Introduction
For a long time, an active, targeted intervention to maintain health into old age was terra incognita. It had no priority, and few, if any, reliable data were available to implement it in everyday life. Today, however, systematically established diagnostic hints become available for the individual, based on family history and biomarker data, including genetic variants (polymorphisms). To assess and prevent premature health deterioration successfully, it would therefore be useful (1) to dissect “health” into a set of its most important features, (2) to specify biomarkers and corresponding supportive interventions for the various features of health and for health itself, and (3) to detail its molecular basis and to map out molecular “healthspan pathways”. Arguably, the increase in life expectancy in the last 100 years has not been accompanied by an increase in disease-free life expectancy [1, 2]. Cardiovascular disease, type-2 diabetes and neurodegenerative disorders are highly prevalent in the elderly, and these diseases frequently coexist in the same aged individual, often with mutual reinforcement [3, 4]. Extending healthspan may thus enable economic, societal and individual gains on a large scale [5, 6].
Intervention studies to prolong healthspan based on compound exposure in humans are limited to relatively few compounds. Resveratrol for instance, being one of the best-studied polyphenols in humans and animals, has been tested in several clinical studies [7]. These include studies focused on biomarkers, like the level of blood glucose [8] and cholesterol [9], or glutathione S-transferase expression [10]. Moreover, data about long-term effects on overall health in human are missing in general, and given the average human life expectancy, they are difficult to obtain. Therefore, model organisms are of great relevance to uncover the molecular basis of healthspan and to identify supporting compounds. The nematode Caenorhabditis elegans (C. elegans) is a widely used model organism for studying ageing which guided the discovery of fundamental ageing-related findings, e.g., on calorie restriction and Insulin/IGF-1 like signaling [11]. Last not least, around 40% of the genes found in C. elegans have human orthologs and, vice versa, about 50% of the human protein-coding genome has recognizable worm orthologs [12]. Studies revealing the role of metabolism on health conducted in C. elegans have been subsequently strengthened in murine models [13, 14], rendering this nematode a valuable model for human ageing processes. Furthermore, the effects on lifespan, when manipulating orthologous lifespan-associated genes in different model organisms, are mostly concordant, despite high evolutionary distances between them [15]. Most recently, C. elegans has come to enjoy increasing popularity as a model for health [16, 17], and an ever increasing number of compounds [18–21] and diets [22, 23] are tested in C. elegans for their anti-ageing and health effects.
Here, we assemble and explore “healthspan pathway maps”, that is, annotated sets of interacting genes implicated in health. To create these, we follow a stepwise procedure: first, we dissect health into its various features, based on disease and dysfunction. Second, we compile lists of genes associated with health based on the literature, for humans and C. elegans. Third, we organize these genes into maps of healthspan pathways, based on gene/protein interaction and annotation data. Fourth, we create an overlay of health-related gene expression data onto the resulting healthspan pathway maps, highlighting corroborating knowledge that was not used as input. Finally, we investigate the overlap of the healthspan pathways in humans and C. elegans.
Health is a term in biology and medicine that is hard to define. We propose that the best definition of health must be based on an aggregation of the literature, see also Fuellen et al. [5] and Luyten et al. [17]. Then, healthspan is simply the time spent in good health. Supplementary Tables 1–3 list features of human health as discussed in the literature, referring to lack of dysfunction, lack of multiple diseases, and lifespan/longevity mediated by lack of disease. In principle, at least for human, dysfunction can be operationalized with the help of a codified classification of function (such as the ICF, the International Classification of Functioning, Disability and Health, https://www.who.int/classifications/icf/en/). This classification provides criteria to establish that an individual is affected by a dysfunction. As described and discussed in Fuellen et al. [5], we can filter the “body function” part of the ICF by looking for follow-up in the literature on health and healthspan. The result is a pragmatic community consensus definition of dysfunction, centering around the lack of physiological, physical, cognitive and reproductive function; a lack of physiological, physical and cognitive functions is often called frailty. To a large degree, this consensus definition can be used for non-human species as well. Further, disease can also be operationalized by a codified classification (such as ICD-11, International Statistical Classification of Diseases and Related Health Problems, https://www.who.int/classifications/icd/en/). Again, the classification provides criteria to establish that an individual is affected by a disease. In this paper, affection by a single disease is not considered, as in old age, single-disease morbidity rarely exists, and in terms of interventions, we are interested in preventing more than one disease. As described and discussed in Fuellen et al. [5], not all parts of the ICD feature diseases related to health and healthspan. However, we note that all diseases referred to in Supplementary Tables 1–3 qualify as age-associated diseases.
The main sources of knowledge about health, that is, about features, biomarkers and interventions regarding health-related phenotypes, are
observational genetic investigations, usually in the form of genome-wide association studies, looking for associations between health and polymorphisms of specific genes [24],
observational studies of non-genetic biomarkers, which are dynamic in time and are usually related to known canonical pathways, and their longitudinal or cross-sectional correlation with health [25],
interventional studies, most often in model organisms, where interventions affecting health may be genetic or based on food or (pharmaceutical) compounds, and the intervention effects are measured on the molecular level, implicating particular genes or pathways [26].
Like genetic studies, compound intervention studies can, in principle, elucidate the causative basis of health. Studies of type (b) may only be revealing correlative evidence and can sometimes not be linked to particular genes; therefore, we will not consider these further. A biomarker of health is any (composite) feature that allows to predict future health better than chronological age [5]; it may be genetic (polymorphisms; such a biomarker is essentially static over lifetime), molecular but not genetic (epigenetic or transcript or protein or metabolic markers, etc.), cellular (blood counts, etc.) or organismic (such as grip strength). Based on studies of types (a) and (c), in this work we will only deal with genes and sets of genes (that is, genes organized into networks or pathways) as candidate biomarkers of health.
For humans, we thus consider that knowledge of the causal basis of health may be best derived from genetic association studies [27]. Based on an extensive review of the literature, we identify a core set of 12 genes that are genetically associated with a lack of frailty [28, 29] and the Healthy Aging Index [30], and another set of 40 genes genetically associated with (a lack of) multiple diseases, or with longevity mediated by a lack of disease (see Supplementary Tables 1–3). In contrast to humans, genetic intervention studies on healthspan are available for C. elegans, as well as compound intervention data. A lack of dysfunction exemplified by stress resistance, locomotion, pharyngeal pumping and reproduction are taken as the key health features in C. elegans [31]. On this basis, a core set of 11 genes is directly implicated in improvements of locomotion by genetics, and another set of 20 genes is indirectly implicated in improvements of the key health features by studies that investigate effects of compounds (see Supplementary Tables 4, 5). While there is a strong overlap between genes affecting healthspan and genes affecting lifespan, the genes we selected may or may not be associated with survival/lifespan. In turn, we do not consider survival/lifespan genes that are not (known to be) directly associated with health. In other words, what we selected is a list of genes related to health(span), without explicitly considering lifespan.
We then place the genes implicated in health into context by adding gene/protein interaction and gene annotation knowledge. Specifically, we turn the lists of genes into gene/protein interaction networks, to which 20 closely interacting genes are added, employing GeneMania [32]. Gene ontology annotation data are then used to annotate clusters of strongly connected genes within the network, employing AutoAnnotate [33]. Then, we elaborate how the resulting healthspan pathways can be interpreted in plausible ways, specifically in the light of independent health-related gene expression data describing effects of caloric restriction and of rapamycin, and in the light of gene expression data describing aging and disease. Given the incomplete and sometimes inaccurate knowledge we use as input, our healthspan pathway map, and its interpretation can only be a first sketch to drive the development of models of this polygenetic phenotype. For example, not much weight should be given to the small pathways (clusters of 2-3 genes) in the pathway maps, as the clustering is entirely based on high-throughput data such as protein interaction data.
We also predict microRNAs that may be potential regulators of healthspan [34, 35]. Finally, we find that if we construct an overlap between the healthspan pathways in C. elegans and humans, genes involved in transcription, proliferation/biosynthesis and lipids are highlighted, but this overlap is not straightforward to interpret in the light of the independent health-related gene expression data that we used to test plausibility of the single-species healthspan pathway maps. Further, “lipids” come up by way of the Gene Ontology annotation data for both species.
All healthspan pathways discussed in this manuscript, as well as the overlaps we found between species, are available for interactive exploration at http://pathways.h2020awe.eu.
Results and Discussion
Based on the considerations in the introduction, we first justify the gene lists we used to construct the healthspan pathway maps for humans and C. elegans. Second, we describe the healthspan pathway maps in detail, specifically in light of gene expression data that we overlaid onto the pathway maps. We then consider the human - C. elegans overlap, followed by some general discussion of our approach, including its strengths and limitations.
Genes associated with health
Health genes in humans may be discovered based on genetic association, and with some probability it can be assumed that these correlations indicate a causal relevance. This is not a certain inference, because of the intrinsic ambiguities in assigning genetic polymorphisms (in the form of SNPs, single-nucleotide polymorphisms) to genes, e.g. in intergenic regions or in intronic regions with overlapping non-coding RNA on the complementary strand [36]. In turn, for C. elegans, only few studies report effects of genetic interventions on health, although these are increasingly becoming available. However, as of early 2018, Sutphin et al. [16] is the only large-scale genetic study that we could identify, even though it is basically a small-scale study of healthspan based on a large-scale study of lifespan. Many more studies in C. elegans refer to canonical aging-related pathways, and in contrast to studies in humans, these studies often directly report the molecular effects of compound intervention. The C. elegans genes listed in the Supplementary Tables 4, 5 are thus based on the effects of genetic intervention and on the effects (on the gene level) of compound intervention, and we can assume a high probability of causality in both cases. For C. elegans, Supplementary Tables 4, 5 list features of health based on the literature, referring to lack of dysfunction in the form of stress resistance (in response to thermal and oxidative stress), (stimulated) locomotion, pharyngeal pumping, and reproduction. These features dominate the literature, and they cover the aspects of physiological function, physical and cognitive function, and reproductive function, as in human [5]. Of note, genetic analyses of health in C. elegans have focused up to now mostly on (stimulated) locomotion. Stimulated locomotion integrates some aspects of strength (physical function) and cognition (cognitive function).
Additional genes associated with C. elegans health
For C. elegans, we generated an additional list (see Supplementary Table 8) of health-associated genes (which differ from genes listed in Supplementary Tables 4, 5 to a large extent) that cannot be generated for humans (see Methods), using WormBase to systematically identify health-related compound interventions with associated gene expression data, and compiling the list of genes with strongest differential expression that are well-annotated by Gene Ontology terms.
From gene lists to maps of healthspan pathways
We used Cytoscape with selected plugins to obtain and annotate a connected network of the human healthspan associated genes from Supplementary Tables 1–3 and the C. elegans genes from Supplementary Tables 4, 5. Specifically, we used GeneMANIA to establish a gene/protein interaction network and to add connecting genes, and subsequently we clustered all genes based on their connectivity, and added GeneOntology-based annotations using AutoAnnotate. The resulting healthspan pathway maps are presented in the following. Moreover, health-related gene expression data are overlaid onto all healthspan pathway maps and will be discussed as well; these data are describing the effects of caloric restriction (CR) in humans [37] and of rapamycin in C. elegans [38], as examples of health-promoting interventions, or they describe the effects of aging and disease in specific tissues.
For humans, we derived a gene list (Supplementary Table 6) summarizing all genes associated with healthspan. (see Supplementary Tables 1–3 to trace back these genes to their origin). This list yielded the network of Figure 1, where the two largest pathways/clusters (15 and 13 genes) are specifically labeled by NOTCH and transcription initiation, and by proliferation, and the smaller pathways/clusters (4, 3, 3 and 3 genes) are labeled by cholesterol and lipid processes, by thymus activation, by myotube (striate muscle) regulation, and by Wnt signaling. In Figure 1 bottom, the list of pathways/clusters is given, and the details of the largest pathway are zoomed in.
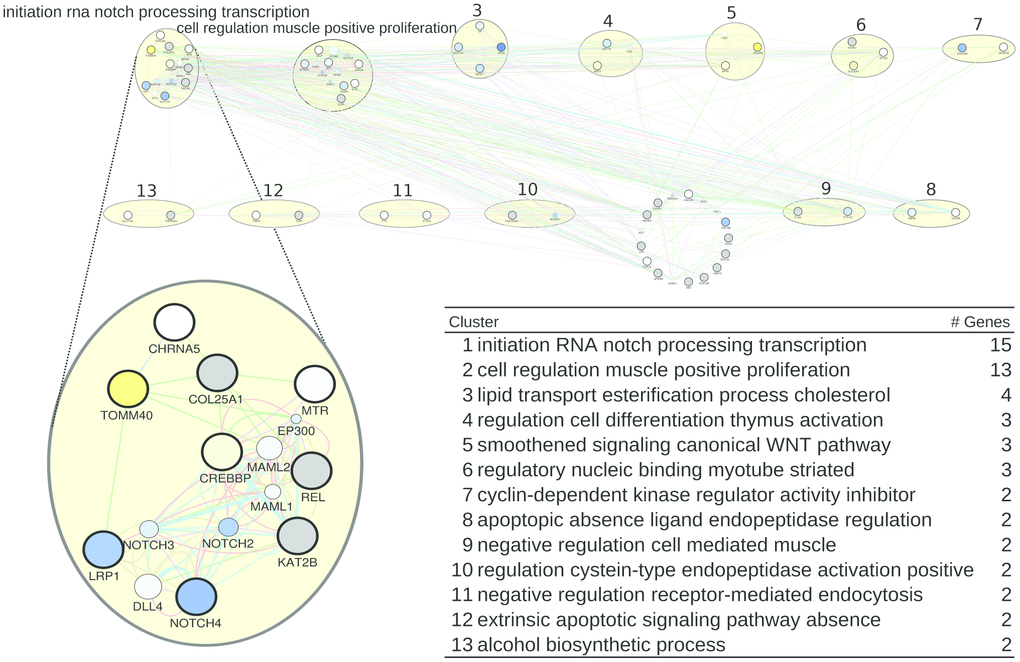
Figure 1. A healthspan pathway map for humans, based on Supplementary Tables 1–3, including the list of pathways/clusters with their labels as assigned by AutoAnnotate and their size (number of genes). The largest pathway is zoomed in to reveal details. The size of a gene node is proportional to its GeneMANIA score, which indicates the relevance of the gene with respect to the original list of genes to which another 20 genes are added by GeneMANIA, based on the network data. Genes upregulated by CR are shown in yellow, downregulated genes are shown in blue, and grey denotes genes for which no expression values are available in the caloric restriction dataset [37]. The color of an edge refers to the source of the edge in the underlying network, that is co-expression (pink), common pathway (green), physical interactions (red), shared protein domains (brown), co-localization (blue), predicted (orange), and genetic interaction (green). The thickness of an edge is proportional to its GeneMANIA “normalized max weight”, based on the network data. Genes from the GeneMANIA input list feature a thick circle, while genes added by GeneMANIA do not.
In the largest pathway/cluster, in light of the CR-triggered gene expression changes, the most prominent findings are an induced downregulation of NOTCH4 (and to a lesser extent of NOTCH 2 and 3), as well as of LRP1, and an upregulation of TOMM40 and CREBBP (also known as CBP). The family of NOTCH proteins has various functions, including a pro-inflammatory one [39, 40]. NOTCH4 is upregulated in kidney failure [41], and promotes vascularization/angiogenesis, which includes its upregulation in malignancy [40, 42]. A downregulation of NOTCH4 by CR can thus be taken as beneficial effect. This is less obvious for LRP1, the low-density lipoprotein receptor-related protein 1, which is responsible for membrane integrity and membrane cholesterol homeostasis, thus being involved in proper myelination [43] and vascular integrity [44]. A downregulation of LRP1 during CR could therefore be seen as deleterious. However, LRP1 expression mainly depends on cholesterol levels [45] – and these are lower during fasting. Hence, lower LRP1 expression actually reflects a lower LDL level, which per se has been found to be protective. The upregulations observed for TOMM40 and CREBBP during CR can also be seen as protective. TOMM40 is part of a mitochondrial membrane protein translocase, supporting mitochondrial function [46], and low expression and/or particular risk alleles of this protein are associated with Huntington’s and Alzheimer’s Disease [47, 48]. Of note, TOMM40 upregulation during CR goes together with APOE4 downregulation. Although both genes are closely located on chromosome 19, prompting the speculation that this linkage could imply concordant expression changes, this is obviously not the case here. CREBBP is a transcriptional co-activator with histone-acetyltransferase activity [49], acting primarily on histones 3 and 4, and thus it acts in concert with a range of transcription factors. Its downregulation is deleterious, resulting in, e.g., MHCII expression loss on lymphocytes [50], rendering the lymphocytes dysfunctional for antigen presentation, and in inflammatory signaling [51]. An upregulation of CREBBP by CR is thus likely beneficial. We further investigated the miRNAs that are statistically enriched in the largest healthspan pathway using the TFmir webserver [52], revealing regulation of NOTCH genes implicated in the epithelial-mesenchymal transition, cancer, heart failure and obesity, see Supplementary Results. The genes in the next-largest pathway/clusters, related to cell proliferation and lipids, are also described there in detail, as well as further evidence provided by mapping aging- and disease-related gene expression data onto them, as published or collected by Aramillo Irizar et al. [53].
For C. elegans, the gene list representing all healthspan associated genes is shown in Supplementary Table 7 (see Supplementary Tables 4–5 to trace back these genes to their origin). This list yielded the network of Figure 2, where the largest clusters (9 and 6 genes, respectively) are labeled by immune response process and by terms related to the mitochondrion. Three clusters (of 4 genes each) specifically feature dauer/dormancy, hormone response, and regulation. In Figure 2 bottom, the list of pathways/clusters, and the details of the largest pathway are zoomed in. Regarding the first pathway, rapamycin reduces ets-7 transcription, which was shown to be necessary for the healthspan-promoting effects of salicylamine [54]. Furthermore, rapamycin upregulates the transcription factor daf-16 (a homolog to Foxo) and downregulates the daf-16 inhibitors akt-1 and akt-2, putatively leading to an improved stress- and immune-response and prolonged lifespan via the Insulin/IGF-1 pathway [55]. Along the same lines, the akt-1 and akt-2 activator pdk-1 is also downregulated by rapamycin, further promoting daf-16 activity [56]. In contrast, the daf-16 inhibitor sgk-1 (a homolog to Nrf) is upregulated; however, its inhibitory role is subject of discussion [57]. Finally, the transcription factors hsf-1 and skn-1, both important in stress response processes [58, 59], are slightly downregulated in rapamycin-treated C. elegans. Thus, the stress defense system of C. elegans seems to play a central role in healthspan prolongation. Indeed, stress resistance is frequently discussed as a key to a long and healthy life. Vitagenes, which are genes involved in preserving cellular homeostasis during stress conditions, were shown to be crucial for the beneficial effects of dietary phytochemicals [60]. Furthermore, mild stress, which stimulates repair pathways and the stress defense of an organism including vitagenes, is able to promote healthy ageing in numerous ways [61]. This phenomenon, called hormesis, was held responsible for beneficial effects observed by many compound interventions [62–64]. More specific concepts, like mitohormesis which explains how reactive oxygen species can increase life- and healthspan [65] or the xenohormesis hypothesis which links evolutionary processes to the health-promoting abilities of plant-derived food [66] allow deeper insights into the entanglement of stress and health. In the Supplementary Results, the next-largest pathway/clusters, related to the mitochondrion, to dauer/dormancy, to regulation, and to hormone response are described in detail.
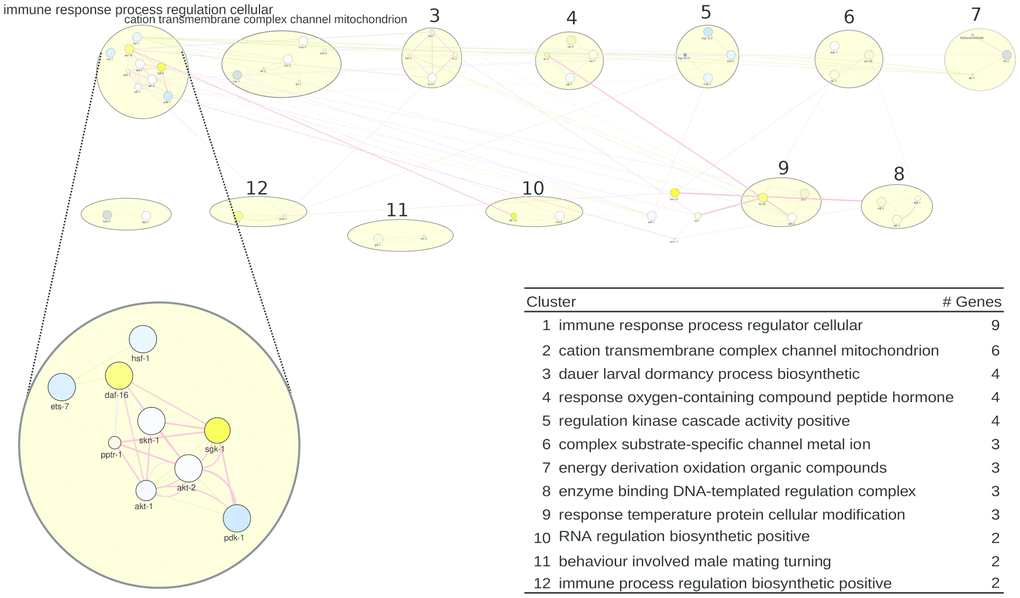
Figure 2. A healthspan pathway map for C. elegans, based on Supplementary Tables 4, 5. See also Figure 1. Gene expression data reflect the effect of rapamycin [38].
For C. elegans, we also derived a gene list from WormBase, taking the genes that are most differentially regulated by healthspan-extending interventions and, at the same time, are annotated with a sufficient number of GO terms (see Methods; Supplementary Table 8). We obtained the network of Figure 3. Curiously, the top healthspan pathways of 11, 9 and 8 genes are related to the endoplasmic reticulum (ER), lipid and membrane, to the peroxisome, macrobody and ER, and to the lysosome. The endoplasmic reticulum, the peroxisome and the lysosome are part of the endomembrane system, together with the mitochondria, contributing to healthspan and longevity in mammals and beyond [67]. Peroxisomal and lysosomal functions connect this pathway to dietary effects on lifespan [68, 69], and to liver disease [70]. The second tier of healthspan pathways (6 or 5 genes) are related to morphogenesis, biosynthesis and transcription.
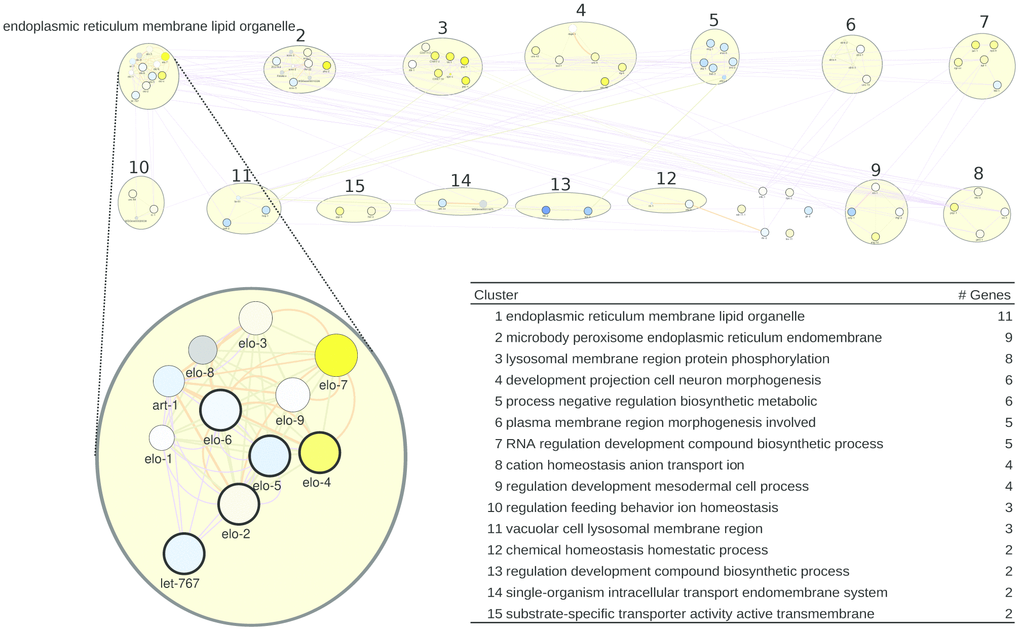
Figure 3. A healthspan pathway map for C. elegans, based on genes affected the most by healthspan-extending interventions, using WormBase gene expression data. See also Figures 1, 2.
For the WormBase data, the list of pathways/clusters, and the details of the largest pathway, are given in Figure 3, bottom. The ER/lipid-related pathway includes genes involved in fatty acid elongation/production (elo-1 to elo-9; let-767; art-1). Overlaying the rapamycin gene expression data, the well-characterized elo-1 and let-767 genes show some downregulation. However, the importance of elongase genes for health maintenance in general was repeatedly documented. Vásquez and colleagues [71] demonstrated the impairment of touch response in elo-1 mutants. They argue that elo-1 has a crucial role in the synthesis of C20 polyunsaturated fatty acids which are required for mechanosensation. Moreover, elo-1 mutants showed increased resistance to Pseudomonas aeruginosa infections due to the accumulation of gamma-linolenic acid and stearidonic acid [72] and knockdown of elo-1 or elo-2 extend survival during oxidative stress [73]. Finally, art-1 is a steroid reductase that is downregulated by rapamycin in our case, but also in long-lived eat-2 mutants [74]. In the Supplementary Results, the next-largest pathway/clusters, related to the ER, the peroxisome, the lysosome, morphogenesis, biosynthesis and transcription, are described in detail.
Overlap between human and C. elegans health genes and healthspan pathways
Based on reciprocal best orthologs, we found no direct overlap between the human health genes based on genetic associations and the C. elegans healthspan genes based in part on genetic interventions, but mostly on expert analysis of intervention effects (Figure 2), or on gene expression changes related to healthspan-extending interventions (Figure 3). We found some hints at an overlap on the level of the healthspan pathway annotations, considering that “proliferation” is listed for human, and “biosynthesis”, “immune response”, and “mitochondrion” for C. elegans, while “transcription” as well as “lipid” are found for both. Due to the post-mitotic nature of the adult C. elegans, proliferation processes have only minor impact on healthspan in C. elegans. In contrast, given that deregulated cell proliferation is the basis for cancer [75] and that cancer is one of the four main reasons for morbidity and mortality in humans according to the WHO (https://www.who.int/gho/ncd/mortality_morbidity/en/; status as of August 2019), it is not surprising that proliferation is a fundamental part of the human healthspan map. Furthermore, since C. elegans is usually fed on bacteria, which cause pathogenic stress in older nematodes [76, 77], the immune system is of particular importance for the health of nematodes. Finally, differences of the healthspan pathway maps regarding annotations such as “mitochondrion” could also be due to differences in how the underlying data were generated, in addition to species-specific differences.
Regarding lipids, for humans, specific reference is made to APOE/APOC (implicated in cholesterol metabolism); for C. elegans, specific reference is made to the elo set of genes (implicated in fatty acid elongation). The dysregulation of cholesterol and its different manifestations such as high- and low-density lipoprotein cholesterol (HDL-C and LDL-C) are one of the main causes for atherosclerotic cardiovascular diseases (CVD), a top ageing-related deadly disease [78, 79]. In contrast to mammals, C. elegans does not exhibit a heart or blood vessels and it cannot synthesize cholesterol by itself. Furthermore, a transgenic cholesterol-heterotrophic line lives 31% longer [80]. Another interesting difference is that cholesterol’s main task in nematodes is probably not its role as a crucial membrane component, but rather its role as a signaling molecule [81, 82]. Further discrepancies regarding the function and regulation of lipids in humans and C. elegans are summarized in Mullaney and Ashrafi [83]. Nevertheless, and quite surprisingly, numerous key components, functions and regulatory pathways regarding lipid metabolism are indeed comparable in C. elegans: Similarities in the regulation of membrane fluidity [84], of fat depletion after consumption of oats [85], legumes [86], and fibrates [87] as well as after exercise [88], and in the genetic background of obesity [89–91] and fat storage [92] are only a few examples. The adult worm is post-mitotic [93] but also many human diseases and cell senescence processes are associated with tissues that no longer divide, e.g., in the brain [94, 95].
In search for other modes of overlap, we additionally constructed and compared two interaction networks, based on mapping genes to their respective orthologs in the other species. Each of the two interaction networks is based on the union set of the health genes of human (based in turn on genetics, Supplementary Tables 1–3, Figure 1) and of C. elegans (based in turn on the gene expression analysis of healthspan-extending interventions using WormBase, Figure 3). Specifically, as outlined in Figure 4, we added the C. elegans orthologs of the human health genes to the list of C. elegans health genes and vice versa, yielding two separate input gene lists for GeneMANIA to enable the construction of the two interaction networks, one per species. We used strict ortholog mapping rules (only reciprocal best hits were accepted). By design, the two gene lists feature a high degree of overlap (with differences due to missing orthologs), and their subsequent comparison, consisting of the partial network alignments that are based on ortholog mapping on the one hand and the species-specific network data on the other hand can only reveal hypotheses for common healthspan pathways, as long as explicit experimental evidence for a relation to health is only found for one species. Moreover, interaction points between a healthspan pathway with evidence in one species and a healthspan pathway with evidence in the other species may be revealed, if a partial alignment of the interaction networks consists of interacting genes for which the relationship to health was demonstrated only in one species for each pair of orthologs.
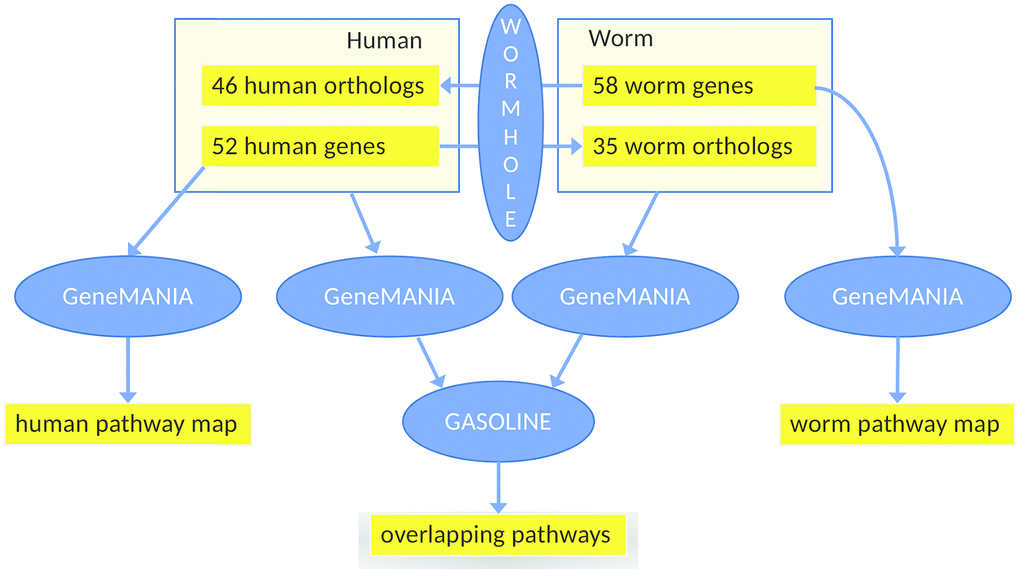
Figure 4. Workflow of the main analysis steps. First, 52 human health genes (Supplementary Tables 1–3) were processed with GeneMANIA and AutoAnnotate to determine the human healthspan pathway map (left, see also Figure 1). Analogously, 58 worm health genes (based on gene expression analysis using WormBase) were studied, yielding the C. elegans healthspan pathway map (right, see also Figure 3). Then, to determine overlap across species, the gene lists were extended by the orthologs (calculated by WORMHOLE, see Supplemental Methods) from the respective other species. We then employed GeneMANIA as before, to generate two interaction networks (one per list). and overlaps between these two networks of health genes were determined by GASOLINE (middle, see also Figure 5).
Of the two interaction networks to be aligned, the first network is based on C. elegans health genes, the C. elegans orthologs of human health genes, and C. elegans gene interaction information provided by GeneMANIA. The second network is based on human health genes, the human orthologs of C. elegans health genes, and human gene interaction information provided by GeneMANIA. Despite using similar lists of genes (with differences due to missing orthologs and due to the genes added by GeneMANIA), we can expect that the two GeneMANIA networks are quite different because the interaction data sources employed by GeneMANIA are strongly species-specific. Moreover, we observe that in both cases, the 20 closely interacting genes added by GeneMANIA for one species included no orthologs of the other species. Nevertheless, to identify joint healthspan pathways and interaction points between healthspan pathways, we used GASOLINE [96] to align the two networks wherever feasible, obtaining two partial (subnetwork) alignments as output, as shown in Figure 5.
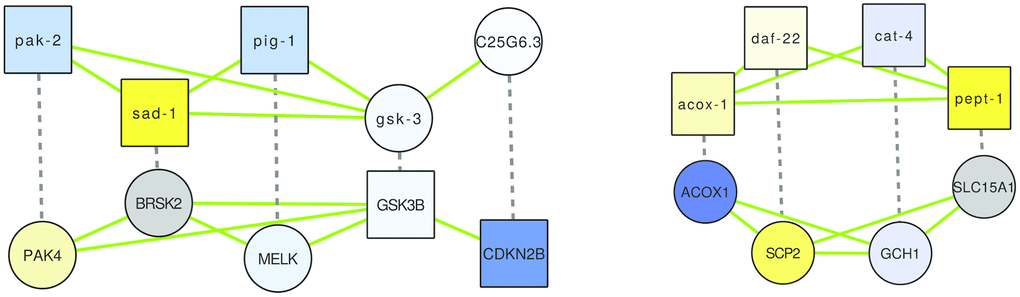
Figure 5. The two alignments demonstrating overlap of (putative) healthspan pathways in human and C. elegans, based on a GASOLINE alignment of the network of genes implicated in health-related gene expression changes in WormBase (top), and in human health based on genetic studies (bottom), and of corresponding orthologs. Dashed edges indicate orthologs, green edges indicate interactions based on GeneMANIA known for the respective species; the node shape is square if the gene originates from the original lists of health genes and it is circular if the gene is an ortholog, and node colors are based on gene expression changes triggered by rapamycin (in case of C. elegans) or by caloric restriction (in case of human), as in Figures 1–3.
In the first alignment (Figure 5, left), we see an alternating pattern of demonstrated health-relatedness, since pak-2, sad-1 and pig-1 are considered health-related by gene expression analysis using WormBase, while CDKN2B and GSK3B are known to be human health genes (Supplementary Tables 1–3; GSK3B was implicated by a GWAS of the Healthy Aging Index, while CDKN2B was in fact one of the few genes implicated by two independent health studies). The C. elegans genes belong to three small clusters in the healthspan pathway map of Figure 3 (pak-2: lysosomal, sad-1: neural, pig-1: biosynthesis), while the human genes belong to one large (GSK3B: proliferation) and one small (CDKN3B: cyclin-dependent kinase) cluster in the human healthspan pathway map of Figure 1. Interactions in C. elegans are all based on shared domains (kinase signaling, except for the predicted interaction of gsk-3 and C25G6.3, which is based on the Interologous Interaction Database), while interactions in human are based on shared domains, genetic interaction (i.e., large-scale radiation hybrid) and pathway data. Essentially, the healthspan pathway overlap suggested by our analysis involves proliferation-related serine/tyrosine kinase signaling (pak-2/sad-1/pig-1 and PAK4/BRSK2/MELK), Wnt signaling (GSK3) and cyclin-dependent kinase signaling (CDKN2B). Both alignments are described further in detail in the Supplementary Results, and a functional analysis of the genes is given in Supplementary Tables 11, 12.
Given lists of genes, there is a plethora of possibilities to organize the genes into groups of related ones. Motivated by the idea of a “healthspan pathway”, we hypothesized that the genes should be known to interact based on functional gene/protein interaction data (provided by GeneMANIA). Here, as in most other studies, pathways are not assumed to be linear [97]. The (higher-level) interaction among the clusters/healthspan pathways (i.e., the pathway map) is given by the individual gene/protein interactions that are shown between the clusters in Figures 1–3. However, we did not investigate these further.
The small amount of healthspan gene/pathway overlap that we found may be seen from a pessimistic or an optimistic perspective, depending in part on expectations. From the pessimistic perspective, the molecular processes may be completely different, and the C. elegans orthologs of the human health genes are involved in different processes as compared to the human health genes, and vice versa. From the optimistic perspective, it may just be that the number and scope of the investigations that yielded the health genes we studied is still insufficient, annotations are still incomplete, and considering only reciprocal best orthologs may be too restrictive. (We tried a less restrictive mapping of orthologs by relaxing the condition that orthologs must be reciprocal, but the overlap was still negligible; results not shown). Nevertheless, future genetic studies are expected to yield more health genes in both species, and their characterizations are expected to improve. Moreover, when we analyze in detail the effects of intervention studies in C.elegans, we do find clear hints to some mechanisms that underlie healthspan also in human [98]. For example, changes in the Ins/IGF-1 pathway genes daf-2 and daf-16 are found to be associated with many of the features described in Supplementary Table 5, suggesting a fundamental role for immune defense mechanisms (and proliferation) in health maintenance, as described by Ermolaeva et al. [99].
Since C. elegans only exhibits an innate immune system and is missing the adaptive immune response, one could argue that the biological relevance of “immune response” in the C. elegans healthspan pathway map is negligible. However, the strict separation of the immune response into an innate and an adaptive system was questioned by Kvell et al. [100] and more recently by Penkov et al. [101], not least because of the discovery of the trained innate immune response [102]. Furthermore, the suitability of C. elegans as a model for the mammalian immune system and for pathogen response was summarized in several reviews [99, 103, 104]. Indeed, based on the expression of antifungal or antibacterial polypeptides in response to pathogenic stress, this nematode is used to find new antimicrobial drugs [105, 106]. Finally, it was demonstrated that immunosenescence, which is one of the most important healthspan parameters, affects the innate immune system in both organisms, nematodes [107–109] and humans [110].
Of course, the precise definition of phenotype is crucial. If the samples are not really about (lack of) health, in human or in C. elegans, then any subsequent molecular or bioinformatics analyses will compare apples and oranges and may thus fail. Therefore, it is important to use a good phenotyping of health in human as well as in C. elegans, and on this basis, to collect data as genome-wide as possible. For most of the age-related diseases that we use to define health in humans, there is no C. elegans counterpart. E.g., as C. elegans has no heart, it cannot have any heart diseases. In addition, the aging process that may underlie most of these age-related diseases is poorly characterized and hard to quantify in humans. Nonetheless, locomotion degrades with age in both species, due to changes at the muscle as well as neural level. Two related features of physical function, that is, grip strength [111] and the ability to sit and rise from the floor [112] are good predictors of all-cause mortality in humans. Likewise, both in humans and in C. elegans, the ability to withstand various forms of stress decreases with age [113, 114]. Thus, at the level of organs or functional systems, both C. elegans and humans show age-related declines in performance, that may well be due to underlying processes that are similar at the cellular and molecular level. Moreover, the investigation of healthspan in C. elegans already identified additional ageing-related genes, e.g. for EGF signaling, which is known for its connection to ageing in mammals [115, 116]. Interestingly, in C. elegans, the EGF-regulator HPA-2 was identified by analyzing locomotion but not lifespan [117] highlighting the usefulness for phenotyping-assays distinct from lifespan. This is underlined by the observation that locomotion is impaired during ageing in mammals and C. elegans in a similar way [118].
Overall, we suggest that within the limitations of currently available data, the health genes we assembled, the healthspan pathways we constructed based on these, and the overlap we then found between species, are a first glimpse of the species-specific and cross-species molecular basis of health.
Materials and Methods
Gene sets associated with health, literature-based
In this work, we conducted a semi-systematic review, including publications until 2018, using health, healthspan and healthy aging, for human and C. elegans, as search terms in Google Scholar, initially filtering for recent reviews and considering only the top hits. For humans, genetic studies of Supplementary Tables 1–3 are often not found using health-related keywords, so we included terms related to dysfunction (such as frailty) and disease (such as multi-morbidity) as well. For the genetics of human frailty, we identified two publications [28, 29]. Overall, a list of 52 genes (Supplementary Table 1, 12 genes; Supplementary Tables 2, 3, 40 genes) was taken as the starting point in humans. For the genetics of C. elegans health, we followed a similar approach (Supplementary Table 4). For compound interventions in C. elegans, we identified a specific set of recent reviews (see Supplementary Table 5). Overall, a list of 31 genes (Supplementary Table 4, 11 genes; Supplementary Table 5, 20 genes) was taken as the starting point in C. elegans. From the original publications and reviews, we extracted the gene names, using iHOP [119] to assign HUGO nomenclature names if necessary. In the Supplement, we describe in detail how a second set of health-associated genes in C. elegans was identified using WormBase.
Construction of maps of clusters/pathways
For all gene sets analyzed, we used the Cytoscape 3.5.1 application GeneMANIA [32], version 3.4.1, downloaded October 2017, with default settings, to create a functional interaction network that is complemented with the GeneMANIA default of 20 connecting genes. For clustering, and for annotating the clusters based on the “annotation name” column of GO annotations collected by GeneMANIA, we used AutoAnnotate [33] v1.2, downloaded October 2017, in Quick start mode to enable to “layout network to prevent cluster overlap”, so that a map of disjoint clusters (i.e., healthspan pathways) was generated. This was supplemented by a second advanced annotation step to increase the “max. number of words per cluster label” to the largest possible value of 5. Cluster annotations were generated using WordCloud [120] v3.1.1, downloaded January 2018.
In the Supplement, we further describe in detail how we overlaid expression data onto the pathway maps, constructed the overlap of healthspan pathways in C. elegans and humans, and programmed the web presentation.
Data accessibility
The accompanying web presentation uses CytoscapeJS to present the pathways, which also offers all pathway maps for download that were exported from Cytoscape. All files contributing to the analysis and to the website are freely available from https://bitbucket.org/ibima/healthspannetworkscytoscapejs. The above described generation of pathway maps was performed manually by interacting with the respective tools. Genes can be selected via their cluster or by the GeneOntology terms they are annotated with. Any such selection of genes is referenced to the MEM [121] and g:Profiler [122] web services.
Author Contributions
Study design: GF, WL, SM, NS. Collection of data: GF, NS, SM. Analysis of data: GF, NS, RK, SSe, HME, CJ, SS, IB, MH. Website: SM, PAd, JV. Manuscript writing: GF, NS, SM, AAC, RK, SSe, HME, LS, BW, FC, AB, PAn, HJG, DR, MB, LJ. All authors reviewed and approved the final manuscript.
Acknowledgments
We thank Yasmeen Quawasmeh for technical assistance. We acknowledge assistance by Giovanni Micale in using GASOLINE.
Conflicts of Interest
Alan A. Cohen is founder and CSO at Oken Health. All other authors declare that they have no conflicts of interest.
Funding
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under Grant agreement No 633589 (Aging with Elegans). This publication reflects only the authors’ views and the Commission is not responsible for any use that may be made of the information it contains. AAC is supported by a CIHR New Investigator Salary Award and is a member of the Fonds de recherche du Québec – Santé funded Centre de recherche du CHUS and Centre de re-cherche sur le vieillissement. Priit Adler and Jaak Vilo acknowledge research and open access funding from Estonian Research Agency grant IUT34-4. Brecht Wouters was supported by FWO Grant 11A5420N. PA was supported by the BME NC TKP2020 grant of NKFIH Hungary, BME-Biotechnology FIKP grant of EMMI (BME FIKP-BIO), OTKA K119866.
References
- 1. Crimmins EM. Lifespan and healthspan: past, present, and promise. Gerontologist. 2015; 55:901–11. https://doi.org/10.1093/geront/gnv130 [PubMed]
- 2. Robine JM, Jagger C, Crimmins E. (2013). Healthy Longevity: A Global Approach: Springer Publishing Company.
- 3. Fillenbaum GG, Pieper CF, Cohen HJ, Cornoni-Huntley JC, Guralnik JM. Comorbidity of five chronic health conditions in elderly community residents: determinants and impact on mortality. J Gerontol A Biol Sci Med Sci. 2000; 55:M84–89. https://doi.org/10.1093/gerona/55.2.m84 [PubMed]
- 4. Wolfson M, Budovsky A, Tacutu R, Fraifeld V. The signaling hubs at the crossroad of longevity and age-related disease networks. Int J Biochem Cell Biol. 2009; 41:516–20. https://doi.org/10.1016/j.biocel.2008.08.026 [PubMed]
- 5. Fuellen G, Jansen L, Cohen AA, Luyten W, Gogol M, Simm A, Saul N, Cirulli F, Berry A, Antal P, Köhling R, Wouters B, Möller S. Health and aging: unifying concepts, scores, biomarkers and pathways. Aging Dis. 2019; 10:883–900. https://doi.org/10.14336/AD.2018.1030 [PubMed]
- 6. Fuellen G, Schofield P, Flatt T, Schulz RJ, Boege F, Kraft K, Rimbach G, Ibrahim S, Tietz A, Schmidt C, Köhling R, Simm A. Living long and well: prospects for a personalized approach to the medicine of ageing. Gerontology. 2016; 62:409–16. https://doi.org/10.1159/000442746 [PubMed]
- 7. Berman AY, Motechin RA, Wiesenfeld MY, Holz MK. The therapeutic potential of resveratrol: a review of clinical trials. NPJ Precis Oncol. 2017; 1:35. https://doi.org/10.1038/s41698-017-0038-6 [PubMed]
- 8. Brasnyó P, Molnár GA, Mohás M, Markó L, Laczy B, Cseh J, Mikolás E, Szijártó IA, Mérei A, Halmai R, Mészáros LG, Sümegi B, Wittmann I. Resveratrol improves insulin sensitivity, reduces oxidative stress and activates the Akt pathway in type 2 diabetic patients. Br J Nutr. 2011; 106:383–89. https://doi.org/10.1017/S0007114511000316 [PubMed]
- 9. Chen S, Zhao X, Ran L, Wan J, Wang X, Qin Y, Shu F, Gao Y, Yuan L, Zhang Q, Mi M. Resveratrol improves insulin resistance, glucose and lipid metabolism in patients with non-alcoholic fatty liver disease: a randomized controlled trial. Dig Liver Dis. 2015; 47:226–32. https://doi.org/10.1016/j.dld.2014.11.015 [PubMed]
- 10. Ghanim H, Sia CL, Korzeniewski K, Lohano T, Abuaysheh S, Marumganti A, Chaudhuri A, Dandona P. A resveratrol and polyphenol preparation suppresses oxidative and inflammatory stress response to a high-fat, high-carbohydrate meal. J Clin Endocrinol Metab. 2011; 96:1409–14. https://doi.org/10.1210/jc.2010-1812 [PubMed]
- 11. Gruber J, Chen CB, Fong S, Ng LF, Teo E, Halliwell B. Caenorhabditis elegans: what we can and cannot learn from aging worms. Antioxid Redox Signal. 2015; 23:256–79. https://doi.org/10.1089/ars.2014.6210 [PubMed]
- 12. Kim W, Underwood RS, Greenwald I, Shaye DD. OrthoList 2: a new comparative genomic analysis of human and Caenorhabditis elegans genes. Genetics. 2018; 210:445–61. https://doi.org/10.1534/genetics.118.301307 [PubMed]
- 13. Berry A, Cirulli F. The p66(shc) gene paves the way for healthspan: evolutionary and mechanistic perspectives. Neurosci Biobehav Rev. 2013; 37:790–802. https://doi.org/10.1016/j.neubiorev.2013.03.005 [PubMed]
- 14. Schumacher B, van der Pluijm I, Moorhouse MJ, Kosteas T, Robinson AR, Suh Y, Breit TM, van Steeg H, Niedernhofer LJ, van Ijcken W, Bartke A, Spindler SR, Hoeijmakers JH, et al. Delayed and accelerated aging share common longevity assurance mechanisms. PLoS Genet. 2008; 4:e1000161. https://doi.org/10.1371/journal.pgen.1000161 [PubMed]
- 15. Yanai H, Budovsky A, Barzilay T, Tacutu R, Fraifeld VE. Wide-scale comparative analysis of longevity genes and interventions. Aging Cell. 2017; 16:1267–75. https://doi.org/10.1111/acel.12659 [PubMed]
- 16. Sutphin GL, Backer G, Sheehan S, Bean S, Corban C, Liu T, Peters MJ, van Meurs JB, Murabito JM, Johnson AD, Korstanje R, and Cohorts for Heart and Aging Research in Genomic Epidemiology (CHARGE) Consortium Gene Expression Working Group. Caenorhabditis elegans orthologs of human genes differentially expressed with age are enriched for determinants of longevity. Aging Cell. 2017; 16:672–82. https://doi.org/10.1111/acel.12595 [PubMed]
- 17. Luyten W, Antal P, Braeckman BP, Bundy J, Cirulli F, Fang-Yen C, Fuellen G, Leroi A, Liu Q, Martorell P, Metspalu A, Perola M, Ristow M, et al. Ageing with elegans: a research proposal to map healthspan pathways. Biogerontology. 2016; 17:771–82. https://doi.org/10.1007/s10522-016-9644-x [PubMed]
- 18. Chen X, Barclay JW, Burgoyne RD, Morgan A. Using C. Elegans to discover therapeutic compounds for ageing-associated neurodegenerative diseases. Chem Cent J. 2015; 9:65. https://doi.org/10.1186/s13065-015-0143-y [PubMed]
- 19. Luo Y, Wu Y, Brown M, Link CD. Caenorhabditis elegans Model for Initial Screening and Mechanistic Evaluation of Potential New Drugs for Aging and Alzheimer’s Disease. In: Buccafusco JJ, editor. Methods of Behavior Analysis in Neuroscience. ed. Boca Raton (FL): CRC Press/Taylor & Francis; 2009. [PubMed]
- 20. Collins JJ, Evason K, Kornfeld K. Pharmacology of delayed aging and extended lifespan of caenorhabditis elegans. Exp Gerontol. 2006; 41:1032–39. https://doi.org/10.1016/j.exger.2006.06.038 [PubMed]
- 21. De Haes W, Frooninckx L, Van Assche R, Smolders A, Depuydt G, Billen J, Braeckman BP, Schoofs L, Temmerman L. Metformin promotes lifespan through mitohormesis via the peroxiredoxin PRDX-2. Proc Natl Acad Sci USA. 2014; 111:E2501–09. https://doi.org/10.1073/pnas.1321776111 [PubMed]
- 22. Verma S, Jagtap U, Goyala A, Mukhopadhyay A. A novel gene-diet pair modulates C. Elegans aging. PLoS Genet. 2018; 14:e1007608. https://doi.org/10.1371/journal.pgen.1007608 [PubMed]
- 23. Revtovich AV, Lee R, Kirienko NV. Interplay between mitochondria and diet mediates pathogen and stress resistance in caenorhabditis elegans. PLoS Genet. 2019; 15:e1008011. https://doi.org/10.1371/journal.pgen.1008011 [PubMed]
- 24. Broer L, Buchman AS, Deelen J, Evans DS, Faul JD, Lunetta KL, Sebastiani P, Smith JA, Smith AV, Tanaka T, Yu L, Arnold AM, Aspelund T, et al. GWAS of longevity in CHARGE consortium confirms APOE and FOXO3 candidacy. J Gerontol A Biol Sci Med Sci. 2015; 70:110–18. https://doi.org/10.1093/gerona/glu166 [PubMed]
- 25. Cohen AA, Legault V, Fuellen G, Fülöp T, Fried LP, Ferrucci L. The risks of biomarker-based epidemiology: associations of circulating calcium levels with age, mortality, and frailty vary substantially across populations. Exp Gerontol. 2018; 107:11–17. https://doi.org/10.1016/j.exger.2017.07.011 [PubMed]
- 26. Barger JL, Kayo T, Vann JM, Arias EB, Wang J, Hacker TA, Wang Y, Raederstorff D, Morrow JD, Leeuwenburgh C, Allison DB, Saupe KW, Cartee GD, et al. A low dose of dietary resveratrol partially mimics caloric restriction and retards aging parameters in mice. PLoS One. 2008; 3:e2264. https://doi.org/10.1371/journal.pone.0002264 [PubMed]
- 27. Martin GM, Bergman A, Barzilai N. Genetic determinants of human health span and life span: progress and new opportunities. PLoS Genet. 2007; 3:e125. https://doi.org/10.1371/journal.pgen.0030125 [PubMed]
- 28. Mekli K, Marshall A, Nazroo J, Vanhoutte B, Pendleton N. Genetic variant of interleukin-18 gene is associated with the frailty index in the english longitudinal study of ageing. Age Ageing. 2015; 44:938–42. https://doi.org/10.1093/ageing/afv122 [PubMed]
- 29. Ho YY, Matteini AM, Beamer B, Fried L, Xue QL, Arking DE, Chakravarti A, Fallin MD, Walston J. Exploring biologically relevant pathways in frailty. J Gerontol A Biol Sci Med Sci. 2011; 66:975–79. https://doi.org/10.1093/gerona/glr061 [PubMed]
- 30. Sanders JL, Minster RL, Barmada MM, Matteini AM, Boudreau RM, Christensen K, Mayeux R, Borecki IB, Zhang Q, Perls T, Newman AB. Heritability of and mortality prediction with a longevity phenotype: the healthy aging index. J Gerontol A Biol Sci Med Sci. 2014; 69:479–85. https://doi.org/10.1093/gerona/glt117 [PubMed]
- 31. Rollins JA, Howard AC, Dobbins SK, Washburn EH, Rogers AN. Assessing health span in caenorhabditis elegans: lessons from short-lived mutants. J Gerontol A Biol Sci Med Sci. 2017; 72:473–80. https://doi.org/10.1093/gerona/glw248 [PubMed]
- 32. Zuberi K, Franz M, Rodriguez H, Montojo J, Lopes CT, Bader GD, Morris Q. GeneMANIA prediction server 2013 update. Nucleic Acids Res. 2013; 41:W115–22. https://doi.org/10.1093/nar/gkt533 [PubMed]
- 33. Kucera M, Isserlin R, Arkhangorodsky A, Bader GD. AutoAnnotate: a cytoscape app for summarizing networks with semantic annotations. F1000Res. 2016; 5:1717. https://doi.org/10.12688/f1000research.9090.1 [PubMed]
- 34. Vora M, Shah M, Ostafi S, Onken B, Xue J, Ni JZ, Gu S, Driscoll M. Deletion of microRNA-80 activates dietary restriction to extend C. Elegans healthspan and lifespan. PLoS Genet. 2013; 9:e1003737. https://doi.org/10.1371/journal.pgen.1003737 [PubMed]
- 35. Aalto AP, Nicastro IA, Broughton JP, Chipman LB, Schreiner WP, Chen JS, Pasquinelli AE. Opposing roles of microRNA argonautes during caenorhabditis elegans aging. PLoS Genet. 2018; 14:e1007379. https://doi.org/10.1371/journal.pgen.1007379 [PubMed]
- 36. Schwarz DF, Hädicke O, Erdmann J, Ziegler A, Bayer D, Möller S. SNPtoGO: characterizing SNPs by enriched GO terms. Bioinformatics. 2008; 24:146–48. https://doi.org/10.1093/bioinformatics/btm551 [PubMed]
- 37. Mercken EM, Crosby SD, Lamming DW, JeBailey L, Krzysik-Walker S, Villareal DT, Capri M, Franceschi C, Zhang Y, Becker K, Sabatini DM, de Cabo R, Fontana L. Calorie restriction in humans inhibits the PI3K/AKT pathway and induces a younger transcription profile. Aging Cell. 2013; 12:645–51. https://doi.org/10.1111/acel.12088 [PubMed]
- 38. Calvert S, Tacutu R, Sharifi S, Teixeira R, Ghosh P, de Magalhães JP. A network pharmacology approach reveals new candidate caloric restriction mimetics in C. Elegans. Aging Cell. 2016; 15:256–66. https://doi.org/10.1111/acel.12432 [PubMed]
- 39. Balistreri CR, Madonna R, Melino G, Caruso C. The emerging role of notch pathway in ageing: focus on the related mechanisms in age-related diseases. Ageing Res Rev. 2016; 29:50–65. https://doi.org/10.1016/j.arr.2016.06.004 [PubMed]
- 40. Zhang J, Kuang Y, Wang Y, Xu Q, Ren Q. Notch-4 silencing inhibits prostate cancer growth and EMT via the NF-κB pathway. Apoptosis. 2017; 22:877–84. https://doi.org/10.1007/s10495-017-1368-0 [PubMed]
- 41. Liu M, Liang K, Zhen J, Zhou M, Wang X, Wang Z, Wei X, Zhang Y, Sun Y, Zhou Z, Su H, Zhang C, Li N, et al. Sirt6 deficiency exacerbates podocyte injury and proteinuria through targeting notch signaling. Nat Commun. 2017; 8:413. https://doi.org/10.1038/s41467-017-00498-4 [PubMed]
- 42. Kofler NM, Shawber CJ, Kangsamaksin T, Reed HO, Galatioto J, Kitajewski J. Notch signaling in developmental and tumor angiogenesis. Genes Cancer. 2011; 2:1106–16. https://doi.org/10.1177/1947601911423030 [PubMed]
- 43. Lin JP, Mironova YA, Shrager P, Giger RJ. LRP1 regulates peroxisome biogenesis and cholesterol homeostasis in oligodendrocytes and is required for proper CNS myelin development and repair. Elife. 2017; 6:e30498. https://doi.org/10.7554/eLife.30498 [PubMed]
- 44. Strickland DK, Au DT, Cunfer P, Muratoglu SC. Low-density lipoprotein receptor-related protein-1: role in the regulation of vascular integrity. Arterioscler Thromb Vasc Biol. 2014; 34:487–98. https://doi.org/10.1161/ATVBAHA.113.301924 [PubMed]
- 45. Llorente-Cortés V, Otero-Viñas M, Sánchez S, Rodríguez C, Badimon L. Low-density lipoprotein upregulates low-density lipoprotein receptor-related protein expression in vascular smooth muscle cells: possible involvement of sterol regulatory element binding protein-2-dependent mechanism. Circulation. 2002; 106:3104–10. https://doi.org/10.1161/01.cir.0000041434.28573.0b [PubMed]
- 46. Zeitlow K, Charlambous L, Ng I, Gagrani S, Mihovilovic M, Luo S, Rock DL, Saunders A, Roses AD, Gottschalk WK. The biological foundation of the genetic association of TOMM40 with late-onset alzheimer’s disease. Biochim Biophys Acta Mol Basis Dis. 2017; 1863:2973–86. https://doi.org/10.1016/j.bbadis.2017.07.031 [PubMed]
- 47. Shirendeb U, Reddy AP, Manczak M, Calkins MJ, Mao P, Tagle DA, Reddy PH. Abnormal mitochondrial dynamics, mitochondrial loss and mutant huntingtin oligomers in huntington’s disease: implications for selective neuronal damage. Hum Mol Genet. 2011; 20:1438–55. https://doi.org/10.1093/hmg/ddr024 [PubMed]
- 48. Chong MS, Goh LK, Lim WS, Chan M, Tay L, Chen G, Feng L, Ng TP, Tan CH, Lee TS. Gene expression profiling of peripheral blood leukocytes shows consistent longitudinal downregulation of TOMM40 and upregulation of KIR2DL5A, PLOD1, and SLC2A8 among fast progressors in early alzheimer’s disease. J Alzheimers Dis. 2013; 34:399–405. https://doi.org/10.3233/JAD-121621 [PubMed]
- 49. Bedford DC, Brindle PK. Is histone acetylation the most important physiological function for CBP and p300? Aging (Albany NY). 2012; 4:247–55. https://doi.org/10.18632/aging.100453 [PubMed]
- 50. Hashwah H, Schmid CA, Kasser S, Bertram K, Stelling A, Manz MG, Müller A. Inactivation of CREBBP expands the germinal center B cell compartment, down-regulates MHCII expression and promotes DLBCL growth. Proc Natl Acad Sci USA. 2017; 114:9701–06. https://doi.org/10.1073/pnas.1619555114 [PubMed]
- 51. Dixon ZA, Nicholson L, Zeppetzauer M, Matheson E, Sinclair P, Harrison CJ, Irving JA. CREBBP knockdown enhances RAS/RAF/MEK/ERK signaling in ras pathway mutated acute lymphoblastic leukemia but does not modulate chemotherapeutic response. Haematologica. 2017; 102:736–45. https://doi.org/10.3324/haematol.2016.145177 [PubMed]
- 52. Hamed M, Spaniol C, Nazarieh M, Helms V. TFmiR: a web server for constructing and analyzing disease-specific transcription factor and miRNA co-regulatory networks. Nucleic Acids Res. 2015; 43:W283–88. https://doi.org/10.1093/nar/gkv418 [PubMed]
- 53. Aramillo Irizar P, Schäuble S, Esser D, Groth M, Frahm C, Priebe S, Baumgart M, Hartmann N, Marthandan S, Menzel U, Müller J, Schmidt S, Ast V, et al. Transcriptomic alterations during ageing reflect the shift from cancer to degenerative diseases in the elderly. Nat Commun. 2018; 9:327. https://doi.org/10.1038/s41467-017-02395-2 [PubMed]
- 54. Nguyen TT, Caito SW, Zackert WE, West JD, Zhu S, Aschner M, Fessel JP, Roberts LJ
2nd . Scavengers of reactive γ-ketoaldehydes extend caenorhabditis elegans lifespan and healthspan through protein-level interactions with SIR-2.1 and ETS-7. Aging (Albany NY). 2016; 8:1759–80. https://doi.org/10.18632/aging.101011 [PubMed] - 55. Henderson ST, Johnson TE. Daf-16 integrates developmental and environmental inputs to mediate aging in the nematode caenorhabditis elegans. Curr Biol. 2001; 11:1975–80. https://doi.org/10.1016/s0960-9822(01)00594-2 [PubMed]
- 56. Paradis S, Ailion M, Toker A, Thomas JH, Ruvkun G. A PDK1 homolog is necessary and sufficient to transduce AGE-1 PI3 kinase signals that regulate diapause in caenorhabditis elegans. Genes Dev. 1999; 13:1438–52. https://doi.org/10.1101/gad.13.11.1438 [PubMed]
- 57. Mizunuma M, Neumann-Haefelin E, Moroz N, Li Y, Blackwell TK. mTORC2-SGK-1 acts in two environmentally responsive pathways with opposing effects on longevity. Aging Cell. 2014; 13:869–78. https://doi.org/10.1111/acel.12248 [PubMed]
- 58. Morton EA, Lamitina T. Caenorhabditis elegans HSF-1 is an essential nuclear protein that forms stress granule-like structures following heat shock. Aging Cell. 2013; 12:112–20. https://doi.org/10.1111/acel.12024 [PubMed]
- 59. Wang J, Robida-Stubbs S, Tullet JM, Rual JF, Vidal M, Blackwell TK. RNAi screening implicates a SKN-1-dependent transcriptional response in stress resistance and longevity deriving from translation inhibition. PLoS Genet. 2010; 6:e1001048. https://doi.org/10.1371/journal.pgen.1001048 [PubMed]
- 60. Calabrese V, Cornelius C, Dinkova-Kostova AT, Iavicoli I, Di Paola R, Koverech A, Cuzzocrea S, Rizzarelli E, Calabrese EJ. Cellular stress responses, hormetic phytochemicals and vitagenes in aging and longevity. Biochim Biophys Acta. 2012; 1822:753–83. https://doi.org/10.1016/j.bbadis.2011.11.002 [PubMed]
- 61. Rattan SI. Hormesis in aging. Ageing Res Rev. 2008; 7:63–78. https://doi.org/10.1016/j.arr.2007.03.002 [PubMed]
- 62. Martel J, Ojcius DM, Ko YF, Ke PY, Wu CY, Peng HH, Young JD. Hormetic effects of phytochemicals on health and longevity. Trends Endocrinol Metab. 2019; 30:335–46. https://doi.org/10.1016/j.tem.2019.04.001 [PubMed]
- 63. Martucci M, Ostan R, Biondi F, Bellavista E, Fabbri C, Bertarelli C, Salvioli S, Capri M, Franceschi C, Santoro A. Mediterranean diet and inflammaging within the hormesis paradigm. Nutr Rev. 2017; 75:442–55. https://doi.org/10.1093/nutrit/nux013 [PubMed]
- 64. Calabrese V, Cornelius C, Trovato A, Cavallaro M, Mancuso C, Di Rienzo L, Condorelli D, De Lorenzo A, Calabrese EJ. The hormetic role of dietary antioxidants in free radical-related diseases. Curr Pharm Des. 2010; 16:877–83. https://doi.org/10.2174/138161210790883615 [PubMed]
- 65. Ristow M. Unraveling the truth about antioxidants: mitohormesis explains ROS-induced health benefits. Nat Med. 2014; 20:709–11. https://doi.org/10.1038/nm.3624 [PubMed]
- 66. Suter S, Lucock M. Xenohormesis: Applying evolutionary principles to contemporary health issues. Exploratory Research and Hypothesis in Medicine. 2017; 2:79–85. https://doi.org/10.14218/ERHM.2017.00023
- 67. Nisoli E, Valerio A. Healthspan and longevity in mammals: a family game for cellular organelles? Curr Pharm Des. 2014; 20:5663–70. https://doi.org/10.2174/1381612820666140306093651 [PubMed]
- 68. Gelino S, Chang JT, Kumsta C, She X, Davis A, Nguyen C, Panowski S, Hansen M. Intestinal autophagy improves healthspan and longevity in C. Elegans during dietary restriction. PLoS Genet. 2016; 12:e1006135. https://doi.org/10.1371/journal.pgen.1006135 [PubMed]
- 69. Weir HJ, Yao P, Huynh FK, Escoubas CC, Goncalves RL, Burkewitz K, Laboy R, Hirschey MD, Mair WB. Dietary restriction and AMPK increase lifespan via mitochondrial network and peroxisome remodeling. Cell Metab. 2017; 26:884–96.e5. https://doi.org/10.1016/j.cmet.2017.09.024 [PubMed]
- 70. Cai M, Sun X, Wang W, Lian Z, Wu P, Han S, Chen H, Zhang P. Disruption of peroxisome function leads to metabolic stress, mTOR inhibition, and lethality in liver cancer cells. Cancer Lett. 2018; 421:82–93. https://doi.org/10.1016/j.canlet.2018.02.021 [PubMed]
- 71. Vásquez V, Krieg M, Lockhead D, Goodman MB. Phospholipids that contain polyunsaturated fatty acids enhance neuronal cell mechanics and touch sensation. Cell Rep. 2014; 6:70–80. https://doi.org/10.1016/j.celrep.2013.12.012 [PubMed]
- 72. Nandakumar M, Tan MW. Gamma-linolenic and stearidonic acids are required for basal immunity in caenorhabditis elegans through their effects on p38 MAP kinase activity. PLoS Genet. 2008; 4:e1000273. https://doi.org/10.1371/journal.pgen.1000273 [PubMed]
- 73. Shmookler Reis RJ, Xu L, Lee H, Chae M, Thaden JJ, Bharill P, Tazearslan C, Siegel E, Alla R, Zimniak P, Ayyadevara S. Modulation of lipid biosynthesis contributes to stress resistance and longevity of C. Elegans mutants. Aging (Albany NY). 2011; 3:125–47. https://doi.org/10.18632/aging.100275 [PubMed]
- 74. Yuan Y, Kadiyala CS, Ching TT, Hakimi P, Saha S, Xu H, Yuan C, Mullangi V, Wang L, Fivenson E, Hanson RW, Ewing R, Hsu AL, et al. Enhanced energy metabolism contributes to the extended life span of calorie-restricted caenorhabditis elegans. J Biol Chem. 2012; 287:31414–26. https://doi.org/10.1074/jbc.M112.377275 [PubMed]
- 75. Evan GI, Vousden KH. Proliferation, cell cycle and apoptosis in cancer. Nature. 2001; 411:342–48. https://doi.org/10.1038/35077213 [PubMed]
- 76. Lithgow GJ. Does anti-aging equal anti-microbial? Sci Aging Knowledge Environ. 2003; 2003:PE16. https://doi.org/10.1126/sageke.2003.25.pe16 [PubMed]
- 77. Garigan D, Hsu AL, Fraser AG, Kamath RS, Ahringer J, Kenyon C. Genetic analysis of tissue aging in caenorhabditis elegans: a role for heat-shock factor and bacterial proliferation. Genetics. 2002; 161:1101–12. [PubMed]
- 78. Abdullah SM, Defina LF, Leonard D, Barlow CE, Radford NB, Willis BL, Rohatgi A, McGuire DK, de Lemos JA, Grundy SM, Berry JD, Khera A. Long-term association of low-density lipoprotein cholesterol with cardiovascular mortality in individuals at low 10-year risk of atherosclerotic cardiovascular disease. Circulation. 2018; 138:2315–25. https://doi.org/10.1161/CIRCULATIONAHA.118.034273 [PubMed]
- 79. Mc Auley MT. The interplay between cholesterol metabolism and intrinsic ageing. Subcell Biochem. 2018; 90:99–118. https://doi.org/10.1007/978-981-13-2835-0_4 [PubMed]
- 80. Lee EY, Shim YH, Chitwood DJ, Hwang SB, Lee J, Paik YK. Cholesterol-producing transgenic caenorhabditis elegans lives longer due to newly acquired enhanced stress resistance. Biochem Biophys Res Commun. 2005; 328:929–36. https://doi.org/10.1016/j.bbrc.2005.01.050 [PubMed]
- 81. Zečić A, Dhondt I, Braeckman BP. The nutritional requirements of Caenorhabditis elegans. Genes Nutr. 2019; 14:15. https://doi.org/10.1186/s12263-019-0637-7 [PubMed]
- 82. Kurzchalia TV, Ward S. Why do worms need cholesterol? Nat Cell Biol. 2003; 5:684–88. https://doi.org/10.1038/ncb0803-684 [PubMed]
- 83. Mullaney BC, Ashrafi K. C. Elegans fat storage and metabolic regulation. Biochim Biophys Acta. 2009; 1791:474–78. https://doi.org/10.1016/j.bbalip.2008.12.013 [PubMed]
- 84. Ruiz M, Bodhicharla R, Svensk E, Devkota R, Busayavalasa K, Palmgren H, Ståhlman M, Boren J, Pilon M. Membrane fluidity is regulated by the C. Elegans transmembrane protein FLD-1 and its human homologs TLCD1/2. Elife. 2018; 7:e40686. https://doi.org/10.7554/eLife.40686 [PubMed]
- 85. Gao C, Gao Z, Greenway FL, Burton JH, Johnson WD, Keenan MJ, Enright FM, Martin RJ, Chu Y, Zheng J. Oat consumption reduced intestinal fat deposition and improved health span in caenorhabditis elegans model. Nutr Res. 2015; 35:834–43. https://doi.org/10.1016/j.nutres.2015.06.007 [PubMed]
- 86. Finley J, Sandlin C, Holliday D, Keenan MJ, Prinyawiwatkul W, Zheng J. Legumes reduced intestinal fat deposition in the Caenorhabditis elegans model system. Journal of Functional Foods. 2013; 5:1487–93.
- 87. Brandstädt S, Schmeisser K, Zarse K, Ristow M. Lipid-lowering fibrates extend C. Elegans lifespan in a NHR-49/PPARalpha-dependent manner. Aging (Albany NY). 2013; 5:270–75. https://doi.org/10.18632/aging.100548 [PubMed]
- 88. Teo E, Batchu KC, Barardo D, Xiao L, Cazenave-Gassiot A, Tolwinski N, Wenk M, Halliwell B, Gruber J. A novel vibration-induced exercise paradigm improves fitness and lipid metabolism of caenorhabditis elegans. Sci Rep. 2018; 8:9420. https://doi.org/10.1038/s41598-018-27330-3 [PubMed]
- 89. Zheng J, Greenway FL. Caenorhabditis elegans as a model for obesity research. Int J Obes (Lond). 2012; 36:186–94. https://doi.org/10.1038/ijo.2011.93 [PubMed]
- 90. Jones KT, Ashrafi K. Caenorhabditis elegans as an emerging model for studying the basic biology of obesity. Dis Model Mech. 2009; 2:224–29. https://doi.org/10.1242/dmm.001933 [PubMed]
- 91. Suh JM, Zeve D, McKay R, Seo J, Salo Z, Li R, Wang M, Graff JM. Adipose is a conserved dosage-sensitive antiobesity gene. Cell Metab. 2007; 6:195–207. https://doi.org/10.1016/j.cmet.2007.08.001 [PubMed]
- 92. McKay RM, McKay JP, Avery L, Graff JM. C elegans: a model for exploring the genetics of fat storage. Dev Cell. 2003; 4:131–42. https://doi.org/10.1016/s1534-5807(02)00411-2 [PubMed]
- 93. Olsen A, Vantipalli MC, Lithgow GJ. Checkpoint proteins control survival of the postmitotic cells in caenorhabditis elegans. Science. 2006; 312:1381–85. https://doi.org/10.1126/science.1124981 [PubMed]
- 94. Tan FC, Hutchison ER, Eitan E, Mattson MP. Are there roles for brain cell senescence in aging and neurodegenerative disorders? Biogerontology. 2014; 15:643–60. https://doi.org/10.1007/s10522-014-9532-1 [PubMed]
- 95. Sapieha P, Mallette FA. Cellular senescence in postmitotic cells: beyond growth arrest. Trends Cell Biol. 2018; 28:595–607. https://doi.org/10.1016/j.tcb.2018.03.003 [PubMed]
- 96. Micale G, Continella A, Ferro A, Giugno R, Pulvirenti A. GASOLINE: a cytoscape app for multiple local alignment of PPI networks. F1000Res. 2014; 3:140. https://doi.org/10.12688/f1000research.4537.2 [PubMed]
- 97. Kanehisa M, Furumichi M, Tanabe M, Sato Y, Morishima K. KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 2017; 45:D353–61. https://doi.org/10.1093/nar/gkw1092 [PubMed]
- 98. Sun T, Wu H, Cong M, Zhan J, Li F. Meta-analytic evidence for the anti-aging effect of hormesis on Caenorhabditis elegans. Aging (Albany NY). 2020; 12:2723–46. https://doi.org/10.18632/aging.102773 [PubMed]
- 99. Ermolaeva MA, Schumacher B. Insights from the worm: the C. Elegans model for innate immunity. Semin Immunol. 2014; 26:303–09. https://doi.org/10.1016/j.smim.2014.04.005 [PubMed]
- 100. Kvell K, Cooper EL, Engelmann P, Bovari J, Nemeth P. Blurring borders: innate immunity with adaptive features. Clin Dev Immunol. 2007; 2007:83671. https://doi.org/10.1155/2007/83671 [PubMed]
- 101. Penkov S, Mitroulis I, Hajishengallis G, Chavakis T. Immunometabolic crosstalk: an ancestral principle of trained immunity? Trends Immunol. 2019; 40:1–11. https://doi.org/10.1016/j.it.2018.11.002 [PubMed]
- 102. Netea MG, Joosten LA, Latz E, Mills KH, Natoli G, Stunnenberg HG, O’Neill LA, Xavier RJ. Trained immunity: a program of innate immune memory in health and disease. Science. 2016; 352:aaf1098. https://doi.org/10.1126/science.aaf1098 [PubMed]
- 103. Sorathia N, Rajadhyaksha MS. Caenorhabditis elegans: a model for studying human pathogen biology. Recent Pat Biotechnol. 2016; 10:217–25. https://doi.org/10.2174/1872208310666160727151853 [PubMed]
- 104. Sifri CD, Begun J, Ausubel FM. The worm has turned—microbial virulence modeled in caenorhabditis elegans. Trends Microbiol. 2005; 13:119–27. https://doi.org/10.1016/j.tim.2005.01.003 [PubMed]
- 105. Kim W, Hendricks GL, Lee K, Mylonakis E. An update on the use of C. Elegans for preclinical drug discovery: screening and identifying anti-infective drugs. Expert Opin Drug Discov. 2017; 12:625–33. https://doi.org/10.1080/17460441.2017.1319358 [PubMed]
- 106. Ewbank JJ, Zugasti O. C. Elegans: model host and tool for antimicrobial drug discovery. Dis Model Mech. 2011; 4:300–04. https://doi.org/10.1242/dmm.006684 [PubMed]
- 107. Komura T, Ikeda T, Hoshino K, Shibamura A, Nishikawa Y. Caenorhabditis elegans as an alternative model to study senescence of host defense and the prevention by immunonutrition. Adv Exp Med Biol. 2012; 710:19–27. https://doi.org/10.1007/978-1-4419-5638-5_3 [PubMed]
- 108. Papp D, Csermely P, Sőti C. A role for SKN-1/nrf in pathogen resistance and immunosenescence in caenorhabditis elegans. PLoS Pathog. 2012; 8:e1002673. https://doi.org/10.1371/journal.ppat.1002673 [PubMed]
- 109. Youngman MJ, Rogers ZN, Kim DH. A decline in p38 MAPK signaling underlies immunosenescence in caenorhabditis elegans. PLoS Genet. 2011; 7:e1002082. https://doi.org/10.1371/journal.pgen.1002082 [PubMed]
- 110. Solana R, Tarazona R, Gayoso I, Lesur O, Dupuis G, Fulop T. Innate immunosenescence: effect of aging on cells and receptors of the innate immune system in humans. Semin Immunol. 2012; 24:331–41. https://doi.org/10.1016/j.smim.2012.04.008 [PubMed]
- 111. Leong DP, Teo KK, Rangarajan S, Lopez-Jaramillo P, Avezum A
Jr , Orlandini A, Seron P, Ahmed SH, Rosengren A, Kelishadi R, Rahman O, Swaminathan S, Iqbal R, et al, and Prospective Urban Rural Epidemiology (PURE) Study investigators. Prognostic value of grip strength: findings from the prospective urban rural epidemiology (PURE) study. Lancet. 2015; 386:266–73. https://doi.org/10.1016/S0140-6736(14)62000-6 [PubMed] - 112. Brito LB, Ricardo DR, Araújo DS, Ramos PS, Myers J, Araújo CG. Ability to sit and rise from the floor as a predictor of all-cause mortality. Eur J Prev Cardiol. 2014; 21:892–98. https://doi.org/10.1177/2047487312471759 [PubMed]
- 113. Dues DJ, Andrews EK, Schaar CE, Bergsma AL, Senchuk MM, Van Raamsdonk JM. Aging causes decreased resistance to multiple stresses and a failure to activate specific stress response pathways. Aging (Albany NY). 2016; 8:777–95. https://doi.org/10.18632/aging.100939 [PubMed]
- 114. Bajorat J, Oberacker T, Ziola S, Edgar BA, Gülow K, Krammer PH. AF-1 a novel regulator of the redox equilibrium during aging. Free Radic Biol Med. 2014 (Suppl 1); 75:S22–23. https://doi.org/10.1016/j.freeradbiomed.2014.10.737 [PubMed]
- 115. Siddiqui S, Fang M, Ni B, Lu D, Martin B, Maudsley S. Central role of the EGF receptor in neurometabolic aging. Int J Endocrinol. 2012; 2012:739428. https://doi.org/10.1155/2012/739428 [PubMed]
- 116. Xu MF, Zhou H, Hu CY, Liang YQ, Hu L, Chen D. The mechanisms of EGFR in the regulation of axon regeneration. Cell Biochem Funct. 2014; 32:101–05. https://doi.org/10.1002/cbf.2977 [PubMed]
- 117. Yu S, Driscoll M. EGF signaling comes of age: promotion of healthy aging in C. Elegans. Exp Gerontol. 2011; 46:129–34. https://doi.org/10.1016/j.exger.2010.10.010 [PubMed]
- 118. Marck A, Berthelot G, Foulonneau V, Marc A, Antero-Jacquemin J, Noirez P, Bronikowski AM, Morgan TJ, Garland T
Jr , Carter PA, Hersen P, Di Meglio JM, Toussaint JF. Age-related changes in locomotor performance reveal a similar pattern for caenorhabditis elegans, mus domesticus, canis familiaris, equus caballus, and homo sapiens. J Gerontol A Biol Sci Med Sci. 2017; 72:455–63. https://doi.org/10.1093/gerona/glw136 [PubMed] - 119. Hoffmann R, Valencia A. A gene network for navigating the literature. Nat Genet. 2004; 36:664. https://doi.org/10.1038/ng0704-664 [PubMed]
- 120. Oesper L, Merico D, Isserlin R, Bader GD. WordCloud: a cytoscape plugin to create a visual semantic summary of networks. Source Code Biol Med. 2011; 6:7. https://doi.org/10.1186/1751-0473-6-7 [PubMed]
- 121. Adler P, Kolde R, Kull M, Tkachenko A, Peterson H, Reimand J, Vilo J. Mining for coexpression across hundreds of datasets using novel rank aggregation and visualization methods. Genome Biol. 2009; 10:R139. https://doi.org/10.1186/gb-2009-10-12-r139 [PubMed]
- 122. Raudvere U, Kolberg L, Kuzmin I, Arak T, Adler P, Peterson H, Vilo J. g:Profiler: a web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Res. 2019; 47:W191–98. https://doi.org/10.1093/nar/gkz369 [PubMed]


