Introduction
Aging affects individuals both personally and societally [1]. Recognizing that relying solely on chronological age is insufficient to comprehend the internal physiological states nor the inter-individual variation in the rate and manner of aging [2], extensive efforts have been made to develop measures to capture the underlying aging processes at the biological level, also termed biological age [3]. These measures involve the use of individual or composite biomarkers that demonstrate associations with typically chronological age or mortality [4]. Of particular interest, clinical-parameter biological-age algorithms (i.e., PhenoAge [5] and BioAge [6]) and telomere length have been validated as among the most reliable predictors of aging outcomes and hold promise for routine application in large populations [7–9].
While the determinants of the rate of aging are intricate, it is possible to moderate the aging process through lifestyle interventions that influence metabolic processes. As one of these potentials, sleep assumes a significant role in daily life and functions as a restorative process facilitating both physical and mental recovery [10]. Accumulating evidence suggests that deviations from normal sleep duration may contribute to the premature development and progression of age-related conditions [11, 12]. However, the specific impact of insufficient or excessive sleep on accelerated biological aging remains uncertain [10, 13, 14]. Previous observational studies, often limited in scale and focused on linear effects, have yielded conflicting findings within specific populations [15–22]. Notably, existing studies have predominantly relied on binary categorizations of “short” and “long” sleep, rather than exploring the potentially diverse patterns across the entire duration continuum. A comprehensive investigation for potential non-linear dose-response relationships may elucidate the need for tailored interventions or recommendations across different ranges of sleep duration to extend a healthy lifespan and mitigate the risks of age-related health outcomes.
The lack of consensus in the existing evidence can be attributed, at least in part, to the vulnerability of conventional observational studies to residual confounding and reverse causality. While these limitations can be tackled by implementing an experimental study, it is unethical to conduct experiment of long-term sleep deprivation to confirm its causal effect in accelerating aging. In such case, Mendelian randomization (MR) provides an alternative means of uncovering the causal nature underlying a phenotypic association by using genetic variants (randomly distributed at conception) as proxies for life-long exposure risks [23]. Recent methodological advancements have introduced the doubly ranked method, a novel nonlinear MR approach that provides more robust stratum-specific estimates compared to conventional approaches [24]. Despite these developments, few nonlinear MR studies have been conducted in the realm of sleep duration and biological aging [25, 26].
Therefore, the present study aimed to provide a parametric visualization of the relationship between sleep duration and three key markers of biological aging status - PhenoAge acceleration, BioAge acceleration, and telomere length, utilizing extensive phenotypic and genotyping data from the UK Biobank. Multivariable regressions and restricted cubic spline analyses were first performed to examine phenotypic associations and whether phenotypic data support nonlinear effects. Linear and non-linear MR were then applied to delineate the shape of the causal relationships of genetically-predicted sleep duration with biological age measurements. Integrating genome-wide association study (GWAS) summary statistics and cell-type-specific annotations, functional annotation analyses were finally undertaken to interrogate potential genetic mechanisms relating sleep duration to the aging process. A graphical abstract is provided in Figure 1.
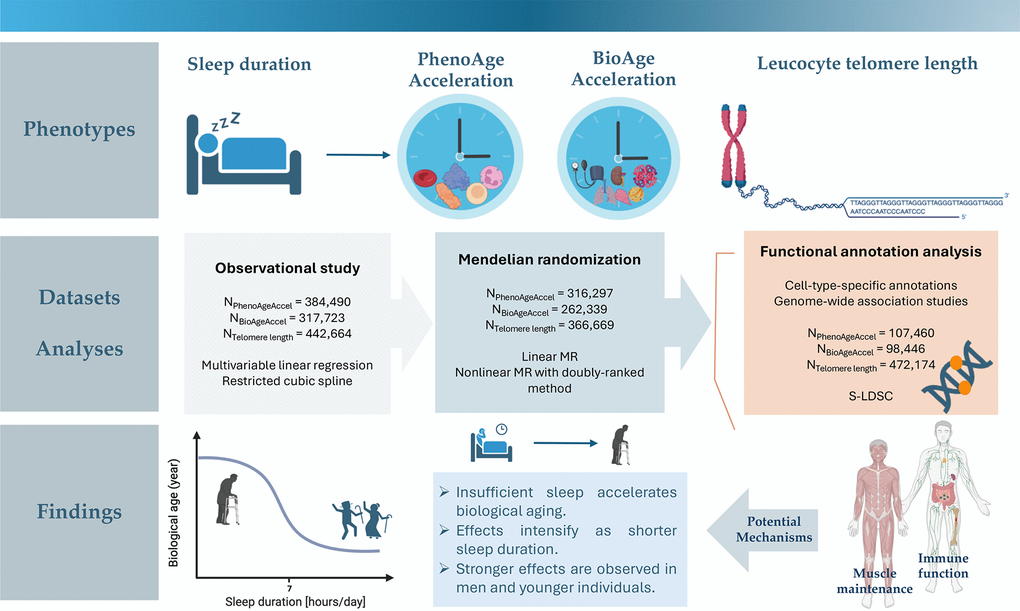
Figure 1. Graphical abstract.
Materials and Methods
The flowchart of participant selection and overall study design is outlined in Figure 2. In general, observational and MR analyses were conducted based on individual participant data from the UK Biobank, while functional annotation analyses were conducted using summary statistics from the hitherto largest GWAS of each phenotype. All analyses were performed in R (version 4.1.0) unless otherwise specified, with a two-sided P-value of 0.05 used as the threshold for statistical significance.
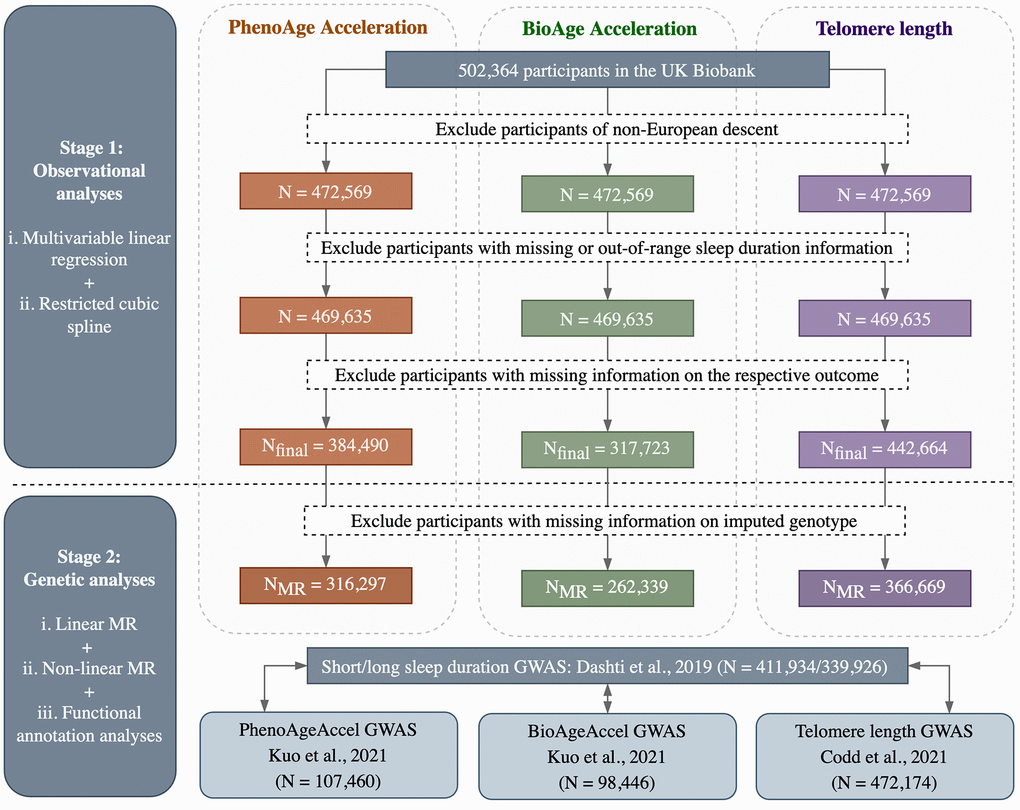
Figure 2. Flowchart of participant selection and overall study design.
Study participants
Participants were drawn from the UK Biobank, a large population-based prospective cohort study which recruited more than 500,000 individuals aged 40-69 years between 2006 and 2010. The study protocol is available online (www.ukbiobank.ac.uk/wp-content/uploads/2011/11/UK-Biobank-Protocol.pdf) and more details are published elsewhere [27]. At the initial visit, participants completed online questionnaire and physical measurements, and their biological samples were collected for genotyping and biochemistry tests. In our study, we used information on habitual sleep duration, biological traits for PhenoAge and BioAge construction, telomere length measurements, relevant confounding factors, and genetic variants. UK Biobank received ethical approval from the North West Multi-centre Research Ethics Committee and obtained written informed consent from all participants. Our study was performed under application number 99713.
Self-reported habitual sleep duration
In the UK Biobank, participants’ habitual sleep duration was assessed at the baseline assessment via a standardized question: “About how many hours sleep do you get in every 24 hours? (please include naps)”, with responses in hour increment. Following the methods of previous studies [28, 29], participants with sleep durations below 2 hours or above 12 hours (extreme responses) were treated similarly to those who did not respond to the question. These participants along with those who responded with “Do not know” or “Prefer not to answer” were excluded from our analysis.
PhenoAge, BioAge, and age accelerations
We computed two validated biological age predictors, PhenoAge [5] and BioAge [6], using two sets of nine clinical-based biomarkers involving a total of 12 blood chemistry and blood count traits as well as assessments of lung function and blood pressure. Details of the included biomarkers can be found in Supplementary Table 1. Both PhenoAge and BioAge were initially trained using data from the third National Health and Nutrition Examination Survey (NHANES III), but with different objectives. While PhenoAge was derived from an algorithm based on multivariate analysis of mortality hazards in the reference population to provide an estimate of the risk of death, BioAge was computed using an algorithm that involved a series of regressions of individual biomarkers on chronological age in the reference population, aiming to quantify the decline in overall system integrity. We applied the previously trained algorithms to the UK Biobank biomarker data to calculate PhenoAge and BioAge for each participant, using the R package “BioAge” [30]. To better quantify the differences between participants in biological aging, we utilized linear regression models to estimate the residual of the computed PhenoAge or BioAge after subtracting the effect of chronological age. These residuals were referred to as PhenoAge acceleration (PhenoAgeAccel) and BioAge acceleration (BioAgeAccel), respectively, and served as the primary outcomes in our analysis. Any observations with missing biomarkers values were excluded.
Leukocyte telomere length
Telomeres are nucleoprotein complexes located at the ends of chromosomes that undergo shortening with each cell division. The length of telomere has been proposed as a biological measure of aging, reflecting the degree of cellular senescence and oxidative stress [31]. In this study, we focused on telomere length measured in leukocyte (LTL), a practical measure correlating well with telomere length across different tissues within individuals. In the UK Biobank, LTL measurements were ascertained on DNA from peripheral blood leukocytes collected at baseline assessment using a well-validated multiplex qPCR assay [32]. Measurements were reported as a ratio of telomere repeat copy number relative to that of a single copy gene (T/S ratios), which were then log-transformed to obtain a normal distribution (logeLTL). Multiple quality checks were applied to control and adjust for technical factors, as described elsewhere [32]. To allow direct comparison to other studies, we used z-standardized logeLTL as our primary outcome. Participants with missing LTL measurements were excluded.
Genetic risk score of self-reported sleep duration
Genetic variants used in MR analyses were extracted genotypes from the UKB imputation dataset (n = 487,150 at the time of our study). Detailed information on genotyping, imputation, and quality control in the UK Biobank has been described previously [33]. We took 85 independent single-nucleotide polymorphisms (SNPs) robustly associated with continuous sleep duration (P-value < 5×10−8), obtained by applying PLINK’s clumping function [34] (parameters: -clump-r2 0.001 --clump-kb 1000) to results from the largest published GWAS of self-reported habitual sleep duration (n = 446,118) [35], as our genetic instruments. Details of the included SNPs are shown in Supplementary Table 2. Following SNP quality control [36], two palindromic SNPs (rs17732997 and rs4333549) were detected and subsequently excluded. To avoid one-sample bias towards confounded observational associations, we calculated an unweighted genetic risk score (GRS) by directly summing the number of sleep duration-increasing alleles across the 83 SNPs. Collectively, the unweighted GRS explained 6.22% of the genome-wide SNP-based heritability of sleep duration, as determined by comparing residual variance in linear regression models of sleep duration on GRS (F-statistic = 2,239).
GWAS data sources
The largest published GWAS of self-reported habitual sleep duration also conducted separate GWAS for short (< 7 h/d; n = 106,192 cases) and long (> 8 h/d; n = 34,184 cases) sleep relative to 7–8 h/d sleep duration (n = 305,742 controls) [35]. We retrieved the full sets of summary statistics for the three sleep duration phenotypes for further functional annotation analyses. We also obtained the largest available GWAS of PhenoAgeAccel (n = 107,460) [37], BioAgeAccel (n = 98,446) [37], and LTL (n = 472,174) [32]. All original GWAS analyses were performed using imputed genotype data from the UK Biobank, involving only participants of European descent.
Statistical analyses
Observational analyses
Participants of European ancestry (to mirror the genetic analyses) with complete (and within range) exposure and outcome data were included. Separate multivariable linear regression models were employed to assess the relationships of continuous sleep duration with PhenoAgeAccel, BioAgeAccel, and LTL. In addition to the top five genetic principal components (PCs), factors previously described as associated with biological age measurements were included as covariates, including age, sex, educational qualifications (degree, no degree), body mass index, smoking history (current, former, or never), drinking history (current, former, or never), physical activity status (low, moderate, high), histories of cardiovascular disease (angina, heart attack, or stroke), hypertension, diabetes mellitus, and leucocyte count (for LTL as an outcome).
To assess the potential for nonlinear associations, we entered the levels of self-reported sleep duration as indicator variables, and obtained estimates comparing each of < 5 h/d, 5 h/d, 6 h/d, 8 h/d, 9 h/d, and > 9 h/d to our chosen reference category of 7 h/d. Restricted cubic spline regressions were further employed to model the non-linear relationships between continuous sleep duration and biological age measurements with four knots (located at the 5th, 35th, 65th, and 95th percentiles) after adjustments.
Considering the previously observed sex differences in biological aging [32, 38] and the age-dependent variations in sleep duration [39], all analyses were additionally stratified by sex and age (using the median age of 58 years as the cutoff point).
Mendelian randomization analyses
Participants of European ancestry (to avoid population stratification) with complete data on sleep duration, aging outcomes, and genotypes were further included in the MR analyses. We first applied a two-stage method to estimate the average causal effect of sleep duration on the outcomes. This conventional MR approach tests for the presence of a causal relationship, yielding effect estimates under a linear modeling framework. In the first-stage analysis, we regressed the exposure (sleep duration) on the unweighted GRS. In the second stage, we regressed the outcomes on the fitted values of the exposure from the first stage. The regression models in both stages were adjusted with age at baseline, sex, age-squared, age-by-sex, age-squared-by-sex, the top five PCs of ancestry, and genotyping array.
While linear MR was conducted in the overall population to estimate averaged causal effects, nonlinear MR involves generating strata within the study sample and undertaking MR analyses within each stratum [40]. We applied the fractional polynomial method to examine nonlinearity and used the recently developed doubly-ranked stratification approach [24] to construct five equal-sized strata of the population. Compared to the conventional residual-based method [40], the doubly-ranked approach offers increased reliability when the genetic effects on the exposure vary across the population, or when the exposure measurement is coarsened (e.g., self-reported sleep duration measured in hourly units) [24]. We first stratified all participants into preliminary strata (each containing 50 participants) according to the levels of the GRS, and then stratified them into final strata based on the levels of observed sleep duration within each pre-stratum. This ensures that the constructed strata are uncorrelated with the GRS, while also guaranteeing that the average exposure level increases monotonically across the strata.
For each final stratum, we calculated a linear MR estimate of the causal effect of increasing sleep duration using the two-stage method described above. To stabilize estimates, we performed a bootstrap averaging approach. We randomly removed a small number of participants (n = 12) from the analysis and performed the doubly-ranked approach using this dataset. We repeated this procedure 100 times and then combined the estimates using Rubin’s rules. To evaluate the presence of a trend in the stratum-specific estimates, we performed a meta-regression of these estimates on the mean value of observed sleep duration in each stratum [40, 41], considering a P-value < 0.05 as evidence supporting nonlinearity. Sex- and age-stratified MR analyses were also performed.
Two sensitivity analyses were performed. First, we incorporated all additional confounding factors included in the multivariable regression models into the two-stage MR analyses to address potential residual confounding. Second, to assess the MR assumption that genetic variants influence biological aging solely through sleep duration, we recalculated the unweighted GRS after disregarding six potential pleiotropic SNPs that demonstrated associations (P-value < 5×10−8) with phenotypes other than sleep duration, as indicated by the GWAS catalog [42], and repeated MR analyses. The potential pleiotropic SNPs and their corresponding related phenotypes are shown in Supplementary Table 2. Given the methods for exploring horizontal pleiotropy in one-sample MR can be underpowered in each stratum, we compared the effect estimates derived from sensitivity analyses with those obtained from the primary analyses to assess the consistency of patterns. All MR analyses were performed using the R packages “ivreg” and “SUMnlmr”.
Functional annotation analyses
To elucidate the potential shared biological mechanisms underlying the observed relationships between sleep duration and biological aging, we conducted functional annotation analyses by partitioning the SNP-heritability of each phenotype based on cell-type-specific annotations and examining their clustering patterns. A total of 396 annotations from the Roadmap Epigenomics project encompassing six histone marks (DNase, H3K27ac, H3K36me3, H3K4me1, H3K4me3, and H3K9ac) across 88 primary cell types or tissues [43] were utilized. These annotations were further categorized into nine broad groups, including adipose, central nervous system (CNS), digestive system, cardiovascular, musculoskeletal and connective tissue, immune and blood, liver, pancreas, and other. For each phenotype, annotation-specific enrichment values were calculated using stratified-LDSC [44], which were then transformed into a color scale and visualized through hierarchical clustering. FDR-adjusted P-value was applied based on the specific numbers of comparisons made in each analysis.
Data availability
The data underlying the results presented in the study are available to researchers upon application to UK Biobank (https://www.ukbiobank.ac.uk/).
Consent to participate
The participants in the UK Biobank study provided written informed consent for their data to be used in health-related research. As this study analyzes aggregated GWAS summary statistics that do not contain any personally identifiable information, no additional consent for publication is required.
Consent to publish
The participants in the UK Biobank study provided written informed consent for their data to be used in publications.
Results
Study population
Table 1 summarizes the baseline characteristics of study participants included in the observational analyses of PhenoAgeAccel (n = 384,490), BioAgeAccel (n = 317,723), and LTL (n = 442,664). The mean age of participants ranged from 56.6 to 56.8 years, and 53.7% to 54.2% of participants were women. More detailed characteristics of the three partially overlapping groups of participants according to self-reported sleep duration are presented in Supplementary Tables 3–5.
Table 1. Baseline characteristics of UK Biobank participants included in observational analyses.
| Characteristics | Analysis of PhenoAgeAccel | Analysis of BioAgeAccel | Analysis of LTL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No. of participants | 384490 | 317723 | 442664 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep duration, h/d | 7.16 (1.08) | 7.16 (1.06) | 7.16 (1.08) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at recruitment, y | 56.8 (8.02) | 56.6 (8.04) | 56.8 (8.03) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex (women), n (%) | 206642 (53.7%) | 171835 (54.1%) | 239936 (54.2%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Degree, n (%) | 315151 (82.7%) | 263167 (83.5%) | 363075 (82.7%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No degree, n (%) | 66008 (17.3%) | 51954 (16.5%) | 75785 (17.3%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body mass index, kg/m2 | 27.4 (4.75) | 27.3 (4.67) | 27.4 (4.76) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never, n (%) | 206510 (53.9%) | 172605 (54.5%) | 237954 (53.9%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Previous, n (%) | 136638 (35.7%) | 112269 (35.5%) | 157078 (35.6%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current, n (%) | 40062 (10.5%) | 31820 (10.0%) | 46137 (10.5%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drinking status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never, n (%) | 12340 (3.21%) | 10002 (3.15%) | 14243 (3.22%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Previous, n (%) | 13271 (3.45%) | 10414 (3.28%) | 15299 (3.46%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current, n (%) | 358575 (93.3%) | 297067 (93.6%) | 412778 (93.3%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IPAQ activity group | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| High, n (%) | 126921 (40.5%) | 106018 (40.9%) | 146179 (40.5%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Moderate, n (%) | 127813 (40.8%) | 105833 (40.8%) | 147130 (40.8%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low, n (%) | 58433 (18.7%) | 47654 (18.4%) | 67260 (18.7%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Leukocyte count, 10^9 cells/Litre | / | / | 6.90 (2.04) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Major diseases | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cardiovascular disease, n (%) | 22051 (5.74%) | 16096 (5.07%) | 25440 (5.76%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension, n (%) | 103404 (26.9%) | 82698 (26.1%) | 118845 (26.9%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes mellitus, n (%) | 18386 (4.79%) | 14409 (4.54%) | 21267 (4.81%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of biological ages | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lymphocyte (%) | 28.7 (7.34) | / | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean cell volume (fL) | 82.9 (5.25) | / | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Serum glucose (mmol/L) | 5.11 (1.21) | / | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Red cell distribution width (%) | 13.5 (0.95) | / | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| White blood cell count (1000 cells/uL) | 6.89 (1.93) | / | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Albumin (g/L) | 45.2 (2.61) | 45.3 (2.59) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creatinine (umol/L) | 72.2 (16.2) | 72.2 (17.1) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C-reactive protein (mg/dL) | 0.26 (0.44) | 0.25 (0.41) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alkaline phosphatase (U/L) | 83.5 (26.1) | 83.0 (25.9) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FEV1 (L) | / | 2.85 (0.80) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SBP (mm Hg) | / | 138 (18.5) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total Cholesterol (mg/dL) | / | 221 (43.9) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Glycated hemoglobin (%) | / | 3.58 (0.63) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Blood urea nitrogen (mg/dL) | / | 15.2 (3.82) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological ages, y | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PhenoAge | 50.8 (9.42) | / | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PhenoAge acceleration | 0.00 (4.66) | / | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioAge | / | 53.9 (8.67) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioAge acceleration | / | 0.00 (3.31) | / | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Z-standardized leucocyte telomere length | / | / | -0.01 (0.99) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Characteristics were presented as mean values (standard deviation) for continuous variables and n (%) for categorical variables. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PhenoAgeAccel, PhenoAge acceleration; BioAgeAccel, BioAge acceleration; LTL, leucocyte telomere length. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Linear and nonlinear phenotypic associations
Linear observational analyses revealed significantly positive associations between continuous sleep duration with both PhenoAgeAccel (β = 0.08 years per additional hour of sleep duration, 95%CI = 0.07 to 0.09) and BioAgeAccel (β = 0.02 years per additional hour of sleep duration, 95%CI = 0.01 to 0.03), but not with LTL (β = 0.0003 SD change per additional hour of sleep duration, 95%CI = –0.0024 to 0.0029) (Supplementary Table 6). Compared to participants reporting 7 hours of sleep per day, those reporting shorter (< 7 h/d) or longer (> 7 h/d) sleep durations showed significantly higher PhenoAgeAccel and BioAgeAccel, as well as significantly shorter LTL (Figure 3). Restricted cubic spline regressions also demonstrated U-shaped nonlinear associations of sleep duration with PhenoAgeAccel and BioAgeAccel, while suggested an inverted reverse J-shaped association of sleep duration with LTL (Supplementary Figure 1). All spline analyses yielded a P-value for nonlinearity below 0.001.
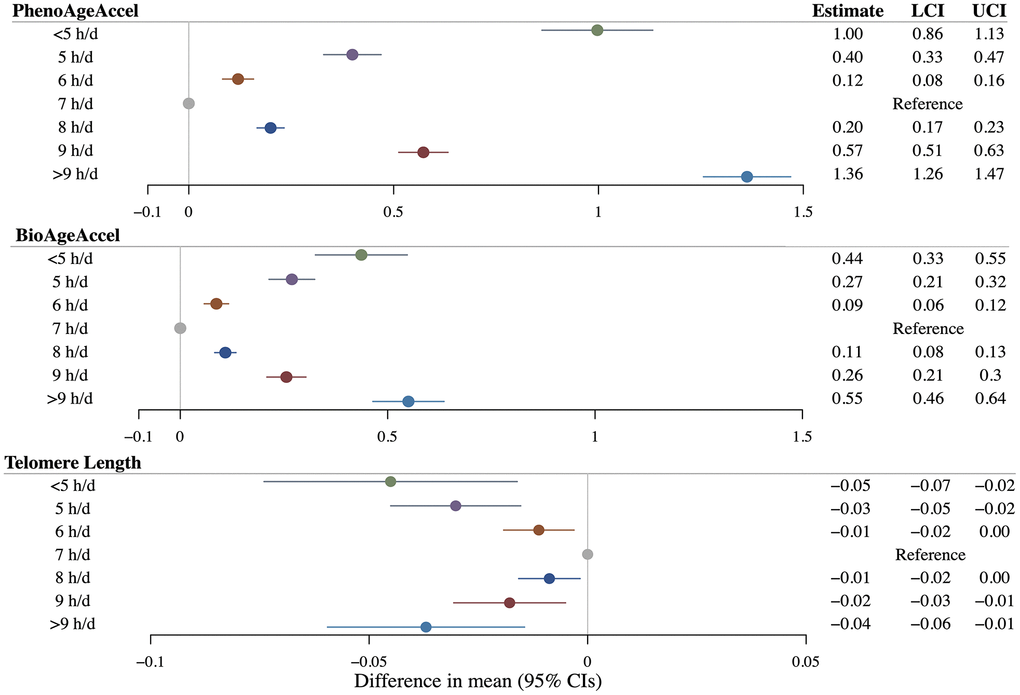
Figure 3. Results of multivariable linear regressions of PhenoAge acceleration (PhenoAgeAccel), BioAge acceleration (BioAgeAccel), and leukocyte telomere length on sleep duration. Circles denote point estimates. Error bars denote 95% confidence intervals. Regression models adjusted for: age at baseline, sex, the top five genetic principal components, educational qualifications, body mass index, smoking history, drinking history, physical activity status, leucocyte count (for telomere length as an outcome), and histories of cardiovascular diseases, hypertension, and diabetes mellitus.
Sex- and age-stratified observational analyses demonstrated similar patterns of nonlinear associations, despite slightly larger statistical uncertainties (Supplementary Table 6 and Supplementary Figure 1). Notably, the magnitude of associations was consistently higher in men than in women across all outcomes. Insufficient sleep exhibited seemingly stronger associations with both PhenoAgeAccel and BioAgeAccel in younger (< 58 years) compared to older participants (≥ 58 years).
Linear and nonlinear mendelian randomization
Utilizing 83 SNPs to instrument self-reported sleep duration, linear MR within the overall UK Biobank population found significant causal associations between genetically-predicted sleep duration and PhenoAgeAccel (β = –0.31 years per 1 hour increase in genetically-predicted sleep duration, 95%CI = –0.50 to –0.12), BioAgeAccel (β = –0.38 years per 1 hour increase in genetically-predicted sleep duration, 95%CI = –0.53 to –0.24), and LTL (β = 0.07 SD change per 1 hour increase in genetically-predicted sleep duration, 95%CI = 0.03 to 0.11) (Figure 4).
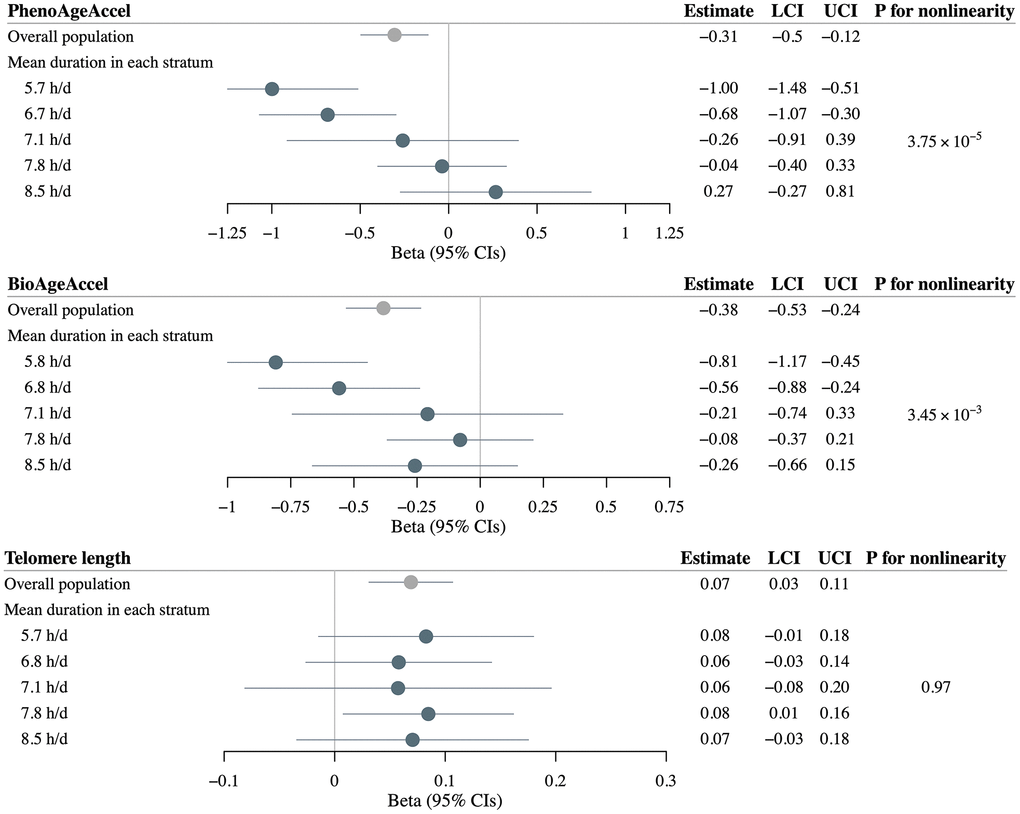
Figure 4. Mendelian randomization estimates for PhenoAge acceleration (PhenoAgeAccel), BioAge acceleration (BioAgeAccel), and leukocyte telomere length in overall population and strata of population defined by residual sleep duration. Grey circles denote overall estimates, and blue circles denote stratum estimates. Error bars denote 95% confidence intervals. Models adjusted for: age at baseline, sex, the top five genetic principal components, and genotyping array.
Stratifying the overall study sample into five strata, nonlinear MR demonstrated threshold nonlinear relationships for sleep duration with both PhenoAgeAccel and BioAgeAccel. We observed a strong inverse causal association of increasing genetically-predicted sleep duration with PhenoAgeAccel in the lowest strata (β = –1.00 years per 1 hour increase in genetically-predicted sleep duration, 95%CI = –1.48 to –0.51), an inverse but slightly weaker association in the second lowest stratum (β = –0.68, 95%CI = –1.07 to –0.30), whereas no significant association in the other three strata with mean observed durations > 7.1 h/d. Similarly, the strongest inverse causal association of increasing genetically-predicted sleep duration with BioAgeAccel was found in the lowest stratum (β = –0.81 years per 1 hour increase in genetically-predicted sleep duration, 95%CI = –1.17 to –0.45), followed by the second lowest stratum (β = –0.56, 95%CI = –0.88 to –0.24). The trends in estimates were significant, as evidenced by the significant P-values for nonlinearity for both PhenoAgeAccel (P = 3.75×10−5) and BioAgeAccel (P = 3.45×10−3) (Figure 4).
Nonlinear MR demonstrated a significant positive causal association of increasing genetically-predicted sleep duration with LTL in the second highest stratum (β = 0.08 SD change per 1 hour increase in genetically-predicted sleep duration, 95%CI = 0.01 to 0.16). No significant association was observed in the other duration groups, likely due to lower power arising from reduced sample size, nor a significant trend across stratum estimates (P = 0.97) (Figure 4).
We found largely consistent results in the sex-stratified nonlinear MR analyses of each biological age measurement, although some confidence intervals included the null (particularly in analyses of LTL) due to reduced statistical power. Notably, compared to women-specific analyses, men-specific nonlinear MR yielded higher effect magnitudes of extremely insufficient sleep duration on PhenoAgeAccel (β = –0.92 vs. β = –0.90) and BioAgeAccel (β = –1.44 vs. β = –0.71) (Supplementary Tables 7, 8). Additionally, insufficient sleep duration demonstrated stronger effects in younger participants compared to older participants for PhenoAgeAccel (β = –0.86 vs. β = –0.65), BioAgeAccel (β = –0.96 vs. β = –0.72), and LTL (β = 0.15 vs. β = 0.12) (Supplementary Tables 9, 10).
Sensitivity analyses incorporating additional confounders or utilizing a reconstructed GRS largely recapitulated the primary results (Supplementary Tables 11, 12).
Cell-type-specific heritability enrichments
Functional annotation analyses further revealed extensively distributed heritability enrichments for sleep duration phenotypes, predominantly in cell types of the CNS, as well as in other components related to blood/immune, pancreas, and musculoskeletal/connective tissues. Comparing the heritability enrichments between sleep duration and biological age measurements, we found that both short sleep (< 7 h/d) and LTL were significantly enriched in the fetal thymus annotation, a component of the blood/immune system. Additionally, short sleep showed significant enrichment in the fetal muscle leg annotation, while BioAgeAccel displayed enrichment in the foreskin fibroblast primary cells annotation, both representing components of the musculoskeletal/connective system. In contrast, no common significant enrichment was identified between long sleep (> 8 h/d) and any of the biological aging outcomes (Supplementary Figure 2).
Discussion
To the best of our knowledge, this is the first phenotypic and genetic analysis that systematically interrogates a nonlinear relationship between sleep duration and accelerated biological aging. Our observational study suggests U-shaped phenotypic associations of sleep duration with PhenoAgeAccel and BioAgeAccel and an inverted reverse J-shaped phenotypic association of sleep duration with LTL, with approximately 7 h/d as the optimal sleep duration. A comprehensive GRS-based MR study further confirmed the detrimental roles of insufficient sleep across all outcomes (in a dose-response manner for PhenoAgeAccel and BioAgeAccel), while finding no evidence to support deleterious impacts of excessive sleep on any of the biological aging measurements.
In utilizing various analytical approaches that designed specifically for linear relationships, we found that the direction of the effect estimates for sleep duration on biological age measurements largely differs when comparing the results of linear MR and multivariable linear regressions. Such disparity suggests that evaluating continuous sleep duration as a whole may lead to unreliable findings underscoring the need to consider potential nonlinear effects when studying its health impact.
PhenoAgeAccel and BioAgeAccel, recognized as the best validated measures for biological aging that could also be implemented with data available in the UK Biobank [6, 9, 45], were designed based on different assumptions to capture distinct aspects of the aging process. BioAge models biological age as the average physiology of an individual with the same age as the research subjects [6], while PhenoAge estimates biological age as the average physiology of an individual with the same risk of death as the research subjects [5]. Therefore, the U-shaped relationship between sleep duration and PhenoAgeAccel observed in our observational study aligns with previous findings that a sleep duration of 7 hours represents the optimal duration associated with lowest all-cause and other-cause mortality [46, 47]. However, leveraging robust genetic instruments, our nonlinear MR analyses failed to confirm a causative role of long sleep duration in increasing PhenoAgeAccel, indicating that the observed associations are likely due to residual confounding or/and reverse causation. Indeed, given long sleep itself can be a consequence of underlying health conditions or comorbidities, and that accelerated aging may cause fatigue or other symptoms that lead to increased sleep duration [39], the association between excessive sleep and accelerated biological aging may be more susceptible to these common limitations inherent in conventional observational studies. Notably, such disagreement between observational and MR findings was consistently observed for PhenoAgeAccel and BioAgeAccel, providing reassurance that the associations reflect genuine biological aging rather than artifacts specific to a particular method of biological age measurement.
Previous observational studies have demonstrated a detrimental role of both insufficient and excessive sleep in PhenoAgeAccel/BioAgeAccel. For example, a study involving 615 participants from the Baltimore Longitudinal Study of Aging reported significantly lower DNA methylation-based PhenoAge among individuals sleeping ≤ 6 h/d compared to those sleeping > 7 h/d [17]. Another study involving 29,309 participants from the Korean NHANES V-VI found significantly higher metabolism diagnostic parameters-based BioAgeAccel among individuals who sleep > 8 h/d compared to those sleeping between 6 to 8 h/d [15]. It is important to note that these previous studies, like many others investigating sleep duration, employed a block classification of “short” and “long” sleep duration that imposed a linear relationship between aging and more or less sleep around a chosen cut-off (e.g., 6, 7, or 8 h/d). This approach, coupled with relatively small sample sizes, limited their ability to detect the impact of different duration patterns or the magnitude of effects associated with more extreme sleep durations. Building upon previous findings, our work based on triangulated evidence supporting that an insufficient sleep duration (defined as < 7 h/d) may exert increasingly pronounced detrimental effects on accelerated biological aging as sleep insufficiency becomes more severe.
While PhenoAge and BioAge aim to capture integrated multi-system dysregulation during aging, LTL and its natural shortening primarily reflect cellular proliferative capacity [31]. To date, epidemiological evidence regarding the relationship of sleep duration and telomere length has remained highly inconclusive. As one of the pioneering studies in this field, an investigation involving 4,117 female participants from the Nurses’ Health Study reported a positive association between sleep duration (9 h/d compared to ≤ 6 h/d) and LTL [18]. However, a separate study with 245 women reported no significant association [19]. Similar mixed results have further been reported in samples of both men and women [13, 14], particularly regarding long sleep duration, which has been linked to both significantly longer [20] and shorter telomere length [21]. A previous exploratory study involving 154 participants indicated a U-shaped association between sleep duration and telomere length [22], which was recapitulated by our large-scale multivariable linear regressions. Nonetheless, stratified MR findings again indicate no causative role of excessive sleep in shortening LTL. Unlike the nonlinear relationships identified for the composite biological age measurements, the stratum estimates for LTL were more consistent across strata and centered around the population-averaged estimate, suggesting a linear effect. While such discrepancy may support the notion that these measurements capture different hallmarks of aging [48], additional investigations are warranted to corroborate our findings and provide deeper insights into the reasons behind the distinct associations observed.
Through the integration of genomics and epigenomics data, our study reveals genetic similarities between short sleep and BioAgeAccel in the musculoskeletal system, as well as between short sleep and LTL in the immune system, providing additional evidence for potential shared genetic mechanisms that contribute to these causal links. Sleep plays a crucial role in connective tissue repair and muscle growth [49], and inadequate sleep has been associated with increased risks of muscle mass reduction and functional decline commonly observed during aging [50]. Moreover, laboratory studies have demonstrated that both acute and chronic sleep loss can have an impact on a wide range of immune function [51]. The immune system is intricately connected to telomere shortening, as its proper functioning relies on the renewal and clonal expansion of T- and B-cell populations [52]. Emerging evidence suggests that immune dysfunctions also contribute to telomere and telomerase deficiency [52]. Taken together, our findings, along with the previous research, support the hypothesis that insufficient sleep adversely affects the musculoskeletal system and immune function, consequently accelerating biological aging. To gain a deeper understanding of these hypothesized mechanisms, future in-depth experimental studies are needed.
A notable strength of our study lies in the comprehensive interrogation of potential sex-heterogeneous effects of sleep duration on biological aging, revealing that short sleep duration accelerates aging more prominently in men than women. This finding aligns with prior research indicating that men are more susceptible to immunosenescence, inflammaging, and higher mortality risk at comparable frailty levels [38]. Importantly, while prior work reported sex-specific association between sleep duration and LTL in men [53], our findings extend this by demonstrating that sufficient sleep slows biological aging in both sexes. Similarly, our age-stratified analyses indicate that insufficient sleep accelerates biological aging in both younger (< 58 years) and older (≥58 years) adults, with a seemingly more pronounced effect observed in the younger group. This is consistent with evidence that younger individuals typically require longer sleep durations and demonstrate reduced resilience to acute sleep deprivation compared to their older counterparts [54]. Overall, these findings emphasize the importance of adequate sleep duration for healthy aging across the population, with particular attention to the needs of men and younger adults. Nevertheless, validation in larger, independent cohorts is necessary before these subgroup findings can inform clinical recommendations.
Several limitations need to be acknowledged. Our sample only comprises individuals of White European ethnic background, necessitating future investigations to ensure the generalizability of findings across diverse racial or ethnic groups. Additionally, the potential presence of a healthy volunteer bias within the UK Biobank may introduce a null bias, given the anticipated lower age accelerations relative to the general UK population. Sleep duration was assessed using a single self-administrated question in the UK Biobank. However, the potential for misclassification error is unlikely to influence our MR estimates as sleep duration is measured in 1-hour increments [55]. Future studies with objective sleep measures are essential to validate our findings. Our study specifically examined telomere length measurements in leukocytes, and further research is needed to determine how well these measurements reflect telomere length in other organ tissues. Finally, it is important to recognize the cross-sectional design of our observational study and approach the interpretation of causality between sleep duration and aging with caution. While MR offers some potential for causal inference, it relies on several assumptions [23] that we have tried to scrutinize, albeit residual uncertainty inevitably remains. For example, it remains possible that some genetic instruments exert direct effects on biological aging, which would violate the exclusion restriction assumption.
Conclusions
Taken together, our study, utilizing extensive phenotypic, genomic, and epigenomic data, consistently demonstrates a significant association between insufficient sleep duration and increased acceleration of biological aging, as indicated by PhenoAgeAccel, BioAgeAccel, and LTL measurements. Further investigation is needed to determine if long sleep is a causal risk factor for accelerating biological aging. Findings suggest that interventions aimed at addressing insufficient sleep may serve as a pathway towards alleviating the burden of aging.
Supplementary Materials
Author Contributions
X.W and X.J. conceptualized the study. X.W. and X.Z. performed the primary analyses with the assistance of S.B., A.G., Z.H., C.H., Y.H., J.X., and M.F. X.W. wrote the manuscript with significant input and comments from X.J., S.B., X.Z., A.G., Z.H., C.H., Y.H., M.F., J.X., and J.L. X.J., J.L., and S.B. supervised the study. All authors contributed to interpreting the findings and critically revising the manuscript.
Conflicts of Interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical Statement and Consent
UK Biobank received ethical approval from the Research Ethics Committee (reference 11/NW/0382). The original GWAS studies used in this research obtained ethical approval from relevant ethics review committees. The written informed consent was obtained from all participants. This study utilizes GWAS summary statistics, which are aggregated data and do not contain any personal information.
Funding
X.J. was supported by the Sichuan Medical Association (S21003), the Science Fund for Creative Research Groups of Science and Technology Bureau of Sichuan Province (2024NSFTD0030), and the National Natural Science Foundation of China (82204170). S.B. is supported by the Wellcome Trust (225790/Z/22/Z). The funders of the study had no role in the conceptualization, design, data collection, analysis, decision to publish, or preparation of the manuscript.
References
- 1. Kennedy BK, Berger SL, Brunet A, Campisi J, Cuervo AM, Epel ES, Franceschi C, Lithgow GJ, Morimoto RI, Pessin JE, Rando TA, Richardson A, Schadt EE, et al. Geroscience: linking aging to chronic disease. Cell. 2014; 159:709–13. https://doi.org/10.1016/j.cell.2014.10.039 [PubMed]
- 2. Gott A, Andrews C, Larriva Hormigos M, Spencer K, Bateson M, Nettle D. Chronological age, biological age, and individual variation in the stress response in the European starling: a follow-up study. PeerJ. 2018; 6:e5842. https://doi.org/10.7717/peerj.5842 [PubMed]
- 3. Ferrucci L, Gonzalez-Freire M, Fabbri E, Simonsick E, Tanaka T, Moore Z, Salimi S, Sierra F, de Cabo R. Measuring biological aging in humans: A quest. Aging Cell. 2020; 19:e13080. https://doi.org/10.1111/acel.13080 [PubMed]
- 4. Jylhävä J, Pedersen NL, Hägg S. Biological Age Predictors. EBioMedicine. 2017; 21:29–36. https://doi.org/10.1016/j.ebiom.2017.03.046 [PubMed]
- 5. Levine ME, Lu AT, Quach A, Chen BH, Assimes TL, Bandinelli S, Hou L, Baccarelli AA, Stewart JD, Li Y, Whitsel EA, Wilson JG, Reiner AP, et al. An epigenetic biomarker of aging for lifespan and healthspan. Aging (Albany NY). 2018; 10:573–91. https://doi.org/10.18632/aging.101414 [PubMed]
- 6. Levine ME. Modeling the rate of senescence: can estimated biological age predict mortality more accurately than chronological age? J Gerontol A Biol Sci Med Sci. 2013; 68:667–74. https://doi.org/10.1093/gerona/gls233 [PubMed]
- 7. Haycock PC, Burgess S, Nounu A, Zheng J, Okoli GN, Bowden J, Wade KH, Timpson NJ, Evans DM, Willeit P, Aviv A, Gaunt TR, Hemani G, et al, and Telomeres Mendelian Randomization Collaboration. Association Between Telomere Length and Risk of Cancer and Non-Neoplastic Diseases: A Mendelian Randomization Study. JAMA Oncol. 2017; 3:636–51. https://doi.org/10.1001/jamaoncol.2016.5945 [PubMed]
- 8. Scheller Madrid A, Rasmussen KL, Rode L, Frikke-Schmidt R, Nordestgaard BG, Bojesen SE. Observational and genetic studies of short telomeres and Alzheimer’s disease in 67,000 and 152,000 individuals: a Mendelian randomization study. Eur J Epidemiol. 2020; 35:147–56. https://doi.org/10.1007/s10654-019-00563-w [PubMed]
- 9. Liu Z, Kuo PL, Horvath S, Crimmins E, Ferrucci L, Levine M. A new aging measure captures morbidity and mortality risk across diverse subpopulations from NHANES IV: A cohort study. PLoS Med. 2018; 15:e1002718. https://doi.org/10.1371/journal.pmed.1002718 [PubMed]
- 10. Carroll JE, Prather AA. Sleep and Biological Aging: A Short Review. Curr Opin Endocr Metab Res. 2021; 18:159–164. https://doi.org/10.1016/j.coemr.2021.03.021 [PubMed]
- 11. Watson NF, Badr MS, Belenky G, Bliwise DL, Buxton OM, Buysse D, Dinges DF, Gangwisch J, Grandner MA, Kushida C, Malhotra RK, Martin JL, Patel SR, et al, and Consensus Conference Panel. Joint Consensus Statement of the American Academy of Sleep Medicine and Sleep Research Society on the Recommended Amount of Sleep for a Healthy Adult: Methodology and Discussion. Sleep. 2015; 38:1161–83. https://doi.org/10.5665/sleep.4886 [PubMed]
- 12. Liu TZ, Xu C, Rota M, Cai H, Zhang C, Shi MJ, Yuan RX, Weng H, Meng XY, Kwong JS, Sun X. Sleep duration and risk of all-cause mortality: A flexible, non-linear, meta-regression of 40 prospective cohort studies. Sleep Med Rev. 2017; 32:28–36. https://doi.org/10.1016/j.smrv.2016.02.005 [PubMed]
- 13. Barragán R, Ortega-Azorín C, Sorlí JV, Asensio EM, Coltell O, St-Onge MP, Portolés O, Corella D. Effect of Physical Activity, Smoking, and Sleep on Telomere Length: A Systematic Review of Observational and Intervention Studies. J Clin Med. 2021; 11:76. https://doi.org/10.3390/jcm11010076 [PubMed]
- 14. Sabot D, Lovegrove R, Stapleton P. The association between sleep quality and telomere length: A systematic literature review. Brain Behav Immun Health. 2023; 28:100577. https://doi.org/10.1016/j.bbih.2022.100577 [PubMed]
- 15. Han KT, Kim DW, Kim SJ. Is Sleep Duration Associated with Biological Age (BA)?: Analysis of (2010−2015) South Korean NHANES Dataset South Korea. Int J Environ Res Public Health. 2018; 15:2009. https://doi.org/10.3390/ijerph15092009 [PubMed]
- 16. Carskadon MA, Chappell KR, Barker DH, Hart AC, Dwyer K, Gredvig-Ardito C, Starr C, McGeary JE. A pilot prospective study of sleep patterns and DNA methylation-characterized epigenetic aging in young adults. BMC Res Notes. 2019; 12:583. https://doi.org/10.1186/s13104-019-4633-1 [PubMed]
- 17. Smail E, Maher B, Moore A. Links of Sleep Duration with Biomarkers of Accelerated Aging: the Baltimore Longitudinal Study of Aging. Innov Aging. 2021; 5:665. https://doi.org/10.1093/GERONI/IGAB046.2512
- 18. Liang G, Schernhammer E, Qi L, Gao X, De Vivo I, Han J. Associations between rotating night shifts, sleep duration, and telomere length in women. PLoS One. 2011; 6:e23462. https://doi.org/10.1371/journal.pone.0023462 [PubMed]
- 19. Prather AA, Puterman E, Lin J, O’Donovan A, Krauss J, Tomiyama AJ, Epel ES, Blackburn EH. Shorter leukocyte telomere length in midlife women with poor sleep quality. J Aging Res. 2011; 2011:721390. https://doi.org/10.4061/2011/721390 [PubMed]
- 20. Zhao H, Han L, Chang D, Ye Y, Shen J, Daniel CR, Gu J, Chow WH, Wu X. Social-demographics, health behaviors, and telomere length in the Mexican American Mano a Mano Cohort. Oncotarget. 2017; 8:96553–67. https://doi.org/10.18632/oncotarget.19903 [PubMed]
- 21. Tempaku P, Hirotsu C, Mazzotti D, Xavier G, Maurya P, Brietzke E, Belangero S, Poyares D, Bittencourt L, Tufik S. Long Sleep Duration, Insomnia, and Insomnia With Short Objective Sleep Duration Are Independently Associated With Short Telomere Length. J Clin Sleep Med. 2018; 14:2037–45. https://doi.org/10.5664/jcsm.7532 [PubMed]
- 22. Cribbet MR, Carlisle M, Cawthon RM, Uchino BN, Williams PG, Smith TW, Gunn HE, Light KC. Cellular aging and restorative processes: subjective sleep quality and duration moderate the association between age and telomere length in a sample of middle-aged and older adults. Sleep. 2014; 37:65–70. https://doi.org/10.5665/sleep.3308 [PubMed]
- 23. Smith GD, Ebrahim S. ‘Mendelian randomization’: can genetic epidemiology contribute to understanding environmental determinants of disease? Int J Epidemiol. 2003; 32:1–22. https://doi.org/10.1093/ije/dyg070 [PubMed]
- 24. Tian H, Mason AM, Liu C, Burgess S. Relaxing parametric assumptions for non-linear Mendelian randomization using a doubly-ranked stratification method. PLoS Genet. 2023; 19:e1010823. https://doi.org/10.1371/journal.pgen.1010823 [PubMed]
- 25. Hu J, Lu J, Lu Q, Weng W, Guan Z, Wang Z. Mendelian randomization and colocalization analyses reveal an association between short sleep duration or morning chronotype and altered leukocyte telomere length. Commun Biol. 2023; 6:1014. https://doi.org/10.1038/s42003-023-05397-7 [PubMed]
- 26. Gao X, Huang N, Guo X, Huang T. Role of sleep quality in the acceleration of biological aging and its potential for preventive interaction on air pollution insults: Findings from the UK Biobank cohort. Aging Cell. 2022; 21:e13610. https://doi.org/10.1111/acel.13610 [PubMed]
- 27. Sudlow C, Gallacher J, Allen N, Beral V, Burton P, Danesh J, Downey P, Elliott P, Green J, Landray M, Liu B, Matthews P, Ong G, et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 2015; 12:e1001779. https://doi.org/10.1371/journal.pmed.1001779 [PubMed]
- 28. Henry A, Katsoulis M, Masi S, Fatemifar G, Denaxas S, Acosta D, Garfield V, Dale CE. The relationship between sleep duration, cognition and dementia: a Mendelian randomization study. Int J Epidemiol. 2019; 48:849–60. https://doi.org/10.1093/ije/dyz071 [PubMed]
- 29. Yang Q, Magnus MC, Kilpi F, Santorelli G, Soares AG, West J, Magnus P, Wright J, Håberg SE, Sanderson E, Lawlor DA, Tilling K, Borges MC. Investigating causal relations between sleep duration and risks of adverse pregnancy and perinatal outcomes: linear and nonlinear Mendelian randomization analyses. BMC Med. 2022; 20:295. https://doi.org/10.1186/s12916-022-02494-y [PubMed]
- 30. Xu Y, Wang X, Belsky DW, McCall WV, Liu Y, Su S. Blunted Rest-Activity Circadian Rhythm Is Associated With Increased Rate of Biological Aging: An Analysis of NHANES 2011-2014. J Gerontol A Biol Sci Med Sci. 2023; 78:407–13. https://doi.org/10.1093/gerona/glac199 [PubMed]
- 31. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013; 153:1194–217. https://doi.org/10.1016/j.cell.2013.05.039 [PubMed]
- 32. Codd V, Denniff M, Swinfield C, Warner SC, Papakonstantinou M, Sheth S, Nanus DE, Budgeon CA, Musicha C, Bountziouka V, Wang Q, Bramley R, Allara E, et al. Measurement and initial characterization of leukocyte telomere length in 474,074 participants in UK Biobank. Nat Aging. 2022; 2:170–79. https://doi.org/10.1038/s43587-021-00166-9 [PubMed]
- 33. Bycroft C, Freeman C, Petkova D, Band G, Elliott LT, Sharp K, Motyer A, Vukcevic D, Delaneau O, O’Connell J, Cortes A, Welsh S, Young A, et al. The UK Biobank resource with deep phenotyping and genomic data. Nature. 2018; 562:203–09. https://doi.org/10.1038/s41586-018-0579-z [PubMed]
- 34. Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D, Maller J, Sklar P, de Bakker PI, Daly MJ, Sham PC. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007; 81:559–75. https://doi.org/10.1086/519795 [PubMed]
- 35. Dashti HS, Jones SE, Wood AR, Lane JM, van Hees VT, Wang H, Rhodes JA, Song Y, Patel K, Anderson SG, Beaumont RN, Bechtold DA, Bowden J, et al. Genome-wide association study identifies genetic loci for self-reported habitual sleep duration supported by accelerometer-derived estimates. Nat Commun. 2019; 10:1100. https://doi.org/10.1038/s41467-019-08917-4 [PubMed]
- 36. Collister JA, Liu X, Clifton L. Calculating Polygenic Risk Scores (PRS) in UK Biobank: A Practical Guide for Epidemiologists. Front Genet. 2022; 13:818574. https://doi.org/10.3389/fgene.2022.818574 [PubMed]
- 37. Kuo CL, Pilling LC, Liu Z, Atkins JL, Levine ME. Genetic associations for two biological age measures point to distinct aging phenotypes. Aging Cell. 2021; 20:e13376. https://doi.org/10.1111/acel.13376 [PubMed]
- 38. Hägg S, Jylhävä J. Sex differences in biological aging with a focus on human studies. Elife. 2021; 10:e63425. https://doi.org/10.7554/eLife.63425 [PubMed]
- 39. Li J, Vitiello MV, Gooneratne NS. Sleep in Normal Aging. Sleep Med Clin. 2018; 13:1–11. https://doi.org/10.1016/j.jsmc.2017.09.001 [PubMed]
- 40. Staley JR, Burgess S. Semiparametric methods for estimation of a nonlinear exposure-outcome relationship using instrumental variables with application to Mendelian randomization. Genet Epidemiol. 2017; 41:341–52. https://doi.org/10.1002/gepi.22041 [PubMed]
- 41. Sun YQ, Burgess S, Staley JR, Wood AM, Bell S, Kaptoge SK, Guo Q, Bolton TR, Mason AM, Butterworth AS, Di Angelantonio E, Vie GÅ, Bjørngaard JH, et al. Body mass index and all cause mortality in HUNT and UK Biobank studies: linear and non-linear mendelian randomisation analyses. BMJ. 2019; 364:l1042. https://doi.org/10.1136/bmj.l1042 [PubMed]
- 42. MacArthur J, Bowler E, Cerezo M, Gil L, Hall P, Hastings E, Junkins H, McMahon A, Milano A, Morales J, Pendlington ZM, Welter D, Burdett T, et al. The new NHGRI-EBI Catalog of published genome-wide association studies (GWAS Catalog). Nucleic Acids Res. 2017; 45:D896–901. https://doi.org/10.1093/nar/gkw1133 [PubMed]
- 43. Finucane HK, Reshef YA, Anttila V, Slowikowski K, Gusev A, Byrnes A, Gazal S, Loh PR, Lareau C, Shoresh N, Genovese G, Saunders A, Macosko E, et al, and Brainstorm Consortium. Heritability enrichment of specifically expressed genes identifies disease-relevant tissues and cell types. Nat Genet. 2018; 50:621–29. https://doi.org/10.1038/s41588-018-0081-4 [PubMed]
- 44. Finucane HK, Bulik-Sullivan B, Gusev A, Trynka G, Reshef Y, Loh PR, Anttila V, Xu H, Zang C, Farh K, Ripke S, Day FR, Purcell S, et al, ReproGen Consortium, Schizophrenia Working Group of the Psychiatric Genomics Consortium, and RACI Consortium. Partitioning heritability by functional annotation using genome-wide association summary statistics. Nat Genet. 2015; 47:1228–35. https://doi.org/10.1038/ng.3404 [PubMed]
- 45. Klemera P, Doubal S. A new approach to the concept and computation of biological age. Mech Ageing Dev. 2006; 127:240–48. https://doi.org/10.1016/j.mad.2005.10.004 [PubMed]
- 46. Yin J, Jin X, Shan Z, Li S, Huang H, Li P, Peng X, Peng Z, Yu K, Bao W, Yang W, Chen X, Liu L. Relationship of Sleep Duration With All-Cause Mortality and Cardiovascular Events: A Systematic Review and Dose-Response Meta-Analysis of Prospective Cohort Studies. J Am Heart Assoc. 2017; 6:e005947. https://doi.org/10.1161/JAHA.117.005947 [PubMed]
- 47. Svensson T, Saito E, Svensson AK, Melander O, Orho-Melander M, Mimura M, Rahman S, Sawada N, Koh WP, Shu XO, Tsuji I, Kanemura S, Park SK, et al. Association of Sleep Duration With All- and Major-Cause Mortality Among Adults in Japan, China, Singapore, and Korea. JAMA Netw Open. 2021; 4:e2122837. https://doi.org/10.1001/jamanetworkopen.2021.22837 [PubMed]
- 48. Belsky DW, Moffitt TE, Cohen AA, Corcoran DL, Levine ME, Prinz JA, Schaefer J, Sugden K, Williams B, Poulton R, Caspi A. Eleven Telomere, Epigenetic Clock, and Biomarker-Composite Quantifications of Biological Aging: Do They Measure the Same Thing? Am J Epidemiol. 2018; 187:1220–30. https://doi.org/10.1093/aje/kwx346 [PubMed]
- 49. Stich FM, Huwiler S, D’Hulst G, Lustenberger C. The Potential Role of Sleep in Promoting a Healthy Body Composition: Underlying Mechanisms Determining Muscle, Fat, and Bone Mass and Their Association with Sleep. Neuroendocrinology. 2022; 112:673–701. https://doi.org/10.1159/000518691 [PubMed]
- 50. Li X, He J, Sun Q. Sleep Duration and Sarcopenia: An Updated Systematic Review and Meta-Analysis. J Am Med Dir Assoc. 2023; 24:1193–206.e5. https://doi.org/10.1016/j.jamda.2023.04.032 [PubMed]
- 51. Besedovsky L, Lange T, Born J. Sleep and immune function. Pflugers Arch. 2012; 463:121–37. https://doi.org/10.1007/s00424-011-1044-0 [PubMed]
- 52. Kordinas V, Ioannidis A, Chatzipanagiotou S. The Telomere/Telomerase System in Chronic Inflammatory Diseases. Cause or Effect? Genes (Basel). 2016; 7:60. https://doi.org/10.3390/genes7090060 [PubMed]
- 53. Jackowska M, Hamer M, Carvalho LA, Erusalimsky JD, Butcher L, Steptoe A. Short sleep duration is associated with shorter telomere length in healthy men: findings from the Whitehall II cohort study. PLoS One. 2012; 7:e47292. https://doi.org/10.1371/journal.pone.0047292 [PubMed]
- 54. Duffy JF, Willson HJ, Wang W, Czeisler CA. Healthy older adults better tolerate sleep deprivation than young adults. J Am Geriatr Soc. 2009; 57:1245–51. https://doi.org/10.1111/j.1532-5415.2009.02303.x [PubMed]
- 55. Pierce BL, VanderWeele TJ. The effect of non-differential measurement error on bias, precision and power in Mendelian randomization studies. Int J Epidemiol. 2012; 41:1383–93. https://doi.org/10.1093/ije/dys141 [PubMed]



