Introduction
Globally, lung cancer is a malignant tumor with the highest mortality rates. Lung cancer associated mortalities in China account for one-third of the global lung cancer mortality [1]. At the time of clinical diagnosis, many lung cancer patients are already at a locally advanced or metastatic stages, resulting in a poor disease prognosis. The diagnosis, treatment, and prognosis of lung cancer is inhibited by various challenges. Elucidating the molecular mechanisms associated with carcinogenesis can help reveal specific cancer biomarkers that can be utilized in personalized therapy. Clinically, lung cancer is divided into two forms: small-cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC), with adenocarcinoma being the most common subtype in China. LncRNAs studies can unravel the multi-level expression regulation network in living organisms, and provide new bio-markers for targeted therapy as well as diagnostic prognostic, and treatment targets. Although the role of several lncRNAs in tumorigenesis has been elucidated, the molecular biological mechanism of lncRNAs in lung cancer pathogenesis is poorly studied. The Human Genome Project revealed that only 1–2% of the genome of cancer cells encodes proteins, whereas the rest of 98% of the genome generate non-coding RNAs (ncRNA) [2]. Approximately two-thirds of human genome does not encode protein, and many are intergenic with most coding for long (>200 nucleotides) non-coding RNAs (lncRNAs). Although lncRNAs lack a protein-coding ability, they are directly involved in a regulatory role at the RNA level, including growth and development, bone marrow hematopoiesis, apoptosis, and cell proliferation [3]. These transcriptional non-coding sequences are widely involved in human physiological and pathological activities and even tumor progression, increasing the complexity of eukaryotes [4]. We initially hypothesized that the majority of ncRNAs could be transcriptional noises and mere by-products of the transcription of RNA polymerase II without biological functions [5]. However, recent developments have revealed the critical role ncRNAs play in cell proliferation, cycle, apoptosis, invasion, and metastasis. Regardless to whether the fragment is longer than 200 nucleotides or less, ncRNAs can be classified into two groups: small non-coding RNAs are less than 200 nt, whereas lncRNAs are longer than 200 nt. Several small non-coding RNAs have been identified as potential markers for tumor-targeted chemotherapy. LncRNAs are involved in the gene life cycle process and are closely associated with the pathogenesis of many complex diseases. As such, they have been identified as potential disease biomarkers and drug target. The Encode project identified nearly 16,000 lncRNAs, demonstrating the diversity of lncRNAs [6]. However, only a few hundred lncRNAs have been reported to have specific functions. Besides regulating gene expression and the biological mechanisms underlying tumorigenesis, lncRNAs influence prognosis of NSCLC. Therefore, this study evaluated the role of lncRNAs in diagnosis, prognosis and treatment of NSCLC.
The classification of lncRNAs
LncRNAs play an important role in various biological processes. Based on their biological functions, ncRNAs with limited or no protein-coding ability are divided into housekeeping ncRNAs and regulatory ncRNAs. Housekeeping RNA is necessary for cell survival and is always present in cells in constant levels. It is less affected by environs, and is essential for maintaining cell function. They mainly include tRNA, rRNA, snRNA, snoRNA and telomerase RNA, etc. Regulatory ncRNAs are ubiquitously expressed in a spatial and/or temporal specific pattern. Moreover, regulatory ncRNAs can be further classified into two subcategories based on their size: small ncRNAs (sncRNAs) and lncRNAs. Short non-coding RNAs including miRNAs, siRNAs, and piRNAs are less than 200 nt long. Long non-coding RNAs and bigger than 200 nt, do not have long open reading frames, and do not code for proteins. Under specific conditions, certain lncRNAs code for micropeptides. LncRNAs constitute 70% of ncRNAs and regulate gene expression via a several mechanisms. Poning et al. divided lncRNAs into 5 types according to their positions in the genome relative to protein-coding genes: (1) Sense lncRNAs, located in the same strand of a protein-coding gene; (2) Antisense lncRNAs, when overlapping one or more exons of other transcript on the same, or opposite, strand, respectively; (3) Bidirectional lncRNAs, when the expression of it and a neighboring coding transcript on the opposite strand is initiated in close genomic proximity; (4) Intronic lncRNAs, when it is derived wholly from within an intron of a second transcript (although these, as noted above, sometimes may represent pre-mRNA sequences), or (5) Intergenic lncRNAs (lincRNAs), located between protein coding regions, when it is derived wholly from within an intron of a second transcript (although these, as noted above, sometimes may represent pre-mRNA sequences) [7]. Based on subcellular localization, lncRNAs can also be classified into two classes: (1) nuclear lncRNAs, are involved in gene transcription and chromatin remodeling, and (2) cytoplasmic lncRNAs, typically regulate RNA-mediated functions [8]. Recent research, however, has shown that lncRNA participate in the processes of silencing X chromosome, chromosome modification, and genome modification, transcription activation and interference, and intranuclear transport, etc. [9]. Given that lncRNAs are expressed cell- and tissue-specific, they are excellent biomarkers for ongoing biological events [10]. The role of lncRNAs in tumor development continues to receive significant attention.
Structure of lncRNAs
LncRNAs can be spliced, capped, and/or polyadenylated molecules widely distributed in the nucleus or cytoplasm of eukaryotes. LncRNAs are mainly transcribed by RNA polymerase II, and structurally similar to mRNA, equipped with a poly (A) tail and promoter regions. LncRNAs also often display a secondary stem-loop-like structures or tertiary structures, occasioned by base complementation. The secondary structure consists of a helix, hairpin, bulge and pseudoknot, formed by complementary base pairing. In many cases, the structure, rather than the original sequence, determines the RNA's function. Depending on its base specificity or structure specificity, lncRNAs strongly regulate gene expression by interacting with nucleic acids or proteins. LncRNAs are tissue-specific and space-time specific and interact with RNA, DNA, and protein to regulate gene expression at multiple levels, including epigenetically, transcriptionally, post-transcriptionally and through splicing. The majority of the lncRNAs are polyadenylated in the nucleus or cytoplasm, showing a strong ability to regulate gene expression based on their base specificity or structure specificity in interacting with nucleic acids or proteins [11]. LncRNAs can form more complex structures than protein-coding genes, and their structural diversity enables them to participate in establishing binding sites that interact with proteins, DNA, and other RNA molecules. This implies that the structure of lncRNAs is closely related to their biological function. Nevertheless, the study of the lncRNA folding process, including the search for cofactors and the relationship between non-coding structures and functions, is still in the initial stage and it could be an important step for future studies in revealing the regulatory mechanism of lncRNAs [12].
Primary structure of lncRNAs
The original structure of the lncRNAs is composed of the nucleotide array with 5-methylguanosine cap structure and a 3-polyadenylation tail structure. To perform their regulatory functions, lncRNAs must be located at special positions in the nucleus. They regulate gene function in several ways, the most importantly through direct interaction with target gene and post transcriptionally by complementary base pairing with target genes or regulating genes transcriptional translation upstream or downstream of the target gene. Base pairing is based on its primary structure. It has been reported that lncRNA Gas5 can directly bind to the glucocorticoid DNA binding domain, then compete with the target genes containing the glucocorticoid response element, thus and regulating its expression [13]. Complementary pairing with the target bases is based on the primary structure of lncRNA. The lncRNAs either target miRNA or mRNA. For example, Linc-MD1 binds miR133 and miR135 through base complementary pairing, competitively inhibiting interaction of the two miRNAs with target genes [14]. LncRNAs termed 1/2sbsRNAs can incompletely pair with the Alu element in the 3'-UTR region of the mRNA to form binding site of the RNA binding protein Stau1. This promotes the binding of Stau1 to mRNA, and degrading mRNA through the stau1-mediated mRNA decay (SMD) pathway [15].
Secondary structure of lncRNAs
The secondary structure of lncRNAs is the center of lncRNA function. In 2012, a study reported the secondary structure information of lncRNA steroid receptor RNA activator (SRA). LncRNA SRA can activate expression of several sex hormones receptors and is closely related to the development of breast cancer [16]. Some studies indicate that secondary structure may be the key to lncRNA function. For instance, MALAT1, a highly conserved uracil-rich region, increases RNA stability by forming a triple helix [17]. The study of two secondary collapsible motifs can contribute to the anticancer effect of LncRNA MEG3 [18]. Novikova et al. found that due to compensatory mutation, the specific folding of SRA can be preserved in the human species [19]. In conclusion, there is increasing evidence that the structure of lncRNAs is important to its function. However, the sequence evolution of lncRNAs with low effective population size remains to be determined within the whole genome sequences regardless of the schema of secondary structure.
Molecular mechanism of lncRNAs
LncRNA is increasingly recognized as a major regulator of gene expression, existing at almost all levels of the gene's life cycle, including chromatin modification, transcription, and post-transcriptional processing. The biological function of lncRNAs is strictly dependent on its cellular location. LncRNAs are usually responsible for chromatin modification, transcription regulation, and mRNA or miRNA processing in the cell nucleus. In the cytoplasm, lncRNAs regulate translation, or act as miRNA. LncRNAs also act as molecular sponges, where they alter miRNA levels and accelerate or suppress the transcription of key genes, including albumen genes [20, 21]. Unlike highly conserved sncRNAs, lncRNAs regulate expression of target genes via various mechanisms [22, 23]. LncRNAs are a class of ubiquitous genes that are not only involved in the normal biological functions of the body but also multiple processes of disease. LncRNAs realize their molecular functions through signals, decoy, guides, and scaffold: ①The Signal lncRNAs can be used as markers in many biological processes because they display cell type, time and interspace specificity. As signaling molecules, lncRNAs regulate numerous signaling pathways. Some lncRNAs regulate the transcription of downstream genes and can reflect the temporal and spatial expression of genes. For example, the lncRNA homeobox (HOX) transcriptional antisense inter-gene RNA (HOTAIR) located at the HOXC site is present in the posterior and distal cells, while the other lncRNA HOXC are expressed in the fore-end pattern [24]. In contrast, the lncRNA HOXA transcript is located in distal cells [25]. ②The Decoy lncRNAs are those that adjust transcription by combining and carrying protein targets but do not cause other effects. The decoy lncRNAs which are known as “molecular sensors”, reduce the availability of regulatory factors including catalytic proteins, subunits of chromatin modifying complexes, transcription factors, and miRNAs by presenting binding sites. The decoy lncRNAs include p21-associated non-coding RNAs (PANDA), non-coding RNAs containing telomere repeats (TERRA), miRNAs, and molecular baits splicing silver [26]. For example, by immediately combining nuclear transcription factor Y that forces apoptosis induced by DNA injury, activated lncRNA p21 damage inhibits apoptotic gene, promoting progression of the cell cycle [27]. ③The Guide lncRNAs can induce chromatin decorating proteins and administration of ribonucleoprotein complexes to specific targets in a cis or trans manner. The famous cis mechanism involves the mammalian X-inactivation center (XIC), and X-inactive-specific transcript (XIST). A 1.6-kb ncRNA, (RepA) recruits poly comb repressive complex 2 (PRC2) in cis on the Xist, as its direct target. PRC2 is associated with extra X-chromosome inactivation [28]. LncRNAs influence their chromosome-wide transcription in trans unlike in cis-regulated lncRNAs. The lncRNA HOTAIR, for instance, is equipped to guide PRC2 target genes [29]. ④The scaffold lncRNAs can be used as a terrace for packaging components to accurately regulate the complex molecular interactions and signal transductions involved in several biological signaling processes [30]. The telomerase catalytic process, for example, requires telomerase RNA (TERC) and telomerase reverse transcriptase (TERT) attachment. TERC is a fundamental lncRNA unit that initiates the combination template, and also reveals the intricate territories that promote TERT banding, catalytic activity, and stability [31]. Dyskeratosis is a congenital disorder caused by mutations that alter the equilibrium between various TERC structures, due to the destruction of the structure of the RNA scaffold where telomeric regulatory protein modular binding sites are located [32].
LncRNAs mediate epigenetic regulation
Epigenetics refer to genetic alterations in gene expression without changes in the underlying DNA sequences. They include DNA methylation, chromatin remodeling, histone modification, and gene-related non-coding RNA silencing [33]. The most important function of lncRNAs is to regulate the epigenetic modification of the target gene. During embryonic development, epigenetic modification regulate several important life activities processes, such as genomic imprinting, chromosomal silencing, and chromosomal dosage compensation effects, all necessary for the normal embryonic development and differentiation of tissue cells [34]. Histones can be regulated and modified in various ways, including methylation, acetylation, and ubiquitination. The modifications affect gene expression differently. Regulatory methylation modification is the major form of epigenetic regulation, and affects the expression of related genes by regulating the CpG island methylation level in the gene promoter region. Chromatin remodeling studies have shown that epigenetic regulation can be combined with chromatin modification complexes to regulate chromatin remodeling in human species genes, influence gene expression, and participate in various life processes in vivo, such as the development of diseases, including tumors [35]. Currently, there are no accurate methods for predicting the pathogenesis and prognosis of NSCLC. Epigenetic gene expression regulation mechanisms also play an important role in tumorigenesis. Besides genetic changes in DNA sequences, several epigenetic changes in tumors diversify gene expression diversification and signaling pathways in malignant cells [36]. Studies have shown that lncRNAs can regulate the progression of lung cancer [37]. They initiate epigenetic changes that induce chromosomal modification complexes to specific genomic sites. Chromosomal instability is an important factor associated with tumor progression and metastasis. Overall, lncRNAs can function as oncogenes and tumor suppressor genes. Increasing evidences has implicated that lncRNA in the occurrence and development of NSCLC [38]. However, in the role of lncRNA in the prognosis of lung cancer has not been established (Figure 1A).
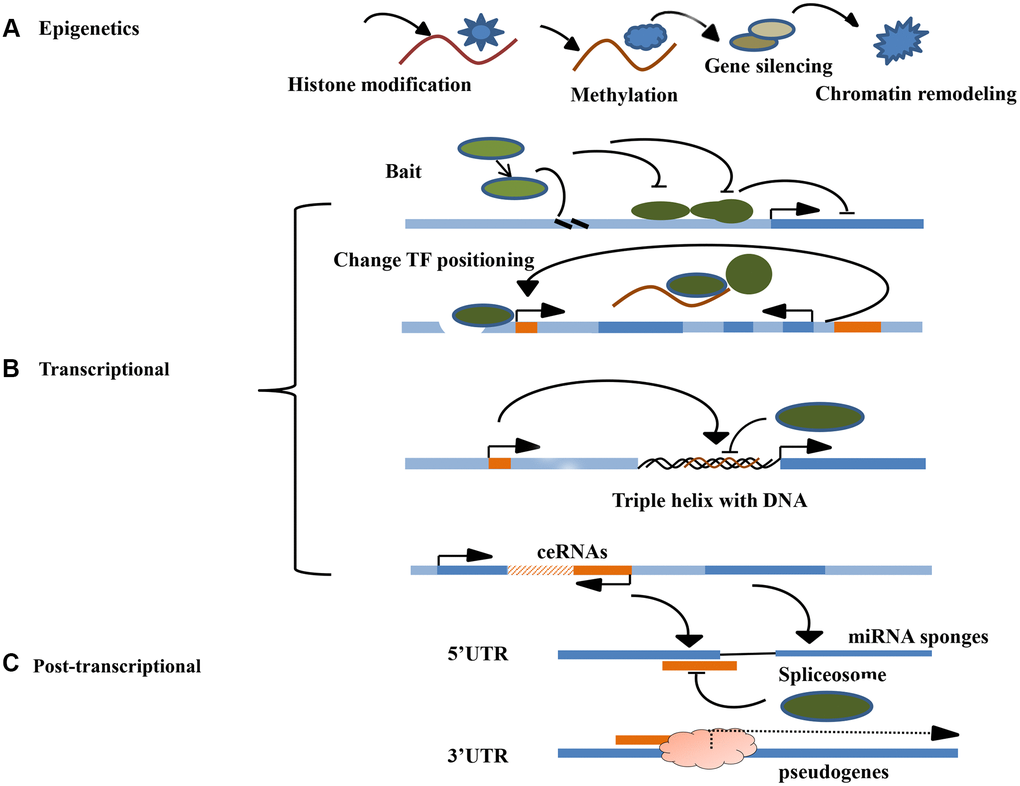
Figure 1. Molecular mechanism of LncRNA. (A) The main mechanisms of lncRNA epigenetics include histone modification, DNA methylation, gene silencing, and chromatin remodeling. (B) The main if lncRNA transcription level include signal induction, regulation of transcription factor sites, DNA triple helix structure and co-expression of ceRNA. (C) The main mechanisms of lncRNA post-transcriptional level include guiding the selective splicing of miRNA subtypes and targeting the protein receptor complex to recognize righteous chain mRNA transcripts.
LncRNAs mediate transcriptional regulation
Regulation of adjacent mRNA transcriptional expression by lncRNAs is common in eukaryotes. Several lncRNAs combine with suitable transcription factors to regulate their transcriptional activity. Studies show that certain lncRNAs can act as receptors to modulate base-pairing interchanges for these transcription-related elements, thus guiding the lncRNA including complex targeting spots for specific RNA or DNA. By acting as transcription regulatory factors, lncRNAs can regulate the transcription of cis-acting elements (adjacent genes) or trans-acting elements (distal genes) positively or negatively as transcription regulatory factors. The specific processes are: ①As decoys for RNA polymerase II or transcription factors (TFs), inhibiting its binding to the enhancer could become the promoter of the target gene, thereby, directly promoting or inhibiting the target gene manifestation [39]; ②Changing the location or modification of TF to accelerate or suppress gene transcription; ③Interacting with DNA to form a triple helix structure to mediate target gene transcription; ④Controlling target gene transcription in the form of competitive endogenous RNAs(ceRNAs). Studies have shown that lncRNAs can silence miRNA expression to promote tumor progression. Over-expression of LncRNA HOTAIR promotes transformation of malignant tumors, leading to a poor prognosis [40]. Furthermore, HOTAIR combines with EZH2 and SUZ12, and directly binds miR-34a promoter region, regulating expression level of miRNA [41]. Suppressing miR-34a expression activates the PI3K/AKT signaling that enhances lung cancer development [42]. LncRNAs can directly regulate gene expression by binding transcription factors and/or polymerase II, driving them away from or guiding them to target genes sites; or indirectly affecting gene expression by regulating transcription localization of proteins, such as promoting or inhibiting their nuclear localization (Figure 1B).
LncRNAs mediate post-transcriptional regulation of gene expression
lncRNAs regulates gene expression at the post-transcriptional level through manifold mechanisms, including mRNA compilation and commutative joint, and serves as a storage for sncRNAs and miRNA sponge layers. RNA compilation is a significant post-transcriptional process that embellishes, inserts, or deletes nucleotides to change the RNA molecule. The most common editing process is the transformation of adenosine to inosine on a double-stranded RNA by the ADAR enzyme. In certain cases, lncRNAs are paired with these protein-coding mRNAs, which appear to anonymously codify protein genes. The double-stranded RNA region can involve ADAR enzymes that help to catalyze adenosine to inosine conversion. For example, lncRNA-PVTL5 can act as a competitive endogenous miR-126 RNA to accelerate cell proliferation by regulating the miR-126/SLC7A5 axis [43]. LncRNA PVTL5 plays a significant role in lung cancer progression. Besides, the regulatory network could be one of the molecular mechanisms for the occurrence and malignant transformation of lung cancer. Another example is the lncRNAs pseudogene, which influences gene expression at the post-transcription level. Pseudogenes are non-functional molecular relics produced in genomes that are similar to the coding gene sequence from genome family and non-functional DNA copy. For example, lncRNA PTENP1 (pseudogene PTEN) can influence the expression levels of PTEN RNA and PTEN protein by competitively binding PTEN3 and miRNA reactive elements in the UTR region [44]. The nitric oxide synthase (NOS) pseudogene (lncRNA pseudo-NOS) inhibits the translation of neuronal nitric oxide synthase (NOS) by regulating binding of ribosomes to the nitric oxide synthase (NOS) pseudogene(lncRNA pseudo-NOS) complex [45]. In conclusion, lncRNAs play important roles in human cancer development, cell cycle control, apoptosis, among other aspects. As a gene suppressor or oncogenic molecule, lncRNAs regulate gene expression transcriptionally, post-transcriptionally, and through epigenetic molecular mechanisms, such as chromatin modification, transcription, splicing, and translation. Therefore, lncRNAs regulate various physiological and pathological processes, including cell proliferation, apoptosis, heat shock response, carcinogenesis, and drug resistance. Among these functions, gene-expression can be used to elucidate how lncRNAs promote or inhibit tumorigenesis. LncRNAs have been implicated in somatic cell mutations and development of malignant tumors. LncRNAs mediate these functions by dysregulating gene expression and networks. Although the biological functions of several lncRNAs have not been established, new techniques such as Sweeping Genetic Analysis, RNA-immunoprecipitation and high-throughput sequencing, as well as gene target sequence detection and gene knockdown/knockout experiments can be used to identify the functions of lncRNAs in cancer. Besides participating in carcinogenesis, tumor invasion and metastasis, lncRNAs play a major role in tumorigenesis and the development of new regulatory factors [46]. (Figure 1C).
Signaling pathways in lung cancer
The Wnt/β-catenin signaling pathway
The Wnt signaling pathway is a complex network of protein-protein interactions that predominates embryonic and cancer development. It is also regulate biological processes of adult animals [47]. It is one of the cell signaling systems, which involves multiple aspects such as signal transduction, cell cycle, cell proliferation, apoptosis, and cell adhesion. There are three main Wnt signaling pathways: ①The Wnt/β-catenin signaling pathway is the most classical signal pathway, activated in various solid tumors, including lung cancer; ②Wnt/Ca2+ signaling pathway and; ③The Wnt/planar cell polarity signaling pathway (PCP signaling pathway). The function of the Wnt/β-catenin signaling pathway is important in tumor pathogenesis [48]. Studies have shown that oncogenic β-catenin is essential in regulating the occurrence and development of tumors and other physiological processes [49, 50]. In lung cancer tissues, the expression of various lncRNAs is up-regulated or down-regulated by various signaling pathways, with Wnt/β-catenin being one of the most important [51]. Xia et al. [52] documented that down-regulated MEG3 expression through the Wnt/β-catenin signaling pathway can enhance the immunity of lung cancer cell lines to cisplatin. Wei et al. [53] found that lncRNA-SVUGP2 overexpression suppressed the Wnt/β-catenin signaling pathway activity in H1975 cells. In addition, suppressed lncRNA-SVUGP2 interacts with EZH2 and activates the Wnt/β-catenin signaling pathway, promoting occurrence and development of NSCLC. It has also been shown that inhibiting miR-101-3p and activating the Wnt/β-catenin signaling pathway upregulates lncRNA SNHG1, promoting NSCLC progression. The Wnt/β-catenin signaling pathway is abnormally activated in tumor cells, which induces the β-catenin protein to enter the nucleus and interact with the T cell transcription factor/lymphoid-enhancer factor (TCF/LEF) to stimulate downstream target genes. This leads to malignant cell proliferation and tumorigenesis of malignant cells (Figure 2).
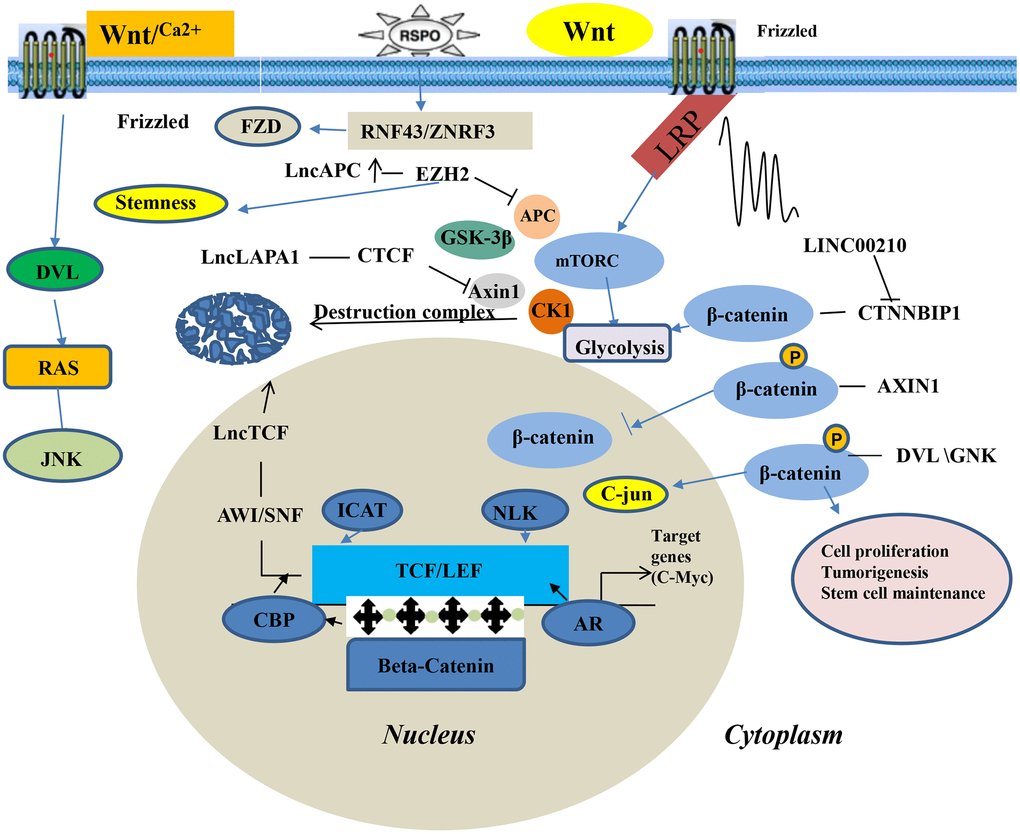
Figure 2. Wnt/β-catenin signaling pathway of LncRNA in lung cancer.
The MAPK signaling pathway
The mitogen-activated protein kinases (MAPKs) are intracellular protein kinases that transduce extracellular stimulation signals into the cell or nucleus. In so doing, they regulate numerous biochemical reactions such as cell proliferation, differentiation, apoptosis, and stress. The MAPKs are divided into extracellular c-Jun-N-terminal kinases (JNKs), extracellular signal-regulated kinases (ERKs), and P38 kinase [54]. Phosphorylation of chemicals, oxidative stress, cytokines, neurotransmitters, and other factors activate the MAPK signaling pathway. Transient excitation of ERKs promotes cell survival and proliferation, whereas their continuous excitation of ERKs promotes cell differentiation [55]. Transient excitation of JNKs promotes cell proliferation or differentiation, whereas their continuous excitation of JNKs promotes apoptosis [56]. Activation of processes downstream of proto-oncogene c-myc and nuclear factor-κB(NF-κB) by P38 induces cell death [57]. The activated MAPKs pathway enhance the occurrence of oxidative stress, inhibits the expression of the anti-apoptotic protein Bcl-2, promotes the expression of the apoptotic regulatory protein Bax, stimulates the release of cytochrome C, and finally, up-regulates cellular caspase-3,8,9 to kill the tumor cells. Upregulated expression of lncRNA TUC338 has been found to be upregulated and is associated with the prognosis of lung cancer. MAPK signaling pathway, regulated by LncRNA TUC338, is involved in of lung cancer to surrounding tissues [44]. Human TUC338 is a 590 bp gene located on chromosome12. Dysregulated TUC338 expression has also been reported in hepatocellular carcinoma and tongue squamous cell carcinoma. [58, 59]. A recent study showed that expression of MAPK correlates with drug-fast lung cancer, and some scholars have also reported that lncRNA NNT-AS1 is strongly expressed in drug-resistant NSCLC tissues and cells. This promotes NSCLC cells resistance to DDP via the MAPK/Slug signaling pathway. Ghafouri-Fard et al. [60] showed that TINCR can interact with BRAF to enhance its kinase activity, which leads to the excitation of the oncogenic mitogen-activated protein kinase (MAPK) signaling pathway, thus promoting the occurrence of NSCLC. In addition, a surgeon found that lncRNA SNHG12 up-regulates the expression of miR-181a to silence the expressions of MAPK1 and MAP2K1, and inhibits the MAPK/Slug signaling pathway by suppressing the level of phosphorylation MAPK1 (p-MAPK1), MAP2K1 (p-MAP2K1), and Slug phosphorylation [61]. SNHG12-miR-181a-MAPK/Slug axis has been established in their research, and the role of lncRNA SNHG12 in NSCLC multidrug resistance (MDR) was partially elucidated, providing a new therapeutic target for the treatment of NSCLC MDR (Figure 3).
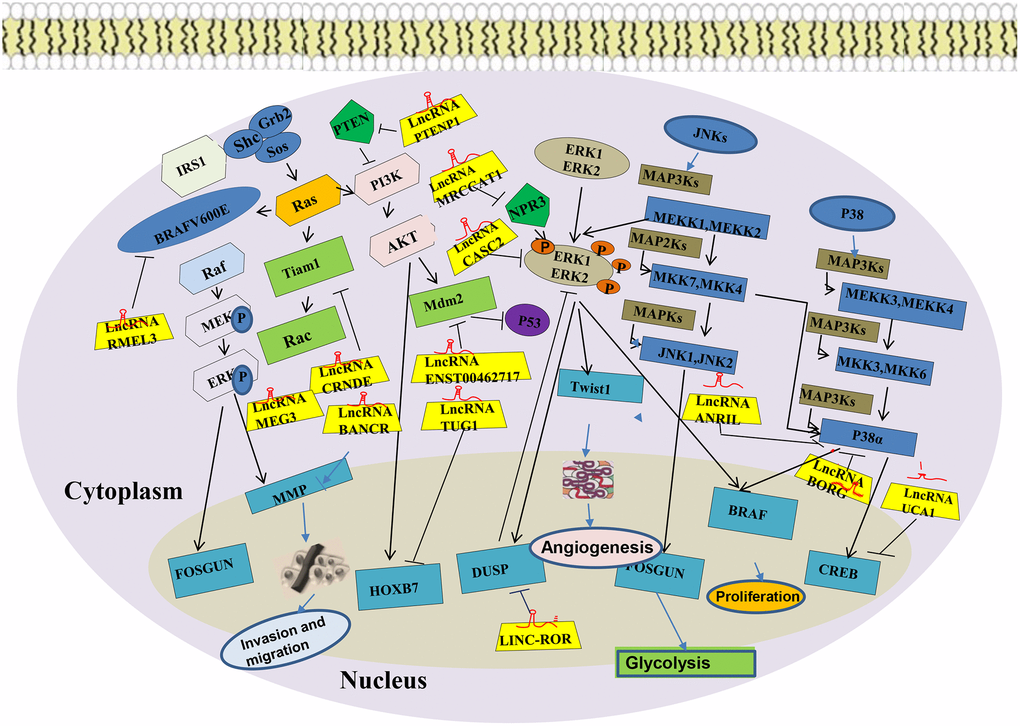
Figure 3. MAPK signaling pathway of LncRNA in lung cancer.
The STAT3 signaling pathway
The Signal Transducers and Activators of Transcription (STATs) related pathway is important in cytokine signal transduction. As a transcription factor, STATS3 is stimulated by cytokines such as interleukins, interferons, tumor necrosis factor, growth factors, and is activated by Janus Activated kinase (JAK), epithelial growth factor receptor (EGFR), etc. It then enters the nucleus to regulate transcription of downstream target genes [62]. Continuous activation of STAT3 is closely associated with the development of various tumors. The dysregulated epigenetic modification of STAT3 mainly consists of DNA and histone modifications. STAT3 transcription regulates epigenetic modification-related enzymes. It also regulates downstream gene expression by forming functional complexes with these enzymes. Seven mammalian members (STAT 1-4, 5a, 5b, and 6) of the STAT family were closely related in tumor progression. They are overactivated in more than 70% of human tumors and are involved in abnormal proliferation, invasion and metastasis, angiogenesis, as well immune dysregulation in tumor cells by regulating oncogenic genes. The STAT3 pathway is an important role in lung cancer. Studies have shown that lncRNA TSLNC8 significantly inhibits lung cancer cell progression and metastasis by targeting the IL-6 /STAT3/ HIF-1alpha signaling pathway. Zhao et al. [63] showed that lncRNA PICART1 induces anti-growth and anti-metastasis effects on lung cancer cells by regulating the JAK2 /STAT3 signaling pathway. While Zhang et al. [64] found that activated EGFR can upregulate the expression of PD-L1 via IL-6/JAK/STAT3 signaling pathway in non-small cell lung cancer (NSCLC) cells. The STAT3 signaling pathway may be aberrantly activated and have crucial roles in various tumor, including lung cancer (Figure 4).
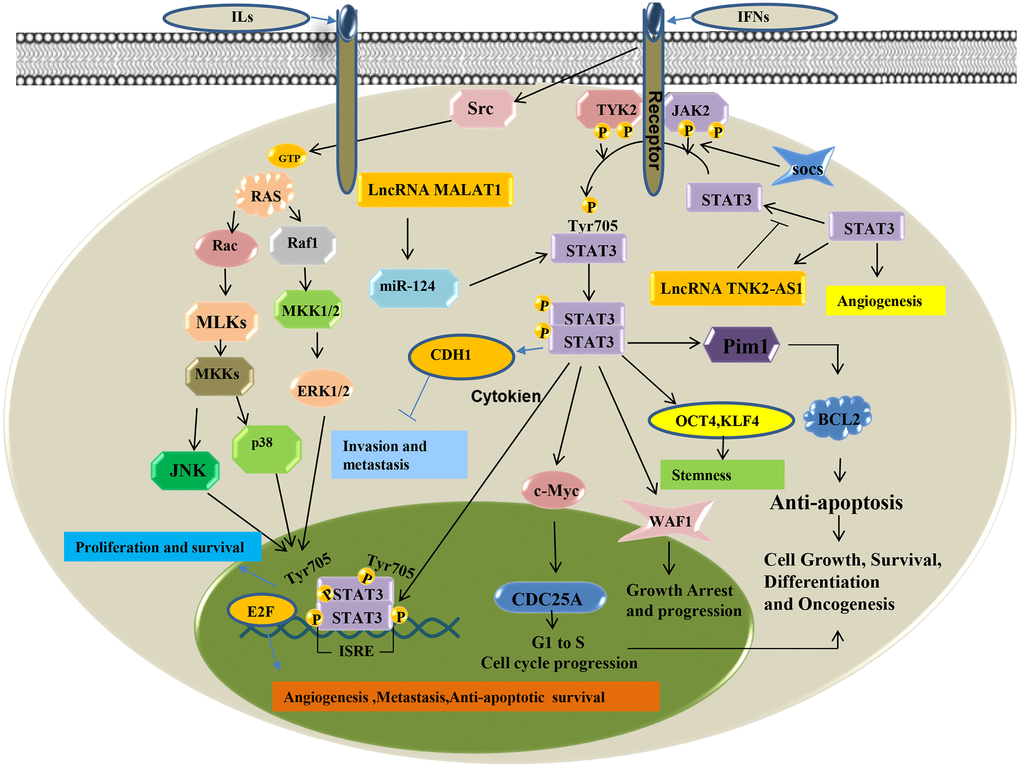
Figure 4. STAT3 signaling pathway of LncRNA in lung cancer.
P53 signaling pathway
P53 is a tumor suppressor protein that regulates various genes, inhibits and promotes DNA senescence in response to genotoxicity or cellular stress reaction including apoptosis, proliferation, and cell cycle [65]. P53 is a transcription factor and is also a component of the N-terminal activation domain. P53 has a specific DNA binding center and C-terminal tetramerization domain. It has a peculiar regulatory domain that is rich in basic amino acids, and a transitory half-life, maintained by continuous ubiquitylation and subsequent degradation by the 26S proteasome at lower standards in mammalian cells. In lung cancer tissues, the expression of various lncRNAs is up-regulated or down-regulated by different signaling pathways, of which P53 is one of the important signaling pathways. Alterations in lncRNA and the signal pathway of p53 are associated with the formation of idiopathic pulmonary fibrosis and lung cancer [66]. CDKN2B-AS1 and its adjacent gene CDKN2A are down-regulated in the peripheral blood of idiopathic pulmonary fibrosis patients. This effect activates the P53 signaling pathway, which promotes the formation of lung cancer development [67]. Zhou et al. [68] found a new prognostic indicator lncRNA LOC285194 that can inhibit tumors by targeting P53 signaling pathway. Ma et al. [69] reported that knock-down lncRNA TRPM2-AS induces cell apoptosis by activating the p53-p66shc pathway, which alters the cell cycle distribution and participation in the cisplatin resistance of NSCLC cells. The P53 gene participates in several human tumors. Moreover, P53 regulates various physiological activities in cells (Figure 5).
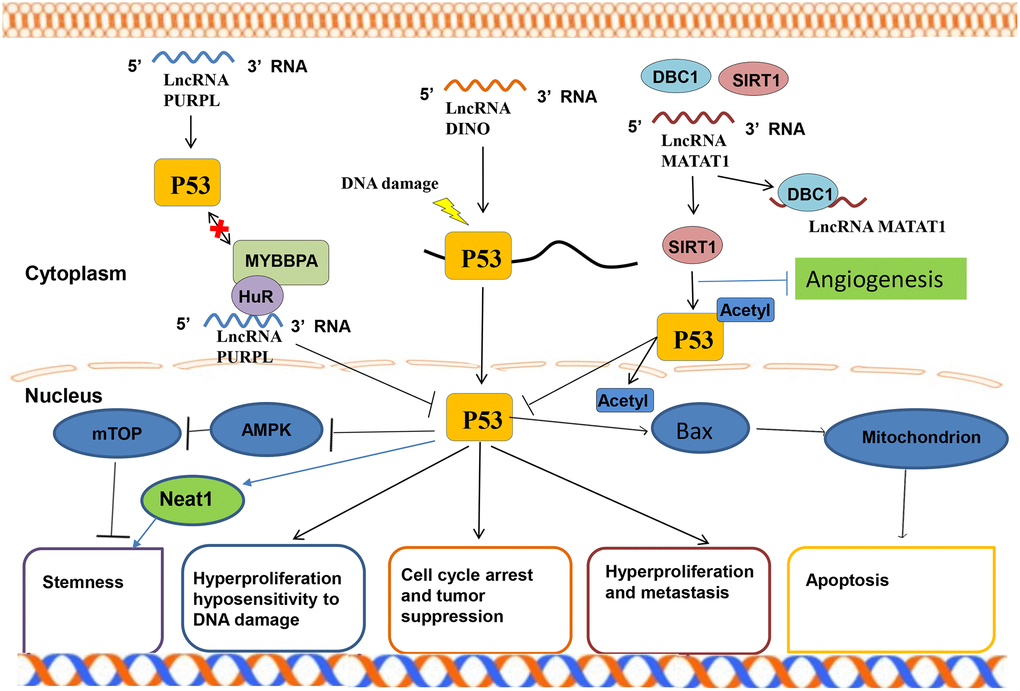
Figure 5. P53 signaling pathway of LncRNA in lung.
AKT signaling pathway
The AKT signaling pathway regulates multiple physiological processes that influence tumor occurrence and development, including metastasis, growth, proliferation, survival, transcription, and protein synthesis. In general, GF combines with receptor tyrosine kinase (RTK), then induces PIP2 to form a second messenger PIP3 through PI3K (phosphatase PTEN has the opposite effect), which activates key pathway molecules such as AKT, mTOR, and downstream gene transcriptions [70]. AKT is a proto-oncogene with important roles in regulating various cellular functions. In lung cancer, the AKT pathway is one of the major signaling pathways of lncRNAs. Dysregulation of the AKT signaling pathway contributes to the high incidences of human disease and also leads to the peak development of small molecule inhibitors of PI3K and AKT [71]. It has also been reported that lncRNA BC200 rapidly regulates the proliferation and cisplatin drug resistance of NSCLC cells through the PI3K/AKT signaling pathway [72]. Liu et al. [73] reported that the lncRNA HULC overexpression can promote cell proliferation and inhibit cell apoptosis by up-regulating the expression of SPHK1 protein expression in NSCLC, which induces the activation of its downstream PI3K/AKT signaling pathway. The cell experiments of Zhang et al. [74] revealed that lncRNA00152 inhibits the biological activities in NSCLC through the EGFR/PI3K/AKT pathway. Therefore, AKT is important in regulating various cell functions, making it an important target for human tumor diseases (Figure 6).
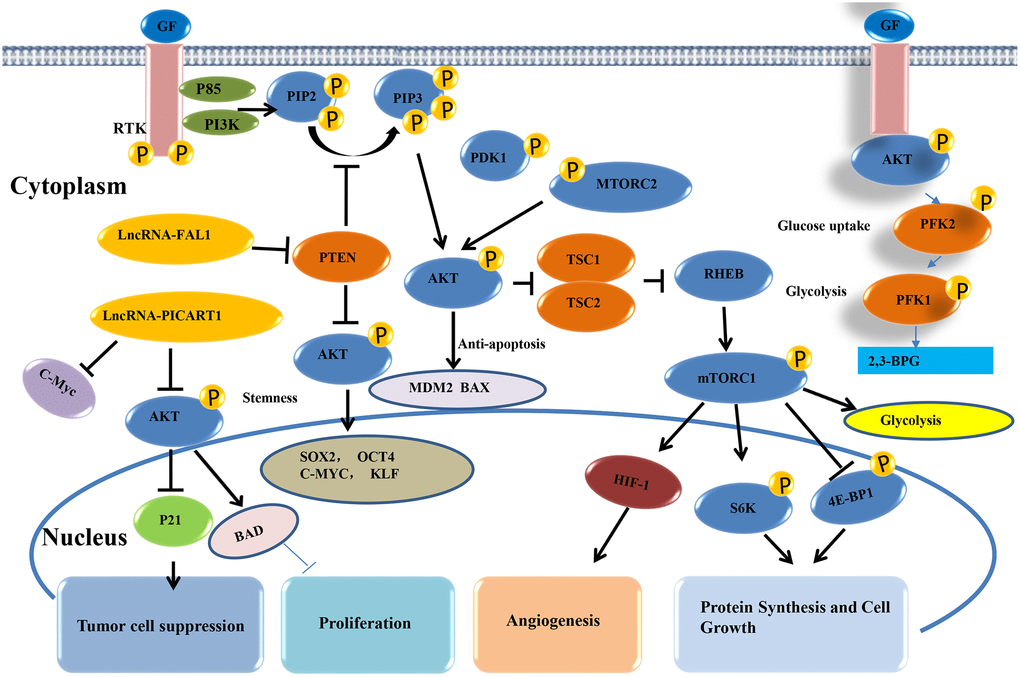
Figure 6. AKT signaling pathway of lncRNAs in lung cancer.
Differential expression and significance of lncRNA in malignant tumors
To identify the published lncRNA biomarkers for diagnosis in lung cancer patients, entry terms of “(long non-coding RNA OR lncRNA) AND (lung cancer OR LC) AND (diagnosis)” were firstly searched in the NCBI PubMed database. A total of 765 studies were obtained after removing any duplication of article content. Then we excluded some studies rigorously according to our criteria. The criteria of record exclusion were as follows: i) Reviews and meta-analysis and bioinformatics; ii) Case report; iii) Article not in English; iv) No lncRNA study; v) Non-human study: cell lines or/and mice; vi) Other tumor and disease. Finally, 35 up-regulated lncRNAs and 12 down-regulated lncRNAs in lung cancer were identified from these studies. And the exclusion criteria for other malignancies were the same as for lung cancer (Figure 7).
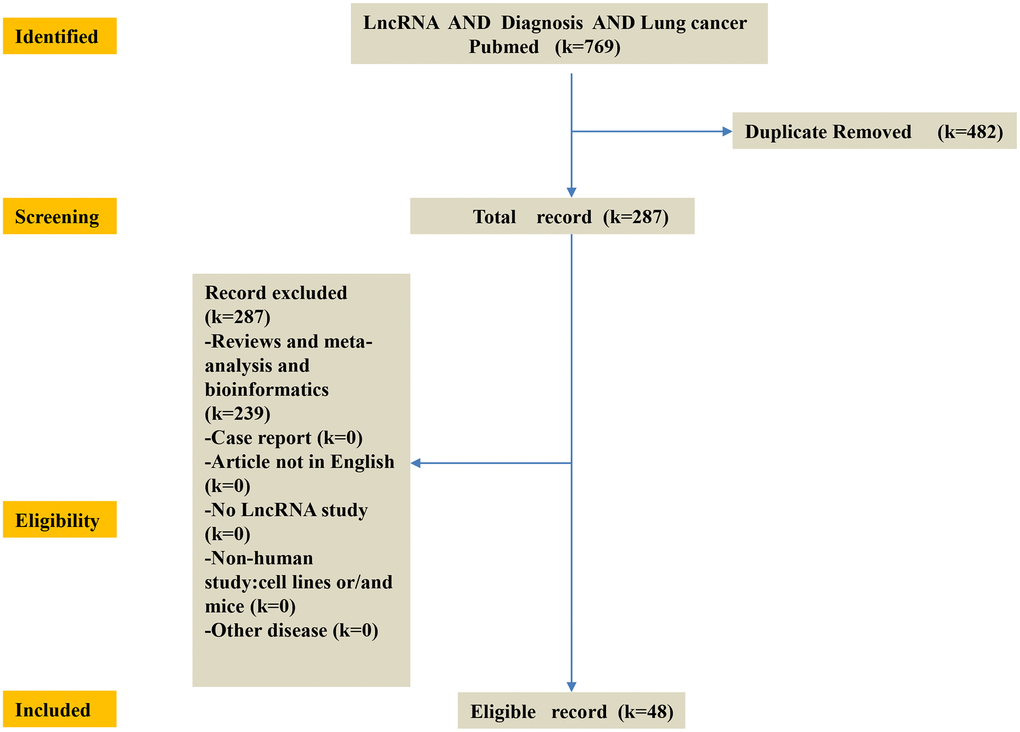
Figure 7. Flow-chart of diagnosis LncRNA of this study. K = number of literature records. First, identify the published lncRNA biomarkers of diagnosis in lung cancer patients in the NCBI PubMed database. Then, screening 769 database and obtain 287 database. The criteria of record exclusion were as follows: i) Reviews and meta-analysis and bioinformatics; ii) Case report; iii) Article not in English; iv) No lncRNA study; v) Non-human study: cell lines or/and mice; vi) Other disease. Finally, 35 up-regulated lncRNAs and 13 down-regulated lncRNAs in lung cancer were identified from these studies.
LncRNAs expression and significance in malignant tumors
Significance of highly expressed lncRNAs in lung cancer
Globally, lung cancer is among the leading causes of morbidity and mortality. The molecular mechanism of lung cancer occurrence and metastasis is relatively complex. Thus, further studies are needed to discover more accurate lncRNAs-based diagnostic biomarkers for lung cancer. Jiang et al. [75] identified diagnosis biomarker lncRNA XLOC_009167 to predict lung cancer through the lncRNAs expression chip. In addition, lncRNA XLOC_009167 was found to be significantly up-regulated in primary lung cancer tissues and cell lines compared to para-carcinoma tissues and cell lines, and its sensitivity and specificity were higher than the conventional markers commonly used in clinical practice (CYFR21-1 and NSE). The study also showed that lncRNA XLOC_009167 can not only be used as a marker for distinguishing lung cancer patients from healthy people, and to distinguish pneumonia from early-stage lung cancer. Zhu et al. [76] found that the expression levels of lncRNA16 in lung cancer tissues are significantly elevated than in para-carcinoma tissues. Due to its high sensitivity and specificity, lncRNA16 is an appropriate plasma target for lung cancer diagnosis. Gupta et al. [77] compared the sputum of lung cancer patients with those of cancer-free smokers with benign diseases and found that three lncRNAs (SNHG1, H19, and HOTAIR) as a biomarker panel were abnormally expressed at different levels. However, the diagnostic rate of this lncRNA was low. More studies should determine whether other lncRNAs can be used as biomarkers to improve the diagnostic accuracy of lung cancer. Lin et al. [78] showed that the expression of lncRNA Small Nucleolar RNA Host Gene 1 (SNHG1) and RNA component of mitochondrial RNA processing (RMRP) in the plasma of lung cancer were higher than those of the cancer-free control group. In addition, the AUC values of lncRNA SNHG1 and RMRP were found to be 0.90 and 0.80, respectively, which could be used to distinguish between NSCLC patients from normal people. The combined diagnostic value of the combined lncRNAs was higher than that of any single lncRNA. Li et al. found that the plasma HOTAIR level in NSCLC patients was significantly higher than that of a normal person, and that the diagnostic efficiency of HOTAIR higher than that of miRNA. In addition, the composite combination of HOTAIR and CEA showed a more precise diagnosis compared to the use of HOTAIR or CEA alone. Zhang et al. [79] evaluated Distal-less homeobox 6 antisense RNA 1 (DLX6-AS1) expression levels in serum and exosomes. Compared to normal matched people, the expression of DLX6-AS1 in NSCLC tissues was higher. ROC analysis also showed that the diagnostic efficiency of DLX6-AS1 for NSCLC outperformed CYFRA21-1. Cui et al. revealed that the expression of lncRNA PVT1 significantly increased in NSCLC tissues and cell lines, with a sensitivity of 0.815 and a specificity of 0.617, and this can be used as an NSCLC diagnostic marker. Li et al. [80] also evaluated the role of P15 and P21 in PVT1-mediated proliferation via the regulation of E2F2 signaling pathway. Li et al. [81] reported that lncRNA AGAP2 antisense RNA1 (AGAP2-AS1) gene expression in NSCLC tissues was up-regulated when compared to the para-carcinoma tissue via RT-PCR. ROC curve analysis showed that AGAP2-AS1 (AGAP2 antisense RNA 1) had a potential diagnostic value for NSCLC. Wang et al. [82] discovered that the expression of lncRNA urothelial carcinoma associated 1 (UCA1) in NSCLC tissues was higher than that of paired para-carcinoma tissues. Furthermore, Receiver operating curve (ROC) analysis revealed that plasma UCA1 can be used as a diagnostic marker for NSCLC. Zhang et al. [83] found that the LINC00152 was up-regulated in lung adenocarcinoma tissues, and LINC00152 promotes lung adenocarcinoma progression. Jing et al. [84] found that both lncRNA Colon Cancer-Associated Transcript 1 (CCAT1) and SOX2 overlapping transcript (SOX2OT) can be detected in human peripheral blood cell components. Serum CCAT1 and SOX2OT levels were found to be elevated in cancer patients, and they both had certain diagnostic values. In addition, the combined application of CCAT1 and SOX2OT was more effective in the diagnosis of NSCLC than CCAT1 or SOX2OT alone. Tang et al. [85] found that three lncRNAs (RP11-397D12.4, AC007403.1, ERICH1-AS1) were up-regulated in in NSCLC patients compared to healthy individuals, and the merged area under the curve of the three lncRNAs was 0.942. More studies should however be done to ascertain their diagnostic values of NSCLC in the future. Tao et al. [86] discovered that serum exosome lncRNA TBILA and AGAP2-AS1 have a strong diagnostic effect on NSCLC. The combination of lncRNA exosomes could enhance diagnostic efficacy. Tang et al. [87] showed that LINC00342 was up-regulated in the tissues, serum, and PBMC of NSCLC patients, while the specificity and sensitivity of LINC00342 exceeded that of CYFRA21-1 for the diagnosis of NSCLC. Wan et al. [88] evaluated the expression levels and diagnostic values of PCAT6 in 349 NSCLC tissues obtained from five GEO datasets (GSE19804, GSE18842, GSE30219, GSE19188, and GSE27262). Lung cancer tissue PCAT6 exhibited the highest diagnostic value. Tan et al. [89] analyzed lncRNA X-inactive specific transcript (XIST) and HIF1A-AS1 in the serum and tumor tissues of NSCLC patients and found the levels of XIST and HIF1A-AS1 in tumor tissues or serum to exceed those of a normal person. Therefore, elevated serum XIST and HIF1A-AS1 levels can be used as predictive biomarkers for NSCLC. The positive diagnostic rate of the combination of XIST and HIF1A-AS1 exceeded that of XIST or HIF1A-AS1 alone. Wang et al. [90] reported that compared to the normal control group, lncRNA SOX2 overlapping transcripts (SOX2OT) and CCAT1 were significantly up-regulated in NSCLC serum. They combined lncRNA SOX2OT and CCAT1 to establish a diagnostic network for NSCLC that has a high efficiency. Hu et al. [91] found that during development and validation tests, circulating SPRY4-IT1 (SPRY4 Intronic Transcript 1), ANRIL, and NEAT1 levels in the plasma specimens of NSCLC patients were above normal. Receiver operating characteristic curve (ROC) results revealed that the diagnostic efficacy of ANRIL is the highest (0.798); the greater the combination of the three elements, the higher the diagnostic value (AUC: 0.876; sensitivity: 82.8%; specificity: 92.3%). Li et al. reported that the expression level of lncRNA AFAP1 Antisense RNA 1 (AFAP1-AS1) in NSCLC patients is above normal. Serum AFAP1-AS1 can be used as a molecular marker to distinguish between NSCLC patients from healthy people, with an AUC of 0.759. When AFAP1-AS1 and CyFRA21-1 were combined, their AUC was 0.860. Further analysis revealed that serum AFAP1-AS1 could be a perfect combined diagnostic biomarker for NSCLC. Luo et al. [92] evaluated the expression and clinical significance of lncRNA H19 in the plasma of NSCLC patients. Plasma H19 levels in NSCLC patients were significantly increased, and can be used as a serological marker for the subsidiary diagnosis of NSCLC. Wang et al. [93] found that lncRNA FAM83H antisense RNA 1 (FAM83H-AS1) was elevated in lung adenocarcinoma, and its expression could significantly differentiate tumors from normal lung tissues. Therefore, it can be used as a diagnostic marker. Liu et al. [94] used RT-qPCR to determine the up-regulated expression of lncRNA taurine up-regulated 1(TUG1)in LAD serum samples and cell lines, which was found to be upregulated ROC was used to evaluate the diagnostic value of TUG1 in LAD patients, and TUG1 was considered as a potential diagnostic marker for LAD (Table 1).
Table 1. LncRNAs are highly expressed in lung cancer.
| Order (high expression) | Pathology type | Sample | Method | Control Type | Cut-off value | AUC | Sensitivity (%) | Specificity (%) | PMID |
| 1 H19 | NSCLC | Plasma | RT-PCR | BLD | 6.62 | 0.73 | 67.74 | 63.08 | 29970666 |
| 2 HOTAIR | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.806 | 76.2 | 71.9 | 28784052 |
| HOTAIR, CEA | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.841 | Unclear | Unclear | 28784052 |
| 3 SNHG1 | LC | Plasma | ddPCR | CFC | 1.11 | 0.9 | 77.78 | 87.88 | 30098474 |
| RMRP | LC | Plasma | ddPCR | CFC | 0.12 | 0.8 | 61.9 | 90.91 | 30098474 |
| SNHG1, RMRP | LC | Plasma | ddPCR | CFC | Unclear | Unclear | 84.13 | 87.5 | 30098474 |
| 4 SNHG1, H19, HOTAIR | NSCLC | Sputum | RT-PCR | CFC | 0.36 | 0.9 | 82.09 | 89.23 | 31450489 |
| LUAD | Sputum | RT-PCR | SCC, CFC | 0.36 | Unclear | 87.5 | 89.23 | 31450489 | |
| LUSC | Sputum | RT-PCR | AD, CFC | 0.36 | Unclear | 79.07 | 89.23 | 31450489 | |
| 5 PVT1 | NSCLC | Tissues | qRT-PCR | ANT | 4.38 | 0.736 | 81.50 | 61.70 | 26490983 |
| 6 TUG1 | LUAD | Cell, Serum | qRT-PCR | HC | Unclear | 0.756 | 78.33 | 60.00 | 29254212 |
| 7 LINC00173 | NSCLC | Serum | qRT-PCR | HD | 0.5197 | 0.809 | 62.96 | 89.01 | 32623390 |
| LINC00173, CEA | NSCLC | Serum | qRT-PCR | HD | Unclear | Unclear | 76.85 | 97.8 | 32623390 |
| LINC00173, Cyfra21-1 | NSCLC | Serum | qRT-PCR | HD | Unclear | Unclear | 82.41 | 95.6 | 32623390 |
| LINC00173, CEA, Cyfra21-1 | NSCLC | Serum | qRT-PCR | HD | Unclear | 0.914 | 90.74 | 68.13 | 32623390 |
| 8 FEZF1-AS1 | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.855 | 67.90 | 85.50 | 32590821 |
| FEZF1-AS1, NSE | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.932 | 0.932 | 90.30 | 32590821 |
| 9 A panel of five lncRNAs | LUSC | Tissue | qRT-PCR | NT | Unclear | 0.912 | 85 | 87 | 28076325 |
| ENST00000453324 | LUSC | Tissue | qRT-PCR | NT | Unclear | 0.77 | 64 | 80 | 28076325 |
| NR_028500 | LUSC | Tissue | qRT-PCR | NT | Unclear | 0.73 | 75 | 68 | 28076325 |
| UC011CLY.2 | LUSC | Tissue | qRT-PCR | NT | Unclear | 0.8 | 73 | 80 | 28076325 |
| NR_046326 | LUSC | Tissue | qRT-PCR | NT | Unclear | 0.77 | 79 | 71 | 28076325 |
| ENST00000441841 | LUSC | Tissue | qRT-PCR | NT | Unclear | 0.8 | 86 | 70 | 28076325 |
| 10 RP11-397D12.4, AC007403.1, ERICH1-AS1 | NSCLC | Plasma | qRT-PCR | CFS | 8.626 | 0.942 | 93 | 90 | 26393913 |
| 11 AGAP2-AS1 | NSCLC | Tissue | qRT-PCR | AT | Unclear | 0.846 | 80 | 75 | 28617550 |
| 12 TBILA | NSCLC | Serum | qRT-PCR | HC | 0.923 | 0.775 | 64.7 | 80.7 | 32015683 |
| AGAP2-AS1 | NSCLC | Serum | qRT-PCR | HC | 1.12 | 0.734 | 66.7 | 73.3 | 32015683 |
| TBILA, AGAP2-AS1 | NSCLC | Serum | qRT-PCR | HC | Unclear | 0.799 | 81.3 | 69.3 | 32015683 |
| TBILA, AGAP2-AS1, Cyfra21-1 | NSCLC | Serum | qRT-PCR | HC | Unclear | 0.853 | 91.4 | 80.7 | 32015683 |
| 13 LINC00342, Cyfra21-1 | NSCLC | Tissue, Serum, PBMC | qRT-PCR | HC | 4.21 | 0.582 | 80.3 | 71.4 | 30320899 |
| 14 LINC00673 | LUAD | Tissue, Plasma | qRT-PCR | NS | Unclear | 0.717 | unclear | unclear | 28849087 |
| 15 LINC00152 | NSCLC | Plasma | qRT-PCR | BD | Unclear | 0.816 | 80 | 72 | 29375177 |
| LINC00152, CEA | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.881 | 76 | 83 | 29375177 |
| 16 UCA1 | NSCLC | Plasma | qRT-PCR | NS | Unclear | 0.912 | 80 | 88 | 26380024 |
| NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.886 | 80 | 88 | 26380024 | |
| 17 LncRNA16 | LC | Tissue | qRT-PCR | AT | 1.945 | 0.858 | 73.97 | 100 | 27999202 |
| 18 XLOC_009167 | LC | Tissue, | qRT-PCR | HC | Unclear | 0.7398 | 78.7 | 61.8 | 30025752 |
| LC | Tissue | qRT-PCR | Pneumonia | Unclear | 0.7005 | 90.1 | 50 | 30025752 | |
| 19 PCAT6 | LUAD | Tissue | qRT-PCR | GSE19804 | Unclear | 0.921 | 96.67 | 85 | 29238201 |
| LUAD | Tissue | qRT-PCR | GSE27262 | Unclear | Unclear | 92 | 96 | 29238201 | |
| LUAD | Tissue | qRT-PCR | GSE30219 | Unclear | Unclear | 98.82 | 78.57 | 29238201 | |
| LUAD | Tissue | qRT-PCR | GSE19188 | Unclear | Unclear | 86.67 | 90.77 | 29238201 | |
| LUAD | Tissue | qRT-PCR | NC | Unclear | Unclear | 95.89 | 87.67 | 29238201 | |
| LUAD | Plasma | qRT-PCR | HD | Unclear | Unclear | 87.67 | 97.44 | 29238201 | |
| LUSC | Tissue | qRT-PCR | GSE30219 | Unclear | 0.9567 | 100 | 85.71 | 29238201 | |
| LUSC | Tissue | qRT-PCR | GSE19188 | Unclear | Unclear | 96.3 | 92.31 | 29238201 | |
| LUSC | Tissue | qRT-PCR | NC | Unclear | Unclear | 100 | 98.04 | 29238201 | |
| LUSC | Plasma | qRT-PCR | HD | Unclear | Unclear | 94.12 | 100 | 29238201 | |
| 20 DLX6-AS1 | NSCLC | Serum | qRT-PCR | HC | Unclear | 0.806 | 77.5 | 85.9 | 31612030 |
| 21 XIST | NSCLC | Tissue Serum | qRT-PCR | NC | Unclear | 0.834 | 82 | 80 | 26339353 |
| HIF1A-AS1 | NSCLC | Tissue Serum | qRT-PCR | NC | Unclear | 0.876 | 78 | 80 | 26339353 |
| XIST, HIF1A-AS1 | NSCLC | Tissue Serum | qRT-PCR | NC | Unclear | 0.931 | 86 | 90 | 26339353 |
| 22 SOX2OT | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.731 | 76.3 | 78.6 | 31077615 |
| 23 SOX2OT, ANRIL | NSCLC | Tissue Serum | qRT-PCR | NC | Unclear | 0.853 | 77.1 | 79.2 | 29504701 |
| 24 SOX2OT | NSCLC | Serum | qRT-PCR | CFS | Unclear | 0.846 | Unclear | Unclear | 31933793 |
| CCAT1 | NSCLC | Serum | qRT-PCR | CFS | Unclear | 0.787 | Unclear | Unclear | 31933793 |
| SOX2OT, CCAT1 | NSCLC | Serum | qRT-PCR | CFS | Unclear | 0.894 | Unclear | Unclear | 31933793 |
| 25 NEAT1 | NSCLC | Tissue | qRT-PCR | AT | Unclear | 0.684 | 80 | 50 | 25854373 |
| 26 SPRY4-IT1, ANRIL, NEAT1 | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.876 | 82.8 | 92.3 | 26453113 |
| 27 AFAP1-AS1 | NSCLC | Serum | qRT-PCR | HC | Unclear | 0.759 | 69.3 | 88.3 | 29080690 |
| AFAP1-AS1, Cyfra21-1 | NSCLC | Serum | qRT-PCR | HC | Unclear | 0.86 | 79.3 | 91 | 29080690 |
| 28 A panel of 64 lncRNAs | NSCLC | Tissue | qRT-PCR | AT | unclear | Unclear | 100 | 95.6 | 25590602 |
| 29 uc001gzl.3 | LUAD | Tissue | qRT-PCR | AT | Unclear | 0.719 | 79.4 | 60.3 | 25758555 |
| 30 CLDN10-AS1 | LUAD | Tissue | MA | NT | 0.078 | 0.847 | 72.03 | 96.3 | 32149133 |
| 31 ZEB2-AS1 | NSCLC | Tissue | qRT-PCR | NS | Unclear | 0.8793 | Unclear | Unclear | 32611283 |
| 32 RMRP | NSCLC | Plasma | qRT-PCR | HC | ≤0.660 | 0.70 | 72.03 | 68.97 | 33425713 |
| NEAT1 | NSCLC | Plasma | qRT-PCR | HC | >1.338 | 0.73 | 67.91 | 68.97 | 33425713 |
| TUG1 | NSCLC | Plasma | qRT-PCR | HC | ≤0.959 | 0.65 | 79.51 | 45.79 | 33425713 |
| MALAT1 | NSCLC | Plasma | qRT-PCR | HC | >0.507 | 0.66 | 82.86 | 45.74 | 33425713 |
| 4-lncRNA panel (RMRP, NEAT1, TUG1, and MALAT1) | NSCLC | Plasma | qRT-PCR | HC | >0.679 | 0.86 | 85.32 | 76.19 | 33425713 |
| 33 SOX2OT | LSCC | Plasma | qRT-PCR | HC | 0.815 | Unclear | 76 | 73.17 | 30986097 |
| 34 SCAL1 | LC | Blood | qRT-PCR | HC | Unclear | Unclear | Unclear | Unclear | 31410144 |
| NR-026689 | LC | Blood | qRT-PCR | HC | Unclear | Unclear | Unclear | Unclear | 31410144 |
| 35 LINC00487, LINC01927, LINC00959 | LSCC | Tissue | Unclear | SN | 0.7274 | Unclear | Unclear | Unclear | 31617686 |
| LINC02315, LINC00491, LINC07049 | LSCC | Tissue | Unclear | SN | 0.7049 | Unclear | Unclear | Unclear | 31617686 |
LncRNA are highly expressed in other tumors
Highly expressed lncRNAs including SNHG1, SPRY4-IT1, H19, LINC00152, MALAT1, AFAP1-AS1, NEAT1, UCA1, HOTAIR, CCAT1, FAM83H-AS1, TUG1, ANRIL, HIF1A-AS1, PVT1 that are associated with the diagnosis of lung cancer also have a diagnostic value in other malignant tumors. Gao et al. [95] found that lncRNA SNHG1 is highly expressed in plasma and tissues from hepatocellular carcinoma (HCC) patients. Therefore, it can be used differentiate HCC patients from HCH (hepatitis B virus positive chronic hepatitis and cirrhosis) and healthy individuals. Yin et al. [96] analyzed the serum lncRNA TUG1 levels in the healthy control individuals and multiple myeloma (MM) patients and found that the serum TUG1 expression levels were up-regulated in MM patients, proving that the serum TUG1 levels is a potential diagnostic biomarker. Jing et al. [97] studied the increased expression of lncRNA SPRY4 intron transcription 1 (SPRY4-IT1) in HCC and its key role in the occurrence of liver cancer and considered it to be as a potential diagnostic indicator for liver cancer. Zhan et al. developed a urinary exosome-derived lncRNA panel (MALAT1, PCAT-1, and SPRY4-IT1) to forecast the diagnosis and relapse of bladder cancer (BC). It was found to have a great clinical value in the diagnosis and prognosis of BC. Ebru et al. [98] studied the role of plasma lncRNA H19 in the diagnosis of gastric cancer and compared it with healthy people. The level of H19 in circulation was higher than normal, showing that H19 is a potential diagnostic marker for gastric cancer. Pan et al. [99] reported that the expression of serum lncRNA H19 was up-regulated in multiple myeloma (MM), and this lncRNA could be a new biomarker for the early diagnosis and clinical treatment of MM. Zhong et al. [100] determined the diagnostic potential of lncRNA H19 for breast cancer (BC). They reported that serum exosome lncRNA H19 is a potential diagnostic biomarker for BC. Li et al. [101] found that in the plasma samples of HCC patients, HULC and LINC00152 were up-regulated in the plasma samples of HCC patients and that they have a high diagnostic value for the occurrence and metastasis of liver cancer, can be considered as a new biomarker for liver cancer. Liu et al. [102] discussed the clinical roles of LINC00152 and SNHG152 in thyroid papillary carcinoma (PTC). Notably, expressions levels of SNHG12 and LINC00152 in PTC tissues were found to be substantially higher than those in the adjacent normal tissues, and also significantly higher than those in benign thyroid nodules. ROC analysis revealed that they have a certain diagnostic value. He et al. [103] evaluated the diagnostic spectrum of lncRNA (MALAT1, AFAP1-AS1, and AL359062). From the receiver operating characteristic (ROC) curve, they found that MALAT1, AFAP1-AS1, and AL359062 can be used as new serum biomarkers for predicting the prognosis of nasopharyngeal carcinoma after diagnosis and treatment. Wen et al. [104] examined the latent value of lncRNA urothelial carcinoma associated 1 (UCA1) as a diagnostic prognostic biomarker of osteosarcoma and reported that lncRNA UCA1 is a potential specific non-invasive candidate biomarker for osteosarcoma. Liu et al. [105] documented that the plasma levels of lncRNA 91H, PVT-1, and MEG3 in CRC patients are higher than normal. A combined application can differentiate CRC patients from non-cancer control patients; and this could be a diagnostic biomarker for early CRC. Yang et al. [106] demonstrated that serum PVT1 levels in cervical cancer tissues was higher than in adjacent normal tissues, forming it important diagnostic marker. Sun et al. [107] found that four lncRNA (HOTAIR, PVT1, XLOC_000303, and AL592284.1) were up-regulated in legacy cancer (CC) compared to the cervical cancer control group. The risk scoring formula was used to analyze ROC, and it was found that a combination of the four factors have a higher diagnostic value. Chen et al. [108] evaluated the published lncRNA PVT1 expression data and found that PVT1 was up-regulated in melanoma tissues compared with para-carcinoma tissues. Serum PVT1 levels were significantly higher in melanoma patients compared with age and sex-matched nonmelanoma and melanocytic nevus controls. The ROC analysis showed that the serum PVT1 level can distinguish the melanoma patients and the control group. Luo et al. [109] selected 6 lncRNAs can be used for the diagnosis of HCC based on RT-qPCR. The level of plasma lncRNA ZFAS1 in HCC was substantially higher than the average level. The results indicated that ZFAS1 can be used as a marker for HCC diagnosis (Table 1).
LncRNA expression is suppressed in malignant tumors
LncRNA expression is suppressed in lung cancer
Compared with the lncRNAs with high expression in lung cancer, the number of lncRNAs with low expression in lung cancer is relatively small. Wang et al. [110] found that the expression levels of lncRNA (C6orf176-TV1 and C6orf176-TV2) in NSCLC tissues were significantly lower than those in paired adjacent tissues. Then, they also found that C6orf176 can be applied in the clinical diagnosis of NSCLC. Liang et al. [111] found that the plasma lncRNA Growth Arrest Specific transcript 5 (GAS5) levels in NSCLC patients were down-regulated. They further found that a combination of GAS5 and CEA provided a more accurate diagnosis for NSCLC patients. Kamel et al. [112] reported that, compared to CEA, lncRNA GAS5 and SOX2OT expression are more sensitive and specific NSCLC biomarkers. Moreover, their diagnostic sensitivity and specificity when combined are higher than that of GAS5 and SOX2OT used alone. Hao et al. [113]. found that lncRNA MAG12 antisense RNA 3 (MAG12-AS3) in NSCLC were significantly down-regulated compared to normal controls. Zeng et al. [114] found that knockdown of lncRNA ZNFX1 antisense RNA1 (ZFAS1) can suppress NSCLC cell proliferation, as well as invasive potentials, increased NSCLC cell apoptotic rates in vitro. Tan et al. [115] found that LINC00312 was significantly down-regulated in NSCLC tissues compared to normal tissues. They further evaluated the significance of LINC00312 in NSCLC subtypes such as ADC and SCC using the ROC curve. The results revealed that LINC00312 can effectively identify NSCLC. Wang et al. [116] reported that lncRNA PRAL levels in most NSCLC tumor tissues are significantly lower than those in para-carcinoma and healthy tissues. ROC curve analysis showed that there is a strong difference between NSCLC and the control group, implying that PRAL expression levels can be used as a latent biomarker for diagnosis for NSCLC diagnosis. Guo et al. [117] evaluated the expression levels of lncRNA MALAT1 in the whole blood of NSCLC and found that they were low compared to normal controls, while the AUC was 0.718. Further results showed that MALAT1 can be used as a predictive biomarker of lung cancer. Weber et al. [118] also evaluated the effectiveness of lncRNA MALAT1 as a blood marker for NSCLC. They found that MALAT1 was not sensitive enough to be used as a single biomarker for the diagnosis of the NSCLC in the blood, but could be used as an auxiliary biomarker within a group to improve the overall diagnostic performance. Chen et al. [119] found that the plasma lncRNA RP11-438N5.3 levels in NSCLC patients were low than normal. ROC analysis showed that lncRNA RP11-438N5.3 could be a new biomarker for NSCLC. Wang et al. [120] reported many significant differences in the expressions of lncRNA and mRNA expression in early lung adenocarcinoma and the corresponding adjacent nontumorous tissues (NT). They later established a lncRNA prediction panel (ENST00000540136, NR_034174, uc001gzl. 3, uc004bbl. 1, ENST00000434223). Further validation showed that these lncRNAs had a high sensitivity and specificity in the identification of early lung adenocarcinoma and NT samples. Jin et al. [121] found that CLDN10-AS1 was up-regulated in lung adenocarcinoma tissues (LUAD) from the GEO database, while SFTA1P, SRGAP3-AS2 and ADAMTS9-AS2 were down-regulated. According to the sensitivity and specificity indices and ROC curve, lncRNA SFTA1P, ADAMTS9-AS2, CLDN10-AS1 (up-regulated) and SRGAP3-AS2 all have a diagnostic value for LUAD. Currently, lncRNAs that have been found to be suppressed have previously not been shown to be viable diagnostic markers for lung squamous cell carcinoma. In this study, we found that suppressed lncRNAs have a certain significance in lung squamous cell carcinoma, and their potential as diagnostic markers should be evaluated. Li et al. [122] reported that lncRNA SFTA1P (Surfactant associated with 1, pseudogene) is significantly down-regulated in LSCC tissues compared to para-carcinoma tissues. In addition, HNRNP-U (heterogeneous nuclear ribonucleoprotein U) expression was shown to be down-regulated in LSCC, and was positively associated with poor prognosis as well as SFTA1P. Li et al. [123] evaluated biomarkers of smoking- related lung squamous cell carcinoma (LSCC) and found that LINC00094 was down-regulated in tumor tissues of smoking patients, suggesting that LINC00094 could be a biomarker or therapeutic target for smoking-related LSCC. He et al. [124] showed that MAGI2-AS3 is significantly down-regulated in the tumor tissues in lung squamous cell carcinoma (LSCC) compared to para-carcinoma tissue. Further analysis revealed that MAGI2-AS3 inhibits LSCC by regulating the miR374a/b-5p /CADM2 axis, hence it may a potential biomarker of LSCC. Xiong et al. [125] found that lncRNA FEZF1 antisense RNA 1(FEZF1-AS1) in lung squamous cell carcinoma (LSCC) tissues was significantly lower than that in the para-carcinoma tissues; and its low expression was correlated with a poor prognosis of LSCC tissues, indicating that FEZF1-AS1 could be a specific biomarker for LSCC. Studies have revealed that the above lncRNAs are down-regulated in lung squamous cell carcinoma. Table 2).
Table 2. LncRNAs are lowly expressed in lung cancer.
| Order (Low expression) | Pathology type | Sample | Method | Control Type | Cut-off value | AUC | Sensitivity (%) | Specificity (%) | PMID |
| 1 C6orf176-TV1 | NSCLC | Tissue | qRT-PCR | PLT | 24.239 | 0.708 | 51 | 88 | 27374438 |
| C6orf176-TV2 | NSCLC | Tissue | qRT-PCR | PLT | 25.646 | 0.64 | 49 | 75 | 27374438 |
| 2 GAS5 | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.832 | 82.2 | 72.7 | 27631209 |
| GAS5, CEA | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.909 | 86.7 | 90.9 | 27631209 |
| 3 GAS5 | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.814 | 82.5 | 80 | 31077615 |
| 4 GAS5 | NSCLC | Tissue | qRT-PCR | PLT | Unclear | 0.653 | 60 | 70 | 28339045 |
| NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.638 | 42.31 | 77.78 | 28339045 | |
| 5 MAGI2-AS3 | LUAD | Plasma | qRT-PCR | HC | Unclear | 0.866 | 82.4 | 85 | 29922089 |
| LUSC | Plasma | qRT-PCR | HC | Unclear | 0.887 | 90.9 | 85 | 29922089 | |
| LUAD | Platelets | qRT-PCR | HC | Unclear | 0.853 | 77.9 | 81.7 | 29922089 | |
| LUSC | Platelets | qRT-PCR | HC | Unclear | 0.892 | 78.7 | 88.3 | 29922089 | |
| LUAD | Plasma | qRT-PCR | HC | Unclear | 0.806 | 75 | 73.3 | 29922089 | |
| ZFAS1 | LUSC | Plasma | qRT-PCR | HC | Unclear | 0.77 | 66.7 | 80 | 29922089 |
| LUAD | Platelets | qRT-PCR | HC | Unclear | 0.78 | 75 | 66.7 | 29922089 | |
| LUSC | Platelets | qRT-PCR | HC | Unclear | 0.744 | 93.9 | 43.3 | 29922089 | |
| MAGI2-AS3, ZFAS1 | LUAD | Plasma | qRT-PCR | HC | Unclear | 0.89 | 76.5 | 86.7 | 29922089 |
| LUSC | Plasma | qRT-PCR | HC | Unclear | 0.902 | 84.9 | 88.3 | 29922089 | |
| LUAD | Platelets | qRT-PCR | HC | Unclear | 0.908 | 89.7 | 86.7 | 29922089 | |
| LSCC | Platelets | qRT-PCR | HC | Unclear | 0.919 | 81.8 | 96.7 | 29922089 | |
| 6 LINC00312 | NSCLC | Tissue | qRT-PCR | NT | Unclear | 0.803 | Unclear | Unclear | 28849087 |
| 7 PRAL | NSCLC | Tissue | qRT-PCR | NT | Unclear | 0.8546 | Unclear | Unclear | 30274002 |
| 8 MALAT-1 | NSCLC | Plasma | qRT-PCR | CFC | –0.41 | 0.79 | 56 | 96 | 24313945 |
| LUAD | Plasma | qRT-PCR | CFC | –1.44 | 0.75 | 81 | 64 | 24313945 | |
| LUSC | Plasma | qRT-PCR | CFC | –0.41 | 0.82 | 63 | 96 | 24313945 | |
| 9 MALAT-1 | LC | Blood | qRT-PCR | HC | 10.344 | 0.718 | 70 | 60 | 26137228 |
| 10 RP11-438N5.3 | NSCLC | Plasma | qRT-PCR | HC | Unclear | 0.814 | Unclear | Unclear | 32184656 |
| 11 ENST00000434223 | LUAD | Tissue | qRT-PCR | PLT | Unclear | 0.843 | 81 | 79.4 | 25758555 |
| uc004bbl.1 | LUAD | Tissue | qRT-PCR | PLT | Unclear | 0.821 | 85.2 | 62.4 | 25758555 |
| ENST00000540136 | LUAD | Tissue | qRT-PCR | PLT | Unclear | 0.882 | 79.4 | 84.1 | 25758555 |
| NR 034174 | LUAD | Tissue | qRT-PCR | PLT | Unclear | 0.835 | 77.8 | 79.4 | 25758555 |
| 12 SFTAP1 | LUAD | Tissue | MA | NT | 39.89 | 0.9126 | 82.29 | 100 | 32149133 |
| ADAMTS9-AS2 | LUAD | Tissue | MA | NT | 0.212 | 0.9116 | 81.49 | 96.3 | 32149133 |
| SRGAP3-AS2 | LUAD | Tissue | MA | NT | 0.921 | 0.7945 | 73.04 | 75.93 | 32149133 |
| 13 Exo-GAS5 | NSCLC | Serum | qRT-PCR | HC | Unclear | 0.857 | 85.94 | 70 | 31032916 |
| CEA+Exo-GAS5 | NSCLC | Serum | qRT-PCR | HC | Unclear | 0.929 | 89.06 | 90 | 31032916 |
LncRNA is down-regulated in malignant tumors
LncRNAs such as AFAP1-AS1, ADAMTSP-AS2, and GAS5 with suppressed expression levels in lung cancer are rarely used as diagnostic markers for other malignant tumors. Esfandi et al. [126] evaluated the clinical value of lncRNA AFAP1-AS1 in gastric cancer and confirmed that AFAP1-AS1 is down-regulated in gastric cancer compared to para-carcinoma tissues, and could therefore, be used as a diagnostic biomarker. Chen et al. [127] evaluated the expression and diagnostic significance of the two new ceRNAs in esophageal squamous cell carcinoma (ESCC). CADM2 and ADAMTS9-AS2 were under-expressed in ESCC compared to para-carcinoma tissues. Du et al. [128] evaluated the diagnostic efficacy of urine lncRNAs for bladder cancer (BC). They constructed a two-lncRNA panel (UC004COX.4, GAS5), and the area under the curve showed that the two-lncRNA panel had a high BC diagnostic accuracy. Permuth et al. [129] developed a NanoString nCounter® technology for the diagnosis of intraductal papillary mucinous neoplasms (IPMNs) cases for cystic pancreatic ductal adenocarcinoma (PDAC) precursors. They found that lncRNA GAS5 expression was suppressed when compared to the healthy control group, and that two lncRNAs (GAS5 (low expression) and SRA (high expression) could distinguish IPMN cases from the non-diseased control group (Table 2).
Future outlook
Early screening and diagnosis were an effective way for enhancing survival outcomes for patients with malignant tumors. Studies have shown that, when compared to non-tumor tissues many lncRNAs are significantly up-regulated or down-regulated in tumor tissues compared with non-tumor tissues, and their differences in expression have a clinical diagnostic value. Novel diagnostic lncRNAs play a significant role in the early screening and of malignant tumors, which improves prognosis. Most studies have combined lncRNAs with micro-RNA and other tumor markers to improve their diagnostic performance. However, more comprehensive studies are still needed to clarify the diagnostic value of lncRNAs in cancers. Studies have proposed various clinical applications for these molecules. Even though some challenges are yet to be solved, lncRNAs play important roles in tumor development [130]. Large-scale and high-quality studies are needed to validate their clinical applications as cancer biomarkers.
Data availability statement
All data generated or analyzed during this study are included in this article.
Author Contributions
Li Li, Hai tao Wei. designed the experiments and wrote draft of the manuscript; Yi wei, Zhang, Shi zhe, Zhao acquired and analyze data; Guo wei, Che, Yun, Wang and Long qi, Chen made critical revision of the manuscript for intellectual content. All authors provided critical feedback and helped shape the research, analysis, and manuscript.
Conflicts of Interest
The authors declare that there is no conflict of interests. No animal or human studies were carried out by the authors for this article.
Funding
This study was supported by Program for Scientific and Technological Research of Henan Education Department (No.18B320012, No.18B320014).
References
- 1. Li J, Liang R, Song C, Xiang Y, Liu Y. Prognostic and clinicopathological significance of long non-coding RNA in glioma. Neurosurg Rev. 2020; 43:1–8. https://doi.org/10.1007/s10143-018-0965-x [PubMed]
- 2. Bhan A, Soleimani M, Mandal SS. Long Noncoding RNA and Cancer: A New Paradigm. Cancer Res. 2017; 77:3965–81. https://doi.org/10.1158/0008-5472.CAN-16-2634 [PubMed]
- 3. Chen T, Wang H, Yang P, He ZY. Prognostic role of long noncoding RNA NEAT1 in various carcinomas: a meta-analysis. Onco Targets Ther. 2017; 10:993–1000. https://doi.org/10.2147/OTT.S128588 [PubMed]
- 4. Zhang L, Fang F, He X. Long noncoding RNA TP73-AS1 promotes non-small cell lung cancer progression by competitively sponging miR-449a/EZH2. Biomed Pharmacother. 2018; 104:705–11. https://doi.org/10.1016/j.biopha.2018.05.089 [PubMed]
- 5. Tang H, Zhao L, Li M, Li T, Hao Y. Investigation of LINC00342 as a poor prognostic biomarker for human patients with non-small cell lung cancer. J Cell Biochem. 2019; 120:5055–61. https://doi.org/10.1002/jcb.27782 [PubMed]
- 6. Gupta C, Su J, Zhan M, Stass SA, Jiang F. Sputum long non-coding RNA biomarkers for diagnosis of lung cancer. Cancer Biomark. 2019; 26:219–27. https://doi.org/10.3233/CBM-190161 [PubMed]
- 7. Marques AC, Ponting CP. Intergenic lncRNAs and the evolution of gene expression. Curr Opin Genet Dev. 2014; 27:48–53. https://doi.org/10.1016/j.gde.2014.03.009 [PubMed]
- 8. Peng Z, Liu C, Wu M. New insights into long noncoding RNAs and their roles in glioma. Mol Cancer. 2018; 17:61. https://doi.org/10.1186/s12943-018-0812-2 [PubMed]
- 9. Cheng Z, Bai Y, Wang P, Wu Z, Zhou L, Zhong M, Jin Q, Zhao J, Mao H, Mao H. Identification of long noncoding RNAs for the detection of early stage lung squamous cell carcinoma by microarray analysis. Oncotarget. 2017; 8:13329–37. https://doi.org/10.18632/oncotarget.14522 [PubMed]
- 10. Choudhari R, Sedano MJ, Harrison AL, Subramani R, Lin KY, Ramos EI, Lakshmanaswamy R, Gadad SS. Long noncoding RNAs in cancer: From discovery to therapeutic targets. Adv Clin Chem. 2020; 95:105–47. https://doi.org/10.1016/bs.acc.2019.08.003 [PubMed]
- 11. Tong J, Ma X, Yu H, Yang J. SNHG15: a promising cancer-related long noncoding RNA. Cancer Manag Res. 2019; 11:5961–69. https://doi.org/10.2147/CMAR.S208054 [PubMed]
- 12. Zhang H, Liang Y, Han S, Peng C, Li Y. Long Noncoding RNA and Protein Interactions: From Experimental Results to Computational Models Based on Network Methods. Int J Mol Sci. 2019; 20:1284. https://doi.org/10.3390/ijms20061284 [PubMed]
- 13. Gharesouran J, Taheri M, Sayad A, Ghafouri-Fard S, Mazdeh M, Omrani MD. The Growth Arrest-Specific Transcript 5 (GAS5) and Nuclear Receptor Subfamily 3 Group C Member 1 (NR3C1): Novel Markers Involved in Multiple Sclerosis. Int J Mol Cell Med. 2018; 7:102–10. https://doi.org/10.22088/IJMCM.BUMS.7.2.102 [PubMed]
- 14. Zheng L, Liu X, Chen P, Xiao W. Expression and role of lncRNAs in the regeneration of skeletal muscle following contusion injury. Exp Ther Med. 2019; 18:2617–27. https://doi.org/10.3892/etm.2019.7871 [PubMed]
- 15. Paul S, Dansithong W, Figueroa KP, Scoles DR, Pulst SM. Staufen1 links RNA stress granules and autophagy in a model of neurodegeneration. Nat Commun. 2018; 9:3648. https://doi.org/10.1038/s41467-018-06041-3 [PubMed]
- 16. Arshi A, Sharifi FS, Khorramian Ghahfarokhi M, Faghih Z, Doosti A, Ostovari S, Mahmoudi Maymand E, Ghahramani Seno MM. Expression Analysis of MALAT1, GAS5, SRA, and NEAT1 lncRNAs in Breast Cancer Tissues from Young Women and Women over 45 Years of Age. Mol Ther Nucleic Acids. 2018; 12:751–57. https://doi.org/10.1016/j.omtn.2018.07.014 [PubMed]
- 17. Zhang Y, Wang F, Chen G, He R, Yang L. LncRNA MALAT1 promotes osteoarthritis by modulating miR-150-5p/AKT3 axis. Cell Biosci. 2019; 9:54. https://doi.org/10.1186/s13578-019-0302-2 [PubMed]
- 18. He Y, Luo Y, Liang B, Ye L, Lu G, He W. Potential applications of MEG3 in cancer diagnosis and prognosis. Oncotarget. 2017; 8:73282–95. https://doi.org/10.18632/oncotarget.19931 [PubMed]
- 19. Novikova MV, Khromova NV, Kopnin PB. Components of the Hepatocellular Carcinoma Microenvironment and Their Role in Tumor Progression. Biochemistry (Mosc). 2017; 82:861–73. https://doi.org/10.1134/S0006297917080016 [PubMed]
- 20. Huntley MA, Srinivasan K, Friedman BA, Wang TM, Yee AX, Wang Y, Kaminker JS, Sheng M, Hansen DV, Hanson JE. Genome-Wide Analysis of Differential Gene Expression and Splicing in Excitatory Neurons and Interneuron Subtypes. J Neurosci. 2020; 40:958–73. https://doi.org/10.1523/JNEUROSCI.1615-19.2019 [PubMed]
- 21. Olgun G, Sahin O, Tastan O. Discovering lncRNA mediated sponge interactions in breast cancer molecular subtypes. BMC Genomics. 2018; 19:650. https://doi.org/10.1186/s12864-018-5006-1 [PubMed]
- 22. Moretti F, Thermann R, Hentze MW. Mechanism of translational regulation by miR-2 from sites in the 5' untranslated region or the open reading frame. RNA. 2010; 16:2493–502. https://doi.org/10.1261/rna.2384610 [PubMed]
- 23. Hu X, Feng Y, Zhang D, Zhao SD, Hu Z, Greshock J, Zhang Y, Yang L, Zhong X, Wang LP, Jean S, Li C, Huang Q, et al. A functional genomic approach identifies FAL1 as an oncogenic long noncoding RNA that associates with BMI1 and represses p21 expression in cancer. Cancer Cell. 2014; 26:344–57. https://doi.org/10.1016/j.ccr.2014.07.009 [PubMed]
- 24. Li L, Wang Y, Song G, Zhang X, Gao S, Liu H. HOX cluster-embedded antisense long non-coding RNAs in lung cancer. Cancer Lett. 2019; 450:14–21. https://doi.org/10.1016/j.canlet.2019.02.036 [PubMed]
- 25. Lin S, Zhang R, An X, Li Z, Fang C, Pan B, Chen W, Xu G, Han W. LncRNA HOXA-AS3 confers cisplatin resistance by interacting with HOXA3 in non-small-cell lung carcinoma cells. Oncogenesis. 2019; 8:60. https://doi.org/10.1038/s41389-019-0170-y [PubMed]
- 26. Paulus JK, Kourelis J, van der Hoorn RAL. Bodyguards: Pathogen-Derived Decoys That Protect Virulence Factors. Trends Plant Sci. 2017; 22:355–57. https://doi.org/10.1016/j.tplants.2017.03.004 [PubMed]
- 27. Wang R, Ma Z, Feng L, Yang Y, Tan C, Shi Q, Lian M, He S, Ma H, Fang J. LncRNA MIR31HG targets HIF1A and P21 to facilitate head and neck cancer cell proliferation and tumorigenesis by promoting cell-cycle progression. Mol Cancer. 2018; 17:162. https://doi.org/10.1186/s12943-018-0916-8 [PubMed]
- 28. Yan J, Dutta B, Hee YT, Chng WJ. Towards understanding of PRC2 binding to RNA. RNA Biol. 2019; 16:176–84. https://doi.org/10.1080/15476286.2019.1565283 [PubMed]
- 29. Tang Q, Hann SS. HOTAIR: An Oncogenic Long Non-Coding RNA in Human Cancer. Cell Physiol Biochem. 2018; 47:893–913. https://doi.org/10.1159/000490131 [PubMed]
- 30. Hou L, Heilbronner U, Degenhardt F, Adli M, Akiyama K, Akula N, Ardau R, Arias B, Backlund L, Banzato CEM, Benabarre A, Bengesser S, Bhattacharjee AK, et al. Genetic variants associated with response to lithium treatment in bipolar disorder: a genome-wide association study. Lancet. 2016; 387:1085–93. https://doi.org/10.1016/S0140-6736(16)00143-4 [PubMed]
- 31. Shallis RM, Ahmad R, Zeidan AM. Aplastic anemia: Etiology, molecular pathogenesis, and emerging concepts. Eur J Haematol. 2018; 101:711–20. https://doi.org/10.1111/ejh.13153 [PubMed]
- 32. Arish N, Petukhov D, Wallach-Dayan SB. The Role of Telomerase and Telomeres in Interstitial Lung Diseases: From Molecules to Clinical Implications. Int J Mol Sci. 2019; 20:2996. https://doi.org/10.3390/ijms20122996 [PubMed]
- 33. Kashyap MP, Sinha R, Mukhtar MS, Athar M. Epigenetic regulation in the pathogenesis of non-melanoma skin cancer. Semin Cancer Biol. 2020. [Epub ahead of print]. https://doi.org/10.1016/j.semcancer.2020.11.009 [PubMed]
- 34. Jo SS, Choi SS. Enrichment of rare alleles within epigenetic chromatin marks in the first intron. Genomics Inform. 2019; 17:e9. https://doi.org/10.5808/GI.2019.17.1.e9 [PubMed]
- 35. Cabot B, Cabot RA. Chromatin remodeling in mammalian embryos. Reproduction. 2018; 155:R147–58. https://doi.org/10.1530/REP-17-0488 [PubMed]
- 36. Pollack IF, Agnihotri S, Broniscer A. Childhood brain tumors: current management, biological insights, and future directions. J Neurosurg Pediatr. 2019; 23:261–73. https://doi.org/10.3171/2018.10.PEDS18377 [PubMed]
- 37. Luo CL, Xu ZG, Chen H, Ji J, Wang YH, Hu W, Wang K, Zhang WW, Yuan CH, Wang FB. LncRNAs and EGFRvIII sequestered in TEPs enable blood-based NSCLC diagnosis. Cancer Manag Res. 2018; 10:1449–59. https://doi.org/10.2147/CMAR.S164227 [PubMed]
- 38. Pan LJ, Zhong TF, Tang RX, Li P, Dang YW, Huang SN, Chen G. Upregulation and clinicopathological significance of long non-coding NEAT1 RNA in NSCLC tissues. Asian Pac J Cancer Prev. 2015; 16:2851–55. https://doi.org/10.7314/apjcp.2015.16.7.2851 [PubMed]
- 39. Ambele MA, Pepper MS. Identification of transcription factors potentially involved in human adipogenesis in vitro. Mol Genet Genomic Med. 2017; 5:210–22. https://doi.org/10.1002/mgg3.269 [PubMed]
- 40. Zheng P, Yin Z, Wu Y, Xu Y, Luo Y, Zhang TC. LncRNA HOTAIR promotes cell migration and invasion by regulating MKL1 via inhibition miR206 expression in HeLa cells. Cell Commun Signal. 2018; 16:5. https://doi.org/10.1186/s12964-018-0216-3 [PubMed]
- 41. Li S, Chen YC. Abstract 4788: Enhancer of zeste homolog 2 couples with HOTAIR to inhibit tumor suppressor miR-34a in human pancreatic ductal adenocarcinoma. Cancer Res. 2015 (Suppl 15); 75:4788. https://doi.org/10.1158/1538-7445.AM2015-4788
- 42. Parfett CL, Desaulniers D. A Tox21 Approach to Altered Epigenetic Landscapes: Assessing Epigenetic Toxicity Pathways Leading to Altered Gene Expression and Oncogenic Transformation In Vitro. Int J Mol Sci. 2017; 18:1179. https://doi.org/10.3390/ijms18061179 [PubMed]
- 43. Chan JJ, Tay Y. Noncoding RNA:RNA Regulatory Networks in Cancer. Int J Mol Sci. 2018; 19:1310. https://doi.org/10.3390/ijms19051310 [PubMed]
- 44. Liu L, Liao JZ, He XX, Li PY. The role of autophagy in hepatocellular carcinoma: friend or foe. Oncotarget. 2017; 8:57707–22. https://doi.org/10.18632/oncotarget.17202 [PubMed]
- 45. Timmins MA, Wagner SD, Ahearne MJ. The new biology of PTCL-NOS and AITL: current status and future clinical impact. Br J Haematol. 2020; 189:54–66. https://doi.org/10.1111/bjh.16428 [PubMed]
- 46. Tang X, Qiao X, Chen C, Liu Y, Zhu J, Liu J. Regulation Mechanism of Long Noncoding RNAs in Colon Cancer Development and Progression. Yonsei Med J. 2019; 60:319–25. https://doi.org/10.3349/ymj.2019.60.4.319 [PubMed]
- 47. Lee SH, Zhu C, Peng Y, Johnson DT, Lehmann L, Sun Z. Identification of a novel role of ZMIZ2 protein in regulating the activity of the Wnt/β-catenin signaling pathway. J Biol Chem. 2013; 288:35913–24. https://doi.org/10.1074/jbc.M113.529727 [PubMed]
- 48. Redelman-Sidi G, Binyamin A, Gaeta I, Palm W, Thompson CB, Romesser PB, Lowe SW, Bagul M, Doench JG, Root DE, Glickman MS. The Canonical Wnt Pathway Drives Macropinocytosis in Cancer. Cancer Res. 2018; 78:4658–70. https://doi.org/10.1158/0008-5472.CAN-17-3199 [PubMed]
- 49. Ahmad R, Kumar B, Chen Z, Chen X, Müller D, Lele SM, Washington MK, Batra SK, Dhawan P, Singh AB. Loss of claudin-3 expression induces IL6/gp130/Stat3 signaling to promote colon cancer malignancy by hyperactivating Wnt/β-catenin signaling. Oncogene. 2017; 36:6592–604. https://doi.org/10.1038/onc.2017.259 [PubMed]
- 50. Jia C, Li H, Fu D, Lan Y. GFAT1/HBP/O-GlcNAcylation Axis Regulates β-Catenin Activity to Promote Pancreatic Cancer Aggressiveness. Biomed Res Int. 2020; 2020:1921609. https://doi.org/10.1155/2020/1921609 [PubMed]
- 51. Ghandadi M, Valadan R, Mohammadi H, Akhtari J, Khodashenas S, Ashari S. Wnt-β-catenin Signaling Pathway, the Achilles' Heels of Cancer Multidrug Resistance. Curr Pharm Des. 2019; 25:4192–207. https://doi.org/10.2174/1381612825666191112142943 [PubMed]
- 52. Chiabrando D, Bertino F, Tolosano E. Hereditary Ataxia: A Focus on Heme Metabolism and Fe-S Cluster Biogenesis. Int J Mol Sci. 2020; 21:3760. https://doi.org/10.3390/ijms21113760 [PubMed]
- 53. Wei S, Liu J, Li X, Liu X. Repression of lncRNA-SVUGP2 mediated by EZH2 contributes to the development of non-small cell lung cancer via brisking Wnt/β-catenin signal. Artif Cells Nanomed Biotechnol. 2019; 47:3400–09. https://doi.org/10.1080/21691401.2019.1648279 [PubMed]
- 54. Zhu ZJ, He JK. TINCR facilitates non-small cell lung cancer progression through BRAF-activated MAPK pathway. Biochem Biophys Res Commun. 2018; 497:971–77. https://doi.org/10.1016/j.bbrc.2018.02.059 [PubMed]
- 55. Li B, Xu N, Wan Z, Ma L, Li H, Cai W, Chen X, Huang Z, He Z. Isobavachalcone exerts anti-proliferative and pro-apoptotic effects on human liver cancer cells by targeting the ERKs/RSK2 signaling pathway. Oncol Rep. 2019; 41:3355–66. https://doi.org/10.3892/or.2019.7090 [PubMed]
- 56. Busquets O, Ettcheto M, Cano A, R Manzine P, Sánchez-Lopez E, Espinosa-Jiménez T, Verdaguer E, Dario Castro-Torres R, Beas-Zarate C, X Sureda F, Olloquequi J, Auladell C, Folch J, Camins A. Role of c-Jun N-Terminal Kinases (JNKs) in Epilepsy and Metabolic Cognitive Impairment. Int J Mol Sci. 2019; 21:255. https://doi.org/10.3390/ijms21010255 [PubMed]
- 57. Kojonazarov B, Novoyatleva T, Boehm M, Happe C, Sibinska Z, Tian X, Sajjad A, Luitel H, Kriechling P, Posern G, Evans SM, Grimminger F, Ghofrani HA, et al. p38 MAPK Inhibition Improves Heart Function in Pressure-Loaded Right Ventricular Hypertrophy. Am J Respir Cell Mol Biol. 2017; 57:603–14. https://doi.org/10.1165/rcmb.2016-0374OC [PubMed]
- 58. Ouyang KX, Zou R, Liang J, Bai ZB, Li ZQ, Zhao JJ. TUC338 Overexpression Leads to Enhanced Proliferation and Reduced Apoptosis in Tongue Squamous Cell Carcinoma Cells In Vitro. J Oral Maxillofac Surg. 2017; 75:423–28. https://doi.org/10.1016/j.joms.2016.08.009 [PubMed]
- 59. Jin W, Chen L, Cai X, Zhang Y, Zhang J, Ma D, Cai X, Fu T, Yu Z, Yu F, Chen G. Long non-coding RNA TUC338 is functionally involved in sorafenib-sensitized hepatocarcinoma cells by targeting RASAL1. Oncol Rep. 2017; 37:273–80. https://doi.org/10.3892/or.2016.5248 [PubMed]
- 60. Ghafouri-Fard S, Dashti S, Taheri M, Omrani MD. TINCR: An lncRNA with dual functions in the carcinogenesis process. Noncoding RNA Res. 2020; 5:109–15. https://doi.org/10.1016/j.ncrna.2020.06.003 [PubMed]
- 61. Wang P, Chen D, Ma H, Li Y. LncRNA SNHG12 contributes to multidrug resistance through activating the MAPK/Slug pathway by sponging miR-181a in non-small cell lung cancer. Oncotarget. 2017; 8:84086–101. https://doi.org/10.18632/oncotarget.20475 [PubMed]
- 62. Zhang CN, Zhang JL, Huang Y, Ren HT, Guan SH, Zeng QH. Dibutyltin depressed immune functions via NF-κB, and JAK/STAT signaling pathways in zebrafish (Danio rerio). Environ Toxicol. 2018; 33:104–11. https://doi.org/10.1002/tox.22502 [PubMed]
- 63. Zhao JM, Cheng W, He XG, Liu YL, Wang FF, Gao YF. Long non-coding RNA PICART1 suppresses proliferation and promotes apoptosis in lung cancer cells by inhibiting JAK2/STAT3 signaling. Neoplasma. 2018; 65:779–89. https://doi.org/10.4149/neo_2018_171130N778 [PubMed]
- 64. Zhang N, Zeng Y, Du W, Zhu J, Shen D, Liu Z, Huang JA. The EGFR pathway is involved in the regulation of PD-L1 expression via the IL-6/JAK/STAT3 signaling pathway in EGFR-mutated non-small cell lung cancer. Int J Oncol. 2016; 49:1360–68. https://doi.org/10.3892/ijo.2016.3632 [PubMed]
- 65. Kamada R, Toguchi Y, Nomura T, Imagawa T, Sakaguchi K. Tetramer formation of tumor suppressor protein p53: Structure, function, and applications. Biopolymers. 2016; 106:598–612. https://doi.org/10.1002/bip.22772 [PubMed]
- 66. Zheng ZH, Wu DM, Fan SH, Zhang ZF, Chen GQ, Lu J. Upregulation of miR-675-5p induced by lncRNA H19 was associated with tumor progression and development by targeting tumor suppressor p53 in non-small cell lung cancer. J Cell Biochem. 2019; 120:18724–35. https://doi.org/10.1002/jcb.29182 [PubMed]
- 67. Du Y, Hao X, Liu X. Low expression of long noncoding RNA CDKN2B-AS1 in patients with idiopathic pulmonary fibrosis predicts lung cancer by regulating the p53-signaling pathway. Oncol Lett. 2018; 15:4912–18. https://doi.org/10.3892/ol.2018.7910 [PubMed]
- 68. Zhou H, Chen A, Shen J, Zhang X, Hou M, Li J, Chen J, Zou H, Zhang Y, Deng Q, She K, Shi X, He J. Long non-coding RNA LOC285194 functions as a tumor suppressor by targeting p53 in non-small cell lung cancer. Oncol Rep. 2019; 41:15–26. https://doi.org/10.3892/or.2018.6839 [PubMed]
- 69. Ma Y, Li Q, Du Y, Chen W, Zhao G, Liu X, Li H, Liu J, Shen Z, Ma L, Zhou Y. Oncogenic Genetic Alterations in Non-Small-Cell Lung Cancer (NSCLC) in Southwestern China. Cancer Manag Res. 2020; 12:10861–74. https://doi.org/10.2147/CMAR.S266069 [PubMed]
- 70. Xia P, Xu XY. PI3K/Akt/mTOR signaling pathway in cancer stem cells: from basic research to clinical application. Am J Cancer Res. 2015; 5:1602–09. [PubMed]
- 71. Liu Y, Wan WZ, Li Y, Zhou GL, Liu XG. Recent development of ATP-competitive small molecule phosphatidylinostitol-3-kinase inhibitors as anticancer agents. Oncotarget. 2017; 8:7181–200. https://doi.org/10.18632/oncotarget.12742 [PubMed]
- 72. Gao BB, Wang SX. LncRNA BC200 regulates the cell proliferation and cisplatin resistance in non-small cell lung cancer via PI3K/AKT pathway. Eur Rev Med Pharmacol Sci. 2019; 23:1093–101. https://doi.org/10.26355/eurrev_201902_16999 [PubMed]
- 73. Liu L, Zhou XY, Zhang JQ, Wang GG, He J, Chen YY, Huang C, Li L, Li SQ. LncRNA HULC promotes non-small cell lung cancer cell proliferation and inhibits the apoptosis by up-regulating sphingosine kinase 1 (SPHK1) and its downstream PI3K/Akt pathway. Eur Rev Med Pharmacol Sci. 2018; 22:8722–30. https://doi.org/10.26355/eurrev_201812_16637 [PubMed]
- 74. Zhang Y, Xiang C, Wang Y, Duan Y, Liu C, Jin Y, Zhang Y. lncRNA LINC00152 knockdown had effects to suppress biological activity of lung cancer via EGFR/PI3K/AKT pathway. Biomed Pharmacother. 2017; 94:644–51. https://doi.org/10.1016/j.biopha.2017.07.120 [PubMed]
- 75. Jiang N, Meng X, Mi H, Chi Y, Li S, Jin Z, Tian H, He J, Shen W, Tian H, Pan J, Fang S, Jin X, et al. Circulating lncRNA XLOC_009167 serves as a diagnostic biomarker to predict lung cancer. Clin Chim Acta. 2018; 486:26–33. https://doi.org/10.1016/j.cca.2018.07.026 [PubMed]
- 76. Zhu H, Zhang L, Yan S, Li W, Cui J, Zhu M, Xia N, Yang Y, Yuan J, Chen X, Luo J, Chen R, Xing R, et al. LncRNA16 is a potential biomarker for diagnosis of early-stage lung cancer that promotes cell proliferation by regulating the cell cycle. Oncotarget. 2017; 8:7867–77. https://doi.org/10.18632/oncotarget.13980 [PubMed]
- 77. Tan N, Li L, Bai L, Zhao K. [Expression of Serum LncRNA HOTAIR in Non-small Cell Lung Cancer and Its Clinical Significance]. Zhongguo Fei Ai Za Zhi. 2017; 20:402–06. https://doi.org/10.3779/j.issn.1009-3419.2017.06.06 [PubMed]
- 78. Lin Y, Leng Q, Zhan M, Jiang F. A Plasma Long Noncoding RNA Signature for Early Detection of Lung Cancer. Transl Oncol. 2018; 11:1225–31. https://doi.org/10.1016/j.tranon.2018.07.016 [PubMed]
- 79. Zhang RM, Tang T, Yu HM, Yao XD. LncRNA DLX6-AS1/miR-129-5p/DLK1 axis aggravates stemness of osteosarcoma through Wnt signaling. Biochem Biophys Res Commun. 2018; 507:260–66. https://doi.org/10.1016/j.bbrc.2018.11.019 [PubMed]
- 80. Li X, Zhang Z, Jiang H, Li Q, Wang R, Pan H, Niu Y, Liu F, Gu H, Fan X, Gao J. Circular RNA circPVT1 Promotes Proliferation and Invasion Through Sponging miR-125b and Activating E2F2 Signaling in Non-Small Cell Lung Cancer. Cell Physiol Biochem. 2018; 51:2324–40. https://doi.org/10.1159/000495876 [PubMed]
- 81. Li W, Sun M, Zang C, Ma P, He J, Zhang M, Huang Z, Ding Y, Shu Y. Upregulated long non-coding RNA AGAP2-AS1 represses LATS2 and KLF2 expression through interacting with EZH2 and LSD1 in non-small-cell lung cancer cells. Cell Death Dis. 2016; 7:e2225. https://doi.org/10.1038/cddis.2016.126 [PubMed]
- 82. Wang HM, Lu JH, Chen WY, Gu AQ. Upregulated lncRNA-UCA1 contributes to progression of lung cancer and is closely related to clinical diagnosis as a predictive biomarker in plasma. Int J Clin Exp Med. 2015; 8:11824–30. [PubMed]
- 83. Zhang PP, Wang YQ, Weng WW, Nie W, Wu Y, Deng Y, Wei P, Xu MD, Wang CF. Linc00152 promotes Cancer Cell Proliferation and Invasion and Predicts Poor Prognosis in Lung adenocarcinoma. J Cancer. 2017; 8:2042–50. https://doi.org/10.7150/jca.18852 [PubMed]
- 84. Jing X, Lin J, Wang H, Tian L, Tian R, Zhang Y, Chen X, Zhang J. Prognostic value of lncRNA SOX2OT for Chinese cancer patients: A meta-analysis. PLoS One. 2017; 12:e0176889. https://doi.org/10.1371/journal.pone.0176889 [PubMed]
- 85. Tang Q, Ni Z, Cheng Z, Xu J, Yu H, Yin P. Three circulating long non-coding RNAs act as biomarkers for predicting NSCLC. Cell Physiol Biochem. 2015; 37:1002–09. https://doi.org/10.1159/000430226 [PubMed]
- 86. Tao Y, Tang Y, Yang Z, Wu F, Wang L, Yang L, Lei L, Jing Y, Jiang X, Jin H, Bai Y, Zhang L. Exploration of Serum Exosomal LncRNA TBILA and AGAP2-AS1 as Promising Biomarkers for Diagnosis of Non-Small Cell Lung Cancer. Int J Biol Sci. 2020; 16:471–82. https://doi.org/10.7150/ijbs.39123 [PubMed]
- 87. Tang Z, Xie H, Heier C, Huang J, Zheng Q, Eichmann TO, Schoiswohl G, Ni J, Zechner R, Ni S, Hao H. Enhanced monoacylglycerol lipolysis by ABHD6 promotes NSCLC pathogenesis. EBioMedicine. 2020; 53:102696. https://doi.org/10.1016/j.ebiom.2020.102696 [PubMed]
- 88. Wan N, Zhang TT, Hua SH, Lu ZL, Ji B, Li LX, Lu LQ, Huang WJ, Jiang J, Li J. Cost-effectiveness analysis of pembrolizumab plus chemotherapy with PD-L1 test for the first-line treatment of NSCLC. Cancer Med. 2020; 9:1683–93. https://doi.org/10.1002/cam4.2793 [PubMed]
- 89. Tantai J, Hu D, Yang Y, Geng J. Combined identification of long non-coding RNA XIST and HIF1A-AS1 in serum as an effective screening for non-small cell lung cancer. Int J Clin Exp Pathol. 2015; 8:7887–95. [PubMed]
- 90. Wang Z, Li J, Wu Y, Wang H. Combined identification of LncRNA CCAT1 and SOX2OT in serum as an effective screening for non-small cell lung cancer. Int J Clin Exp Pathol. 2019; 12:3975–81. [PubMed]
- 91. Hu Q, Rix LLR, Li X, Welsh EA, Fang B, Yun S, Kroeger J, Lawrence HR, Marusyk A, Koomen JM, Haura EB, Rix U. Dissecting the landscape of CAF-mediated drug resistance mechanisms in ALK-rearranged NSCLC. Eur J Cancer. 2020; 138:S48. https://doi.org/10.1016/S0959-8049(20)31205-3
- 92. Luo J, Li Q, Pan J, Li L, Fang L, Zhang Y. Expression level of long noncoding RNA H19 in plasma of patients with nonsmall cell lung cancer and its clinical significance. J Cancer Res Ther. 2018; 14:860–63. https://doi.org/10.4103/jcrt.JCRT_733_17 [PubMed]
- 93. Wang S, Han C, Liu T, Ma Z, Qiu M, Wang J, Zheng X, Xu W, Xia W, Xu Y, Hu J, Xu L, Yin R. LncRNA FAM83H-AS1 Amplification is Associated With a Poor Prognosis in Lung Adenocarcinoma and Can Serve as A Therapeutic Target. Research Square. 2020. [Preprint]. https://doi.org/10.21203/rs.3.rs-45692/v1
- 94. Liu S, Liu Y, Lu Q, Zhou X, Chen L, Liang W. The lncRNA TUG1 promotes epithelial ovarian cancer cell proliferation and invasion via the WNT/β-catenin pathway. Onco Targets Ther. 2018; 11:6845–51. https://doi.org/10.2147/OTT.S167900 [PubMed]
- 95. Gao S, Xu X, Wang Y, Zhang W, Wang X. Diagnostic utility of plasma lncRNA small nucleolar RNA host gene 1 in patients with hepatocellular carcinoma. Mol Med Rep. 2018; 18:3305–13. https://doi.org/10.3892/mmr.2018.9336 [PubMed]
- 96. Yin Q, Shen X, Cui X, Ju S. Elevated serum lncRNA TUG1 levels are a potential diagnostic biomarker of multiple myeloma. Exp Hematol. 2019; 79:47–55.e2. https://doi.org/10.1016/j.exphem.2019.10.002 [PubMed]
- 97. Jing W, Gao S, Zhu M, Luo P, Jing X, Chai H, Tu J. Potential diagnostic value of lncRNA SPRY4-IT1 in hepatocellular carcinoma. Oncol Rep. 2016; 36:1085–92. https://doi.org/10.3892/or.2016.4859 [PubMed]
- 98. Zhou X, Yin C, Dang Y, Ye F, Zhang G. Identification of the long non-coding RNA H19 in plasma as a novel biomarker for diagnosis of gastric cancer. Sci Rep. 2015; 5:11516. https://doi.org/10.1038/srep11516 [PubMed]
- 99. Pan Y, Chen H, Shen X, Wang X, Ju S, Lu M, Cong H. Serum level of long noncoding RNA H19 as a diagnostic biomarker of multiple myeloma. Clin Chim Acta. 2018; 480:199–205. https://doi.org/10.1016/j.cca.2018.02.019 [PubMed]
- 100. Zhong G, Wang K, Li J, Xiao S, Wei W, Liu J. Determination of Serum Exosomal H19 as a Noninvasive Biomarker for Breast Cancer Diagnosis. Onco Targets Ther. 2020; 13:2563–71. https://doi.org/10.2147/OTT.S243601 [PubMed]
- 101. Li J, Wang X, Tang J, Jiang R, Zhang W, Ji J, Sun B. HULC and Linc00152 Act as Novel Biomarkers in Predicting Diagnosis of Hepatocellular Carcinoma. Cell Physiol Biochem. 2015; 37:687–96. https://doi.org/10.1159/000430387 [PubMed]
- 102. Liu J, Tang X, Lv J, Peng X, Zhang K, Li C, Liu J, Wang G, Li Z. LncRNAs SNHG12 and LINC00152 were associated with progression of patients with papillary thyroid carcinoma. Future Oncol. 2019; 15:4167–79. https://doi.org/10.2217/fon-2019-0016 [PubMed]
- 103. He R, Hu Z, Wang Q, Luo W, Li J, Duan L, Zhu YS, Luo DX. The role of long non-coding RNAs in nasopharyngeal carcinoma: As systemic review. Oncotarget. 2017; 8:16075–83. https://doi.org/10.18632/oncotarget.14211 [PubMed]
- 104. Wen JJ, Ma YD, Yang GS, Wang GM. Analysis of circulating long non-coding RNA UCA1 as potential biomarkers for diagnosis and prognosis of osteosarcoma. Eur Rev Med Pharmacol Sci. 2017; 21:498–503. [PubMed]
- 105. Liu H, Ye D, Chen A, Tan D, Zhang W, Jiang W, Wang M, Zhang X. A pilot study of new promising non-coding RNA diagnostic biomarkers for early-stage colorectal cancers. Clin Chem Lab Med. 2019; 57:1073–83. https://doi.org/10.1515/cclm-2019-0052 [PubMed]
- 106. Yang J, Li C, Mudd A, Gu X. LncRNA PVT1 predicts prognosis and regulates tumor growth in prostate cancer. Biosci Biotechnol Biochem. 2017; 81:2301–06. https://doi.org/10.1080/09168451.2017.1387048 [PubMed]
- 107. Sun W, Wang L, Zhao D, Wang P, Li Y, Wang S. Four Circulating Long Non-Coding RNAs Act as Biomarkers for Predicting Cervical Cancer. Gynecol Obstet Invest. 2018; 83:533–39. https://doi.org/10.1159/000487595 [PubMed]
- 108. Chen Y, Du H, Bao L, Liu W. LncRNA PVT1 promotes ovarian cancer progression by silencing miR-214. Cancer Biol Med. 2018; 15:238–50. https://doi.org/10.20892/j.issn.2095-3941.2017.0174 [PubMed]
- 109. Luo H, Zheng W, Yan Q, Wang B, Ye B, Deng S, He F, Yang X, Wang X. A network meta-analysis: The best Yiqi Fuzheng Chinese herbal injections for use based on the NP regimen to treat NSCLC. Medicine (Baltimore). 2020; 99:e20500. https://doi.org/10.1097/MD.0000000000020500 [PubMed]
- 110. Wang J, Peng Z, Dong Y, He D, Duan C, Li X. [Expression of two transcript variants of long noncoding RNA C6orf176 in non-small cell lung cancer and its clinical significance]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2016; 41:560–65. [PubMed]
- 111. Liang W, Lv T, Shi X, Liu H, Zhu Q, Zeng J, Yang W, Yin J, Song Y. Circulating long noncoding RNA GAS5 is a novel biomarker for the diagnosis of nonsmall cell lung cancer. Medicine (Baltimore). 2016; 95:e4608. https://doi.org/10.1097/MD.0000000000004608 [PubMed]
- 112. Kamel LM, Atef DM, Mackawy AMH, Shalaby SM, Abdelraheim N. Circulating long non-coding RNA GAS5 and SOX2OT as potential biomarkers for diagnosis and prognosis of non-small cell lung cancer. Biotechnol Appl Biochem. 2019; 66:634–42. https://doi.org/10.1002/bab.1764 [PubMed]
- 113. Hao XZ, Yang K. LncRNA MAGI2-AS3 suppresses the proliferation and invasion of non-small cell lung carcinoma through miRNA-23a-3p/PTEN axis. Eur Rev Med Pharmacol Sci. 2019; 23:7399–407. https://doi.org/10.26355/eurrev_201909_18848 [PubMed]
- 114. Zeng Z, Zhao G, Rao C, Hua G, Yang M, Miao X, Ying J, Nie L. Knockdown of lncRNA ZFAS1-suppressed non-small cell lung cancer progression via targeting the miR-150-5p/HMGA2 signaling. J Cell Biochem. 2020; 121:3814–24. https://doi.org/10.1002/jcb.29542 [PubMed]
- 115. Tan Q, Yu Y, Li N, Jing W, Zhou H, Qiu S, Liang C, Yu M, Tu J. Identification of long non-coding RNA 00312 and 00673 in human NSCLC tissues. Mol Med Rep. 2017; 16:4721–29. https://doi.org/10.3892/mmr.2017.7196 [PubMed]
- 116. Wang H, Wang J, Liang CF, Zhou T. Expression of Long Non-Coding RNA PRAL as a Potential Biomarker for Diagnosis in Non-Small-Cell Lung Cancer Patients is Associated with the Inhibition of Cell Proliferation and Metastasis. Clin Lab. 2018; 64:1341–48. https://doi.org/10.7754/Clin.Lab.2018.171237 [PubMed]
- 117. Guo F, Yu F, Wang J, Li Y, Li Y, Li Z, Zhou Q. Expression of MALAT1 in the peripheral whole blood of patients with lung cancer. Biomed Rep. 2015; 3:309–12. https://doi.org/10.3892/br.2015.422 [PubMed]
- 118. Scheffler M, Holzem A, Kron A, Nogova L, Ihle MA, von Levetzow C, Fassunke J, Wömpner C, Bitter E, Koleczko S, Abdulla DSY, Michels S, Fischer R, et al. Co-occurrence of targetable mutations in Non-small cell lung cancer (NSCLC) patients harboring MAP2K1 mutations. Lung Cancer. 2020; 144:40–48. https://doi.org/10.1016/j.lungcan.2020.04.020 [PubMed]
- 119. Chen Q, Zhu C, Jin Y, Si X, Jiao W, He W, Mao W, Li M, Luo G. Plasma Long Non-Coding RNA RP11-438N5.3 as a Novel Biomarker for Non-Small Cell Lung Cancer. Cancer Manag Res. 2020; 12:1513–21. https://doi.org/10.2147/CMAR.S237024 [PubMed]
- 120. Wang P, Lu S, Mao H, Bai Y, Ma T, Cheng Z, Zhang H, Jin Q, Zhao J, Mao H. Identification of biomarkers for the detection of early stage lung adenocarcinoma by microarray profiling of long noncoding RNAs. Lung Cancer. 2015; 88:147–53. https://doi.org/10.1016/j.lungcan.2015.02.009 [PubMed]
- 121. Jin D, Song Y, Chen Y, Zhang P. Identification of Three lncRNAs as Potential Predictive Biomarkers of Lung Adenocarcinoma. Biomed Res Int. 2020; 2020:7573689. https://doi.org/10.1155/2020/7573689 [PubMed]
- 122. Li L, Yin JY, He FZ, Huang MS, Zhu T, Gao YF, Chen YX, Zhou DB, Chen X, Sun LQ, Zhang W, Zhou HH, Liu ZQ. Long noncoding RNA SFTA1P promoted apoptosis and increased cisplatin chemosensitivity via regulating the hnRNP-U-GADD45A axis in lung squamous cell carcinoma. Oncotarget. 2017; 8:97476–89. https://doi.org/10.18632/oncotarget.22138 [PubMed]
- 123. Li S, Sun X, Miao S, Liu J, Jiao W. Differential protein-coding gene and long noncoding RNA expression in smoking-related lung squamous cell carcinoma. Thorac Cancer. 2017; 8:672–81. https://doi.org/10.1111/1759-7714.12510 [PubMed]
- 124. He J, Zhou X, Li L, Han Z. Long Noncoding MAGI2-AS3 Suppresses Several Cellular Processes of Lung Squamous Cell Carcinoma Cells by Regulating miR-374a/b-5p/CADM2 Axis. Cancer Manag Res. 2020; 12:289–302. https://doi.org/10.2147/CMAR.S232595 [PubMed]
- 125. Xiong Y, Zhang X, Lin Z, Xiong A, Xie S, Liang J, Zhang W. SFTA1P, LINC00968, GATA6-AS1, TBX5-AS1, and FEZF1-AS1 are crucial long non-coding RNAs associated with the prognosis of lung squamous cell carcinoma. Oncol Lett. 2019; 18:3985–93. https://doi.org/10.3892/ol.2019.10744 [PubMed]
- 126. Esfandi F, Taheri M, Namvar A, Oskooei VK, Ghafouri-Fard S. AFAP1 and its naturally occurring antisense RNA are downregulated in gastric cancer samples. Biomed Rep. 2019; 10:296–302. https://doi.org/10.3892/br.2019.1207 [PubMed]
- 127. Chen LP, Wang H, Zhang Y, Chen QX, Lin TS, Liu ZQ, Zhou YY. Robust analysis of novel mRNA-lncRNA cross talk based on ceRNA hypothesis uncovers carcinogenic mechanism and promotes diagnostic accuracy in esophageal cancer. Cancer Manag Res. 2018; 11:347–58. https://doi.org/10.2147/CMAR.S183310 [PubMed]
- 128. Du L, Jiang X, Duan W, Wang R, Wang L, Zheng G, Yan K, Wang L, Li J, Zhang X, Pan H, Yang Y, Wang C. Cell-free microRNA expression signatures in urine serve as novel noninvasive biomarkers for diagnosis and recurrence prediction of bladder cancer. Oncotarget. 2017; 8:40832–42. https://doi.org/10.18632/oncotarget.16586 [PubMed]
- 129. Permuth JB, Chen DT, Yoder SJ, Li J, Smith AT, Choi JW, Kim J, Balagurunathan Y, Jiang K, Coppola D, Centeno BA, Klapman J, Hodul P, et al. Linc-ing Circulating Long Non-coding RNAs to the Diagnosis and Malignant Prediction of Intraductal Papillary Mucinous Neoplasms of the Pancreas. Sci Rep. 2017; 7:10484. https://doi.org/10.1038/s41598-017-09754-5 [PubMed]
- 130. Schmitt AM, Chang HY. Long Noncoding RNAs in Cancer Pathways. Cancer Cell. 2016; 29:452–63. https://doi.org/10.1016/j.ccell.2016.03.010 [PubMed]

