Introduction
Fruit flies, which are ectothermic animals, can live more than twice as long at 18°C than at 25°C [1]. Even though it has been thought that this enhanced longevity at a lower temperature (18°C) is caused by a change of metabolic rate [2], the mechanisms that regulate longevity by ambient temperature are poorly understood. Previously, we found that development at 18°C (from embryo to newborn adult) significantly enhances stress resistance of adult flies with more accumulation of nutrients (especially fat) in the body than development at 25°C [3]. This enhanced resistance to stress was similarly observed in both sexes and sustained up to 30 days (middle age) after hatching of the adult flies [3], indicating that development at a lower temperature, 18°C, significantly enhances the mechanism(s) of stress resistance. Higher stress resistance and/or fat accumulation are frequently found in long-lived flies [4-6] such as mutants of the IGF (insulin/insulin-like growth factor) signaling pathway [7,8]. From the RT-PCR tests of representative stress-related genes, we showed that the development at a lower temperature (18°C) downregulates antimicrobial peptide genes, AttA and DptB, of the Immune deficiency (Imd) pathway [3]. The Imd pathway is known to regulate innate immune responses in Drosophila [9], and the Imd protein activates two downstream branches, JNK/basket and NF-kB/Relish, which are subsequently responsible for the upregulation of stress tolerance and antimicrobial peptide genes, respectively [10,11].
The roles of the Imd pathway have been well studied in a humoral response against intruders, which is characterized by the secretion of antimicrobial peptides (AMPs) into the hemolymph [12]. However, whether the Imd pathway is involved in a longevity mechanism has not been reported. Using hypomorphic imd and AttC mutant flies, here, we show that the mild downregulation of the Imd pathway has a beneficial effect for stress resistance with higher fat content in the body even when developed at 25°C. The Imd pathway functions for the immune response in the fat body [13] which is involved in the metabolism and storage of fat in adult flies. Surprisingly, our data show that the fat-body-specific downregulation of Imd AMP genes significantly enhances heat resistance and extends lifespan.
Results
Imd downregulation in flies developed at 25°C enhances stress resistance and extends lifespan with reduced AMP expressions
To investigate how stress resistance in flies is influenced by the developmental temperatures, we compared the RNA expression levels between 2-day-old male flies developed at 18°C and 25°C by microarray analyses (Fig. 1). We first filtered out non-expressed genes that received both of Affymetrix Absent calls in the gene expression of 18°C and 25°C development and, next, removed highly variable genes for which the standard error was bigger than 1.32-fold from three independent experiments. The final 62% of the genes (11,768) among total gene probes (18,952) were considered to have statistically reliable expressions in the 2-day-old male flies developed at 18°C and 25°C (Fig. 1B). With a cut-off value of ± 1.5-fold changes [14-16], we found that 0.9% of the genes (176/18,952) are differentially expressed between the flies developed at 18°C and 25°C (Fig. 1B). The gene ontology analyses of these 176 genes revealed a possible link of those genes to the stress resistance and aging process, such as defense responses, extracellular proteins, and the polysaccharide metabolic process (Fig. 1C). Among the defense response genes, antimicrobial peptide (AMP) genes in the Imd pathway [9] were specifically downregulated with the development at a lower temperature 18°C (Fig. 1D). Imd AMP genes, DptB and AttA, were still downregulated even after 30 days of age at 25°C (Fig. 1E) when the higher stress resistance was sustained (Fig. 1A).
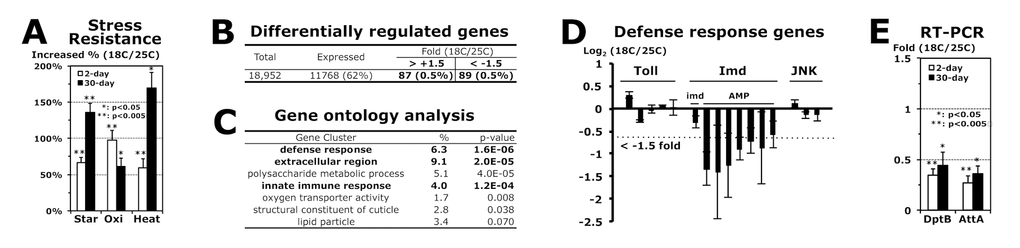
Figure 1. Imd AMP genes are downregulated in adult flies developed at 18°C. (A) Increased stress resistance of 2-day-old (white) and 30-day-old (black) male adult flies developed at 18 °C, which were compared from 2- and 30-day-old flies developed at 25 °C (0%), respectively. The median survival times of flies under each stress (starvation, oxidation or heat) were calculated from the survival curves of 3 ~ 5 independent experiments, and then the changed percentage is represented as average ± standard error of mean (SEM) following normalization with the median of flies developed at 25 °C (starvation: 28.2 and 11.1 hours; oxidation: 15.4 and 8.0 hours; heat: 15.3 and 1.7 hours of 2- and 30-day-old flies, respectively). P-value (*): Student’s t-test. (B) The gene expression analyses between 2-day-old male flies developed at 18°C and 25°C. From three independent microarray experiments, the fold changes of gene expression (18C/25C) were averaged with SEM. (C) With the genes changed more than 1.5 fold (total 176 in B), the gene ontology was analyzed using a DAVID web tool (http://david.abcc.ncifcrf.gov/home.jsp). %: involved genes/total 176 genes; p-value: a modified Fisher Exact. (D) Expressional changes of genes in Toll (Tl, Def, Drs-l, Drs, Mtk), Imd (imd, DptB, AttA, AttB, AttC, CecB, CecC, Dro) and JNK (bsk, GstD1, Thor) pathways from the microarray experiments (B). (E) RT and real-time PCR analyses of AMP genes in Imd pathway. Gene expression folds (18C/25C) of 2- and 30-day-old male flies were averaged from 4 ~ 8 independent experiments using four different RNA batches.
To test if the downregulation of imd (0.76-fold) and AMP genes contributed to higher stress resistance in the flies developed at 18°C (Fig. 1A), we reduced the expression of the imd gene with the hypomorphic imd mutants and compared the stress resistance between the wild-type and imd mutant flies, both developed at 25°C. The imdP mutant, in which a P-element is inserted into the imd promoter, reduced the imd expression (heterozygote imdP/+: 0.88-fold; homozygote imdP: 0.45-fold in Fig. 2A) and then decreased the expression of downstream AMP genes (Fig. 2A). Dependent on the reduced levels of imd and AMP expressions, the stress resistances were enhanced (Fig. 2B-C) with a higher SOD2 (MnSOD) activity (33.8% increase). Interestingly, lifespan of imdP male flies were extended up to 24.8% (Fig. 2D) and these longer lifespans were shown in both male and female flies (Fig. 2E). Previous studies have suggested that attaining higher resistance against stresses may extend the lifespan of fruit flies [17-19]. It was shown that several long-lived mutant flies are more resistant to stresses such as starvation, oxidation, and heat than wild-type flies [5], indicating that lifespan and stress resistance often correlate positively with each other [20]. When we measured the nutrients of fly bodies, only fat (triacylglycerol) content increased ~40% in both hetero and homozygous imdP flies (Fig. 2F), which is commonly observed in long-lived mutant animals including C. elegans, flies, and mice [7,8,21]. These nutrient storages are often interpreted as an adaptation mechanism for increased energy demands during a longer lifespan and against stress resistance [6,22].
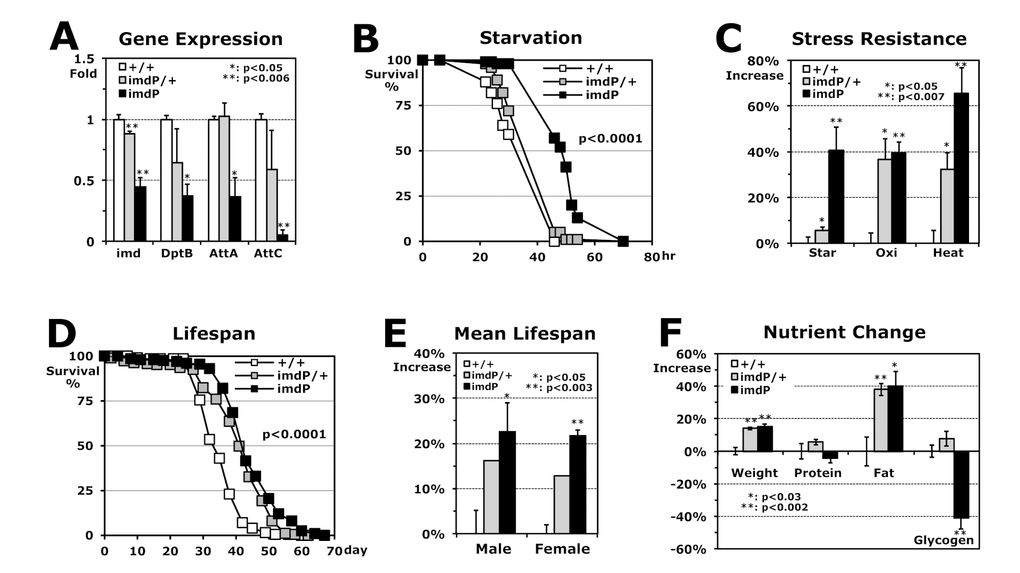
Figure 2. Stress resistance and lifespan enhanced by Imd downregulation in flies developed at 25°C. (A) RT-PCR analyses of imd and AMP genes in Imd pathway. Using total RNAs of 2-day-old male flies developed at 25°C, gene expressions in hetero (imdP/+) and homozygous (imdP) mutants were normalized with those of wild-type (+/+: 1 fold) and then, were averaged with SEM. (B) The survival curve for starvation stress using 2-day-old male flies (p-value: log-rank test). (C) Increased stress resistance of 2-day-old imdP mutant flies from the wild-type flies (+/+: 0%). The median survival times of flies under each stress (starvation, oxidation or heat) were calculated from the several survival curves (B) and then, the changed percentages are represented as average ± SEM after normalization with the median times of wild-type flies (Table 1). (D) The lifespan of adult male flies between the wild-type and imdP mutants. (E) Increased mean lifespan of imdP mutant flies. Percent changes of mean lifespan are indicated as average ± SEM normalized by wild-type lifespan days (Table 1), which were calculated from the several lifespan curves (D). (F) Changes of weight and nutritional contents between 2-day-old wild-type and imdP mutants male flies. After fresh weight per fly was measured (mg/fly), the nutrient contents were normalized by fresh weight of fly (μg/mg). The data represent average ± SEM normalized by nutrient contents of wild-type flies (Table 1).
Mild downregulation of the Imd pathway increases stress resistance and fat content in adult flies developed at 25°C
The imd1 mutant is known to be a stronger hypomorphic mutant than imdP in the immune response to E. coli infection [23]. Although the expression change of the imd gene was not shown due to the missense mutation of imd1 [24], the AMP genes in the Imd pathway were downregulated in the imd1 flies (Table 1). The heterozygous (imd1/+) flies exhibited enhanced stress resistances, higher MnSOD activity, longer lifespans, and increased fat content (Table 1), as shown in the imdP mutants (Fig. 2). However, in imd1 homozygous mutants, the stress resistance was not significantly changed under oxidation or was highly decreased when subjected to heat stress (Table 1). Another imdNP mutant, in which a P-element is inserted into the coding region of the imd gene, also showed the different phenotypes between the heterozygous and homozygous imdNP flies (Fig. S1). A possible explanation is that the JNK/basket pathway, which is activated by the Imd protein and then is subsequently responsible for upregulation of the stress response genes [10,11], is less active due to the severe loss of imd function. The JNK downstream genes, Thor and GstD1, are upregulated for stress tolerance [25]. As expected, their expressions were increased during oxidative stress (24 hours) of the wild-type flies (Fig. 3A). In contrast, AMP genes (DptB and AttC) were downregulated (Fig. 3A), which suggests that reducing Imd AMP expressions may be beneficial for stress tolerance. Compared to the wild-type flies, both imdP and imd1 mutant flies show more reduced DptB and AttC expressions after 24 hours of oxidative stress (Fig. 3B). However, the imd1 flies also showed significantly reduced expressions of Thor and GstD1 (Fig. 3B). Then, we found that the imdP flies were able to survive longer than the wild-type and imd1 flies under the oxidative stress (Fig. 3C), probably due to reduced AMP expressions, but still sustained expressions of Thor and GstD1.
Table 1. The comparison of imd1 hetero and homozygous mutants from the wild-type (+/+).
| Genotype | Gene Expression (fold) | Stress Resistance (hr) | MnSOD | Lifespan (day) | Weight | Nutrients (μg/mg) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| imd | DptB | AttA | Starvation | Oxidation | Heat | (mU/μg) | Male | Female | (mg/fly) | Protein | Fat | Glycogen | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| +/+ | 1.00 | 1.00 | 1.00 | 31.8 | 22.8 | 13.7 | 0.30 | 35.1 | 39.3 | 0.67 | 56.2 | 11.1 | 18.7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| imd1/+ | 1.13 | *0.55* | *0.48* | *46%* | **29%** | 6% | *54%* | *17%* | *16%* | 6% | -4% | *40%* | *32%* | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| imd1 | 0.88 | *0.34* | *0.32* | **37%** | 7% | *-177%** | ND | *-34%** | ND | 8% | -2% | **58%** | *-55%** | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The data of gene expression (RT-PCR), stress resistance (median survival time), MnSOD (activity per mitochondrial proteins), lifespan (mean lifespan), weight (fresh weight per fly), and nutritional contents (amount per fresh fly weight) represent the original values of wild-type and percentages changed from the wild-type in the imd1 mutants (2-day-old male flies developed at 25°C). Bold: significant p-values < 0.05 (*) ~ 0.005 (**). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
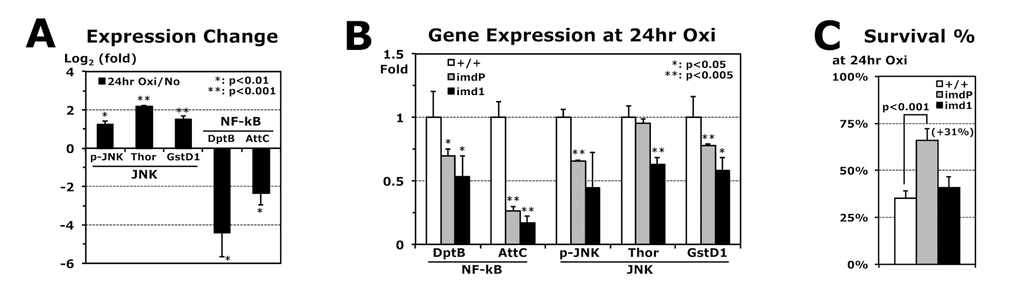
Figure 3. Reduced AMP expressions during paraquat-induced oxidative stress. (A) Gene expressions in 2-day-old male wild-type (+/+) flies after 24 hours of oxidative stress (20 mM paraquat), which are normalized from those of flies without the stress (log21 = 0). p-JNK: average with SEM from Western blots (Phospho-JNK/GAPDH); Thor, GstD1 (JNK pathway), DptB, and AttC (NF-kB pathway): RT-PCR experiments using total RNAs of flies developed at 25°C. (B) Gene expressions in imdP and imd1 homozygous mutants after 24 hours of oxidative stress, which were compared with those of wild-type (+/+: 1 fold). (C) Survival percentages of wild-type (+/+), imdP, and imd1 mutants at 24 hours of oxidative stress. Parenthesis: increased survivorship (%) of imdP mutants from wild-type flies.
AMP downregulations of the Imd pathway directly enhance stress resistance and extend lifespan
Next, to investigate if the expression changes of Imd-driven AMP genes are directly related to stress resistance and longevity, we tested AttC-P mutant flies in which AttC expression is completely removed without any change in the expression of another longevity-related gene, loco [26] (Fig. 4A). The AttC-P flies interestingly enhanced stress resistances against oxidation and heat, significantly (Fig. 4B-C), as did the imdP mutant flies (Fig. 2C). In addition, lifespan was 21% extended with the AttC downregulation (Fig. 4D). Using an UAS/Gal4 system [27], we examined the downregulation effect of another AMP gene (UAS-DptB-dsRNAi: DptBRi) with an arm-Gal4 driver that expresses a target UAS-gene in the whole body. The DptBRi/armGal4 flies also exhibited higher stress resistance (~21% increase) and longer lifespan (+13%) compared to the control DptBRi/+ flies (Fig. 4E-G), indicating that the expression levels of antimicrobial peptide genes in the Imd pathway are directly involved in the stress resistance and the longevity mechanism.
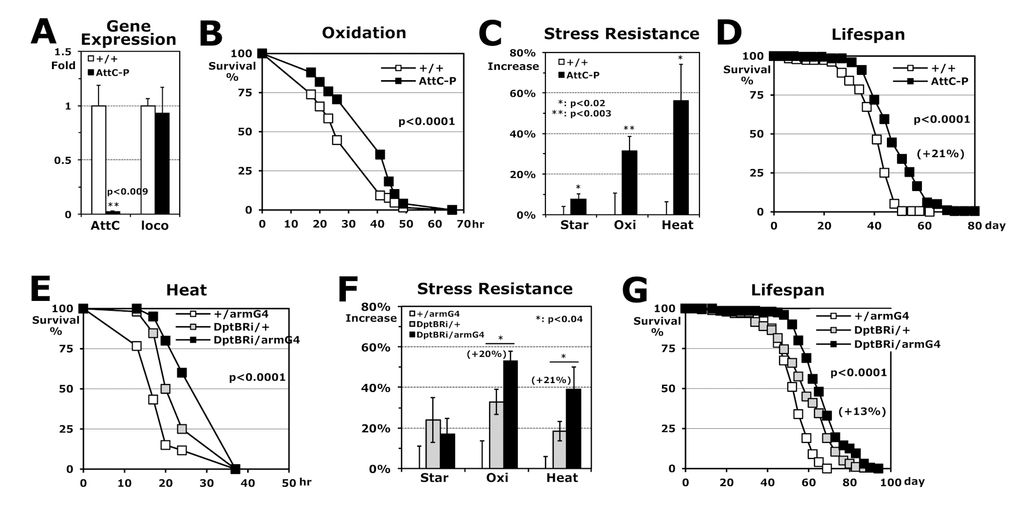
Figure 4. Stress resistance and lifespan enhanced by downregulation of AMP genes in Imd pathway. (A) Gene expressions of AttC and loco in AttC-P homozygous mutant, which were normalized with those of wild-type (+/+: 1 fold) using total RNAs of 2-day-old male flies developed at 25°C. (B) The survival curve for oxidation stress using 2-day-old male flies (p-value: log-rank test). (C) Increased stress resistance of 2-day-old AttC-P mutant flies from the wild-type flies (+/+: 0%). The median survival times of flies under each stress (starvation, oxidation or heat) were calculated from the several survival curves (B) and then, the percentages changed from wild-type flies are represented as average ± SEM. (D) The lifespan of adult male flies between the wild-type and AttC-P mutant. Parenthesis: increased percentage of mean lifespan of AttC-P mutant from the wild-type flies. (E) The survival curve for heat stress using 2-day-old male flies between single transgene controls (+/armG4, DptBRi/+) and DptB downregulation in a whole body (DptBRi/armG4). (F) Increased stress resistance of 2-day-old DptBRi/armG4 flies from the control +/armG4 (0%). Parenthesis: increased percentage of median survival time of the DptBRi/armG4 flies from another control DptBRi/+. (G) The lifespan of adult male flies between the two controls and DptB downregulation. Parenthesis: increase percentage of DptBRi/armG4 flies from the DptBRi/+ control.
The fat body, the fruit fly analogue of mammalian liver and adipose tissues, is known to function in metabolic homeostasis, stress tolerance, growth, and longevity in Drosophila [28,29]. For the antimicrobial response to bacterial infection, the antimicrobial peptide genes are mainly synthesized from fat body and secreted into the hemolymph [13]. We tested how downregulation of AMP genes in the fat body affects stress resistance and lifespan. We found that fat-body-specific (r4-Gal4 [30],) downregulations of imd, DptB, and AttC genes enhanced heat resistances significantly, between 20% and 32% (Fig. 5A-B), compared to the downregulation of another longevity gene, rpd3 [31,32], in the fat body (1.5% in Fig. 5B). Surprisingly, the AttCRi/r4Gal4 flies could exhibit up to 25% extended lifespan compared to the control AttCRi/+ flies (Fig. 5C), indicating that a manipulation of AMP expression in fat body tissues affects longevity of the whole body. Our data propose that the Imd pathway regulates the longevity mechanism through two downstream branches (Fig. 5D): 1) positively by stress tolerance genes of p-JNK [25,29,33,34] and 2) negatively by antimicrobial peptide genes of NF-kB that function positively in the immune response to bacterial infection [9,35].
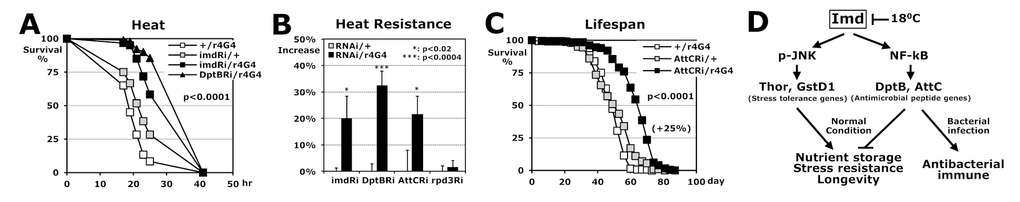
Figure 5. Stress resistance and lifespan enhanced by fat body-specific downregulations of imd and AMP genes in Imd pathway. (A) The survival curve for heat stress using 2-day-old male flies between single transgene controls (+/r4G4, imdRi/+) and fat-body-specific imd or DptB downregulation (imdRi/ or DptBRi/r4G4). (B) Comparison of heat resistance between RNAi single transgenic (Ri/+: 0%) and fat body-specific downregulation (Ri/r4G4). The median survival times of flies under heat stress were calculated from the several survival curves (A) and the median survival time of common control +/r4G4 flies was lower than those of each Ri/+ flies (A). (C) The lifespan of adult male flies between the two controls and a fat-body-specific AttC downregulation (AttCRi/r4G4). Parenthesis: increase percentage of the AttCRi/r4G4 from AttCRi/+ flies. (D) Imd pathway regulates the longevity mechanism through two pathways.
Discussion
The antimicrobial peptide (AMP) genes in the Imd pathway were specifically downregulated in the wild-type flies developed at a lower temperature 18°C (Fig. 1D), which exhibited the stronger resistance to all three applied stressors (starvation, oxidation, and heat) (Fig. 1A) [3]. We found that the flies developed at 25°C also can enhance stress resistances (Fig. 2B-C) when the imd and downstream AMP genes are downregulated (Fig. 2A). However, these additional stress resistances by the imdP mutation in development at 25°C were not observed when the imdP flies were developed at 18°C (Fig. S2), indicating that the stress resistances are already induced by the downregulation of Imd AMP genes in wild-type flies developed at 18°C (Fig. 1). Instead, a heat resistance was significantly reduced in the imdP flies developed at 18°C (Fig. S2B) as shown in the severe hypomorphic imd1 flies (Table 1). It implies that intense downregulation of the Imd pathway has a negative effect on the heat resistance with additional downregulation of another downstream JNK/basket pathway (Fig. 3B) that is required to activate the stress response genes [10,11]. Overall, our data support that a mild downregulation of the Imd pathway enhances stress resistance. However, the increased levels of stress resistance by imdP flies developed at 25°C (Fig. 2B-C) were not enough to reach the levels that the 18°C development of wild-type flies induces (Fig. 1A), suggesting that the downregulation of Imd AMP genes alone cannot fully explain the higher stress resistances in flies developed at 18°C (Fig. 1A).
Long-lived mutant animals, including C. elegans, flies, and mice, commonly show increased fat content [7,8,21], and our imd-downregulated (imdP and imd1/+) flies also show increased fat (triacylgycerol) content with extended lifespan (Fig. 2D-F and Table 1). It is known that Imd-driven AMP proteins dominantly function in the fat body, which is involved in the metabolism and storage of fat in adult flies, for the antimicrobial response to bacterial infection [13]. The increased fat content in imd flies suggests a possibility that Imd AMP proteins are related to the fat metabolism in Drosophila fat body. Accordingly, the fat-body-specific AttC downregulation (AttCRi/r4Gal4) extended lifespan with enhanced heat resistance (Fig. 5). However, the Imd AMP downregulation in fat body did not affect starvation and oxidation resistances (data not shown) in contrast to the increased oxidation resistance shown with the downregulation in the whole body (Fig. 4B-C and F), suggesting that Imd AMP proteins may be involved differently in the several mechanisms against the stressors.
The Drosophila innate immunity AMP genes are upregulated during aging [15], which may prepare pathogen defenses promptly in the old-aged flies that have weak immune systems. However, it was reported that female fecundity is reduced by the induction of innate immunity [36] and lesser expressions of AMP genes are found in longer-lived flies [15], suggesting that less activation of AMP synthesis has a beneficial effect for other biological processes. In summary, our data indicate that mild downregulation of the Imd pathway increases stress resistance, lifespan, and fat content in adult flies, which mimics the enhanced stress resistance caused by a lower developmental temperature [3]. There are many reports showing that stress resistance is intensely related to the aging process [20]. Here, we demonstrate that a delicate modulation of the Imd pathway is important for the regulation of stress resistance and lifespan.
Methods
Fly genotypes and aging assay
The imdP (P{EPgy2}imdEY08573), AttC-P (Mi{ET1}AttCMB05438), UAS-DptBRi (P{TRiP.HM05186}attP2), arm-Gal4, and r4-Gal4 flies were obtained from the Bloomington Drosophila stock center. The UAS-dsRNAi stocks (AttCRi: V47041; imdRi: V9253) and imdNP (P{GawB}imdNP1182) flies were obtained from Vienna and Kyoto stock centers, respectively. The imd1 (imd1) was kindly provided by D. Kimbrell [23]. The y1 w1 (Bloomington) flies were used as wild-type control and the flies obtained outside were six times isogenized with y1 w1 before the stress resistance and aging tests. Virgin flies were collected from a bottle in which larval density was controlled in a standard cornmeal medium, and were used for all fly experiments including stress response, aging, and nutritional content studies [26]. For the aging test, 200 virgin flies (20 flies per vial) were counted and transferred to fresh standard cornmeal vials every 3-4 days [5]. Mean lifespan was calculated from the lifespan curves and averaged with standard error of mean (SEM) from 2 ~ 4 independent experiments.
Stress response assays
To measure stress responses, 100 newly eclosed flies (20 flies per vial) were kept on a standard cornmeal medium at 25°C for 2 days [3,5,26]. For the starvation test, these 2-day-old adult flies were transferred to new vials (2.5 × 9.3 cm) containing two filter circles (2.4-cm diameter, Fisher Scientific) soaked in 300 μl of distilled water, and were maintained at 25°C under moist conditions with 100 μl of water added every 12 hrs. For the oxidative stress test, the 2-day-old adult flies were starved for 6 hrs at 25°C as described above. The flies were then transferred to new vials containing two filter circles wetted with 300 μl of 20 mM methyl viologen hydrate (Paraquat, Fisher Scientific) in a 5% sucrose solution and maintained at 25°C. For the heat test, the 2-day-old adult flies were transferred to new vials containing standard cornmeal medium and maintained at 37°C with 30% humidity. The median survival times of flies under each stress (starvation, oxidation, or heat) were calculated from the survival curves of 4 ~ 8 independent experiments.
Nutrients and MnSOD assays
All 20 flies were weighed and homogenized with specific buffers for nutrients (fat, protein, and glycogen: 0.01 M KH2PO4, 1 mM EDTA pH 7.4) [37] and MnSOD assays (20 mM HEPES, pH 7.2, 1 mM EGTA, 210 mM mannitol, 70 mM sucrose). The supernatant was recovered by centrifugation at 4,000g for 10 min at 4°C and used for nutrient measurement (triacylglycerol: Cayman Chemical; protein: Pierce; glycogen: BioVision). For measuring MnSOD activity, the recovered supernatant above was centrifuged again at 10,000g for 15 min at 4°C to pellet down the mitochondrial fraction containing MnSOD. This pellet was homogenized with the same buffer and used for the MnSOD assay (Cayman Chemical) in the presence of 2 mM KCN (Cu/ZnSOD inhibitor). Each assay was repeated and averaged (± SEM) from the 5 ~ 20 independent samples.
Microarray and RT-PCR analyses
Total RNA was extracted with the TRIzol Reagent (Invitrogen) from a whole body of male adult flies and used as probes for the hybridization reaction on Affymetrix Drosophila 2.0 microarray chips in the EOHSI facility (http://eohsi.rutgers.edu/). The expression levels of genes were calculated by the RMA (robust multichip analysis) normalization method. For RT-PCR, oligo dT-primed cDNAs (Superscript II RT, Invitrogen) were made from 5 μg total RNA of adult flies and used as templates for the quantitative real-time PCR, which was performed with power SYBR green PCR mix (Applied Biosystems). The rp49 gene was used as an internal reference for normalizing the quality of total RNA purified from each flies. The fold changes in gene expression were determined by comparative CT method (ABI Prism 7700 Sequence Detection System User Bulletin #2, Applied Biosystems) and then, were averaged with SEM from 4 ~ 8 independent experiments using at least two different RNA batches.
Supplementary Materials
Acknowledgements
We thank Dr. Deborah Kimbrell for imd1 flies; Zachary Kopp and Ayush Parikh for research assistance; Christopher Brady for critical reading; Dr. Hyangyee Oh for research support.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
This work was supported by the New Jersey Health Foundation (NJHF) and National Institute on Aging (NIA) grant AG042021.
References
- 1. Miquel J, Lundgren PR, Bensch KG, Atlan H. Effects of temperature on the life span, vitality and fine structure of Drosophila melanogaster.. Mech Ageing Dev. 1976; 5:347–70. https://doi.org/10.1016/0047-6374(76)90034-8 [PubMed]
- 2. Helfand SL, Rogina B. Genetics of aging in the fruit fly, Drosophila melanogaster.. Annu Rev Genet. 2003; 37:329–48. https://doi.org/10.1146/annurev.genet.37.040103.095211 [PubMed]
- 3. Kim K, Lin YR, Park Y. Enhancement of stress resistances and downregulation of Imd pathway by lower developmental temperature in Drosophila melanogaster.. Exp Gerontol. 2010; 45:984–87. https://doi.org/10.1016/j.exger.2010.08.033 [PubMed]
- 4. Clancy DJ, Gems D, Harshman LG, Oldham S, Stocker H, Hafen E, Leevers SJ, Partridge L. Extension of life-span by loss of CHICO, a Drosophila insulin receptor substrate protein. Science. 2001; 292:104–06. https://doi.org/10.1126/science.1057991 [PubMed]
- 5. Lin YJ, Seroude L, Benzer S. Extended life-span and stress resistance in the Drosophila mutant methuselah.. Science. 1998; 282:943–46. https://doi.org/10.1126/science.282.5390.943 [PubMed]
- 6. Djawdan M, Sugiyama TT, Schlaeger LK, Bradley TJ, Rose MR. Metabolic aspects of the trade-off between fecundity and longevity in Drosophila melanogaster. Physiol Zool. 1996; 69:1176–95. https://doi.org/10.1086/physzool.69.5.30164252
- 7. Tatar M, Kopelman A, Epstein D, Tu MP, Yin CM, Garofalo RS. A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science. 2001; 292:107–10. https://doi.org/10.1126/science.1057987 [PubMed]
- 8. Kimura KD, Tissenbaum HA, Liu Y, Ruvkun G. daf-2, an insulin receptor-like gene that regulates longevity and diapause in Caenorhabditis elegans.. Science. 1997; 277:942–46. https://doi.org/10.1126/science.277.5328.942 [PubMed]
- 9. Aggarwal K, Silverman N. Positive and negative regulation of the Drosophila immune response. BMB Rep. 2008; 41:267–77. https://doi.org/10.5483/BMBRep.2008.41.4.267 [PubMed]
- 10. Silverman N, Zhou R, Erlich RL, Hunter M, Bernstein E, Schneider D, Maniatis T. Immune activation of NF-kappaB and JNK requires Drosophila TAK1. J Biol Chem. 2003; 278:48928–34. https://doi.org/10.1074/jbc.M304802200 [PubMed]
- 11. Boutros M, Agaisse H, Perrimon N. Sequential activation of signaling pathways during innate immune responses in Drosophila.. Dev Cell. 2002; 3:711–22. https://doi.org/10.1016/S1534-5807(02)00325-8 [PubMed]
- 12. Myllymäki H, Valanne S, Rämet M. The Drosophila imd signaling pathway. J Immunol. 2014; 192:3455–62. https://doi.org/10.4049/jimmunol.1303309 [PubMed]
- 13. Hoffmann JA, Reichhart JM. Drosophila innate immunity: an evolutionary perspective. Nat Immunol. 2002; 3:121–26. https://doi.org/10.1038/ni0202-121 [PubMed]
- 14. Hamada FN, Park PJ, Gordadze PR, Kuroda MI. Global regulation of X chromosomal genes by the MSL complex in Drosophila melanogaster.. Genes Dev. 2005; 19:2289–94. https://doi.org/10.1101/gad.1343705 [PubMed]
- 15. Landis GN, Abdueva D, Skvortsov D, Yang J, Rabin BE, Carrick J, Tavaré S, Tower J. Similar gene expression patterns characterize aging and oxidative stress in Drosophila melanogaster.. Proc Natl Acad Sci USA. 2004; 101:7663–68. https://doi.org/10.1073/pnas.0307605101 [PubMed]
- 16. Chamilos G, Lewis RE, Hu J, Xiao L, Zal T, Gilliet M, Halder G, Kontoyiannis DP. Drosophila melanogaster as a model host to dissect the immunopathogenesis of zygomycosis. Proc Natl Acad Sci USA. 2008; 105:9367–72. https://doi.org/10.1073/pnas.0709578105 [PubMed]
- 17. Parkes TL, Elia AJ, Dickinson D, Hilliker AJ, Phillips JP, Boulianne GL. Extension of Drosophila lifespan by overexpression of human SOD1 in motorneurons. Nat Genet. 1998; 19:171–74. https://doi.org/10.1038/534 [PubMed]
- 18. Chavous DA, Jackson FR, O’Connor CM. Extension of the Drosophila lifespan by overexpression of a protein repair methyltransferase. Proc Natl Acad Sci USA. 2001; 98:14814–18. https://doi.org/10.1073/pnas.251446498 [PubMed]
- 19. Ruan H, Tang XD, Chen ML, Joiner ML, Sun G, Brot N, Weissbach H, Heinemann SH, Iverson L, Wu CF, Hoshi T. High-quality life extension by the enzyme peptide methionine sulfoxide reductase. Proc Natl Acad Sci USA. 2002; 99:2748–53. https://doi.org/10.1073/pnas.032671199 [PubMed]
- 20. Vermeulen CJ, Loeschcke V. Longevity and the stress response in Drosophila.. Exp Gerontol. 2007; 42:153–59. https://doi.org/10.1016/j.exger.2006.09.014 [PubMed]
- 21. Brüning JC, Gautam D, Burks DJ, Gillette J, Schubert M, Orban PC, Klein R, Krone W, Müller-Wieland D, Kahn CR. Role of brain insulin receptor in control of body weight and reproduction. Science. 2000; 289:2122–25. https://doi.org/10.1126/science.289.5487.2122 [PubMed]
- 22. Djawdan M, Chippindale AK, Rose MR, Bradley TJ. Metabolic reserves and evolved stress resistance in Drosophila melanogaster.. Physiol Zool. 1998; 71:584–94. https://doi.org/10.1086/515963 [PubMed]
- 23. Taylor K, Kimbrell DA. Host immune response and differential survival of the sexes in Drosophila.. Fly (Austin). 2007; 1:197–204. https://doi.org/10.4161/fly.5082 [PubMed]
- 24. Georgel P, Naitza S, Kappler C, Ferrandon D, Zachary D, Swimmer C, Kopczynski C, Duyk G, Reichhart JM, Hoffmann JA. Drosophila immune deficiency (IMD) is a death domain protein that activates antibacterial defense and can promote apoptosis. Dev Cell. 2001; 1:503–14. https://doi.org/10.1016/S1534-5807(01)00059-4 [PubMed]
- 25. Karpac J, Hull-Thompson J, Falleur M, Jasper H. JNK signaling in insulin-producing cells is required for adaptive responses to stress in Drosophila.. Aging Cell. 2009; 8:288–95. https://doi.org/10.1111/j.1474-9726.2009.00476.x [PubMed]
- 26. Lin YR, Kim K, Yang Y, Ivessa A, Sadoshima J, Park Y. Regulation of longevity by regulator of G-protein signaling protein, Loco. Aging Cell. 2011; 10:438–47. https://doi.org/10.1111/j.1474-9726.2011.00678.x [PubMed]
- 27. Brand AH, Perrimon N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development. 1993; 118:401–15. [PubMed]
- 28. Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006; 444:860–67. https://doi.org/10.1038/nature05485 [PubMed]
- 29. Hull-Thompson J, Muffat J, Sanchez D, Walker DW, Benzer S, Ganfornina MD, Jasper H. Control of metabolic homeostasis by stress signaling is mediated by the lipocalin NLaz. PLoS Genet. 2009; 5:e1000460. https://doi.org/10.1371/journal.pgen.1000460 [PubMed]
- 30. DiAngelo JR, Bland ML, Bambina S, Cherry S, Birnbaum MJ. The immune response attenuates growth and nutrient storage in Drosophila by reducing insulin signaling. Proc Natl Acad Sci USA. 2009; 106:20853–58. https://doi.org/10.1073/pnas.0906749106 [PubMed]
- 31. Kopp ZA, Hsieh JL, Li A, Wang W, Bhatt DT, Lee A, Kim SY, Fan D, Shah V, Siddiqui E, Ragam R, Park K, Ardeshna D, et al. Heart-specific Rpd3 downregulation enhances cardiac function and longevity. Aging (Albany NY). 2015; 7:648–63. https://doi.org/10.18632/aging.100806 [PubMed]
- 32. Rogina B, Helfand SL, Frankel S. Longevity regulation by Drosophila Rpd3 deacetylase and caloric restriction. Science. 2002; 298:1745. https://doi.org/10.1126/science.1078986 [PubMed]
- 33. Wang MC, Bohmann D, Jasper H. JNK extends life span and limits growth by antagonizing cellular and organism-wide responses to insulin signaling. Cell. 2005; 121:115–25. https://doi.org/10.1016/j.cell.2005.02.030 [PubMed]
- 34. Wang MC, Bohmann D, Jasper H. JNK signaling confers tolerance to oxidative stress and extends lifespan in Drosophila. Dev Cell. 2003; 5:811–16. https://doi.org/10.1016/S1534-5807(03)00323-X [PubMed]
- 35. Kim T, Kim YJ. Overview of innate immunity in Drosophila.. J Biochem Mol Biol. 2005; 38:121–27. [PubMed]
- 36. Zerofsky M, Harel E, Silverman N, Tatar M. Aging of the innate immune response in Drosophila melanogaster.. Aging Cell. 2005; 4:103–08. https://doi.org/10.1111/j.1474-9728.2005.00147.x [PubMed]
- 37. Clark AG, Keith LE. Variation among extracted lines of Drosophila melanogaster in triacylglycerol and carbohydrate storage. Genetics. 1988; 119:595–607. [PubMed]



