mass (%) | 76.9 ± 7.7 | 72.9 ± 5.8 | NC | 67.6 ± 4.6**,†† | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HOMA-IR | 1.28 ± 0.67 | 1.11 ± 0.51 | NC | 1.31 ± 0.56 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL (mmol/L) | 1.35 ± 0.28 | 1.19 ± 0.28 | NC | 1.30 ± 0.34 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL (mmol/L) | 2.8 ± 0.74 | 3.2 ± 0.94 | NC | 3.01 ± 0.94 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TRIG (mmol/L) | 1.02 ± 0.50 | 1.45 ± 0.60* | NC | 1.19 ± 0.62 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Data are mean ± standard deviation for n=26 per group except for body composition measurements where data was not available for a young (n=25) and middle-aged (n=25) participant. NC=not collected; BMI= body mass index; HDL= high density lipoprotein; LDL=low density lipoprotein; TRIG= triglycerides; *p<0.05 vs Young, **p<0.001 vs Young, †p<0.05 vs Middle-aged, ††p<0.01 vs Middle-aged, ‡‡p<0.01 vs Older 1. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Transcriptional regulation of MOTS-c and MT-RNR1 with age
The coding sequence for MOTS-c is found within the 12S rRNA gene (MT-RNR1) of mtDNA (Figure 4A). To determine if MOTS-c transcripts are present in muscle independent of MT-RNR1 transcripts we filtered total RNA to enrich small mRNA fragments (<200 nt) and used a custom TaqMan small RNA assay with a probe that was designed against the 51-nucleotide sequence of MOTS-c to measure MOTS-c mRNAs in the enriched small RNA fraction. Similar to muscle MOTS-c protein expression, both the middle and older aged groups had increased muscle MOTS-c mRNA compared to the young group (Figure 4B), and MOTS-c mRNA correlated with MOTS-c protein levels (Figure 4C). Due to the amount of RNA required for this approach, only 52 samples could be included in this analysis; 18 each from young and middle-aged, and 16 from the older groups.
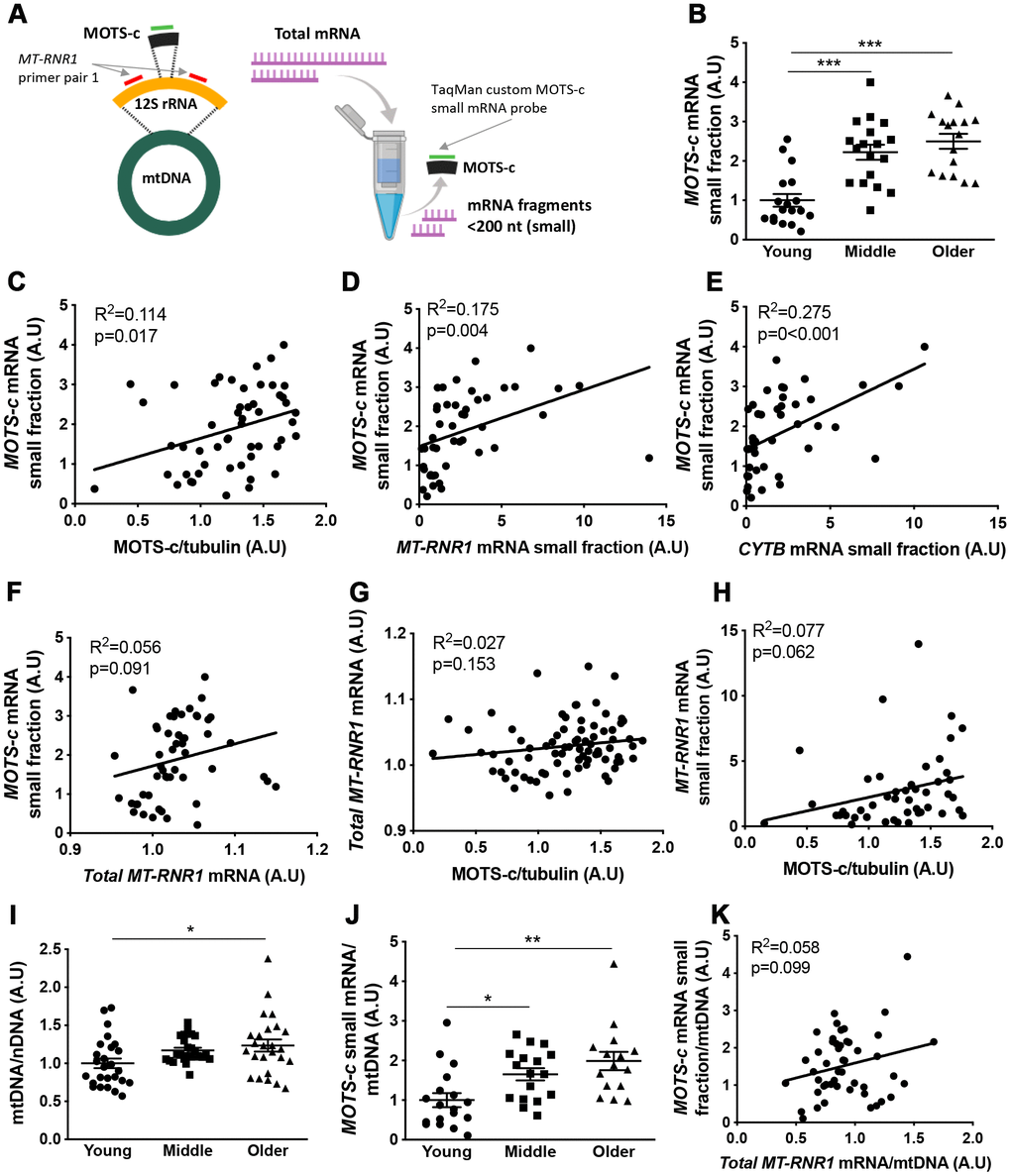
Figure 4. Muscle MOTS-c and 12S rRNA transcription with aging.12S rRNA mRNA (MT-RNR1) and MOTS-c mRNA analysis technique (A). Young, middle-aged and older male muscle MOTS-c mRNA levels in the small RNA fraction were determined (B) and correlated with muscle MOTS-c protein expression (C), MT-RNR1 (D) and CYTB (E) mRNA levels in the small RNA fraction and MT-RNR1 mRNA levels in the total RNA fraction (F). MT-RNR1 mRNA levels in the total (G) and small (H) RNA fraction were correlated with MOTS-c protein expression. Muscle mitochondrial to nuclear DNA (mtDNA/nDNA) (I), and MOTS-c mRNA levels relative to mtDNA (J), and correlation of MOTS-c mRNA and MT-RNR1 mRNA in the total RNA relative to mtDNA (K). Significance was determined using linear regression or one-way ANOVA. Results are shown as means ± SE, due to limited sample availability and assay failure, for small RNA fraction assays n=14-18 per group, and for mtDNA n=26 for young, 24 for middle and 25 for old. *p<0.05, **p<0.01, ***p<0.001.
MOTS-c mRNA correlated with MT-RNR1 and CYTB measured in the small RNA fraction but not in total RNA (Figure 4D–4F; and Supplementary Figure 1). Similar results were seen for a second primer pair designed against different location in the MT-RNR1 gene (Supplementary Figure 1), and as a result of enriching for transcripts <200 nt, the expression of these mRNA’s was substantially lower in the small fraction compared to fractions containing larger transcripts (Supplementary Figure 2). This indicates that small RNA transcript levels are independent of the larger transcript levels, suggesting that they may be independently transcribed. Neither MT-RNR1 mRNA measured in total or small RNA fraction correlated with muscle protein MOTS-c protein levels (Figure 4G, 4H).
To determine whether the increase in MOTS-c mRNA is a function of differences in mtDNA content, mitochondrial-to-nuclear ratio (mtDNA/nDNA) was measured and used to standardize MOTS-c mRNA levels (Figure 4I, 4J). Similar to MOTS-c protein expression, when corrected for mtDNA content, both the middle and older groups had greater MOTS-c mRNA relative to mtDNA than the young group (Figure 4J), and this standardization did not improve the association between MOTS-c mRNA and MT-RNR1 mRNA total RNA fraction (Figure 4K).
MOTS-c has been shown to bind to nuclear DNA in HEK293 cells and interact with Nrf2 (NFE2L2) to regulate ARE-related gene expression [22]. A weak correlation was observed between skeletal muscle NRF2 mRNA and MOTS-c small mRNA levels, and HMOX-1 mRNA and MOTS-c protein expression (Figure 5B and 5E). However, overall there was no association between ARE-responsive genes (HMOX-1, NQO1 and SOD2) and MOTS-c small mRNA, or NQO1 and SOD2 mRNA and MOTS-c protein levels in skeletal muscle (Figure 5A–5H).
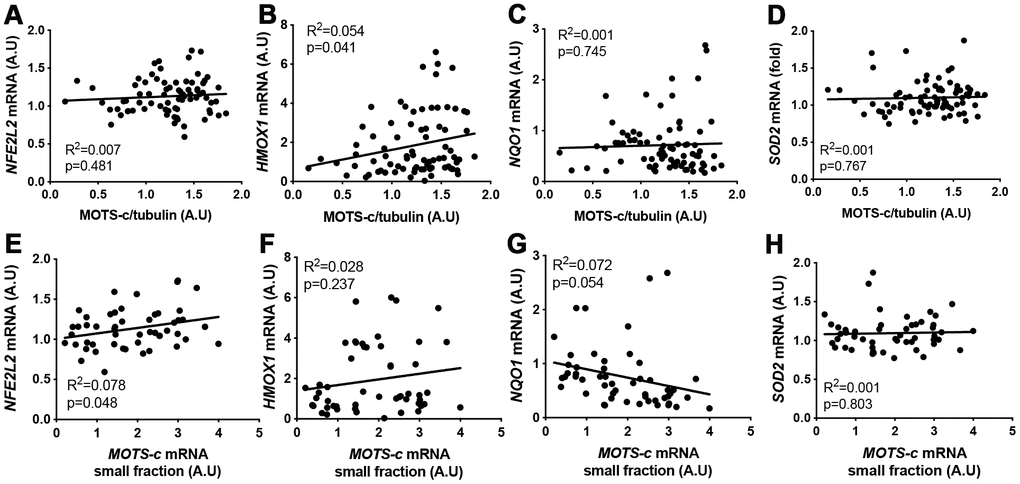
Figure 5. MOTS-c muscle protein or small mRNA expression does not correlate well with antioxidant-response element (ARE) related genes. Correlation between MOTS-c protein (A–D) or small mRNA (E–H) expression and mRNA levels of NFE2L2, HMOX1, NQO1 and SOD2 in muscle samples from young, middle-aged and older men. Significance was determined using linear regression for n=78 (protein correlations) or n=52 (MOTS-c mRNA correlations).
Discussion
The mitochondrial encoded peptide, MOTS-c, has been implicated in regulating the aging process from observations in murine studies [29, 30]. Here, we provide evidence that plasma MOTS-c levels are lower, but skeletal muscle levels are higher in aged human males independent of body composition, HOMA-IR and plasma triglycerides. As such, and because our participants were free from overt aging-related co-morbidities, these changes in MOTS-c expression are likely to be largely independent of aged-associated disease, fat mass, and insulin sensitivity, suggesting that changes in MOTS-c levels are the results of normal aging rather than an artifact of disease development.
While MOTS-c serum levels have recently been reported to negatively correlate with age in a cohort of participants with type 2 diabetes [32], interpretation of this data is confounded by the healthy control group being significantly younger than the diabetic groups. Regardless, our results support this finding and previous observations of lower MOTS-c plasma levels in aged mice, but are in contrast to reported murine skeletal muscle age-associated decline in MOTS-c levels [14]. This may reflect inherent differences between human and mouse skeletal muscle aging [33, 34], but also could be the result of the relative age of the mice (32 months which equates to >80 human years [35]) being older than that of human participants in this study. The middle-aged and older participants were healthy, independent and mobile, and therefore it is possible that the observed increase in muscle MOTS-c in these groups is a compensatory response to age-related muscle metabolic stress [36] aimed at coordinating pathways to maintaining muscle quality and function. This would be consistent with the recent report that metabolic and oxidative stressors increase MOTS-c expression to regulate an adaptive transcriptional response [22], and supports our observation of a positive association between muscle quality and MOTS-c muscle expression. However, when the ability of the muscle cell to tolerate or adapt to this stress is overwhelmed then MOTS-c levels may decrease along with function of the cell. In support, some studies have reported reduced plasma levels of MOTS-c in people with obesity, type 2 diabetes [32, 37] or impaired coronary endothelial function [38], and exogenous MOTS-c treatment restores skeletal muscle insulin sensitivity in mice [14, 27].
Plasma and muscle MOTS-c levels show opposing responses to aging, perhaps suggesting that at rest in a fasted state the primary source of plasma MOTS-c is not skeletal muscle or the pharmacokinetics of MOTS-c changes with age. However, interpretation of this result is limited by plasma and muscle samples from the older men being from different participants. Nevertheless, mice express MOTS-c in a wide range of tissues [25], being particularly highly expressed in the liver, an organ that is sensitive to age-related decline in function. A number of hepatokines are now suggested as being age- and metabolic-sensitive [39–41], and MOTS-c may fall into this category. Interestingly, however, we observed a strong correlation between plasma and skeletal muscle MOTS-c in young but not middle-aged groups. An intriguing alternative hypothesis is that the ability for MOTS-c to leave the muscle cell is lost with age, and this could be the result of either the loss of an export process and/or a greater need for mitochondrial retrograde signaling in aging muscle. It is also tempting to further speculate that this inability to release MOTS-c is tied to the greater expression of MOTS-c in slow-type fibers and age-related fast-to-slow fiber type transition. Indeed, fiber-type specific regulation of myokines is common with brain-derived neurotrophic factor (BDNF) promoting glycolytic fiber type transition [42] and myonectin being more highly expressed in oxidative muscle fibers [43]. Further work is required to determine whether MOTS-c has fiber-type specific roles and to identify the molecular targets of MOTS-c in human skeletal muscle. Our analysis showed little association between previously reported MOTS-c transcriptional targets and either MOTS-c protein or mRNA expression suggesting targets may differ in humans in vivo, or with aging. However, recently published plasma metabolomic data from MOTS-c treated obese mice suggest the lipid metabolism pathway as a key target [24].
Mitochondrial derived peptides are transcribed from sORF within mitochondrial genes and hypothesized to be translated in the cytoplasm [14]. Whether MOTS-c is translated as part of 12S rRNA transcript or independently is not known. Using a small RNA probe against the MOTS-c sequence and filtering total RNA to enrich short RNA fragments (<200 nt; lowering full length 12S rRNA mRNA) we provide evidence that MOTS-c transcripts may be differentially regulated from that of the full parent gene sequence, but similar to that of other small transcripts from the mitochondrial genome. This could be the result of sequence specific transcription and export, an artifact of lower MOTS-c mRNA stability compared to full length 12S mRNA, or reflect the formation of smaller functional mRNA transcripts during the multiple steps of mitochondrial RNA processing and export [44]. Whether the latter of these options is responsive to mitochondrial perturbations and could be a mechanism underpinning MOTS-c sensitivity to metabolic stress is yet to be determined, along with the full sequence of the MOTS-c transcript.
Taken together, our data indicate that the mitochondrial derived peptide, MOTS-c, is differentially regulated in the skeletal muscle and plasma of healthy (free of serious comorbidities) males during normal aging. We suggest that an aging-associated fast-to-slow type muscle transition may underpin the increase in muscle MOTS-c seen during aging and that this may be related to the loss of positive association between plasma and muscle in older age.
Materials and Methods
Participants
One hundred and four healthy young, middle-aged or older males were recruited (Table 1 and [45]) and gave written informed consent before the commencement of the study. Participants were sedentary to recreationally active, non-smokers, free of chronic illnesses including cardiovascular or metabolic disease and were not taking any medication. Body composition was assessed by dual X-ray absorptiometry (DXA; model iDXA, GE-Lunar, Madison, WI), and thigh muscle cross-sectional area was determined via anthropometric measurement as previously reported [46]. One repetition maximum (1-RM) for leg press was estimated using the Brzycki equation [47]. Two groups of older participants were recruited as blood samples were not available for the group that had muscle samples collected (Older 1). All experimental procedures were approved by the Health and Disability (New Zealand) and Deakin University Human (Melbourne, Australia) Research Ethics Committee’s, and performed in accordance with the Helsinki declaration.
Murine housing
Mice were maintained in a temperature-controlled animal facility with 12 h light-dark cycle and ad libitum access to water, a standard rodent chow diet (Teklad TB 2018; Harlan, Madison, WI). At 10-12 weeks of age in-house bred male C57BL/6J mice were culled by cervical dislocation and muscle excised, snap frozen in liquid nitrogen and stored at -80°C until analysis. All experiments were approved by the University of Auckland Animal Ethics Committee, New Zealand.
Muscle and blood collection
Following an overnight fast, and having abstained from strenuous physical activity for at least 48h prior to arriving at the laboratory in the morning (before 0900 h), venous blood was drawn into a 10 ml EDTA vacutainer tube, and a percutaneous muscle biopsy was obtained from the vastus lateralis muscle using suction-modified Bergstrom biopsy needle under local anaesthesia. Muscle samples were snap-frozen in liquid nitrogen and blood samples were centrifuged immediately upon collection at 4° C, 1900 x g for 15 min. Plasma and muscle samples were subsequently stored at -80°C until analysis.
Plasma analysis
Glucose (C311 autoanalyzer, Roche, Mannheim, Germany) and insulin (cobas e11, Roche, Mannheim, Germany) were measured in plasma and the homeostatic model assessment (HOMA) was used to estimate insulin resistance (HOMA2-IR2; The Oxford Centre for Diabetes, Endocrinology and Metabolism, UK) [48]. As described previously [14], MOTS-c was extracted from plasma in acetonitrile-hydrochloric acid and measured using an in-house sandwich ELISA developed at the University of Southern California by the laboratory that first described MOTS-c in human and murine plasma. This ELISA has been extensively validated, and peptide blocking experiments have been undertaken to show specificity of the antibody [14].
Immunoblotting
~20 mg of muscle was homogenized for 40s at 20 Hz using a TissueLyser (Qiagen, Venlo, Netherlands) in lysis buffer containing 10 μL/mg 25 mM Tris 0.5% v/v Triton X-100 supplemented with Halt Protease and Phosphatase Inhibitor Cocktail (Thermo Scientific, Waltham, MA). The supernatant was collected after centrifugation to determine protein concentration using the BCA protein assay kit (Thermo Scientific, Waltham, MA). Laemmli's buffer was added to protein homogenates and boiled for 5 min at 95 °C. Denatured proteins were separated by SDS-PAGE on 8%–16% TGX pre-cast gels (Bio-Rad, Hercules, USA) and proteins were transferred to PVDF membranes (Bio-Rad, Hong Kong, China) using a TransBlot semidry transfer apparatus (Bio-Rad, Hongkong, China). Membranes were blocked in 5% BSA in TBST for 1h at room temperature, and probed using specific antibodies for MOTS-c (custom produced by YenZym Antibodies, LLC, South San Francisco, CA and described previously [14], 1:500) COXIV (Molecular probes #A6431, 1:10,000) and α-tubulin (Sigma-Aldrich #T9026, 1:10,000). Primary antibodies were incubated overnight at 4 °C with gentle agitation. The following morning, the membranes were probed with either anti-rabbit or anti-mouse linked to horseradish peroxidase secondary antibodies (Jackson ImmunoResearch, West Grove, PA, USA) (1:10,000 dilution) for 1h at room temperature. Membranes were exposed using enhanced chemiluminescence reagent (Western Lightning® Ultra, Perkin Elmer, Waltham, MA, USA) and chemiluminescent signals were captured using ChemiDoc image device (BioRad, HongKong, China). Densitometry analysis of protein bands were quantified using ImageJ software (National Institutes of Health, Bethesda, MD). The intensity of each band was recorded relative to their respective α-tubulin intensity and then normalized to the ratio of target to loading protein in the pooled control sample ran on each gel, and log transformed if required.
Real-time polymerase chain reaction
Total RNA and DNA was extracted from ~20 mg of tissue using the AllPrep® DNA/RNA/miRNA Universal Kit (QIAGEN GmbH, Hilden, Germany) and small RNA’s (<200 nt) enriched using the Zymo RNA Clean and Concentrator Kit (Cat # R1016, Zymo Research, Irvine, CA) per the manufacturer’s instructions, and enrichment confirmed using RNA NANO Chip using a 2100 Bioanalyzer (Agilent Technologies; data not shown). Total RNA was reverse transcribed using a High-Capacity RNA-to-cDNA™ kit (Life Technologies, Carlsbad, CA) and quantitative real-time PCR was performed on a QuantStudio 6 PCR System using the SYBR green select master mix (Applied Biosystems, Foster City, CA). Reactions were performed in duplicate and relative quantification achieved using the ∆∆Ct method with geometric mean of four reference genes (human sample: C1ORF43, CHMP2A, VCPEMC7; mouse sample, Gapdh, Actb B2mb Rplpo) as an internal control. Primer sequences used are listed in Supplementary Table 3, and for mitochondrial (mtDNA) to nuclear (nDNA) ratio primers were used to MTND4 and 18S rRNA. For small RNA assays, 50 ng of fractioned RNA was converted to cDNA using TaqMan® microRNA RT kit #4366596 (Life Technologies, Carlsbad, CA) and a multiplexed approach [49] was utilized with specific TaqMan small RNA assays (Life Technologies, Carlsbad, CA) for MOTS-c (designed using Custom TaqMan® Small RNA Assay Design Tool against MOTS-c mtDNA sequence), RNU44 (assay ID: 001094) and RNU48 (assay ID: 001006) primers as per the manufactures instruction and as described previously [50]. The expression of MOTS-c mRNA was normalized to the geomean RNU44 and RNU48 using the ∆∆Ct method.
Statistical analyses
Statistical analyses were performed using Prism 8 (GraphPad Software Incorporated, California, USA) and SPSS (IBM SPSS, NY, USA, version 25), with statistical significance determined as p ≤ 0.05, and data are presented as individual data points and mean ± standard error of the mean unless stated otherwise. Statistical significance was determined with Pearson linear regression, one-way ANOVA with Holm-Sidak’s post-hoc analysis or ANCOVA.
Author Contributions
TLM, DC-S, PC, CL HK and CJM conceived the experiments. RFD, JSTW, CPH, NZ, JW and CJM performed the experiments. TLM and RFD analyzed the data. TLM wrote the manuscript and all authors approved the final manuscript version.
Conflicts of Interest
PC and CL are consultants and shareholders of CohBar, Inc. All other authors declare no conflicts of interest.
Funding
This study was funded by a Marsden Fast-start grant, the University of Auckland Faculty Research Development Fund (all to TLM), AFAR BIG AWARD P01AG034906 to PC, and TLM is supported by a Rutherford Discovery Fellowship. CL was supported by the NIA (R01AG052258), Ellison Medical Foundation (EMF), AFAR, and the Hanson-Thorell Family. We would like to thank Caitlin MacRae for her technical assistance and all the participants for donation of their time and samples.
References
- 1. Flatt T. A new definition of aging? Front Genet. 2012; 3:148. https://doi.org/10.3389/fgene.2012.00148 [PubMed]
- 2. Martin GM, Austad SN, Johnson TE. Genetic analysis of ageing: role of oxidative damage and environmental stresses. Nat Genet. 1996; 13:25–34. https://doi.org/10.1038/ng0596-25 [PubMed]
- 3. Jang JY, Blum A, Liu J, Finkel T. The role of mitochondria in aging. J Clin Invest. 2018; 128:3662–70. https://doi.org/10.1172/JCI120842 [PubMed]
- 4. Petersen KF, Befroy D, Dufour S, Dziura J, Ariyan C, Rothman DL, DiPietro L, Cline GW, Shulman GI. Mitochondrial dysfunction in the elderly: possible role in insulin resistance. Science. 2003; 300:1140–42. https://doi.org/10.1126/science.1082889 [PubMed]
- 5. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013; 153:1194–217. https://doi.org/10.1016/j.cell.2013.05.039 [PubMed]
- 6. Shigenaga MK, Hagen TM, Ames BN. Oxidative damage and mitochondrial decay in aging. Proc Natl Acad Sci USA. 1994; 91:10771–78. https://doi.org/10.1073/pnas.91.23.10771 [PubMed]
- 7. Merry TL, Ristow M. Mitohormesis in exercise training. Free Radic Biol Med. 2016; 98:123–30. https://doi.org/10.1016/j.freeradbiomed.2015.11.032 [PubMed]
- 8. Nakahira K, Haspel JA, Rathinam VA, Lee SJ, Dolinay T, Lam HC, Englert JA, Rabinovitch M, Cernadas M, Kim HP, Fitzgerald KA, Ryter SW, Choi AM. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat Immunol. 2011; 12:222–30. https://doi.org/10.1038/ni.1980 [PubMed]
- 9. Korolchuk VI, Miwa S, Carroll B, von Zglinicki T. Mitochondria in Cell Senescence: Is Mitophagy the Weakest Link? EBioMedicine. 2017; 21:7–13. https://doi.org/10.1016/j.ebiom.2017.03.020 [PubMed]
- 10. Chandel NS. Evolution of Mitochondria as Signaling Organelles. Cell Metab. 2015; 22:204–06. https://doi.org/10.1016/j.cmet.2015.05.013 [PubMed]
- 11. Houtkooper RH, Argmann C, Houten SM, Cantó C, Jeninga EH, Andreux PA, Thomas C, Doenlen R, Schoonjans K, Auwerx J. The metabolic footprint of aging in mice. Sci Rep. 2011; 1:134. https://doi.org/10.1038/srep00134 [PubMed]
- 12. Durieux J, Wolff S, Dillin A. The cell-non-autonomous nature of electron transport chain-mediated longevity. Cell. 2011; 144:79–91. https://doi.org/10.1016/j.cell.2010.12.016 [PubMed]
- 13. Gustafsson CM, Falkenberg M, Larsson NG. Maintenance and Expression of Mammalian Mitochondrial DNA. Annu Rev Biochem. 2016; 85:133–60. https://doi.org/10.1146/annurev-biochem-060815-014402 [PubMed]
- 14. Lee C, Zeng J, Drew BG, Sallam T, Martin-Montalvo A, Wan J, Kim SJ, Mehta H, Hevener AL, de Cabo R, Cohen P. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metab. 2015; 21:443–54. https://doi.org/10.1016/j.cmet.2015.02.009 [PubMed]
- 15. Guo B, Zhai D, Cabezas E, Welsh K, Nouraini S, Satterthwait AC, Reed JC. Humanin peptide suppresses apoptosis by interfering with Bax activation. Nature. 2003; 423:456–61. https://doi.org/10.1038/nature01627 [PubMed]
- 16. Cobb LJ, Lee C, Xiao J, Yen K, Wong RG, Nakamura HK, Mehta HH, Gao Q, Ashur C, Huffman DM, Wan J, Muzumdar R, Barzilai N, Cohen P. Naturally occurring mitochondrial-derived peptides are age-dependent regulators of apoptosis, insulin sensitivity, and inflammatory markers. Aging (Albany NY). 2016; 8:796–809. https://doi.org/10.18632/aging.100943 [PubMed]
- 17. Xiao J, Howard L, Wan J, Wiggins E, Vidal A, Cohen P, Freedland SJ. Low circulating levels of the mitochondrial-peptide hormone SHLP2: novel biomarker for prostate cancer risk. Oncotarget. 2017; 8:94900–09. https://doi.org/10.18632/oncotarget.20134 [PubMed]
- 18. Lee C, Yen K, Cohen P. Humanin: a harbinger of mitochondrial-derived peptides? Trends Endocrinol Metab. 2013; 24:222–28. https://doi.org/10.1016/j.tem.2013.01.005 [PubMed]
- 19. Kim SJ, Mehta HH, Wan J, Kuehnemann C, Chen J, Hu JF, Hoffman AR, Cohen P. Mitochondrial peptides modulate mitochondrial function during cellular senescence. Aging (Albany NY). 2018; 10:1239–56. https://doi.org/10.18632/aging.101463 [PubMed]
- 20. Kim SJ, Guerrero N, Wassef G, Xiao J, Mehta HH, Cohen P, Yen K. The mitochondrial-derived peptide humanin activates the ERK1/2, AKT, and STAT3 signaling pathways and has age-dependent signaling differences in the hippocampus. Oncotarget. 2016; 7:46899–912. https://doi.org/10.18632/oncotarget.10380 [PubMed]
- 21. Duan J, Chen Z, Wu Y, Zhu B, Yang L, Yang C. Metabolic remodeling induced by mitokines in heart failure. Aging (Albany NY). 2019; 11:7307–27. https://doi.org/10.18632/aging.102247 [PubMed]
- 22. Kim KH, Son JM, Benayoun BA, Lee C. The Mitochondrial-Encoded Peptide MOTS-c Translocates to the Nucleus to Regulate Nuclear Gene Expression in Response to Metabolic Stress. Cell Metab. 2018; 28:516–524.e7. https://doi.org/10.1016/j.cmet.2018.06.008 [PubMed]
- 23. Ramanjaneya M, Jerobin J, Bettahi I, Bensila M, Aye M, Siveen KS, Sathyapalan T, Skarulis M, Abou-Samra AB, Atkin SL. Lipids and insulin regulate mitochondrial-derived peptide (MOTS-c) in PCOS and healthy subjects. Clin Endocrinol (Oxf). 2019; 91:278–87. https://doi.org/10.1111/cen.14007 [PubMed]
- 24. Kim SJ, Miller B, Mehta HH, Xiao J, Wan J, Arpawong TE, Yen K, Cohen P. The mitochondrial-derived peptide MOTS-c is a regulator of plasma metabolites and enhances insulin sensitivity. Physiol Rep. 2019; 7:e14171. https://doi.org/10.14814/phy2.14171 [PubMed]
- 25. Li Q, Lu H, Hu G, Ye Z, Zhai D, Yan Z, Wang L, Xiang A, Lu Z. Earlier changes in mice after D-galactose treatment were improved by mitochondria derived small peptide MOTS-c. Biochem Biophys Res Commun. 2019; 513:439–45. https://doi.org/10.1016/j.bbrc.2019.03.194 [PubMed]
- 26. Lu H, Tang S, Xue C, Liu Y, Wang J, Zhang W, Luo W, Chen J. Mitochondrial-Derived Peptide MOTS-c Increases Adipose Thermogenic Activation to Promote Cold Adaptation. Int J Mol Sci. 2019; 20:E2456. https://doi.org/10.3390/ijms20102456 [PubMed]
- 27. Lu H, Wei M, Zhai Y, Li Q, Ye Z, Wang L, Luo W, Chen J, Lu Z. MOTS-c peptide regulates adipose homeostasis to prevent ovariectomy-induced metabolic dysfunction. J Mol Med (Berl). 2019; 97:473–85. https://doi.org/10.1007/s00109-018-01738-w [PubMed]
- 28. Kim SJ, Miller B, Kumagai H, Yen K, Cohen P. MOTS-c: an equal opportunity insulin sensitizer. J Mol Med (Berl). 2019; 97:487–90. https://doi.org/10.1007/s00109-019-01758-0 [PubMed]
- 29. Alis R, Lucia A, Blesa JR, Sanchis-Gomar F. The role of mitochondrial derived peptides (MDPs) in metabolism. J Cell Physiol. 2015; 230:2903–04. https://doi.org/10.1002/jcp.25023 [PubMed]
- 30. Fuku N, Pareja-Galeano H, Zempo H, Alis R, Arai Y, Lucia A, Hirose N. The mitochondrial-derived peptide MOTS-c: a player in exceptional longevity? Aging Cell. 2015; 14:921–23. https://doi.org/10.1111/acel.12389 [PubMed]
- 31. Miljkovic N, Lim JY, Miljkovic I, Frontera WR. Aging of skeletal muscle fibers. Ann Rehabil Med. 2015; 39:155–62. https://doi.org/10.5535/arm.2015.39.2.155 [PubMed]
- 32. Ramanjaneya M, Bettahi I, Jerobin J, Chandra P, Abi Khalil C, Skarulis M, Atkin SL, Abou-Samra AB. Mitochondrial-Derived Peptides Are Down Regulated in Diabetes Subjects. Front Endocrinol (Lausanne). 2019; 10:331. https://doi.org/10.3389/fendo.2019.00331 [PubMed]
- 33. Zhuang J, Zhang L, Dai S, Cui L, Guo C, Sloofman L, Yang J. Comparison of multi-tissue aging between human and mouse. Sci Rep. 2019; 9:6220. https://doi.org/10.1038/s41598-019-42485-3 [PubMed]
- 34. Boldrin L, Muntoni F, Morgan JE. Are human and mouse satellite cells really the same? J Histochem Cytochem. 2010; 58:941–55. https://doi.org/10.1369/jhc.2010.956201 [PubMed]
- 35. Flurkey K, Currer JM, Harrison DE. Mouse Models in Aging Research. In: Fox JG, Davisson MT, Quimby FW, Barthold SW, Newcomer CE, Smith AL, eds. The Mouse in Biomedical Research, 2nd Edition: Elsevier). 2007. https://doi.org/10.1016/B978-012369454-6/50074-1
- 36. Barzilai N, Huffman DM, Muzumdar RH, Bartke A. The critical role of metabolic pathways in aging. Diabetes. 2012; 61:1315–22. https://doi.org/10.2337/db11-1300 [PubMed]
- 37. Du C, Zhang C, Wu W, Liang Y, Wang A, Wu S, Zhao Y, Hou L, Ning Q, Luo X. Circulating MOTS-c levels are decreased in obese male children and adolescents and associated with insulin resistance. Pediatr Diabetes. 2018; 19:1058–64. https://doi.org/10.1111/pedi.12685 [PubMed]
- 38. Qin Q, Delrio S, Wan J, Jay Widmer R, Cohen P, Lerman LO, Lerman A. Downregulation of circulating MOTS-c levels in patients with coronary endothelial dysfunction. Int J Cardiol. 2018; 254:23–27. https://doi.org/10.1016/j.ijcard.2017.12.001 [PubMed]
- 39. Salminen A, Kaarniranta K, Kauppinen A. Regulation of longevity by FGF21: interaction between energy metabolism and stress responses. Ageing Res Rev. 2017; 37:79–93. https://doi.org/10.1016/j.arr.2017.05.004 [PubMed]
- 40. Meex RC, Hoy AJ, Morris A, Brown RD, Lo JC, Burke M, Goode RJ, Kingwell BA, Kraakman MJ, Febbraio MA, Greve JW, Rensen SS, Molloy MP, et al. Fetuin B Is a Secreted Hepatocyte Factor Linking Steatosis to Impaired Glucose Metabolism. Cell Metab. 2015; 22:1078–89. https://doi.org/10.1016/j.cmet.2015.09.023 [PubMed]
- 41. Stefan N, Häring HU. The role of hepatokines in metabolism. Nat Rev Endocrinol. 2013; 9:144–52. https://doi.org/10.1038/nrendo.2012.258 [PubMed]
- 42. Delezie J, Weihrauch M, Maier G, Tejero R, Ham DJ, Gill JF, Karrer-Cardel B, Rüegg MA, Tabares L, Handschin C. BDNF is a mediator of glycolytic fiber-type specification in mouse skeletal muscle. Proc Natl Acad Sci USA. 2019; 116:16111–20. https://doi.org/10.1073/pnas.1900544116 [PubMed]
- 43. Seldin MM, Peterson JM, Byerly MS, Wei Z, Wong GW. Myonectin (CTRP15), a novel myokine that links skeletal muscle to systemic lipid homeostasis. J Biol Chem. 2012; 287:11968–80. https://doi.org/10.1074/jbc.M111.336834 [PubMed]
- 44. D’Souza AR, Minczuk M. Mitochondrial transcription and translation: overview. Essays Biochem. 2018; 62:309–20. https://doi.org/10.1042/EBC20170102 [PubMed]
- 45. Zeng N, D’Souza RF, Mitchell CJ, Cameron-Smith D. Sestrins are differentially expressed with age in the skeletal muscle of men: A cross-sectional analysis. Exp Gerontol. 2018; 110:23–34. https://doi.org/10.1016/j.exger.2018.05.006 [PubMed]
- 46. D’Souza RF, Zeng N, Markworth JF, Figueiredo VC, Hedges CP, Petersen AC, Della Gatta PA, Cameron-Smith D, Mitchell CJ. Whey Protein Supplementation Post Resistance Exercise in Elderly Men Induces Changes in Muscle miRNA’s Compared to Resistance Exercise Alone. Front Nutr. 2019; 6:91. https://doi.org/10.3389/fnut.2019.00091 [PubMed]
- 47. Brzycki M. A Practical Approach to Strength Training. Indianapolis (IN): Blue River; 2012.
- 48. Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care. 2004; 27:1487–95. https://doi.org/10.2337/diacare.27.6.1487 [PubMed]
- 49. Le Carré J, Lamon S, Léger B. Validation of a multiplex reverse transcription and pre-amplification method using TaqMan(®) MicroRNA assays. Front Genet. 2014; 5:413. https://doi.org/10.3389/fgene.2014.00413 [PubMed]
- 50. Mitchell CJ, D’Souza RF, Schierding W, Zeng N, Ramzan F, O’Sullivan JM, Poppitt SD, Cameron-Smith D. Identification of human skeletal muscle miRNA related to strength by high-throughput sequencing. Physiol Genomics. 2018; 50:416–24. https://doi.org/10.1152/physiolgenomics.00112.2017 [PubMed]