Introduction
Diabetes mellitus (DM) is a serious metabolic disease with increasing complications and mortality associated with DM. According to epidemiological surveys, nearly 400 million diabetes patients are diagnosed annually in the world, of which 90–95% are type 2 diabetes mellitus (T2DM) [1]. It is expected that by 2035, the number of worldwide diabetic patients will reach 600 million [2]. Multiple serious complications are reported on T2DM, including CI [3]. Therefore, it is urgent to seek methods for treating DM complications. Significant cognitive decline is reportedly observed in DM patients, about 70% of which eventually develop into Alzheimer’s disease (AD). A much higher risk of AD is reported in DM patients [4, 5]. CI refers to the early stage of dementia, in which patients show cognitive dysfunction at the time of testing while retaining their basic ability to live [6]. Compared to subjects with normal blood glucose, the risk of CI in patients with diabetes is 0.5- times higher and the risk of dementia is 1- time higher [7]. Diabetes-related CI has a negative impact on learning ability, memory, attention, and executive ability, and even causes emotional disorders such as depression and anxiety [8, 9]. Inflammatory response is one of the important causes of CI in diabetes. Long-term hyperglycemia is a major feature of diabetes, which induces the inflammatory response by activating the NLRP3 inflammasome to promote the production of interleukin (IL)-1β [10]. Furthermore, the glycolysis intermediates are accumulated by persistent hyperglycemia to induce the accumulation of advanced glycation end products (AGEs), which further activate the NF-κB pathway to induce the secretion of a variety of inflammatory factors by microglia. By binding to the RAGE, AGEs induce the release of reactive oxygen species (ROS) to activate NF-κB signaling, which finally contributes to the excessive production of inflammatory factors [11, 12]. Therefore, controlling neuroinflammatory response may become an important direction for treating diabetes-related CI.
As an inhibitor of DDP-IV, teneligliptin is a novel antidiabetic drug that inhibits the degradation of glucagon-like peptide 1 (GLP-1) via repressing the activity of DDP-IV. It exerts its antidiabetic function by increasing the blood concentration of GLP-1 [13, 14]. Teneligliptin was first developed by Mitsubishi Tanabe Pharma and approved for treating T2DM in Japan in 2012, with its promising antidiabetic function proven in several clinical trials [15, 16]. DPP-4 inhibitors could be classified into peptidomimetic (i.e., sitagliptin, vildagliptin, saxagliptin, and anagliptin) and non-peptidomimetic (i.e., alogliptin and linagliptin) subtypes. Teneligliptin has a different structure and pharmacodynamic characteristics from other gliptins. These features could confer properties diverse from or additive to other DPP-4 inhibitors. Firstly, it is a potent, selective, and long-lasting inhibitor of DPP-4, and exhibits strong inhibitory activity via its J-shaped structure and ‘anchor lock domain’. Secondly, teneligliptin has high tissue distribution [17]. An X-ray co-crystal structure of teneligliptin with DPP-4 demonstrates that the key interaction occurs between the phenyl ring on the pyrazole and the S2 extensive subsite of DPP-4, which not only enhances the potency of the drug but also increases its selectivity [18]. Recently, several researches have claimed that teneligliptin shows prominent inhibitory effects against inflammation [19–21]. Also, teneligliptin was found to ameliorate high glucose-induced ER stress in endothelial cells [22]. However, the function of teneligliptin in diabetes-related CI remains uncertain. Herein, the preliminary investigation of teneligliptin against diabetes-related CI was conducted in db/db mice.
Materials and Methods
Animals and grouping
12 non-diabetic (db/m) and 12 db/db mice (7–9 weeks) were obtained from the Vital River (China). After one week of adaptive feeding, db/m mice and db/db mice were orally administered with teneligliptin (60 mg/kg/day) for 10 weeks. The mice were divided into four groups: db/m, db/m+ teneligliptin, db/db, and db/db+ teneligliptin.
The detection of serum levels of TC, TG, and LDL-C
The peripheral blood was collected to obtain the serum, followed by detecting the TC, TG, and LDL-C levels in the serum using the fully automatic biochemical analyzer (HITACHI, Japan).
Morris water maze test
The black maze was divided into 4 quadrants and the platform was placed in the 3rd quadrant. Water was poured into the maze to cover the platform, and differentiated by shapes and colors. The training was initiated by putting the mouse’s head toward the wall of the pool in the other 3 quadrants however it halted- when the mouse climbed to the platform. The escape latency was recorded as the time the mouse took to find the hidden underwater platform. On the 6th day of training, the mouse was put in the maze to test its spatial memory. The mouse was submerged in water for 60 s, and its spatial memory was assessed based on the time spent in the platform quadrant and the number of platform crossings.
Fear conditioning test
The experiment was divided into two stages: conditioned fear experimental training and situational conditioned fear experimental testing. The activity trajectory of the mouse was automatically tracked using the image automatic monitoring system (XR-XC404, Xinruan Information, China). The conditioned fear test was performed on the second day after the operation. Mice were placed in a sound-proof training box for 180 s, after which they received sound (30 s, 75 dB, 3 000 Hz) and plantar shock (2 s, 0.75 mA, and the last 2 s of shock before the end of the sound). 24 h after training, the situational fear conditioning test was performed. Mice were put back into the original experimental chamber without any stimulation, and the time of freezing (an inactive state with no other behavior except breathing) within 5 min was recorded.
Y-maze test
The equipment consisted of three identical arms (40 cm × 8.5 cm × 15 cm, angle 120°), designated as A, B, and C, respectively. Each mouse was put in the middle area of the maze at the beginning of the experiment. Subsequently, the mouse was allowed to probe freely for 8 mins, and the Noldus movement trajectory tracking system was used to record the time spent in the novel arm and the number of correct rotations (continuously entering 3 different arms, such as ABC, BCA, etc.). The percentage of alterations was calculated as (number of rotations/maximum number of rotations -2) ×100.
Enzyme-linked immunosorbent assay (ELISA) for the cytokine level and glutathione peroxidase (GSH-PX) activity detection
The mice were sacrificed in a sealed container collected with a CO2 tanker. The hippocampus of each mouse was collected and homogenized, followed by centrifugation and collection of the supernatant. 50 μL supernatant was diluted at a 1:1 ratio and loaded into the well. Then, 50 μL Biotin-labeled antibody was introduced and cultured for 60 min at 37°C, followed by removing the reagent and adding 80 μL horseradish peroxidase (HRP)-loaded secondary antibody. Following half an hour of culturing at 37°C, 50 μL TMB substrates were added and cultured at 37°C for 10 min, followed by loading 50 μL stop solution. Lastly, the optical density (OD) value was achieved using a microplate reader (MD, USA).
The measurement of malondialdehyde (MDA) level, Superoxide dismutase (SOD) activity in the hippocampus
The hippocampus was collected and homogenized, followed by centrifugation and collecting the supernatant. The MDA level (Jiancheng, China) and SOD activity (Solarbio, China) in the hippocampus were determined with a commercial kit using the TBA and WST-1 methods, respectively. The instructions were strictly followed.
Real-time polymerase chain reaction (RT-PCR) assay
Total RNAs were obtained from the hippocampus using the TRIzol reagent and were quantified with the ultraviolet spectrophotometer (Hach, USA). A cDNA synthesis kit (SolelyBio, China) was utilized for conducting the transcription from RNAs to cDNAs, followed by performing the PCR reaction utilizing an SYBR Premix Ex TaqII kit (Takara, Japan). The 2−ΔΔCt method was used for the calculation of gene levels. The primer sequences for the genes used in this experiment are listed as follows: IL-1β FW5′-TTCGA GGCAC AAGGC ACAA-3′; RV5′-CCATC ATTTC ACTGG CGAGC-3′; MCP-1 FW5′-GCTCA GCCAG ATGCA AT-3′; RV5′-GCTTG TCCAG GTGGT CCATG-3′; IL-6 FW5′-GACAA AGCCA GAGTC CTTCA GAGAG ATACA G-3′; RV5′-TTGGA TGGTC TTGGT CCTTA GCCAC-3′; β-actin-FW5′-CATGT ACGTT GCTAT CCAG GC-3′; RV5′-CTCCT TAATG TCACG CACGA T-3′.
Western blot analysis
The hippocampus was collected and lysed to obtain total proteins and quantified with the BCA method. After separation using the sodium dodecyl sulfate (SDS)-polyacrylamide gel (PAGE), proteins were delivered to the polyvinylidene fluoride (PVDF) membrane, followed by blocking in 5% skim milk. Primary antibodies against thioredoxin-interacting protein (TXNIP) (1:2000, Cat#abs136992, Absin, China), apoptosis-associated speck-like protein (ASC) (1:1000, Cat#abs155599, Absin, China), NLRP3 (1:2500, Cat#abs151715, Absin, China), C/EBP Homologous Protein (CHOP) (1:1000, Cat#abs131376, Absin, China), phosphorylated-PERK (1:1000, Cat#abs137056, Absin, China), PERK (1:1000, Cat#abs124201, Absin, China), phosphorylated inositol-requiring enzyme 1alpha (p-IRE1α) (1:1000, abs127778, Absin, China), IRE1α (1:1000, abs127778, Absin, China), ATF6 (1:2000, #65880, CST, USA), or β-actin (1:2000, Cat#8457CST, USA), the HRP-linked, Anti-rabbit IgG (1:2000, Cat#7074, CST, USA) were introduced and cultured at 4°C overnight. Subsequently, the HRP-linked, anti-rabbit IgG (1:2000, Cat#7074, CST, USA) was loaded and cultured for 90 min at 37°C. Lastly, ECL solution was utilized for the exposure of the bands and the ImageJ software was used for expression level analysis.
Statistical analysis
Data were listed as mean ± Standard Deviation (SD), which were analyzed using the one-way analysis of variance (ANOVA) method by Tukey’s test with the software SPSS (Version 24.0). P < 0.05 was considered statistically significant.
Data availability
The data is available upon reasonable request from the corresponding author.
Results
Teneligliptin alleviated dyslipidemia in diabetic mice
Firstly, the biochemical indexes were detected following administration. The TC level was maintained around 3.6 mM in the db/m and db/m+ teneligliptin groups and was signally increased to 6.18 mM in the db/db group, then markedly reduced to 4.92 mM by teneligliptin (Figure 1A). Furthermore, the TG levels in the db/m, db/m+ teneligliptin, db/db, and db/db+ teneligliptin groups were 1.25, 1.33, 3.46, and 2.52 mM, respectively (Figure 1B). Moreover, the LDL-C level was slightly altered in the db/m+ teneligliptin group and notably elevated in the db/db group but was dramatically declined in the db/db+ teneligliptin group (Figure 1C).
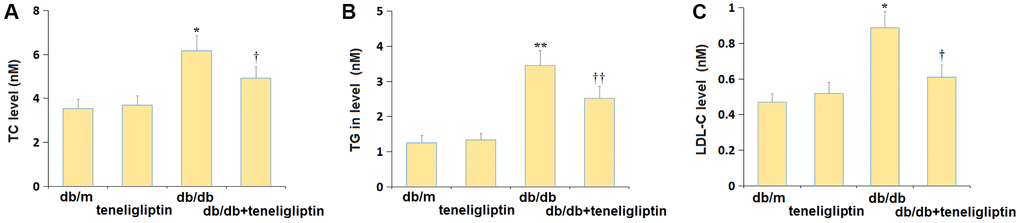
Figure 1. Teneligliptin improved the dyslipidemia in db/db mice. (A) Total cholesterol (TC) level; (B) Triglyceride (TG) in level; (C) Low-density lipoprotein cholesterol (LDL-C) level (*, **P < 0.05, 0.01 vs. db/db mice group; †, ††P < 0.05, 0.01 vs. Teneligliptin group).
Teneligliptin alleviated the behavioral dysfunction of diabetic mice in the Morris water maze test
The escape latency to reach the escape platform in the db/m, db/m+ teneligliptin, db/db, and db/db+ teneligliptin groups was 21.4, 22.7, 38.9, and 29.3 s, respectively (Figure 2A). The time spent in the platform (Figure 2B) was kept around 18.0 s in the db/m and db/m+ teneligliptin groups, and was markedly reduced to 10.8 s in diabetic mice, then signally increased to 16.2 s by teneligliptin. Furthermore, the average number of platform crossings in the db/m, db/m+ teneligliptin, db/db, and db/db+ teneligliptin groups was 3.5, 3.4, 1.3, and 2.5, respectively (Figure 2C).
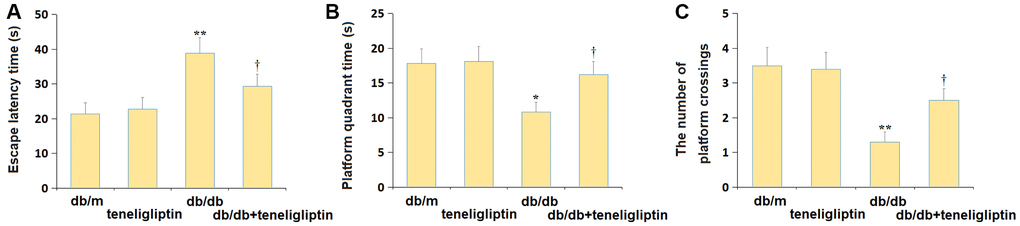
Figure 2. Teneligliptin alleviated the behavioral dysfunction of db/db mice in the Morris water maze test. (A) The escape latency to reach the escape platform; (B) The time spent in the platform quadrant; (C) The number of platform crossings (*, **P < 0.05, 0.01 vs. db/db mice group; †, ††P < 0.05, 0.01 vs. Teneligliptin group).
Teneligliptin mitigated the behavioral dysfunction of diabetic mice in the fear conditioning test
Subsequently, the behavior change in diabetic mice was confirmed using the fear conditioning test. The freezing index was minorly changed from 50.1% to 49.2% in the db/m+ teneligliptin group and largely decreased to 21.8% in the db/db group, then greatly reversed to 40.7% by teneligliptin (Figure 3).
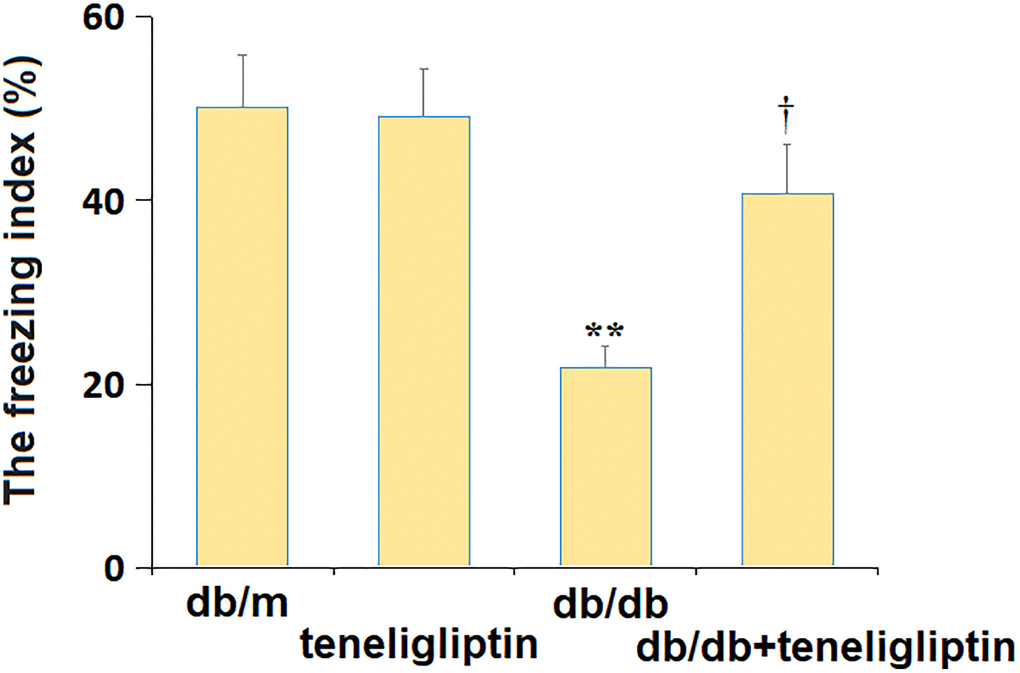
Figure 3. Teneligliptin mitigated the behavioral dysfunction of db/db mice in the fear conditioning test. The freezing index (%) (*, **P < 0.05, 0.01 vs. db/db mice group; †P < 0.05 vs. Teneligliptin group).
Teneligliptin ameliorated the behavioral dysfunction of diabetic mice in the Y-maze test
In the Y-maze test, the time spent in the new arm was minorly changed from 148.2 s to 152.4 s, largely declined to 75.4 s in diabetic mice, then dramatically increased to 107.2 s by teneligliptin (Figure 4A). Moreover, the percentage of alterations in the db/m, db/m+ teneligliptin, db/db, and db/db+ teneligliptin groups was 52.3%, 55.1%, 32.9%, and 44.7%, respectively (Figure 4B).
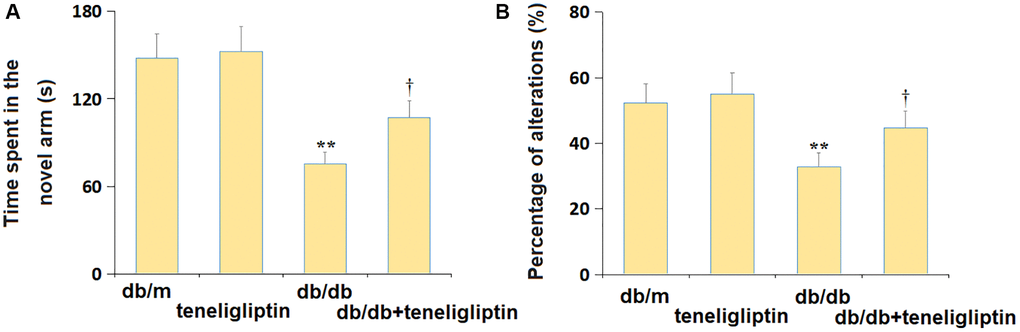
Figure 4. Teneligliptin ameliorated the behavioral dysfunction of db/db mice in the Y-maze test. (A) Time spent in the novel arm; (B) Percentage of alterations (%) (*, **P < 0.05, 0.01 vs. db/db mice group; †P < 0.05 vs. Teneligliptin group).
Teneligliptin repressed the production of inflammatory cytokines in diabetic mice
The state of neuroinflammation in the hippocampus was investigated. The gene levels of IL-1β, MCP-1, and IL-6 were mildly changed in the db/m+ teneligliptin group, markedly elevated in the db/db group, then memorably reduced in the db/db+ teneligliptin group (Figure 5A–5C). The IL-1β levels in the db/m, db/m+ teneligliptin, db/db, and db/db+ teneligliptin groups were 32.4, 33.8, 77.4, and 57.3 pg/mg protein, respectively (Figure 5D). The MCP-1 level was kept around 400.0 ng/mg protein in the db/m and db/m+ teneligliptin groups was markedly increased to 702.5 ng/mg protein in diabetic mice, then greatly decreased to 569.8 ng/mg protein by teneligliptin (Figure 5E). Furthermore, the IL-6 levels in the db/m, db/m+ teneligliptin, db/db, and db/db+ teneligliptin groups were 54.9, 56.3, 114.7, and 71.9 pg/mg protein, respectively (Figure 5F).
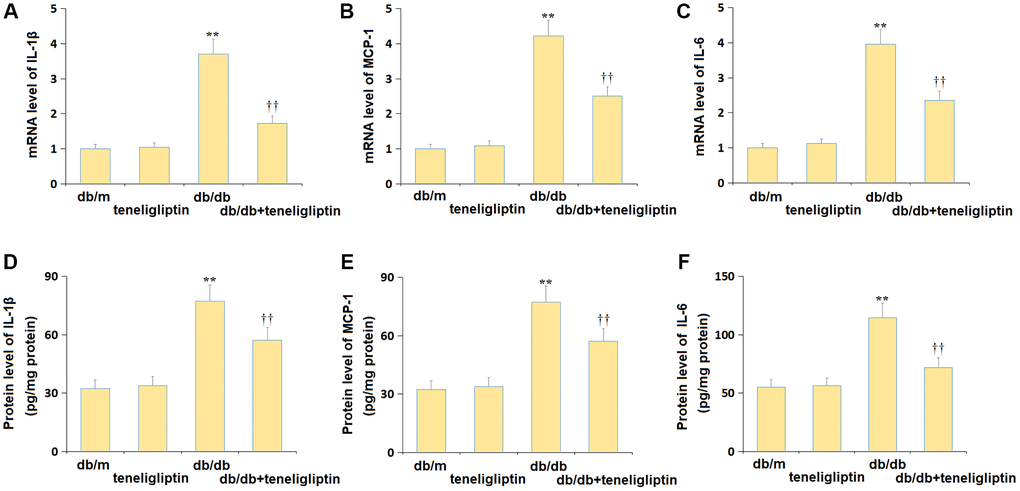
Figure 5. Teneligliptin repressed the production of inflammatory cytokines in the hippocampus of db/db mice. (A) mRNA level of IL-1β, (B) mRNA level of MCP-1, and (C) mRNA level of IL-6; (D) Protein level of IL-1β, (E) Protein level of MCP-1, and (F) Protein level of IL-6 (**P < 0.01 vs. db/db mice group; ††0.01 vs. Teneligliptin group).
Teneligliptin reversed the OS in diabetic mice
The OS state in the hippocampus of each mouse was further studied. The increased MDA level in diabetic mice was found markedly repressed by teneligliptin (Figure 6A). The SOD activity in the db/m, db/m+ teneligliptin, db/db, and db/db+ teneligliptin groups was 42.3, 43.7, 21.5, and 33.6 U/mg protein, respectively (Figure 6B). The GSH-PX activity was retained around 45.0 U/mg protein in the db/m and db/m+ teneligliptin groups was notably decreased to 17.8 U/mg protein in diabetic mice, then markedly reversed to 28.4 U/mg protein by teneligliptin (Figure 6C).
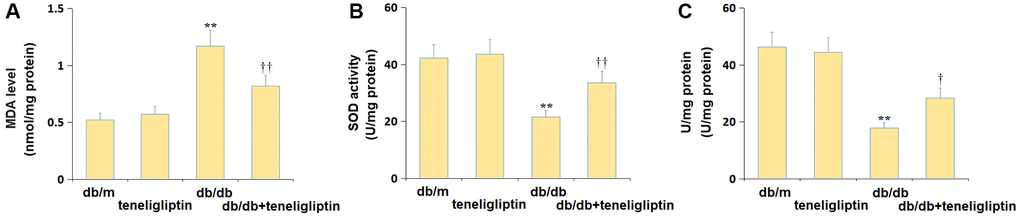
Figure 6. Teneligliptin reversed the oxidative stress in the hippocampus of db/db mice. (A) MDA level; (B) SOD activity; (C) GSH-PX activity (**P < 0.01 vs. db/db mice group; ††0.01 vs. Teneligliptin group).
Teneligliptin inhibited the NLRP3 inflammasome in diabetic mice
NLRP3 signaling has a claimed correlation to the development of diabetes-related CI [23]. Herein, the mildly altered TXNIP, ASC, and NLRP3 levels in the db/m+ teneligliptin group were markedly promoted in diabetic mice, and then signally repressed by teneligliptin (Figure 7A–7D).
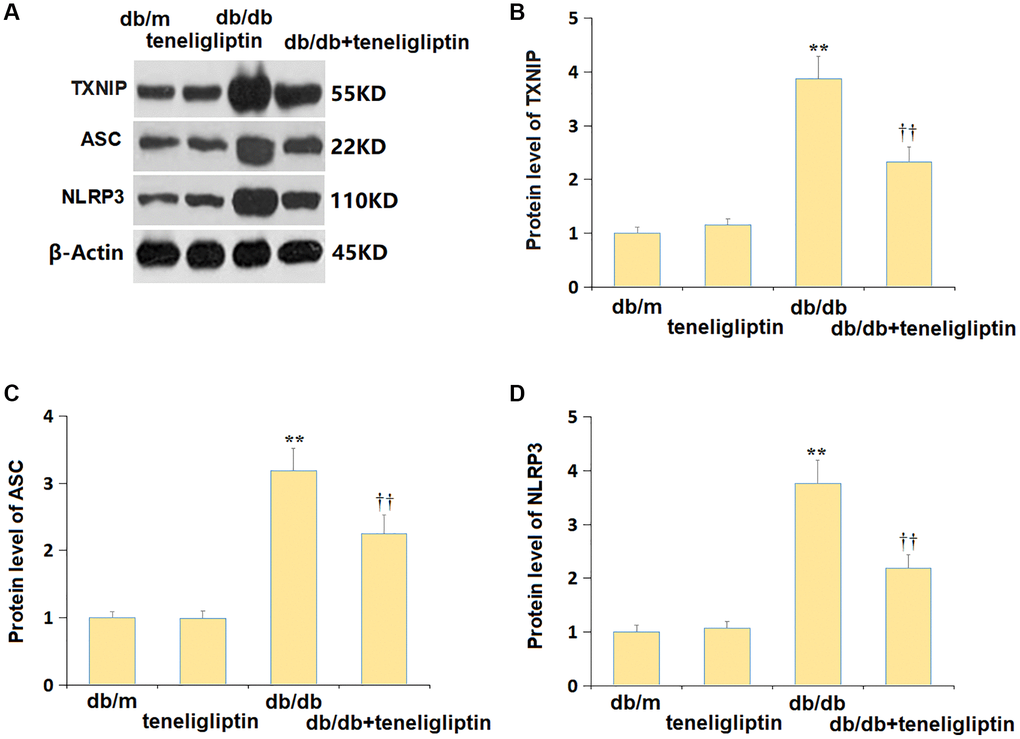
Figure 7. Teneligliptin inhibited the NLRP3 inflammasome in the hippocampus of db/db mice. (A) Protein level of genes were determined using western blots; (B) Protein level of TXNIP; (C) Protein level of ASC; (D) Protein level of NLRP3 (**P < 0.01 vs. db/db mice group; ††0.01 vs. Teneligliptin group).
Teneligliptin alleviated the ER stress in diabetic mice
ER stress is another important mechanism contributing to the pathogenesis of diabetes-related CI [24]. Herein, the levels of ER stress-related proteins, including GRP78, CHOP, p-PERK, p-IRE1α, and ATF6, were minorly altered in the db/m+ teneligliptin group, largely increased in the db/db group, then markedly suppressed in the db/db+ teneligliptin group (Figure 8A–8F).
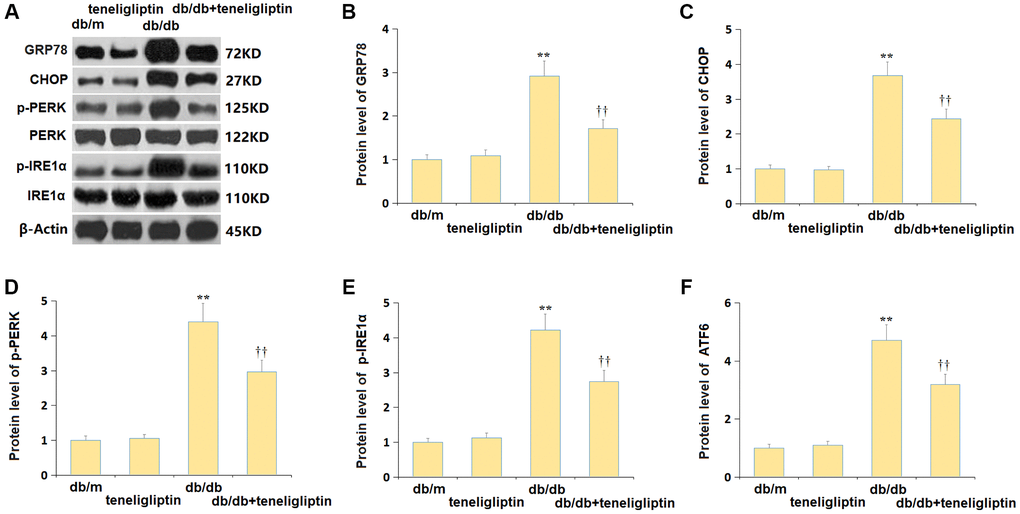
Figure 8. Teneligliptin alleviated the ER stress in the hippocampus of db/db mice. (A) Protein level of genes were detected by western blots; (B) Protein level of GRP78; (C) Protein level of CHOP; (D) Protein level of p-PERK; (E) Protein level of p-IRE1α; (F) Protein level of ATF6 (*, **P < 0.05, 0.01 vs. db/db mice group; †, ††P < 0.05, 0.01 vs. Teneligliptin group).
Discussion
DPP-4 inhibitors are potent drugs used to treat patients with T2DM [25, 26]. Inhibition of DPP-4 increases levels of biologically intact incretins, improving glucose metabolism through the upregulation of insulin secretion and the suppression of glucagon release [27]. Recently, several articles have reported on its neuroprotective effects in both in vivo and in vitro models [28, 22]. The inflammatory response is a critical mechanism in DM and diabetes-related CI [29]. Hyperglycemia and ROS are critical factors inducing an inflammatory response in diabetes-related CI patients. When an inflammatory response occurs, microglia in the hippocampus related to learning and memory function release a large number of inflammatory factors, which further contribute to neuronal degeneration [30]. The persistent occurrence of chronic inflammation changes the permeability of the blood-brain barrier (BBB) [31], and a large number of toxic and harmful substances cross the BBB and enter the brain, increasing the burden on the nervous system and causing neurodegenerative diseases. The secretion of pro-inflammatory factor IL-1β is found to induce neuroinflammation, which is the key to the pathogenesis of diabetes-related CI [32]. The levels of inflammatory factors in diabetes-related CI patients are dramatically higher than those in DM patients [29]. Herein, changed biochemical criteria observed in diabetic mice were consistent with data reported previously [33]. Furthermore, in diabetic mice, behavioral dysfunction was observed in the Morris water maze, the fear conditioning, and the Y-maze tests, in line with the research published by Wu [34]. After the administration of teneligliptin, biochemical criteria were markedly improved, accompanied by alleviation of the behavioral dysfunction, implying that teneligliptin exerted a promising protective function on CI in diabetic mice. Moreover, similar to data presented by Shang [35], severe neuroinflammation and activated OS state were observed in diabetic mice, both of which were sharply ameliorated by teneligliptin, suggesting that the protection of teneligliptin against CI in diabetic mice might be correlated to its repression on the inflammation and OS.
The secretion and maturation of IL-1β is regulated by the inflammatory activation of NLRP3, which induces various forms of inflammatory response, thereby promoting the development of central nervous system diseases [36–38]. IL-1β is also an important mediator of accelerating neurodegenerative diseases and diabetes-related CI [39], and significantly increased hippocampal IL-1β levels are associated with cognitive and emotional changes in diabetic mice [40]. Animal studies have shown that diabetic complications are alleviated by the inhibitor of the NLRP3 inflammasome [41, 42]. Herein, high levels of IL-1β were observed in diabetic mice, accompanied by an activation of the NLRP3 inflammasome, consistent with data reported by Tian [43] and Lu [44]. Moreover, the level of IL-1β and the activation of the NLRP3 inflammasome were notably suppressed by teneligliptin, indicating that the function of teneligliptin in diabetic mice was correlated to the inhibition of NLRP3 signaling. In future work, the association between the NLRP3 inflammasome and teneligliptin in diabetes-related CI will be identified by co-administering the diabetic mice with teneligliptin and the agonist of the NLRP3 inflammasome.
ER is an important organelle that participates in protein folding, transport, and maintaining calcium ion balance. When the pressure load of protein folding in the cell is low, Bip/GRP78 in the ER binds to the luminal segment of the ER transmembrane proteins ATF6, IRE-1, and PERK, which is in an inactive state. However, when the folding capacity of ER cannot meet the demand of newly synthesized unfolded protein or the calcium ion balance is broken, cells will be in the ER stress state [45, 46]. ER stress signaling is closely related to various pathological factors and states of cognitive decline in diabetes, such as metabolic abnormalities, cerebral ischemia, insulin resistance, neuronal calcium homeostasis, neurotransmitter changes, inflammatory response, and oxidative stress [47]. ER stress induces neuronal apoptosis through GSK3/3β, C/EBP-homologous protein (CHOP), and Caspase-12 signaling pathways or interacting with the mitochondrial apoptosis pathway [48–51]. Herein, consistent with the research by He [52], the remarkably activated ER stress pathway in diabetic mice was markedly repressed by teneligliptin, implying that the function of teneligliptin in diabetic mice was correlated to the suppression of the ER stress pathway. The involvement of the ER stress pathway in the regulatory mechanism of teneligliptin in diabetes-related CI will be further confirmed by co-administering diabetic mice with teneligliptin and tunicamycin, the activator of ER stress.
In conclusion, teneligliptin alleviated diabetes-related CI by repressing the ER stress and NLRP3 inflammasome in diabetic mice. Further studies can help clarify whether a sufficient improvement in cognitive impairment could result from long-term use of teneligliptin therapy in diabetic patients.
Author Contributions
Weifeng Wang wrote the main manuscript text; Weifeng Wang, Juanjuan Zhang performed the experiments, collected and analyzed the data, and prepared all the figures; Juanjuan Zhang supplied the reagents and materials; Juanjuan Zhang revised the manuscript; and all the authors reviewed the manuscript and approved the submission.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement
This study is approved by the Animal care committee of Laizhou City People’s Hospital (protocol No. LZ-2020–0019.
Funding
This study was supported by Laizhou City People’s Hospital.
References
- 1. Glovaci D, Fan W, Wong ND. Epidemiology of Diabetes Mellitus and Cardiovascular Disease. Curr Cardiol Rep. 2019; 21:21. https://doi.org/10.1007/s11886-019-1107-y [PubMed]
- 2. The Lancet Diabetes Endocrinology. Sarcopenia: a fate worth challenging. Lancet Diabetes Endocrinol. 2014; 2:183. https://doi.org/10.1016/S2213-8587(14)70055-5 [PubMed]
- 3. Swomley AM, Butterfield DA. Oxidative stress in Alzheimer disease and mild cognitive impairment: evidence from human data provided by redox proteomics. Arch Toxicol. 2015; 89:1669–80. https://doi.org/10.1007/s00204-015-1556-z [PubMed]
- 4. Fiorentino TV, Prioletta A, Zuo P, Folli F. Hyperglycemia-induced oxidative stress and its role in diabetes mellitus related cardiovascular diseases. Curr Pharm Des. 2013; 19:5695–703. https://doi.org/10.2174/1381612811319320005 [PubMed]
- 5. Zhang H, Davies KJA, Forman HJ. Oxidative stress response and Nrf2 signaling in aging. Free Radic Biol Med. 2015; 88:314–36. https://doi.org/10.1016/j.freeradbiomed.2015.05.036 [PubMed]
- 6. Jha JC, Banal C, Chow BS, Cooper ME, Jandeleit-Dahm K. Diabetes and Kidney Disease: Role of Oxidative Stress. Antioxid Redox Signal. 2016; 25:657–84. https://doi.org/10.1089/ars.2016.6664 [PubMed]
- 7. Zhang S, Xue R, Hu R. The neuroprotective effect and action mechanism of polyphenols in diabetes mellitus-related cognitive dysfunction. Eur J Nutr. 2020; 59:1295–311. https://doi.org/10.1007/s00394-019-02078-2 [PubMed]
- 8. van Duinkerken E, Snoek FJ, de Wit M. The cognitive and psychological effects of living with type 1 diabetes: a narrative review. Diabet Med. 2020; 37:555–63. https://doi.org/10.1111/dme.14216 [PubMed]
- 9. van Duinkerken E, Ryan CM. Diabetes mellitus in the young and the old: Effects on cognitive functioning across the life span. Neurobiol Dis. 2020; 134:104608. https://doi.org/10.1016/j.nbd.2019.104608 [PubMed]
- 10. Ward R, Li W, Abdul Y, Jackson L, Dong G, Jamil S, Filosa J, Fagan SC, Ergul A. NLRP3 inflammasome inhibition with MCC950 improves diabetes-mediated cognitive impairment and vasoneuronal remodeling after ischemia. Pharmacol Res. 2019; 142:237–50. https://doi.org/10.1016/j.phrs.2019.01.035 [PubMed]
- 11. Chen Q, Mo R, Wu N, Zou X, Shi C, Gong J, Li J, Fang K, Wang D, Yang D, Wang K, Chen J. Berberine Ameliorates Diabetes-Associated Cognitive Decline through Modulation of Aberrant Inflammation Response and Insulin Signaling Pathway in DM Rats. Front Pharmacol. 2017; 8:334. https://doi.org/10.3389/fphar.2017.00334 [PubMed]
- 12. Pignalosa FC, Desiderio A, Mirra P, Nigro C, Perruolo G, Ulianich L, Formisano P, Beguinot F, Miele C, Napoli R, Fiory F. Diabetes and Cognitive Impairment: A Role for Glucotoxicity and Dopaminergic Dysfunction. Int J Mol Sci. 2021; 22:12366. https://doi.org/10.3390/ijms222212366 [PubMed]
- 13. Goda M, Kadowaki T. Teneligliptin for the treatment of type 2 diabetes. Drugs Today (Barc). 2013; 49:615–29. https://doi.org/10.1358/dot.2013.49.10.2035882 [PubMed]
- 14. Sharma SK, Panneerselvam A, Singh KP, Parmar G, Gadge P, Swami OC. Teneligliptin in management of type 2 diabetes mellitus. Diabetes Metab Syndr Obes. 2016; 9:251–60. https://doi.org/10.2147/DMSO.S106133 [PubMed]
- 15. Kadowaki T, Sasaki K, Ishii M, Matsukawa M, Ushirogawa Y. Efficacy and Safety of Teneligliptin 40 mg in Type 2 Diabetes: A Pooled Analysis of Two Phase III Clinical Studies. Diabetes Ther. 2018; 9:623–36. https://doi.org/10.1007/s13300-018-0372-x [PubMed]
- 16. Pelluri R, Kongara S, Nagasubramanian VR, Mahadevan S, Chimakurthy J. Systematic review and meta-analysis of teneligliptin for treatment of type 2 diabetes. J Endocrinol Invest. 2023; 46:855–67. https://doi.org/10.1007/s40618-023-02003-9 [PubMed]
- 17. Kuthati Y, Rao VN, Busa P, Wong CS. Teneligliptin Exerts Antinociceptive Effects in Rat Model of Partial Sciatic Nerve Transection Induced Neuropathic Pain. Antioxidants (Basel). 2021; 10:1438. https://doi.org/10.3390/antiox10091438 [PubMed]
- 18. Yoshida T, Akahoshi F, Sakashita H, Kitajima H, Nakamura M, Sonda S, Takeuchi M, Tanaka Y, Ueda N, Sekiguchi S, Ishige T, Shima K, Nabeno M, et al. Discovery and preclinical profile of teneligliptin (3-[(2S,4S)-4-[4-(3-methyl-1-phenyl-1H-pyrazol-5-yl)piperazin-1-yl]pyrrolidin-2-ylcarbonyl]thiazol idine): a highly potent, selective, long-lasting and orally active dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes. Bioorg Med Chem. 2012; 20:5705–19. https://doi.org/10.1016/j.bmc.2012.08.012 [PubMed]
- 19. Liu X, Cao Y, Zhang Y, Sun B, Liang H. Teneligliptin inhibits lipopolysaccharide-induced cytotoxicity and inflammation in dental pulp cells. Int Immunopharmacol. 2019; 73:57–63. https://doi.org/10.1016/j.intimp.2019.04.059 [PubMed]
- 20. Peng W, Rao D, Zhang M, Shi Y, Wu J, Nie G, Xia Q. Teneligliptin prevents doxorubicin-induced inflammation and apoptosis in H9c2 cells. Arch Biochem Biophys. 2020; 683:108238. https://doi.org/10.1016/j.abb.2019.108238 [PubMed]
- 21. Salim HM, Fukuda D, Higashikuni Y, Tanaka K, Hirata Y, Yagi S, Soeki T, Shimabukuro M, Sata M. Teneligliptin, a dipeptidyl peptidase-4 inhibitor, attenuated pro-inflammatory phenotype of perivascular adipose tissue and inhibited atherogenesis in normoglycemic apolipoprotein-E-deficient mice. Vascul Pharmacol. 2017; 96-98:19–25. https://doi.org/10.1016/j.vph.2017.03.003 [PubMed]
- 22. Pujadas G, De Nigris V, Prattichizzo F, La Sala L, Testa R, Ceriello A. The dipeptidyl peptidase-4 (DPP-4) inhibitor teneligliptin functions as antioxidant on human endothelial cells exposed to chronic hyperglycemia and metabolic high-glucose memory. Endocrine. 2017; 56:509–20. https://doi.org/10.1007/s12020-016-1052-0 [PubMed]
- 23. Zhang J, Liu L, Zhang Y, Yuan Y, Miao Z, Lu K, Zhang X, Ni R, Zhang H, Zhao Y, Wang X. ChemR23 signaling ameliorates cognitive impairments in diabetic mice via dampening oxidative stress and NLRP3 inflammasome activation. Redox Biol. 2022; 58:102554. https://doi.org/10.1016/j.redox.2022.102554 [PubMed]
- 24. Ferrannini E, DeFronzo RA. Impact of glucose-lowering drugs on cardiovascular disease in type 2 diabetes. Eur Heart J. 2015; 36:2288–96. https://doi.org/10.1093/eurheartj/ehv239 [PubMed]
- 25. Baggio LL, Drucker DJ. Biology of incretins: GLP-1 and GIP. Gastroenterology. 2007; 132:2131–57. https://doi.org/10.1053/j.gastro.2007.03.054 [PubMed]
- 26. Kadowaki T, Haneda M, Ito H, Sasaki K, Matsukawa M, Yamada Y. Long-Term, Real-World Safety and Efficacy of Teneligliptin: A Post-Marketing Surveillance of More Than 10,000 Patients with Type 2 Diabetes in Japan. Adv Ther. 2020; 37:1065–86. https://doi.org/10.1007/s12325-019-01189-w [PubMed]
- 27. Nakamaru Y, Akahoshi F, Iijima H, Hisanaga N, Kume T. Tissue distribution of teneligliptin in rats and comparisons with data reported for other dipeptidyl peptidase-4 inhibitors. Biopharm Drug Dispos. 2016; 37:142–55. https://doi.org/10.1002/bdd.2003 [PubMed]
- 28. Zhu Q. The ER stress-autophagy axis: implications for cognitive dysfunction in diabetes mellitus. Clin Sci (Lond). 2020; 134:1255–8. https://doi.org/10.1042/CS20200235 [PubMed]
- 29. Marioni RE, Strachan MW, Reynolds RM, Lowe GD, Mitchell RJ, Fowkes FG, Frier BM, Lee AJ, Butcher I, Rumley A, Murray GD, Deary IJ, Price JF. Association between raised inflammatory markers and cognitive decline in elderly people with type 2 diabetes: the Edinburgh Type 2 Diabetes Study. Diabetes. 2010; 59:710–3. https://doi.org/10.2337/db09-1163 [PubMed]
- 30. Villegas-Llerena C, Phillips A, Garcia-Reitboeck P, Hardy J, Pocock JM. Microglial genes regulating neuroinflammation in the progression of Alzheimer's disease. Curr Opin Neurobiol. 2016; 36:74–81. https://doi.org/10.1016/j.conb.2015.10.004 [PubMed]
- 31. Acharya NK, Levin EC, Clifford PM, Han M, Tourtellotte R, Chamberlain D, Pollaro M, Coretti NJ, Kosciuk MC, Nagele EP, Demarshall C, Freeman T, Shi Y, et al. Diabetes and hypercholesterolemia increase blood-brain barrier permeability and brain amyloid deposition: beneficial effects of the LpPLA2 inhibitor darapladib. J Alzheimers Dis. 2013; 35:179–98. https://doi.org/10.3233/JAD-122254 [PubMed]
- 32. Pabreja K, Dua K, Sharma S, Padi SS, Kulkarni SK. Minocycline attenuates the development of diabetic neuropathic pain: possible anti-inflammatory and anti-oxidant mechanisms. Eur J Pharmacol. 2011; 661:15–21. https://doi.org/10.1016/j.ejphar.2011.04.014 [PubMed]
- 33. Ye T, Meng X, Wang R, Zhang C, He S, Sun G, Sun X. Gastrodin Alleviates Cognitive Dysfunction and Depressive-Like Behaviors by Inhibiting ER Stress and NLRP3 Inflammasome Activation in db/db Mice. Int J Mol Sci. 2018; 19:3977. https://doi.org/10.3390/ijms19123977 [PubMed]
- 34. Wu XL, Deng MZ, Gao ZJ, Dang YY, Li YC, Li CW. Neferine alleviates memory and cognitive dysfunction in diabetic mice through modulation of the NLRP3 inflammasome pathway and alleviation of endoplasmic-reticulum stress. Int Immunopharmacol. 2020; 84:106559. https://doi.org/10.1016/j.intimp.2020.106559 [PubMed]
- 35. Shang J, Che S, Zhu M. Oleuropein Improves Cognitive Dysfunction and Neuroinflammation in Diabetic Rats through the PI3K/Akt/mTOR Pathway. Appl Bionics Biomech. 2022; 2022:5892463. https://doi.org/10.1155/2022/5892463 [PubMed]
- 36. Broz P, Dixit VM. Inflammasomes: mechanism of assembly, regulation and signalling. Nat Rev Immunol. 2016; 16:407–20. https://doi.org/10.1038/nri.2016.58 [PubMed]
- 37. Bruce KD, Gorkhali S, Given K, Coates AM, Boyle KE, Macklin WB, Eckel RH. Lipoprotein Lipase Is a Feature of Alternatively-Activated Microglia and May Facilitate Lipid Uptake in the CNS During Demyelination. Front Mol Neurosci. 2018; 11:57. https://doi.org/10.3389/fnmol.2018.00057 [PubMed]
- 38. Coll RC, Robertson AA, Chae JJ, Higgins SC, Muñoz-Planillo R, Inserra MC, Vetter I, Dungan LS, Monks BG, Stutz A, Croker DE, Butler MS, Haneklaus M, et al. A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases. Nat Med. 2015; 21:248–55. https://doi.org/10.1038/nm.3806 [PubMed]
- 39. Pan Y, Chen XY, Zhang QY, Kong LD. Microglial NLRP3 inflammasome activation mediates IL-1β-related inflammation in prefrontal cortex of depressive rats. Brain Behav Immun. 2014; 41:90–100. https://doi.org/10.1016/j.bbi.2014.04.007 [PubMed]
- 40. Peiró C, Lorenzo Ó, Carraro R, Sánchez-Ferrer CF. IL-1β Inhibition in Cardiovascular Complications Associated to Diabetes Mellitus. Front Pharmacol. 2017; 8:363. https://doi.org/10.3389/fphar.2017.00363 [PubMed]
- 41. Luo B, Li B, Wang W, Liu X, Xia Y, Zhang C, Zhang M, Zhang Y, An F. NLRP3 gene silencing ameliorates diabetic cardiomyopathy in a type 2 diabetes rat model. PLoS One. 2014; 9:e104771. https://doi.org/10.1371/journal.pone.0104771 [PubMed]
- 42. Devi TS, Lee I, Hüttemann M, Kumar A, Nantwi KD, Singh LP. TXNIP links innate host defense mechanisms to oxidative stress and inflammation in retinal Muller glia under chronic hyperglycemia: implications for diabetic retinopathy. Exp Diabetes Res. 2012; 2012:438238. https://doi.org/10.1155/2012/438238 [PubMed]
- 43. Tian R, Liu X, Jing L, Yang L, Xie N, Hou Y, Tao H, Tao Y, Wu J, Meng X. Huang-Lian-Jie-Du decoction attenuates cognitive dysfunction of rats with type 2 diabetes by regulating autophagy and NLRP3 inflammasome activation. J Ethnopharmacol. 2022; 292:115196. https://doi.org/10.1016/j.jep.2022.115196 [PubMed]
- 44. Lu Z, Yao Y, Wang J, Peng JY. Dioscin ameliorates diabetes cognitive dysfunction via adjusting P2X7R/NLRP3 signal. Int Immunopharmacol. 2021; 101:108314. https://doi.org/10.1016/j.intimp.2021.108314 [PubMed]
- 45. Chen X, Cubillos-Ruiz JR. Endoplasmic reticulum stress signals in the tumour and its microenvironment. Nat Rev Cancer. 2021; 21:71–88. https://doi.org/10.1038/s41568-020-00312-2 [PubMed]
- 46. Oakes SA, Papa FR. The role of endoplasmic reticulum stress in human pathology. Annu Rev Pathol. 2015; 10:173–94. https://doi.org/10.1146/annurev-pathol-012513-104649 [PubMed]
- 47. Kong FJ, Ma LL, Guo JJ, Xu LH, Li Y, Qu S. Endoplasmic reticulum stress/autophagy pathway is involved in diabetes-induced neuronal apoptosis and cognitive decline in mice. Clin Sci (Lond). 2018; 132:111–25. https://doi.org/10.1042/CS20171432 [PubMed]
- 48. Nishitoh H, Matsuzawa A, Tobiume K, Saegusa K, Takeda K, Inoue K, Hori S, Kakizuka A, Ichijo H. ASK1 is essential for endoplasmic reticulum stress-induced neuronal cell death triggered by expanded polyglutamine repeats. Genes Dev. 2002; 16:1345–55. https://doi.org/10.1101/gad.992302 [PubMed]
- 49. Chen G, Fan Z, Wang X, Ma C, Bower KA, Shi X, Ke ZJ, Luo J. Brain-derived neurotrophic factor suppresses tunicamycin-induced upregulation of CHOP in neurons. J Neurosci Res. 2007; 85:1674–84. https://doi.org/10.1002/jnr.21292 [PubMed]
- 50. Nakagawa T, Zhu H, Morishima N, Li E, Xu J, Yankner BA, Yuan J. Caspase-12 mediates endoplasmic-reticulum-specific apoptosis and cytotoxicity by amyloid-beta. Nature. 2000; 403:98–103. https://doi.org/10.1038/47513 [PubMed]
- 51. Meares GP, Mines MA, Beurel E, Eom TY, Song L, Zmijewska AA, Jope RS. Glycogen synthase kinase-3 regulates endoplasmic reticulum (ER) stress-induced CHOP expression in neuronal cells. Exp Cell Res. 2011; 317:1621–8. https://doi.org/10.1016/j.yexcr.2011.02.012 [PubMed]
- 52. He J, Chen Z, Kang X, Wu L, Jiang JM, Liu SM, Wei HJ, Chen YJ, Zou W, Wang CY, Zhang P. SIRT1 Mediates H2S-Ameliorated Diabetes-Associated Cognitive Dysfunction in Rats: Possible Involvement of Inhibiting Hippocampal Endoplasmic Reticulum Stress and Synaptic Dysfunction. Neurochem Res. 2021; 46:611–23. https://doi.org/10.1007/s11064-020-03196-8 [PubMed]



