Introduction
Copper (Cu), as an essential element, is the main metal used in bronze and brass [1–3]. It plays an indispensable role in both animals and plants [4] and also is widely used in pipes, wires, cooking utensils, ornaments and jewellery [5, 6]. Wide industrial use of Cu leads to increased copper pollution in the environment, enhancing the risk of copper toxicity in humans and animals [7–10]. Usually, workers who are continuously exposed to copper face high risk of lung damage, and lower respiratory function is associated with higher serum copper concentrations [11, 12]. It has been reported that oral overdose of copper sulphate (CuSO4) can repress body development and impair organic function [13]. Wistar rats fed on diets contaminated with high CuSO4 levels exhibit growth depression [14].
Oxidative stress results from an imbalance between the oxidative and antioxidant systems of cells and tissues, and is the result of excessive production of oxidative free radicals and related reactive oxygen species (ROS) [15]. It has been identified that high Cu levels have toxic effects including ROS generation leading to DNA damage [16], oxidative damage to biological molecules [17], and peroxidation of cell membrane lipids [18]. In addition, studies have shown that Cu overload leads to oxidative stress, and subsequently damages proteins, lipids and nucleic acids [19–23]. In vivo studies, high dietary copper can inhibit the serum, hepatic [24–27] and nephritic [28] antioxidase activities in ducklings, and induce oxidative stress in the brain [29, 30] and spleen [31] of chickens. Also, it has been reported that intradermal injection of 2% CuSO4 solution in the chicken can increase oxygen-derived free radicals, result in tissue damage and increase vascular permeability [32]. In in vitro studies, Cu and Cu compounds can cause oxidative stress via the production of ROS in human epithelial lung (A549) cells [33] and brain microvascular endothelial cells [34].
Additionally, increased intracellular ROS as a signal of oxidative stress, can break single or double strands of DNA and activate DNA-dependent protein kinase, leading to apoptosis [35, 36]. Reports of in vivo studies have shown that high Cu levels induce the higher percentage of apoptotic cells of the lymphoid organs [37] and kidney [38] in ducklings and lymphoid organs [39], kidney [40], and liver [41] in chickens. The findings of Guo et al. (2017) indicated that Cu can induce oxidative stress and apoptosis in the White Shrimp (Litopenaeus vannamei) [42]. Besides, it has been reported that Cu induces apoptosis by increasing the expression levels of cysteine aspartate specific protease-3(caspase-3), cysteinyl aspartate-specific protease9 (caspase-9), and Bcl-2 associated X protein(Bax) in the rat kidney and liver [43, 44]. In in vitro studies, the findings have indicated that Cu can induce mitochondrial dysfunction in primary culture of chicken hepatocytes [45] and apoptosis in human skin melanoma A-375 cell line [46] and Aedes C6/36 cells [47].
Inflammation is a defensive response to stimulation and usually is beneficial for animals and human beings [48]: however, continued inflammation can cause damage to the body [49]. Inflammation can be triggered by a variety of factors, such as pathogens, damaged cells, toxic compounds, and irradiation [50]. In addition, oxidative damage can lead to cell metabolism disorders, which accelerates the production of inflammation [51]. In in vivo studies, the study has investigated the potential synergistic effects of chronic exposure to Cu in promoting inflammatory and oxidative events in mouse brains [52]. Brand et al. (2019) has found that inhalation of fumes containing Cu can induce asymptomatic systemic inflammation [53]. Pharyngeal aspiration of CuO nanoparticles causes lung inflammation and lung injuries in Wistar rats [54], however, no reports are focused on CuSO4-induced inflammation in the human and animal lung.
The aforementioned studies suggest that oxidative stress, apoptosis, and inflammatory responses are the three main mechanisms involved in Cu-induced toxic effects on organs, tissues and cells, however, there are few studies focused on the relationship between Cu and oxidative stress, apoptosis, inflammatory responses in animal and human lungs. Therefore, the present study aims to assess pulmonary toxicity via oxidative stress, apoptosis and inflammatory responses after CuSO4 intake in mice by using methods of histopathology, qRT-PCR, and ELISA, flow cytometry, and western blot assay.
Results
Histopathological changes in the lung
Figure 1 shows the dose- and time-dependent lesions in lungs at the three CuSO4-treated groups. Histopathologically, alveolar walls were thickened in varying degrees, mainly due to inflammatory cells infiltration and capillary congestion. The aforementioned histopathological lesions were not observed in the control group.
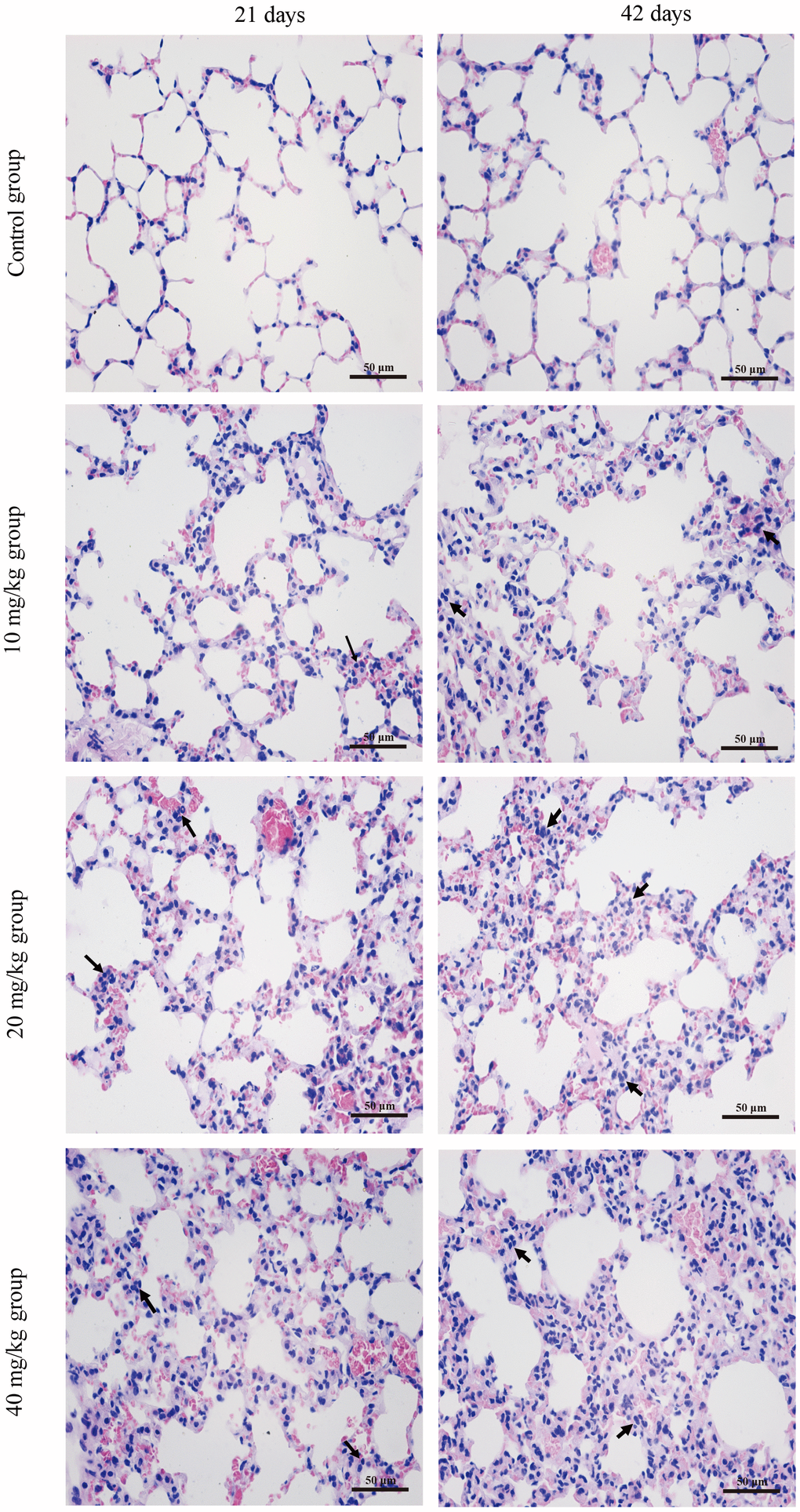
Figure 1. Histopathological changes in the lung at 21 and 42 days of the experiment. (H&E ×400). Control group at 21 and 42 days: no changes are observed. 10 mg/kg group at 21 days: the inflammatory cells (↑) are obviously observed in alveolar walls. 20 mg/kg group at 21 days: the alveolar walls are slightly thickened with inflammatory cell infiltration (↑) 40 mg/kg group at 21 days: the alveolar walls are obviously thickened with inflammatory cell infiltration (↑). 10 mg/kg group at 42 days: the alveolar walls are slightly thickened with inflammatory cell infiltration (↑). 20 mg/kg group at 42 days: the alveolar walls are thickened with inflammatory cell infiltration (↑). 40 mg/kg group at 42 days: the alveolar walls are markedly thickened with inflammatory cell infiltration (↑).
Changes of ROS production levels and Cu contents in the lung
The ROS production levels were increased (p < 0.05 or p < 0.01) in the 20 and 40 mg/kg groups at day 21 of the experiment and in the three CuSO4-treated groups at day 42 of the experiment when compared to the control group. The Cu contents in the lung were increased (p < 0.05 or p < 0.01) in the 20 and 40 mg/kg group at day 42 of the experiment when compared to the control group. The results are illustrated in Figure 2.
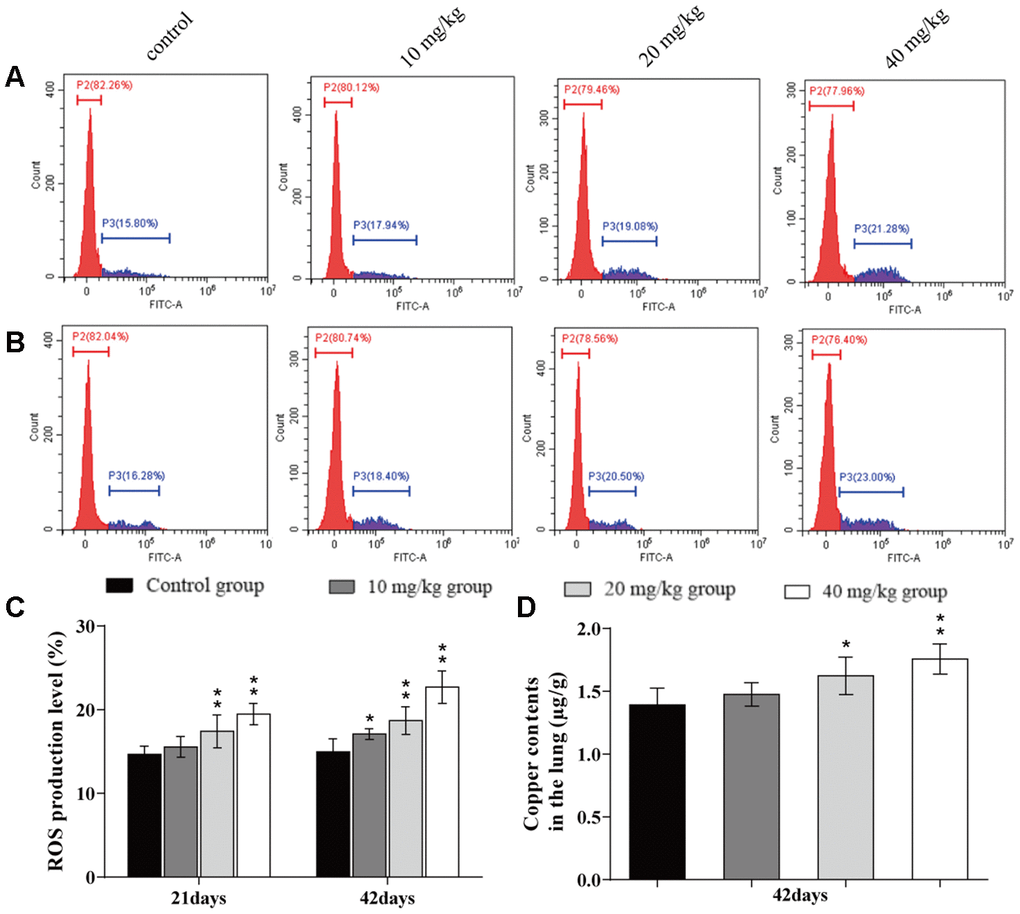
Figure 2. Changes of ROS production levels and Cu contents in the lung. (A) ROS production levels in the lung by flow cytometry at 21 days of the experiment. (B) ROS production levels in the lung by flow cytometry at 42 days. (C) ROS production levels in the lung. (D) Cu contents in the lung at 42 days of the experiment. Data are presented with the mean± standard deviation (n=8). *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
Changes of oxidative damage parameters in the lung
The superoxide dismutase (SOD) activities were significantly reduced (p < 0.05 or p < 0.01) in the 40 mg/kg group at 21 and 42 days of the experiment and in the 20 mg/kg group at 42 days in comparison to the control group. The activities of Catalase (CAT), glutathione peroxidase (GSH-Px), Glutathione (GSH) contents, and the GSH/GSSG ratio were significantly decreased (p < 0.01) in the three CuSO4-treated groups at 21 and 42 days. The oxidised glutathione (GSSG) content was significantly increased (p < 0.05 or p < 0.01) in the 20 and 40 mg/kg groups at days 21 and 42. The mRNA expression levels of antioxidant enzymes (GSH-Px, CAT, MnSOD, and ZnSOD) were significantly lower (p < 0.05 or p < 0.01) in the 40 mg/kg group at day 21 and in the three CuSO4-treated groups at day 42 than in the control group. The results are shown in Figure 4.
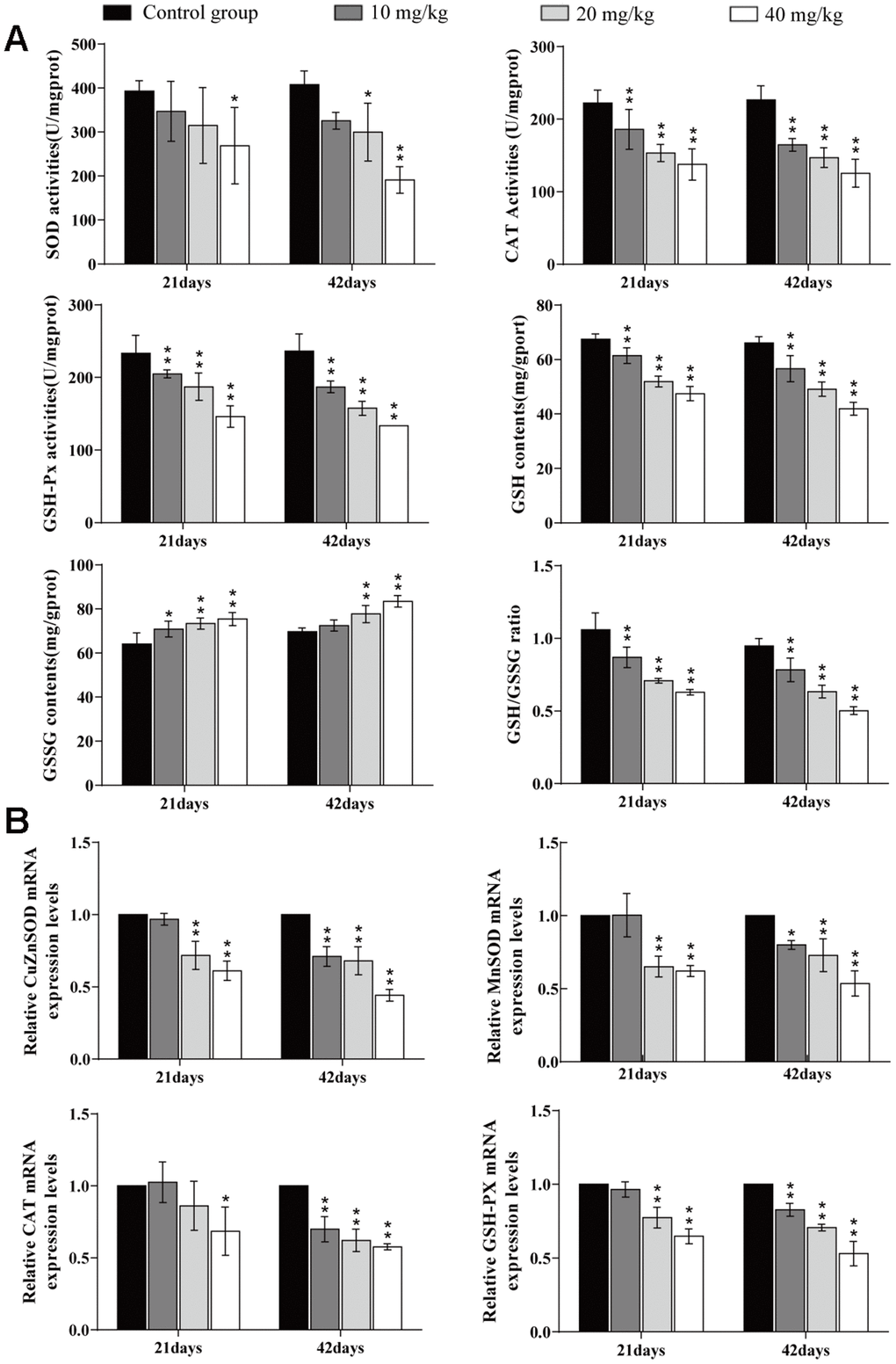
Figure 4. Changes of oxidative damage parameters in the lung. (A) Changes of antioxidant enzyme activities, GSH and GSSG contents, and GSG/GSSG ratio in the lung at 21 and 42 days of the experiment. (B) Changes of mRNA expression levels of antioxidant enzymes in the lung. Data are presented with the mean± standard deviation (n=8). *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
Changes of apoptosis percentages in the lung
The apoptotic percentages were elevated (p < 0.05 or p < 0.01) in the three CuSO4-treated groups at days 21 and 42 of the experiment when compared to the control group, as shown in Figure 5.
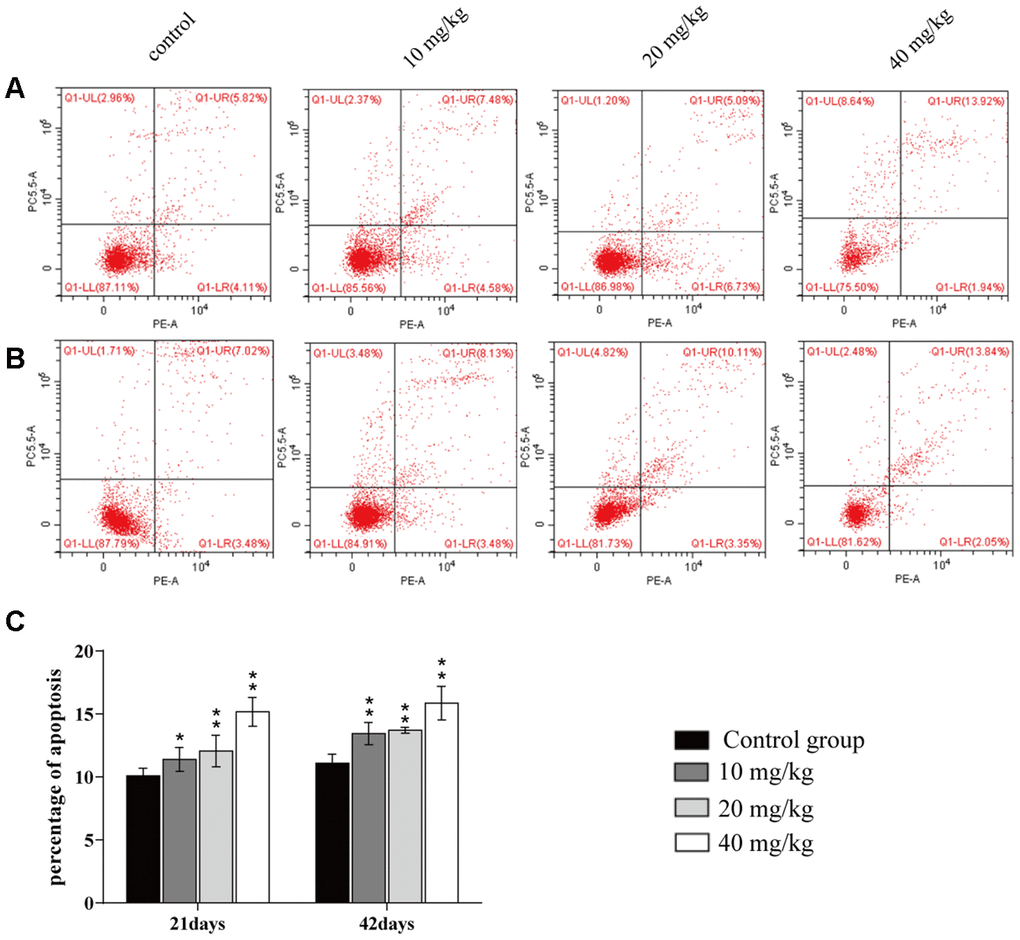
Figure 5. Changes of apoptosis percentages in the lung. (A) Apoptosis in the lung at 21 days of the experiment. (B) Apoptosis in the lung at 42 days. (C) Percentage of apoptosis in the lung. Data are presented with the mean± standard deviation (n=8). *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
Changes of MPO activities and PGE2 contents in the lung
The myeloperoxidase (MPO) activities and prostaglandin E2 (PGE2) contents were significantly higher (p < 0.01) in the 20 and 40 mg/kg groups at days 21 and 42 of the experiment than in the control group, as shown in Figure 7.
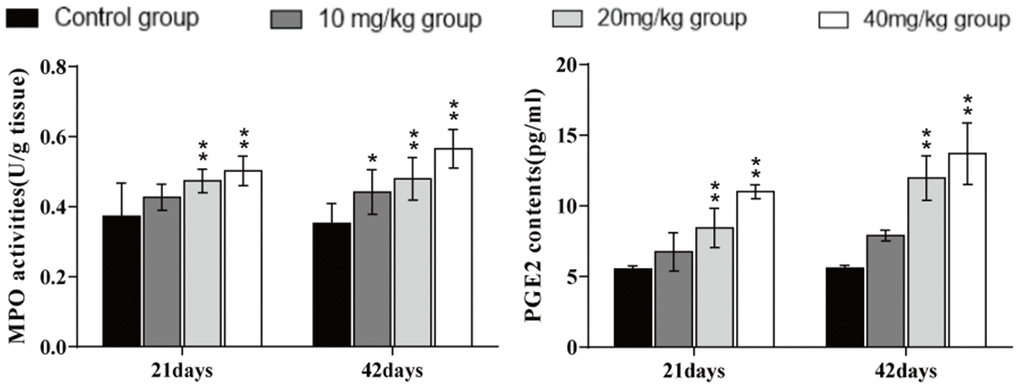
Figure 7. Changes in MPO activities and PGE2 contents in the lung at 21 and 42 days of the experiment. Data are presented with the mean± standard deviation (n=8). *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
Changes of mRNA expression and protein expression levels of NF-κB and IκB in the lung
The mRNA and protein expression levels of nuclear factor-kappa B (NF-κB) were significantly higher (p < 0.01 or p < 0.05) in the 20 and 40 mg/kg groups at 21 and 42 days of the experiment than in the control group. The mRNA and protein expression levels of inhibitory kappa B (IκB) were significantly decreased (p < 0.01 or p < 0.05) in the 20 and 40 mg/kg groups at 21 and 42 days when compared to the control group. The results are shown in Figure 8.
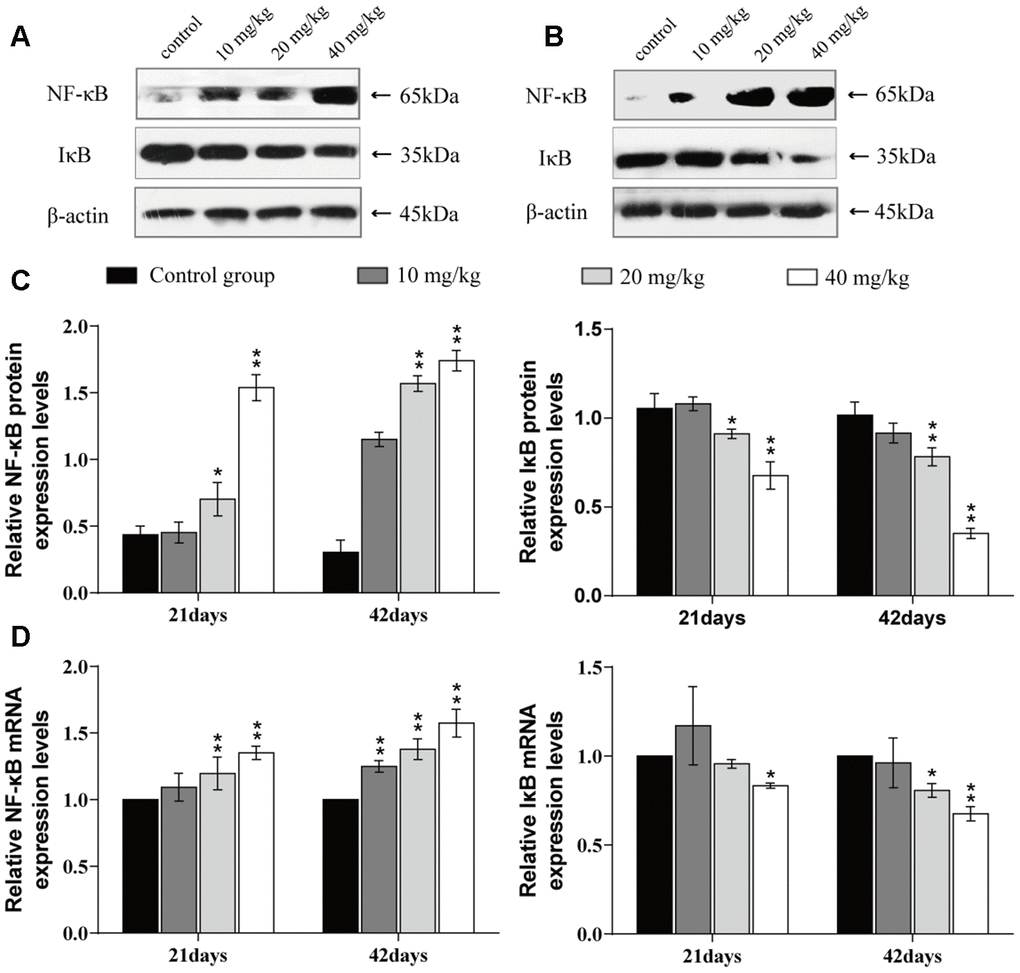
Figure 8. Changes of mRNA expression and protein expression levels of NF-κB and IκB in the lung at 21 and 42 days of the experiment. (A) Western blot assay of NF-κB and IκB at 21 days. (B) Western blot assay of NF-κB and IκB at 42 days. (C) The relative protein expression levels of NF-κB and IκB. (D) The relative mRNA expression levels of NF-κB and IκB. Data are presented with the mean± standard deviation (n=8). *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
Changes in mRNA expression and protein expression levels of inflammatory cytokines in the lung
The mRNA expression levels and protein expression levels of cyclooxygenase-2 (COX-2), interleukin-1β (IL-1β), interleukin-8 (IL-8), and interleukin-6 (IL-6) were significantly increased (p < 0.01 or p < 0.05) in the 20 and 40 mg/kg groups in comparison to the control group at 21 and 42 days of the experiment.
The mRNA and protein expression levels of tumour necrosis factor-α (TNF-α) were significantly higher (p < 0.01) in the 40 mg/kg group at 21 days of the experiment and in the 20 and 40 mg/kg groups at 42 days than in the control group. The mRNA expression levels and protein expression levels of interleukin-2 (IL-2), interleukin-4 (IL-4), and interleukin-10 (IL-10) were significantly decreased (p < 0.01 or p < 0.05) in the 20 and 40 mg/kg groups at 21 days and in the three CuSO4-treated groups at 42 days when compared to in the control group. The results are shown in Figures 9 and 10.
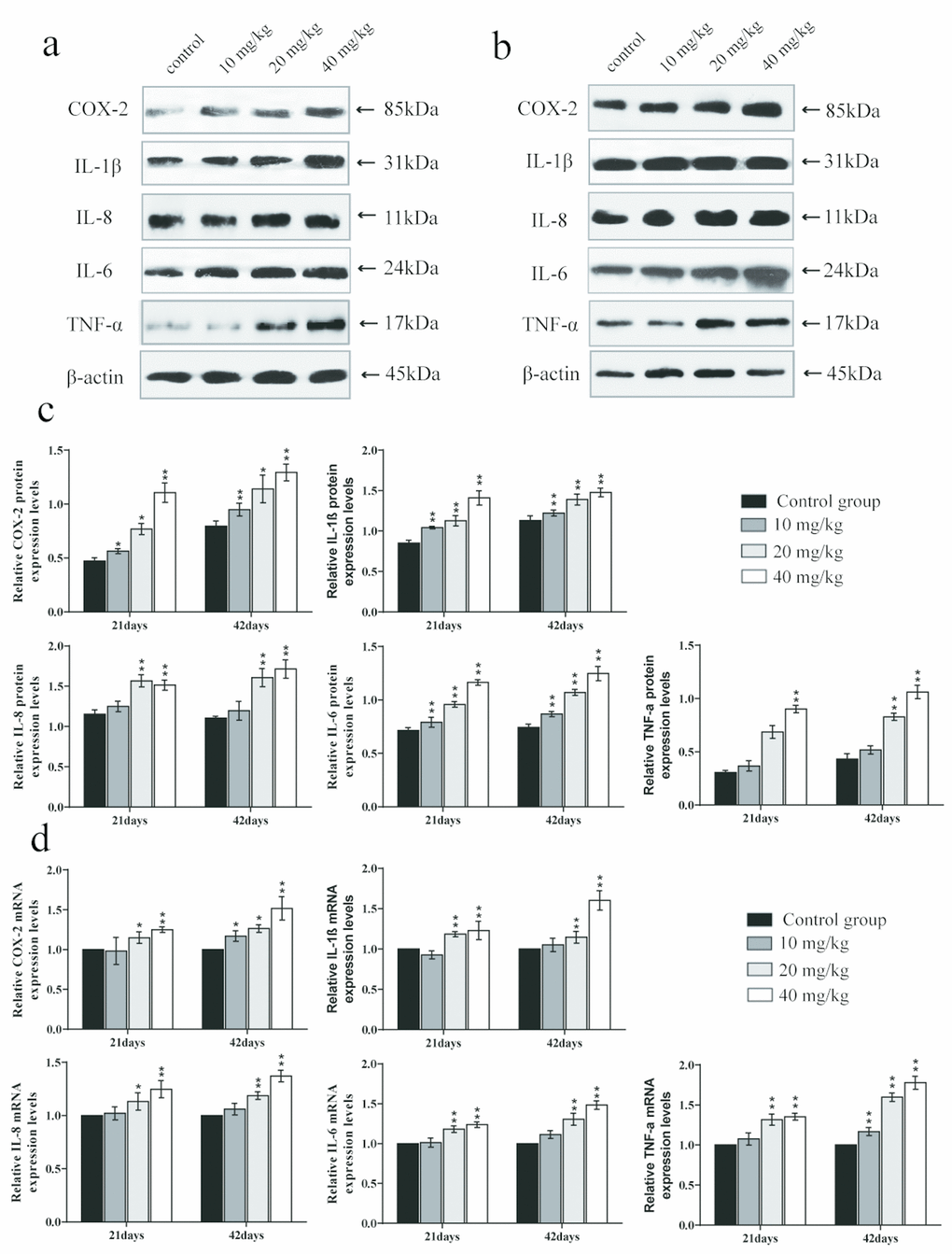
Figure 9. Changes of mRNA expression levels and protein expression levels of pro-inflammatory cytokines in the lung at 21 and 42 days of the experiment. (A) Western blot assay of pro-inflammatory cytokines at 21 days. (B) Western blot assay of pro-inflammatory cytokines at 42 days. (C) The relative protein expression levels of pro-inflammatory cytokines. (D) The relative mRNA expression levels of pro-inflammatory cytokines. Data are presented with the mean± standard deviation (n=8). *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
Figure 10. Changes of mRNA expression levels and protein expression levels of anti-inflammatory cytokines in the lung at 21 and 42 days of the experiment. (A) Western blot assay of anti-inflammatory cytokines at 21 days. (B) Western blot assay of anti-inflammatory cytokines at 42 days. (C) The relative protein expression levels of anti-inflammatory cytokines. (D) The relative mRNA expression levels of anti-inflammatory cytokines. Data are presented with the mean± standard deviation (n=8). *p < 0.05, compared with the control group; **p < 0.01, compared with the control group.
Discussion
In summary, this study firstly found that CuSO4 could induce oxidative stress, apoptosis, and inflammatory responses in the mouse lung.
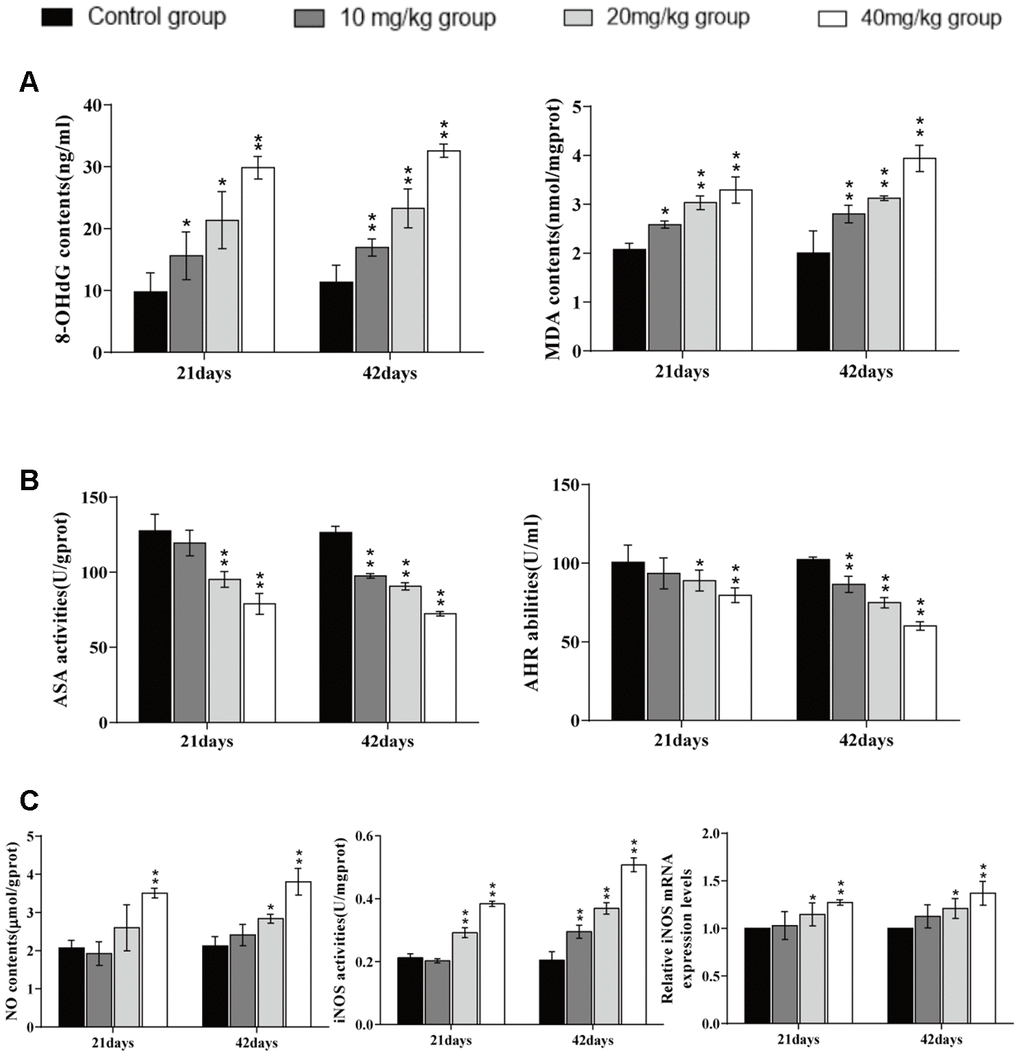
We observed histopathological changes induced by CuSO4, alveolar walls were thickened in varying degrees with the dose- and time-dependent lesions, which mainly due to inflammatory cells infiltration and capillary congestion. Also, the pulmonary injuries were strongly consistent with the accumulation of Cu in the lung. It has been suggested that one possible molecular mechanism involved in the Cu toxicity is the formation of ROS, which leads to lesions via oxidative damage [55]. Our results showed that CuSO4 induced the production of ROS in the pulmonary tissue, and then excessive ROS caused oxidative damage to DNA (increased 8-OHdG contents) and lipid peroxidation (increased MDA contents). To reveal causes of the increased ROS, we tested the ASA and AHR abilities. The results showed that ASA and AHR abilities were reduced, which indicated that CuSO4 decreased the capacity of the lung to scavenge ROS and disturbed the dynamic balance between ROS production and elimination of ROS. Once the antioxidant function is impaired, free radicals (including oxygen free radicals, hydroxyl free radicals and nitrogen free radicals, etc.) are accumulated, which then causes oxidative stress in the body [56]. Kubbw et al. has found that rats consuming the zinc-deficient (0.5 mg/kg), high-copper (over 15 mg/kg) diet have the lowest weight gain and have an increase in endogenous free radical production in lung [57]. We also found an increase in the NO contents, iNOS enzyme activities, and mRNA expression levels, leading to oxidative damage in the lung. NO is an important nitrogen free radical in the body, which can also induce inflammation [58].
The increased ROS may indicate that the functioning of the antioxidant defence system is weakened [59], including decreased antioxidant enzymes (SOD, CAT, and GSH-PX) activities and non-enzymatic scavenger (GSH) content. SOD and CAT are important antioxidant enzymes that play a major role in ROS clearance [60]. GSH is an important intracellular antioxidant in alveolar epithelial cells [61]. GSH-dependent enzymes such as GSH-PX can remove hydrogen peroxide by oxidising GSH to GSSG, but ROS can oxidise GSH into GSSG, too much GSSG will weaken the protective effect of GSH, and the dysfunction of GSH will also aggravate the damage to organs [62, 63]. In the present study, the results showed that CuSO4 decreased CAT, SOD, and GSH-Px activities, GSH contents, and also the GSH/GSSG ratio was decreased in the lung, which were consistent with the report that copper can enhance oxidative stress, reduce the activities of antioxidant enzymes (SOD and CAT) and decrease the contents of GSH in brain tissues of chicken [51]. Cinar et al. (2014) has found that oral intake of CuSO4 can induce oxidative stress by reducing the activity of enzymes such as CAT and SOD in broiler serum [64]. Other studies have shown that the reduced intracellular GSH is associated with activation of NF-κB [65]. The further to explore the molecular mechanism of CuSO4-decreased antioxidant enzyme activities, we also detected mRNA expression levels of antioxidant enzymes in the lung. The results indicated that the mRNA expression levels of CuZnSOD, MnSOD, CAT, and GSH-PX were lower in the CuSO4-treated groups than in the control group, which were consistent with the reduction of these antioxidant enzyme activities. Decreased activities and mRNA expression levels of antioxidant enzymes promote oxidative stress, which plays critical roles in the pathogenesis of various diseases [66, 67].
In conclusion, our results showed that CuSO4 induces oxidative stress in the lung by promoting excessive ROS production and increased NO contents, and reducing antioxidant enzyme activities, GSH contents, and the GSH/GSSG ratio, which contribute to pulmonary lesions and dysfunction. Also, this study is the first to be focused on CuSO4-induced oxidative stress in the lung.
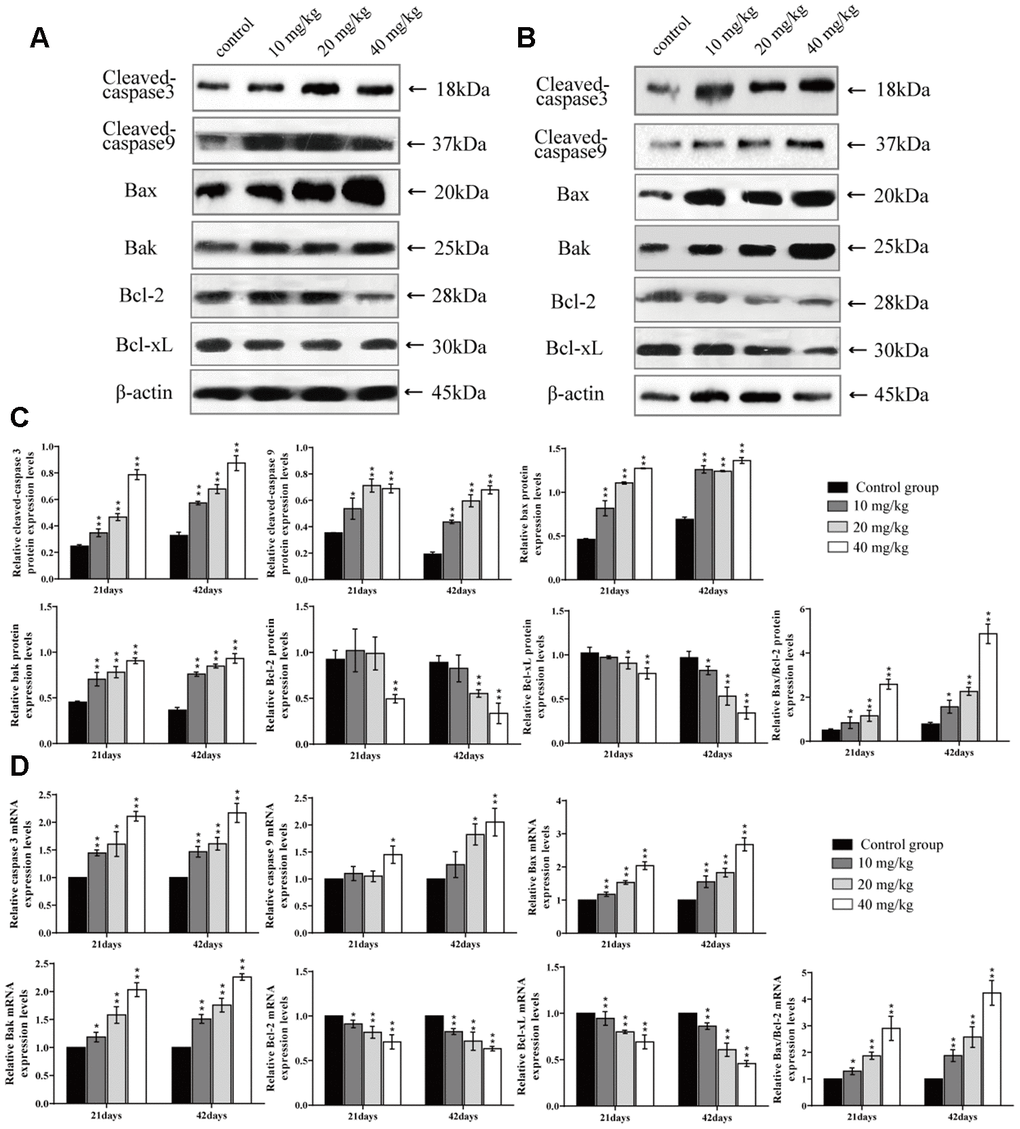
It has been known that ROS and oxidative stress play important roles in the early stages of apoptosis [68]. Next, we explored whether, or not, apoptosis was also involved in the mechanism of CuSO4-cuased toxicology in the lung. After CuSO4 treatment, the results indicated that the percentage of apoptosis was increased in the CuSO4-treated groups at 21 and 42 days. Apoptosis is a process controlled by multiple genes, such as bcl-2 family and caspase family, which are conserved among species [69]. As a group of important apoptotic regulators, the bcl-2 family can indirectly regulate caspases in relevant apoptotic pathways [70, 71]. Results in the present study showed that CuSO4 could cause a significant decrease in the protein expression levels and mRNA expression levels of anti-apoptotic proteins (Bcl-2 and Bcl-xL), while the protein and mRNA expression levels of pro-apoptotic proteins (Bax and Bak) were significantly increased in the lung. Hsien et al. (2008) has also observed that Cu-induced apoptosis was accompanied by the increased Bax and Bak expression levels and a decreased Bcl-2 expression level in neuroblastoma cells [72]. We also observed an increased Bax/Bcl-2 ratio in this research. Lee et al. (2008) has indicated that the increase of the ratio between Bax and Bcl-2 is the key point in the occurrence of apoptosis [73]. This increased Bax/Bcl-2 ratio leads to changes in mitochondrial membrane permeability and the release of apoptotic proteins, such as Cyt c [74]. In addition, excessive ROS production with decreasing concomitant in GSH contents also leads to mitochondrial membrane permeability and induction of apoptotic cell death in cultured cells [75]. Cyt c can cleave and activate caspase-9, which then activates downstream caspase-3 and cell apoptosis [76]. In this study, CuSO4 increased mRNA expression levels of caspase-9 and -3 and protein expression levels of cleaved-caspase-9 and -3, which contributed to apoptotic occurrence. The reports have also found that apoptosis is caused by copper-induced ROS formation in MCF7 cells [77], rat liver [78], and chicken intestine [79]. In summary, the present study was the first in which CuSO4-induced apoptosis was observed in the lung, and this was accompanied by decreasing Bcl-2, Bcl-xL mRNA expression levels and protein expression levels, and increasing Bax, Bak, caspase-3, and caspase-9 mRNA expression levels and protein expression levels, and the Bax/Bcl-2 ratio.
Inflammation is also a crucial toxicological mechanism of copper [80]. Myeloperoxidase (MPO) activity is used to measure the inflammatory degree in tissues and organs, and our results suggested that MPO activities were increased, which was consistent with the increased MPO arising from excessive accumulation of ROS [81]. This study is the first to investigate the mechanism of NF-κB activation in pulmonary inflammation by CuSO4 intake in mice. NF-κB is considered to be a major cellular transcription factor in inflammatory processes, and NF-κB activation has been identified as an important feature of inflammatory pulmonary diseases [82, 83]. Normally, NF-κB and IκB combine to form an inactive NF-κB-IκB complex [84]. In this study, The expression levels of IκB mRNA and protein were decreased, and the expression levels of NF-κB mRNA and NF-κB protein were increased, which demonstrated that CuSO4-induced IκB degradation promoted the activation of NF-κB. Concurrently, we observed that CuSO4 increased mRNA and protein expression levels of pro-inflammatory cytokines, such as TNF-α, COX-2, IL-6, IL-1β, and IL-8, which was in line with activation of the transcription factor NF-κB pathway. Our results are consistent with the report that copper can increase the expression levels of NF-κB, COX-2, IL-1β, TNF-α, IL-1β, and IL-8 mRNA and protein and exacerbate the damage and oxidative stress in zebrafish larvae tissues [85, 86].
It is well known that COX-2 is a classic proinflammatory cytokine, and plays an important role in the regulation of pulmonary inflammation [87]. As a downstream product of COX-2, PGE2 can not only induce the aggregation of inflammatory cells but also accelerate the process of peripheral inflammatory response induced by harmful stimuli [88]. In the present study, we observed that the PGE2 contents as well as the COX-2 mRNA and protein expression levels were significantly increased. Lu et al. (2008) has reported that intake of copper can up-regulate the expression of inflammation-related genes in the mouse brain, such as COX-2 and TNF-α [89]. iNOS, a proinflammatory marker in lung tissue, plays an essential role in exacerbating inflammation and can catalyse the production of NO, while excessive NO can increase the permeability of blood vessels and promote the infiltration of inflammatory cells [90–93]. In the present study, we found that CuSO4 increased the activities and mRNA expression levels of iNOS, and NO contents, which promoted lung inflammation.
On the contrary, IL-2, IL-4, and IL-10 are recognised as cytokines able to mediate immune suppression, which can inhibit the production of Th1 cells and reduce the release of pro-inflammatory cytokine [94, 95]. In the stage of acute pulmonary inflammation, IL-10 has significant anti-lymphocytes and neutrophilic infiltration [96]. Our results showed that CuSO4 decreased the mRNA and protein expression levels of IL-2, IL-4, and IL-10.
In conclusion, CuSO4 can increase MPO activities, activate the NF-κB pathway, and down-regulate anti-inflammatory cytokines, indicating that imbalance between pro-inflammatory and anti-inflammatory cytokine induces inflammatory responses in the lung. CuSO4-induced inflammatory responses contribute to pulmonary lesions and dysfunction.
Materials and Methods
Experimental animals and diets
A total of 240 four-week-old ICR mice (half male and half female) obtained from the Chengdu Dossy Experimental Animals were used in the present study. The animals were housed in separate polypropylene cages, and diet and water were provided ad libitum throughout the experiment. The mice were fed a full-price diet provided by Dossy. After a week of rest and acclimatisation, mice were equally divided into four different groups (each with n = 60). The control group was given orally distilled water only, groups I, II, and III were given CuSO4 orally at the dose of 10, 20, and 40 mg/kg body mass, respectively. Mice were administered their respective doses daily by gavage for 42 consecutive days, and the gavage volume was 1 ml/100 g body mass.
The animal protocols and all procedures of the experiment were performed in compliance with the laws and guidelines of Sichuan Agricultural University Animal Care and Use Committee.
Histopathological observation of the lung
At 21 and 42 days, eight mice (male female half) in each group were humanely killed and their lungs were removed, fixed in 4% paraformaldehyde solution, dehydrated with increasing concentrations of ethanol, cleared with xylene and embedded in paraffin. And then lungs were serial sectioned at 5μm thickness, stained with hematoxylin and eosin (H&E), and observed by optical microscopy.
Determination of pulmonary Cu contents
At 42 days of the experiment, eight mice (male female half) in each group were humanely killed and their lungs were removed, weighed, dried, and collected for the determination of the Cu contents. The Cu contents in the lung were measured according to a reference method [97].
Determination of the oxidative and anti-oxidative parameters in the lung
At 21and 42 days of age during the experiment, eight mice (male female half) in each group were humanely sacrificed, and the lungs were immediately stored at 4 °C cold phosphate buffer saline in a chilled homogeniser, and centrifuged at 3000 rpm for 15 min. Thereafter, the supernatant was transferred into new Eppendorf tubes. The commercial kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China) and used to detect ASA (Cat. No. A052-1) and AHR (Cat. No. A018), MDA (Cat. No. A003-1), CAT (Cat. No. A007-1), T-SOD (Cat. No. A001-1), GSH-Px (Cat. No. A005), and GSH (Cat. No. A061-1) according to the manufacturer’s instructions. Pulmonary 8-OHdG levels were measured using ELISA according to the manufacturer’s instructions.
Detection of MPO, iNOS activities, and NO and PGE2 contents
At 21 and 42 days into the experiment, eight mice (half male, half female) in each group were humanely sacrificed, and the lungs were immediately stored at 4 °C cold phosphate buffer saline in a chilled homogeniser, and centrifuged at 3500 rpm for 10 min. Thereafter, the supernatant was transferred into new Eppendorf tubes. The commercial kits were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China) and used to detect MPO (Cat. No. A044), iNOS (Cat. No. A014-1), and NO (Cat. No. A013-2) according to the manufacturer’s instructions. Pulmonary PGE2 levels were measured using ELISA according to the manufacturer’s instructions.
Apoptosis and ROS analysis by flow cytometry
At 21and 42 days, eight mice (half male, half female) in each group were humanely sacrificed, and the lungs were immediately stored at 4 °C phosphate buffer saline, and then were cut up to make a cell suspension, which was filtered through a 350-mesh nylon screen. The cells were washed twice with ice-cold phosphate buffer saline (PBS, pH 7.2-7.4), and then suspended in PBS at a concentration of 1 × 106 cells/ml.
Thereafter, a total of 100 μL cell suspension were transfer into a 5 mL culture tube and centrifuged at 800g for 5 min for apoptosis testing. PE Annexin V and 7-aminoactinomycin (7-AAd) staining were used to dye the specimens. The mixture was gently shaken and then left in the dark for 15 minutes. Then 400 μl binding buffer was added to the tube, and the apoptosis rate of lung cells was detected and analysed by flow cytometry (FACS Calibur, BD, USA). The results were analysed using the Mod Fit LT for Mac V3.0 computer program.
Some 300 μL of the aforementioned cell suspensions were taken and transferred to another centrifuge tube, and stained with 10 μM DCFH- DA for 20 min at 37 °C for ROS testing. Then the cells were washed with PBS and centrifuged (600g, 5 min) once more. The supernatant was discarded, and cells were resuspended in 0.5 ml PBS and counted by flow cytometry (FACS Calibur, BD, USA).
Determination of mRNA expression levels of antioxidant enzymes, inflammatory cytokines, and apoptotic proteins in the lung by qRT-PCR
At 21 and 42 days, lungs of eight mice in each group (half male, half female) were removed, stored in liquid nitrogen, and then Total RNA was extracted with the RNAiso Plus, and reverse transcribed into cDNA by using the Prim-Script™ RT reagent Kit as per the manufacture’s specification. The gene sequences of CuZnSOD, MnSOD, CAT, GSH-PX, Caspase 3, Caspase 9, Bax, Bak, Bcl-2, Bcl-xL, NF-κB, IκB, COX-2, IL-1β, IL-8, IL-6, and TNF-α IL-2, IL-4 and IL-10 were retrieved from NCBI, and the primers of these genes (Tables 1–3) were synthesised by Sangon Biotech (Shanghai, China). β-actin of mice was chosen as the reference gene. qRT-PCR reaction conducted on a C1000 Thermal Cycler (BIO RAD, USA) by using the SYBR® Premix Ex TaqII (Takara, China) in accordance with the standard steps. All data output from the qRT-PCR experiments were analysed using the 2-ΔΔCT method.
Table 1. List of primers of the antioxidant enzymes in qRT-PCR analysis.
| Gene symbol | Accession number | Primer sequence (5'–3') | Product size | Tm(°C) |
| CuZn- SOD | NM205064 | F: CGCAGGTGCTCACTTTAATCC | 119bp | 57 |
| R: CTATTTCTACTTCTGCCACTCCTCC | ||||
| Mn-SOD | NM204211 | F: CACTCTTCCTGACCTGCCTTACG | 146bp | 57 |
| R: TTGCCAGCGCCTCTTTGTATT | ||||
| CAT | NM001031215 | F: CTGTTGCTGGAGAATCTGGGTC | 160bp | 61 |
| R: TGGCTATGGATGAAGGATGGAA | ||||
| GSH-Px | NM001277853 | F: TTGTAAACATCAGGGGCAAA | 140bp | 61 |
| R: TGGGCCAAGATCTTTCTGTAA |
Table 2. List of primers of the apoptotic genes in qRT-PCR analysis.
| Gene symbol | Accession number | Primer sequence (5′–3′) | Product size | Tm (°C) |
| caspase-3 | NM_009810 | F: ACATGGGAGCAAGTCAGTGG | 149bp | 60 |
| R: CGTCCACATCCGTACCAGAG | ||||
| caspase-9 | NM_015733 | F: GAGGTGAAGAACGACCTGAC | 103bp | 57 |
| R: AGAGGATGACCACCACAAAG | ||||
| Bax | NM_007527 | F: ATGCGTCCACCAAGAAGC | 163bp | 61 |
| R: CAGTTGAAGTTGCCATCAGC | ||||
| Bak | NM_007523 | F: CGCTACGACACAGAGTTCCA | 175bp | 60 |
| R: CACGCTGGTAGACGTACAGG | ||||
| Bcl-2 | NM_009741 | F: AGCCTGAGAGCAACCCAAT | 159bp | 59 |
| R: AGCGACGAGAGAAGTCATCC | ||||
| Bcl-xL | NM_009743 | F: TGTGGATCTCTACGGGAACA | 117bp | 59 |
| R: AAGAGTGAGCCCAGCAGAAC |
Table 3. List of primers of the inflammatory mediators in qRT-PCR analysis.
| Gene symbol | Accession number | Primer sequence (5′–3′) | Product size | Tm (°C) |
| NF-κB | NM205134 | F: CTGAAACTACTGATTGCTGCTGGA | 179bp | 62 |
| R: GCTATGTGAAGAGGCGTTGTGC | ||||
| IκB | NM204588 | F:TGAGGACGAGGACGATAAGC | 146bp | 58.8 |
| R: ACAACGTGATCGCCATTACCTG | ||||
| COX-2 | NM001167718 | F: CTTAAATTGAGACTTCGCAAGGATG | 165bp | 62 |
| R: TGGGACCAAGCCAAACACCT | ||||
| IL-1β | Y15006 | F: CAGCCTCAGCGAAGAGACCTT | 106bp | 60 |
| R: CACTGTGGTGTGCTCAGAATCC | ||||
| IL-8 | HM179639 | F: CTGGCCCTCCTCCTGGTT | 105bp | 60 |
| R: GCAGCTCATTCCCCATCTTTAC | ||||
| IL-6 | NM001314054.1 | F:ACAAAGCCAGAGTCCTTCAGAG | 86bp | 60 |
| R:GCCACTCCTTCTGTGACTCC | ||||
| TNF-α | NM204267 | F: CCCCTACCCTGTCCCACAA | 100bp | 58 |
| R: TGAGTACTGCGGAGGGTTCAT | ||||
| IL-2 | NM_001303244.1 | F: TGTGGAATGGCGTCTCTGTC | 125bp | 60 |
| R: AGTTCAATGGGCAGGGTCTC | ||||
| IL-4 | NM_021283.2 | F: ATGGATGTGCCAAACGTCCT | 78bp | 60 |
| R: AAGCACCTTGGAAGCCCTAC | ||||
| IL-10 | NM_010548.2 | F: TGCCTGCTCTTACTGACTGG | 79bp | 60 |
| R: CTGGGAAGTGGGTGCAGTTAT | ||||
| β-actin | NM_007393 | F:GCTGTGCTATGTTGCTCTAG | 117bp | 60.9 |
| R:CGCTCGTTGCCAATAGTG |
Determination of protein expression levels of apoptotic proteins and inflammatory cytokines in the lung by Western blot assay
At 21 and 42 days, lungs of eight mice in each group (half male, half female) were removed, stored in liquid nitrogen, and then homogenised in liquid nitrogen by using a mortar and pestle. The total protein in each sample was carried out using RIPA lysis buffer, and protein contents of lungs were measured by using the BCA Protein Assay kit. Then the protein samples were separated by sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) (10%-15% gels), and protein standards were used as molecular weight marker. After electrophoresis, proteins were transferred to nitrocellulose filter membranes. The membranes were blocked with 5 % non-fat-dried milk in phosphate-buffered saline with 0.1 % Tween 20 (PBST) for 1 h. After primary antibodies were incubated overnight at 4 °C, the membranes were washed three times with PBS for 10 min and incubated with biotin-conjugated secondary antibodies for 1 h with gentle shaking and washed again with PBST. Blots were visualised by ECLTM (Bio-Rad, Hercules, CA, USA) and X-ray film. The detected indicators were: cleaved-caspase-3, cleaved-caspase-9, Bax, Bak, Bcl-2, Bcl-xL, NF-κB, IκB, COX-2, IL-1β, IL-8, IL-6, TNF-α, IL-2, IL-4, and IL-10.
Statistical analysis
The significance of difference was analyzed by the SPSS version 17.0. The results were shown as means ± standard deviation. The analysis was performed with the one-way analysis of variance (ANOVA). The differences between control and experimental group(s) at p < 0.05 were considered significant.
Author Contributions
Z. Jian, H. Guo, and H. Cui designed the experiments. Z. Jian, H. Guo and H. Liu carried out the experiments. Z. Jian, H. Guo, H. Liu, J. Fang, Z. Zuo, J. Deng, Y. Li, X. Wang and L. Zhao analyzed and interpreted the data. Z. Jian, H. Guo, H. Liu and H. Cui wrote and revised the manuscript. Z. Jian, H. Guo and H. Liu contributed equally to this work.
Conflicts of Interest
The authors declare that there are no conflicts of interest.
Funding
This research was supported by the program for Changjiang Scholars and the University Innovative Research Team (IRT 0848), and the Shuangzhi project of Sichuan Agricultural University (03573050; 1921993267).
References
- 1. Lech T, Sadlik JK. Copper concentration in body tissues and fluids in normal subjects of southern Poland. Biol Trace Elem Res. 2007; 118:10–15. https://doi.org/10.1007/s12011-007-0014-z [PubMed]
- 2. Moynier F, Vance D, Fujii T, Savage P. The isotope geochemistry of zinc and copper. Mineralogy and Geochemistry. 2017; 82:543–600. https://doi.org/10.2138/rmg.2017.82.13
- 3. Feng J, Ma WQ, Gu ZL, Wang YZ, Liu JX. Effects of dietary copper (II) sulfate and copper proteinate on performance and blood indexes of copper status in growing pigs. Biol Trace Elem Res. 2007; 120:171–78. https://doi.org/10.1007/s12011-007-8001-y [PubMed]
- 4. Yang W, Wang J, Liu L, Zhu X, Wang X, Liu Z, Wang Z, Yang L, Liu G. Effect of high dietary copper on somatostatin and growth hormone-releasing hormone levels in the hypothalami of growing pigs. Biol Trace Elem Res. 2011; 143:893–900. https://doi.org/10.1007/s12011-010-8904-x [PubMed]
- 5. Gupta R. Veterinary toxicology basic and clinical principles. Cyanogenic Plants. 2007; 49:873–75.
- 6. Taylor KD. The encyclopaedia of food science, food technology and nutrition. Food Chem. 1994; 51:247. https://doi.org/10.1016/0308-8146(94)90266-6
- 7. Huster D, Purnat TD, Burkhead JL, Ralle M, Fiehn O, Stuckert F, Olson NE, Teupser D, Lutsenko S. High copper selectively alters lipid metabolism and cell cycle machinery in the mouse model of Wilson disease. J Biol Chem. 2007; 282:8343–55. https://doi.org/10.1074/jbc.M607496200 [PubMed]
- 8. Chambers A, Krewski D, Birkett N, Plunkett L, Hertzberg R, Danzeisen R, Aggett PJ, Starr TB, Baker S, Dourson M, Jones P, Keen CL, Meek B, et al. An exposure-response curve for copper excess and deficiency. J Toxicol Environ Health B Crit Rev. 2010; 13:546–78. https://doi.org/10.1080/10937404.2010.538657 [PubMed]
- 9. Krewski D, Yokel RA, Nieboer E, Borchelt D, Cohen J, Harry J, Kacew S, Lindsay J, Mahfouz AM, Rondeau V. Human health risk assessment for aluminium, aluminium oxide, and aluminium hydroxide. J Toxicol Environ Health B Crit Rev. 2007 (Suppl 1); 10:1–269. https://doi.org/10.1080/10937400701597766 [PubMed]
- 10. Bat L, Bilgin S, Öztekin A. Toxicity of copper on marine organisms from the Black Sea. J Coast Life Med. 2017; 5:422–26. https://doi.org/10.12980/jclm.5.2017J7-119
- 11. Dagli CE, Tanrikulu AC, Koksal N, Abakay A, Gelen ME, Demirpolat G, Yuksel M, Atilla N, Tolun FI. Interstitial lung disease in coppersmiths in high serum copper levels. Biol Trace Elem Res. 2010; 137:63–68. https://doi.org/10.1007/s12011-009-8566-8 [PubMed]
- 12. Pearson P, Britton J, McKeever T, Lewis SA, Weiss S, Pavord I, Fogarty A. Lung function and blood levels of copper, selenium, vitamin C and vitamin E in the general population. Eur J Clin Nutr. 2005; 59:1043–48. https://doi.org/10.1038/sj.ejcn.1602209 [PubMed]
- 13. Wideman R
Jr , Kirby Y, Barton T, Clark D, Bayyari G, Huff W, Moore PJr , Dunn P. Excess dietary copper triggers enlargement of the proventriculus in broilers. J Appl Poult Res. 1996; 5:219–30. https://doi.org/10.1093/japr/5.3.219 - 14. Liu JY, Yang X, Sun XD, Zhuang CC, Xu FB, Li YF. Suppressive effects of copper sulfate accumulation on the spermatogenesis of rats. Biol Trace Elem Res. 2016; 174:356–61. https://doi.org/10.1007/s12011-016-0710-7 [PubMed]
- 15. Newsholme P, Cruzat VF, Keane KN, Carlessi R, de Bittencourt PI
Jr . Molecular mechanisms of ROS production and oxidative stress in diabetes. Biochem J. 2016; 473:4527–50. https://doi.org/10.1042/BCJ20160503C [PubMed] - 16. Kawanishi S, Inoue S, Yamamoto K. Hydroxyl radical and singlet oxygen production and DNA damage induced by carcinogenic metal compounds and hydrogen peroxide. Biol Trace Elem Res. 1989; 21:367–72. https://doi.org/10.1007/BF02917277 [PubMed]
- 17. Gaetke LM, Chow CK. Copper toxicity, oxidative stress, and antioxidant nutrients. Toxicology. 2003; 189:147–63. https://doi.org/10.1016/S0300-483X(03)00159-8 [PubMed]
- 18. Assadian E, Zarei MH, Gilani AG, Farshin M, Degampanah H, Pourahmad J. Toxicity of copper oxide (CuO) nanoparticles on human blood lymphocytes. Biol Trace Elem Res. 2018; 184:350–57. https://doi.org/10.1007/s12011-017-1170-4 [PubMed]
- 19. Arnal N, de Alaniz MJ, Marra CA. Cytotoxic effects of copper overload on human-derived lung and liver cells in culture. Biochimica et Biophysica Acta (BBA) -. General Subjects. 2012; 1820:931–39. https://doi.org/10.1016/j.bbagen.2012.03.007
- 20. Hussain SP, Raja K, Amstad PA, Sawyer M, Trudel LJ, Wogan GN, Hofseth LJ, Shields PG, Billiar TR, Trautwein C, Hohler T, Galle PR, Phillips DH, et al. Increased p53 mutation load in nontumorous human liver of wilson disease and hemochromatosis: oxyradical overload diseases. Proc Natl Acad Sci USA. 2000; 97:12770–75. https://doi.org/10.1073/pnas.220416097 [PubMed]
- 21. Dalle-Donne I, Rossi R, Giustarini D, Milzani A, Colombo R. Protein carbonyl groups as biomarkers of oxidative stress. Clin Chim Acta. 2003; 329:23–38. https://doi.org/10.1016/S0009-8981(03)00003-2 [PubMed]
- 22. Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact. 2006; 160:1–40. https://doi.org/10.1016/j.cbi.2005.12.009 [PubMed]
- 23. Gupte A, Mumper RJ. Elevated copper and oxidative stress in cancer cells as a target for cancer treatment. Cancer Treat Rev. 2009; 35:32–46. https://doi.org/10.1016/j.ctrv.2008.07.004 [PubMed]
- 24. Zhao L, Cui HM, Yang F, Peng X, Deng JL. Pathological evaluations of effect of high dietary copper intake on liver in ducklings. Vet Sci China. 2007; 37:990–93.
- 25. Zhao L, Yang F, Peng X, Deng JL, Cui HM. Effects of high copper on the production of the hydroxy radical and nitrogen monoxide in liver of ducklings. Chinese Veterinary Science. 2008; 38:787–90.
- 26. Zhao L, Cui HM, Yang F, Peng X, Deng JL. Effects of high dietary copper on hepatic oxidation and hepatocyte apoptosis in ducklings. Chinese Veterinary Science. 2008; 38:54–58.
- 27. Zhao L, Yang F, Peng X, Deng JL, Cui HM. Effect of high copper dief on the antioxidase activities in ducklings. Chin J Vet Sci. 2009; 29:210–13.
- 28. Wei C, Peng X, Li Z, Yang F, Cui HM. Effect of high copper on the antioxydic function of kidney in ducklings. Chinese Journal of Animal Veterinary Sciences. 2009; 40:572–76.
- 29. Min L, Cui W, Peng X, Bai CM, Cui HM. Effect of dietary high copper on the antioxidase activities of brain tissue in chickens. Chinese Journal of Animal and Veterinary Sciences. 2010; 41:220–23.
- 30. Min L, Cui W, Peng X, Bai CM, Cui HM. Effect of dietary high copper on the oxidization in brain tissue of chickens. Chin J Vet Sci. 2009; 29:1334–37.
- 31. Cui W, Li M, Peng X, Deng JL, Cui HM. Effects of dietary high copper on antioxidative function and observation of pathologic lesion in spleen of chick. Chinese Veterinary Science. 2009; 39:338–43.
- 32. Singh KK, Kumar M, Kumar P, Gupta MK, Jha DK, Kumari S, Roy BK, Kumar S. “Free” copper: a new endogenous chemical mediator of inflammation in birds. Biol Trace Elem Res. 2012; 145:338–48. https://doi.org/10.1007/s12011-011-9198-3 [PubMed]
- 33. Connolly S. Cytotoxicity of copper oxide nanoparticles and associated ions on human epithelial lung cells (A549). The NNIN REU Research Accomplishments; 2010. pp. 6–7.
- 34. Wang J, Chen J, Tang Z, Li Y, Hu L, Pan J. hu L and Pan J. The effects of copper on brain microvascular endothelial cells and claudin via apoptosis and oxidative stress. Biol Trace Elem Res. 2016; 174:132–41. https://doi.org/10.1007/s12011-016-0685-4 [PubMed]
- 35. Bower JJ, Leonard SS, Shi X. Conference overview: molecular mechanisms of metal toxicity and carcinogenesis. Mol Cell Biochem. 2005; 279:3–15. https://doi.org/10.1007/s11010-005-8210-7 [PubMed]
- 36. Khan M, Ding C, Rasul A, Yi F, Li T, Gao H, Gao R, Zhong L, Zhang K, Fang X, Ma T. Isoalantolactone induces reactive oxygen species mediated apoptosis in pancreatic carcinoma PANC-1 cells. Int J Biol Sci. 2012; 8:533–47. https://doi.org/10.7150/ijbs.3753 [PubMed]
- 37. Cui HM, Chen HT. Effect of copper toxicity on lymphocyte apoptosis of lymphoid organs in ducklings. Chinese Journal of Animal Veterinary Sciences. 2005; 3:224–33.
- 38. Cui W, Peng X, Zhao L, Yang F, Cui HM. Effect of dietary high copper on the cell cycle and apoptosis of kidney in ducklings. Chinese Journal of Animal Veterinary Sciences. 2008; 39:980–84.
- 39. Cui HM, Xu ZY, Peng X, Zhu KC, Deng JL. The effect of high copper on lymphocyte apoptosis of lymphoid organs in chickens. Acta Veterinaria et Zootechnica Sinica. 2007; 38:601–07.
- 40. Cui W, Li M, Peng X, Deng JL, Cui HM. Effect of dietary high copper on the apoptosis and cell cycle of kidney in chickens. Chin J Vet Sci. 2009; 29:482–85.
- 41. Cui HM, Zhu KC, Peng X, Deng JL, Xu Zy. Effect of dietary high copper on the cell cycle and apoptosis of liver in chickens. Acta Veterinaria et Zootechnica Sinica. 2006; 37:908–13.
- 42. Guo H, Li K, Wang W, Wang C, Shen Y. Effects of copper on hemocyte apoptosis, ROS production, and gene expression in white shrimp litopenaeus vannamei. Biol Trace Elem Res. 2017; 179:318–26. https://doi.org/10.1007/s12011-017-0974-6 [PubMed]
- 43. Kabak YB, Gülbahar MY. Determination of apoptosis in liver and kidney tissues in experimental copper toxicity in rats. Ankara Univ Vet Fak Derg. 2012; 60:39–45.
- 44. Li YW, Wang XH, Nin Q, Luo XP. [Excessive copper induces hepatocyte apoptosis and affects Bax and Bcl-2 expression in rat liver]. Zhongguo Dang Dai Er Ke Za Zhi. 2008; 10:42–46. [PubMed]
- 45. Su R, Wang R, Guo S, Cao H, Pan J, Li C, Shi D, Tang Z. In vitro effect of copper chloride exposure on reactive oxygen species generation and respiratory chain complex activities of mitochondria isolated from broiler liver. Biol Trace Elem Res. 2011; 144:668–77. https://doi.org/10.1007/s12011-011-9039-4 [PubMed]
- 46. Chakraborty R, Basu T. Metallic copper nanoparticles induce apoptosis in a human skin melanoma A-375 cell line. Nanotechnology. 2017; 28:105101. https://doi.org/10.1088/1361-6528/aa57b0 [PubMed]
- 47. Raes H, Braeckman BP, Criel GR, Rzeznik U, Vanfleteren JR. Copper induces apoptosis in Aedes C6/36 cells. J Exp Zool. 2000; 286:1–12. https://doi.org/10.1002/(SICI)1097-010X(20000101)286:1<1::AID-JEZ1>3.0.CO;2-Z [PubMed]
- 48. Nathan C, Ding A. Nonresolving inflammation. Cell. 2010; 140:871–82. https://doi.org/10.1016/j.cell.2010.02.029 [PubMed]
- 49. Beyer I, Mets T, Bautmans I. Chronic low-grade inflammation and age-related sarcopenia. Curr Opin Clin Nutr Metab Care. 2012; 15:12–22. https://doi.org/10.1097/MCO.0b013e32834dd297 [PubMed]
- 50. Medzhitov R. Inflammation 2010: new adventures of an old flame. Cell. 2010; 140:771–76. https://doi.org/10.1016/j.cell.2010.03.006 [PubMed]
- 51. Sun X, Li J, Zhao H, Wang Y, Liu J, Shao Y, Xue Y, Xing M. Synergistic effect of copper and arsenic upon oxidative stress, inflammation and autophagy alterations in brain tissues of Gallus gallus. J Inorg Biochem. 2018; 178:54–62. https://doi.org/10.1016/j.jinorgbio.2017.10.006 [PubMed]
- 52. Becaria A, Lahiri DK, Bondy SC, Chen D, Hamadeh A, Li H, Taylor R, Campbell A. Aluminum and copper in drinking water enhance inflammatory or oxidative events specifically in the brain. J Neuroimmunol. 2006; 176:16–23. https://doi.org/10.1016/j.jneuroim.2006.03.025 [PubMed]
- 53. Brand P, Beilmann V, Thomas K, Kraus T, Krichel T, Reisgen M, Schmidt K, Krabbe J. The effects of exposure time on systemic inflammation in subjects with exposure to zinc- and copper-containing brazing fumes. J Occup Environ Med. 2019; 61:806–11. https://doi.org/10.1097/JOM.0000000000001676 [PubMed]
- 54. Cho WS, Duffin R, Poland CA, Howie SE, MacNee W, Bradley M, Megson IL, Donaldson K. Metal oxide nanoparticles induce unique inflammatory footprints in the lung: important implications for nanoparticle testing. Environ Health Perspect. 2010; 118:1699–706. https://doi.org/10.1289/ehp.1002201 [PubMed]
- 55. Stohs SJ, Bagchi D. Oxidative mechanisms in the toxicity of metal ions. Free Radic Biol Med. 1995; 18:321–36. https://doi.org/10.1016/0891-5849(94)00159-H [PubMed]
- 56. LI Y, Kong LQ, Gao H, Yan YL. Progress on free radical and diseases. Progress in Veterinary Medicine. 2008; 29:89–92.
- 57. Kubow S, Bray TM, Bettger WJ. Effects of dietary zinc and copper on free radical production in rat lung and liver. Can J Physiol Pharmacol. 1986; 64:1281–85. https://doi.org/10.1139/y86-216 [PubMed]
- 58. Menshikova EB, Zenkov NK, Reutov VP. Nitric oxide and NO-synthases in mammals in different functional states. Biochemistry (Mosc). 2000; 65:409–26. [PubMed]
- 59. Cabiscol E, Tamarit J, Ros J. Oxidative stress in bacteria and protein damage by reactive oxygen species. Int Microbiol. 2000; 3:3–8. [PubMed]
- 60. Muthumani M, Prabu SM. Silibinin potentially protects arsenic-induced oxidative hepatic dysfunction in rats. Toxicol Mech Methods. 2012; 22:277–88. https://doi.org/10.3109/15376516.2011.647113 [PubMed]
- 61. Rahman I, Mulier B, Gilmour PS, Watchorn T, Donaldson K, Jeffery PK, MacNee W. Oxidant-mediated lung epithelial cell tolerance: the role of intracellular glutathione and nuclear factor-kappaB. Biochem Pharmacol. 2001; 62:787–94. https://doi.org/10.1016/s0006-2952(01)00702-x [PubMed]
- 62. Chinoy NJ, Sharma AK, Patel TN, Memon R, Jhala D. Recovery from fluoride and aluminium induced free radical liver toxicity in mice. Fluoride. 2004; 37:257–63.
- 63. Liang Q, Sheng Y, Jiang P, Ji L, Xia Y, Min Y, Wang Z. The gender-dependent difference of liver GSH antioxidant system in mice and its influence on isoline-induced liver injury. Toxicology. 2011; 280:61–69. https://doi.org/10.1016/j.tox.2010.11.010 [PubMed]
- 64. Cinar M, Yildirim E, Yigit AA, Yalcinkaya I, Duru O, Kisa U, Atmaca N. Effects of dietary supplementation with vitamin C and vitamin E and their combination on growth performance, some biochemical parameters, and oxidative stress induced by copper toxicity in broilers. Biol Trace Elem Res. 2014; 158:186–96. https://doi.org/10.1007/s12011-014-9926-6 [PubMed]
- 65. Hehner SP, Breitkreutz R, Shubinsky G, Unsoeld H, Schulze-Osthoff K, Schmitz ML, Dröge W. Enhancement of T cell receptor signaling by a mild oxidative shift in the intracellular thiol pool. J Immunol. 2000; 165:4319–28. https://doi.org/10.4049/jimmunol.165.8.4319 [PubMed]
- 66. Marnett LJ. Lipid peroxidation-DNA damage by malondialdehyde. Mutat Res. 1999; 424:83–95. https://doi.org/10.1016/S0027-5107(99)00010-X [PubMed]
- 67. Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, Nakayama O, Makishima M, Matsuda M, Shimomura I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest. 2004; 114:1752–61. https://doi.org/10.1172/JCI21625 [PubMed]
- 68. Aghvami M, Ebrahimi F, Zarei MH, Salimi A, Pourahmad Jaktaji R, Pourahmad J. Matrine induction of ROS mediated apoptosis in human all B-lymphocytes via mitochondrial targeting. Asian Pac J Cancer Prev. 2018; 19:555–60. [PubMed]
- 69. Adams JM, Cory S. Bcl-2-regulated apoptosis: mechanism and therapeutic potential. Curr Opin Immunol. 2007; 19:488–96. https://doi.org/10.1016/j.coi.2007.05.004 [PubMed]
- 70. Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science. 1997; 275:1132–36. https://doi.org/10.1126/science.275.5303.1132 [PubMed]
- 71. Hausmann G, O’Reilly LA, van Driel R, Beaumont JG, Strasser A, Adams JM, Huang DC. Pro-apoptotic apoptosis protease-activating factor 1 (Apaf-1) has a cytoplasmic localization distinct from Bcl-2 or Bcl-x(L). J Cell Biol. 2000; 149:623–34. https://doi.org/10.1083/jcb.149.3.623 [PubMed]
- 72. Chan HW, Liu T, Verdile G, Bishop G, Haasl RJ, Smith MA, Perry G, Martins RN, Atwood CS. Copper induces apoptosis of neuroblastoma cells via post-translational regulation of the expression of Bcl-2-family proteins and the tx mouse is a better model of hepatic than brain Cu toxicity. Int J Clin Exp Med. 2008; 1:76–88. [PubMed]
- 73. Lee JH, Jung JY, Jeong YJ, Park JH, Yang KH, Choi NK, Kim SH, Kim WJ. Involvement of both mitochondrial- and death receptor-dependent apoptotic pathways regulated by Bcl-2 family in sodium fluoride-induced apoptosis of the human gingival fibroblasts. Toxicology. 2008; 243:340–47. https://doi.org/10.1016/j.tox.2007.10.026 [PubMed]
- 74. Cory S, Adams JM. The Bcl2 family: regulators of the cellular life-or-death switch. Nat Rev Cancer. 2002; 2:647–56. https://doi.org/10.1038/nrc883 [PubMed]
- 75. Ježek P, Hlavatá L. Mitochondria in homeostasis of reactive oxygen species in cell, tissues, and organism. Int J Biochem Cell Biol. 2005; 37:2478–503. https://doi.org/10.1016/j.biocel.2005.05.013 [PubMed]
- 76. Zhang N, Chen Y, Jiang R, Li E, Chen X, Xi Z, Guo Y, Liu X, Zhou Y, Che Y, Jiang X. PARP and RIP 1 are required for autophagy induced by 11′-deoxyverticillin A, which precedes caspase-dependent apoptosis. Autophagy. 2011; 7:598–612. https://doi.org/10.4161/auto.7.6.15103 [PubMed]
- 77. Hosseini MJ, Shaki F, Ghazi-Khansari M, Pourahmad J. Toxicity of copper on isolated liver mitochondria: impairment at complexes I, II, and IV leads to increased ROS production. Cell Biochem Biophys. 2014; 70:367–81. https://doi.org/10.1007/s12013-014-9922-7 [PubMed]
- 78. Ostrakhovitch EA, Cherian MG. Role of p53 and reactive oxygen species in apoptotic response to copper and zinc in epithelial breast cancer cells. Apoptosis. 2005; 10:111–21. https://doi.org/10.1007/s10495-005-6066-7 [PubMed]
- 79. Zhao H, Wang Y, Shao Y, Liu J, Liu Y, Xing M. Deciphering the ionic homeostasis, oxidative stress, apoptosis, and autophagy in chicken intestine under copper(II) stress. Environ Sci Pollut Res Int. 2018; 25:33172–82. https://doi.org/10.1007/s11356-018-3163-z [PubMed]
- 80. Mattie MD, McElwee MK, Freedman JH. Mechanism of copper-activated transcription: activation of AP-1, and the JNK/SAPK and p38 signal transduction pathways. J Mol Biol. 2008; 383:1008–18. https://doi.org/10.1016/j.jmb.2008.08.080 [PubMed]
- 81. Shanmugam T, Selvaraj M, Poomalai S. Epigallocatechin gallate potentially abrogates fluoride induced lung oxidative stress, inflammation via Nrf2/Keap1 signaling pathway in rats: an in-vivo and in-silico study. Int Immunopharmacol. 2016; 39:128–39. https://doi.org/10.1016/j.intimp.2016.07.022 [PubMed]
- 82. Sanz AB, Sanchez-Niño MD, Ramos AM, Moreno JA, Santamaria B, Ruiz-Ortega M, Egido J, Ortiz A. NF-kappaB in renal inflammation. J Am Soc Nephrol. 2010; 21:1254–62. https://doi.org/10.1681/ASN.2010020218 [PubMed]
- 83. Zaynagetdinov R, Sherrill TP, Gleaves LA, Hunt P, Han W, McLoed AG, Saxon JA, Tanjore H, Gulleman PM, Young LR, Blackwell TS. Chronic NF-κB activation links COPD and lung cancer through generation of an immunosuppressive microenvironment in the lungs. Oncotarget. 2016; 7:5470–82. https://doi.org/10.18632/oncotarget.6562 [PubMed]
- 84. Tian Y, Huo M, Li G, Li Y, Wang J. Regulation of LPS-induced mRNA expression of pro-inflammatory cytokines via alteration of NF-κB activity in mouse peritoneal macrophages exposed to fluoride. Chemosphere. 2016; 161:89–95. https://doi.org/10.1016/j.chemosphere.2016.06.035 [PubMed]
- 85. Leite CE, Maboni LO, Cruz FF, Rosemberg DB, Zimmermann FF, Pereira TC, Bogo MR, Bonan CD, Campos MM, Morrone FB, Battastini AM. Involvement of purinergic system in inflammation and toxicity induced by copper in zebrafish larvae. Toxicol Appl Pharmacol. 2013; 272:681–89. https://doi.org/10.1016/j.taap.2013.08.001 [PubMed]
- 86. Pereira TC, Campos MM, Bogo MR. Copper toxicology, oxidative stress and inflammation using zebrafish as experimental model. J Appl Toxicol. 2016; 36:876–85. https://doi.org/10.1002/jat.3303 [PubMed]
- 87. Park GY, Christman JW. Involvement of cyclooxygenase-2 and prostaglandins in the molecular pathogenesis of inflammatory lung diseases. Am J Physiol Lung Cell Mol Physiol. 2006; 290:L797–805. https://doi.org/10.1152/ajplung.00513.2005 [PubMed]
- 88. Lee KM, Kang BS, Lee HL, Son SJ, Hwang SH, Kim DS, Park JS, Cho HJ. Spinal NF-kB activation induces COX-2 upregulation and contributes to inflammatory pain hypersensitivity. Eur J Neurosci. 2004; 19:3375–81. https://doi.org/10.1111/j.0953-816X.2004.03441.x [PubMed]
- 89. Lu J, Wu DM, Zheng YL, Sun DX, Hu B, Shan Q, Zhang ZF, Fan SH. Trace amounts of copper exacerbate beta amyloid-induced neurotoxicity in the cholesterol-fed mice through TNF-mediated inflammatory pathway. Brain Behav Immun. 2009; 23:193–203. https://doi.org/10.1016/j.bbi.2008.09.003 [PubMed]
- 90. Ameeramja J, Perumal E. Protocatechuic acid methyl ester ameliorates fluoride toxicity in A549 cells. Food Chem Toxicol. 2017; 109:941–50. https://doi.org/10.1016/j.fct.2016.12.024 [PubMed]
- 91. Zhang S, Jiang C, Liu H, Guan Z, Zeng Q, Zhang C, Lei R, Xia T, Gao H, Yang L, Chen Y, Wu X, Zhang X, et al. Fluoride-elicited developmental testicular toxicity in rats: roles of endoplasmic reticulum stress and inflammatory response. Toxicol Appl Pharmacol. 2013; 271:206–15. https://doi.org/10.1016/j.taap.2013.04.033 [PubMed]
- 92. Nader M, Vicente G, da Rosa JS, Lima TC, Barbosa AM, Santos AD, Barison A, Dalmarco EM, Biavatti MW, Fröde TS. Jungia sellowii suppresses the carrageenan-induced inflammatory response in the mouse model of pleurisy. Inflammopharmacology. 2014; 22:351–65. https://doi.org/10.1007/s10787-014-0210-3 [PubMed]
- 93. Delker SL, Xue F, Li H, Jamal J, Silverman RB, Poulos TL. Role of zinc in isoform-selective inhibitor binding to neuronal nitric oxide synthase. Biochemistry. 2010; 49:10803–10. https://doi.org/10.1021/bi1013479 [PubMed]
- 94. van Strien ME, Mercier D, Drukarch B, Brevé JJ, Poole S, Binnekade R, Bol JG, Blits B, Verhaagen J, van Dam AM. Anti-inflammatory effect by lentiviral-mediated overexpression of IL-10 or IL-1 receptor antagonist in rat glial cells and macrophages. Gene Ther. 2010; 17:662–71. https://doi.org/10.1038/gt.2010.8 [PubMed]
- 95. Morita Y, Yamamura M, Kawashima M, Aita T, Harada S, Okamoto H, Inoue H, Makino H. Differential in vitro effects of IL-4, IL-10, and IL-13 on proinflammatory cytokine production and fibroblast proliferation in rheumatoid synovium. Rheumatol Int. 2001; 20:49–54. https://doi.org/10.1007/s002960000074 [PubMed]
- 96. Deng J, Guo H, Cui H, Fang J, Zuo Z, Deng J, Wang X, Zhao L. Oxidative stress and inflammatory responses involved in dietary nickel chloride (NiCl2)-induced pulmonary toxicity in broiler chickens. Toxicol Res (Camb). 2016; 5:1421–33. https://doi.org/10.1039/C6TX00197A [PubMed]
- 97. Liu H, Guo H, Jian Z, Cui H, Fang J, Zuo Z, Deng J, Li Y, Wang X, Zhao L. Copper Induces Oxidative Stress and Apoptosis in the Mouse Liver. Oxid Med Cell Longev. 2020; 2020:1359164. https://doi.org/10.1155/2020/1359164 [PubMed]