Introduction
Over the next 50 years, the aging population in developing countries is expected to grow four-fold. Since the end of the 20th century, China has gradually become an aging society. The problems brought on by aging have become more prominent and pose a serious challenge to the development of the entire society. Therefore, it is urgent to deal with the diseases caused by aging and the aging body. Aging is a natural law that cannot be avoided by individuals. The body is chronically affected by various environmental factors inside and outside the body, causing the structure and function of various tissues and organs to gradually degenerate, eventually developing chronic tissue syndromes or organ dysfunction [1]. However, the mechanism of aging has not been thoroughly elucidated to date. In recent years, the rapid development of cell biotherapy has led to new opportunities to elucidate the diseases related to the elderly. The role of cell biotherapy in this regard has attracted the attention of researchers and achieved important progress.
Adult stem cells play an essential role in tissue engineering research and are widely used to treat degenerative diseases. Stem cells can self-renew and differentiate and are seed cells that maintain the structure and function of tissues and organs. The whole process of human growth, development, aging and death involves the proliferation and differentiation of stem cells. The physiological replacement of tissues and the repair of pathological damage are also dependent on the mobilization, proliferation and differentiation of stem cells [2]. The aging of stem cells is an important cause of the decline in tissue and organ function and is also an important factor affecting the efficacy of stem cell therapy in aging patients [3]. The aging of the human body is mainly due to the decrease in the number of stem cells and their activity, resulting in fewer young cells than aged cells, a slow metabolism and, subsequently, degenerative diseases [4]. Signer et al. showed that aging is a gradual, cumulative process. The normal operation and environmental exposure of the body may cause damage. Therefore, the body needs to be repaired continuously. In embryonic development and human growth, stem cells have robust proliferation and differentiation ability; therefore, repairs are easy at these stages. However, in aging tissues and organs, the activity of stem cells is markedly decreased. With increasing age, the number and activity of stem cells in the body are decreased, the appearance of cells in some tissues and organs is lost, and a novel supply of cells is not obtained. This change will affect the function of the organism, eventually leading to functional failure of important organs and even death [5].
Mesenchymal stem cells (MSCs) are heterogeneous adult stem cells that were originally isolated from the bone marrow and later extracted from the connective tissue of almost all organs. Professor P. Bianco and colleagues [6–11] argued that the trilineage MSCs in bone marrow are skeletal progenitors that differ from MSC-like cells in other tissues. It is clear that non-BMSCs may be more accurately referred to as MSC-like cells. MSCs have good self-renewal ability and the ability to differentiate into a variety of mesodermal cells, such as adipocytes, osteoblasts and chondrocytes, which serve as functional criteria for identifying MSCs [12].
MSCs exhibit a variety of biological functions, including proper immune regulation and secretion of growth factors, cytokines and angiogenic mediators. These cells have immunosuppressive functions. Therefore, we believe that MSCs may be an effective therapeutic for immune mediator-related diseases [13, 14]. By expressing a variety of chemokines and chemokine receptors, MSCs migrate towards inflammatory chemokines and cytokines, reach the area where inflammation occurs, and promote tissue regeneration and repair [15, 16]. A phase I multicenter clinical study and phase II clinical trial studies have shown that bone marrow mesenchymal stem cells (BMSCs) can be safely applied to the treatment of hormone-resistant graft-versus-host disease (GVHD) by intravenous injection without acute poisoning [17].
However, with age, the risk of bone marrow failure increases and tolerance to cytotoxic damage is reduced. As the body ages, MSCs will show signs of aging, which limits the use of autologous MSCs in transplant treatment for elderly patients [18]. Understanding the development and mechanism of aging BMSCs is vital in basic research and can be applied to clinical practice.
Macaques diverged from humans approximately 25 million years ago, and the similarity of their genome to that of humans is approximately 93% [19]. Macaques are widely used in basic and applied biomedical research, which has greatly promoted the development of multidisciplinary research.
Understanding the molecular mechanisms of aging BMSCs will help to identify more effective diagnostic and therapeutic strategies. Through the analysis of biological functions and signaling pathways, we can further understand the changes in aging at the molecular level to explore the potential diagnostic, prognostic and biological markers of drug targets, which is of great significance for preclinical research on aging.
This study attempts to compare the differences in miRNA and mRNA gene expression levels between juvenile and aged macaque mononuclear cells by second-generation sequencing technology. The differences in peripheral blood cells and bone marrow cytokines were observed. The bone marrow cells of juvenile and aging macaques were extracted. BMSCs were isolated for culture and biocharacteristics analysis. The aging cell model was established using the strong oxidant t-BHP. The telomere length and telomerase expression in target cells were affected by cell coculture, and the aging of macaque BMSCs and mononuclear cells was explored by modern biological techniques. The changes in biological characteristics provide a valuable theoretical basis for research on and the application of macaques and their BMSCs.
Results
Morphological characteristics of BMSCs
With prolonged in vitro culture, some cells in the aging group became broad, and the number of polygonal and irregularly shaped cells began to increase. The nuclear-to-cytoplasmic ratio of the cells decreased. In addition, there were small particles or small vacuoles observed in the cytoplasm. The shape of the cells in the young group was relatively uniform, with a strong refractive index and optimal growth (Figure 1).
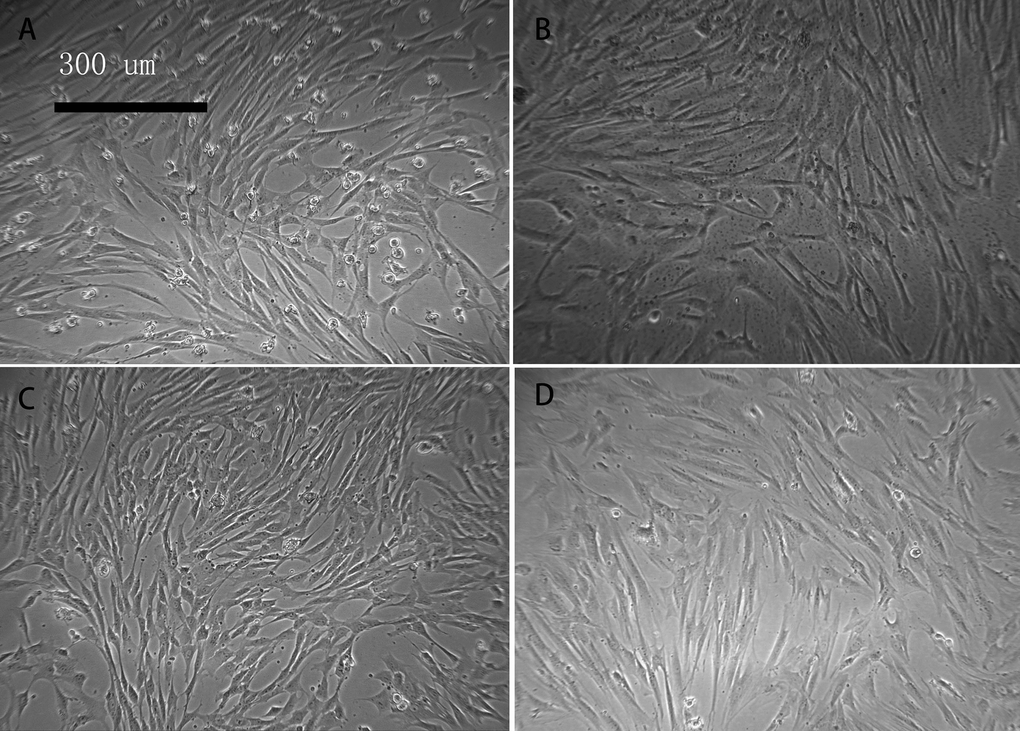
Figure 1. Cell morphology of young and aged macaque BMSCs (×100) (A, B) show the primary BMSCs of young and aged macaques, respectively; (C, D) show the P3 BMSCs of young and aged macaques, respectively. With prolonged in vitro culture, some cells in the aging group became broad, and the number of polygonal and irregularly shaped cells began to increase.
Immunophenotype of macaque BMSCs
Flow cytometry was performed on P3 BMSCs, and the cell surface antigens CD29, CD105, CD73, CD184, CD90, CD45 and HLA-DR were detected. There were several differences in the surface antigens of the BMSCs in the young and aged groups, but these differences were not statistically significant (Table 1). Only CD90 showed a significant difference (P < 0.05). The percentage of CD90-positive cells (99.43 ± 0.45%) of young macaques was significantly higher than that of aged macaques (24.37 ± 17.35%).
Table 1. Flow cytometry analysis of surface antigens in young and aged macaque BMSCs (
| Surface antigens | Groups | Positive cell rate(%) | P-value |
|---|---|---|---|
| CD29 | Young cell group | 100.00 ± 0.00 | 0.423 |
| Aged cell group | 99.97 ± 0.06 | ||
| CD105 | Young cell group | 99.83 ± 0.06 | 0.057 |
| Aged cell group | 99.97 ± 0.06 | ||
| CD73 | Young cell group | 99.97 ± 0.06 | 0.329 |
| Aged cell group | 98.60 ± 1.85 | ||
| CD184 | Young cell group | 96.83 ± 2.85 | 0.409 |
| Aged cell group | 88.2 ± 15.96 | ||
| CD90 | Young cell group | 99.43 ± 0.45 | 0.017 |
| Aged cell group* | 24.37 ± 17.35 | ||
| CD45 | Young cell group | 0.17 ± 0.12 | 0.097 |
| Aged cell group | 3.47 ± 2.65 | ||
| HLA-DR | Young cell group | 0.33 ± 0.23 | 0.066 |
| Aged cell group | 4.53 ± 2.89 |
Ability to induce differentiation of macaque BMSCs
P3 BMSCs in the young and aged groups underwent osteogenic induction. After staining with alizarin red, the dark-red calcium nodules were quantified (Figure 2A, 2B). The number of calcium nodules in the aged group was fewer than that in the young group. In the young group and the aged group, the macaque BMSCs underwent adipogenesis. After adding the oil red O stain, the dye was dissolved in the intracellular lipids, and the lipid droplets in the cells were red (Figure 2C, 2D). Fewer lipid droplets were produced in the aged group than in the young group. In the young group and the aged group, the macaque BMSCs underwent cartilage induction, and alcian blue staining showed the proteoglycans synthesized by the blue chondrocytes (Figure 2E, 2F). The aged group had less proteoglycans than the young group. We then used ImageJ 1.37c software to quantitate the osteogenic, adipogenic and chondrogenic results, which are shown in Figure 2G. Overall, in the aged group, the osteogenic, adipogenic and chondrogenic markers produced were less than those in the young group (*p<0.05, n=3).
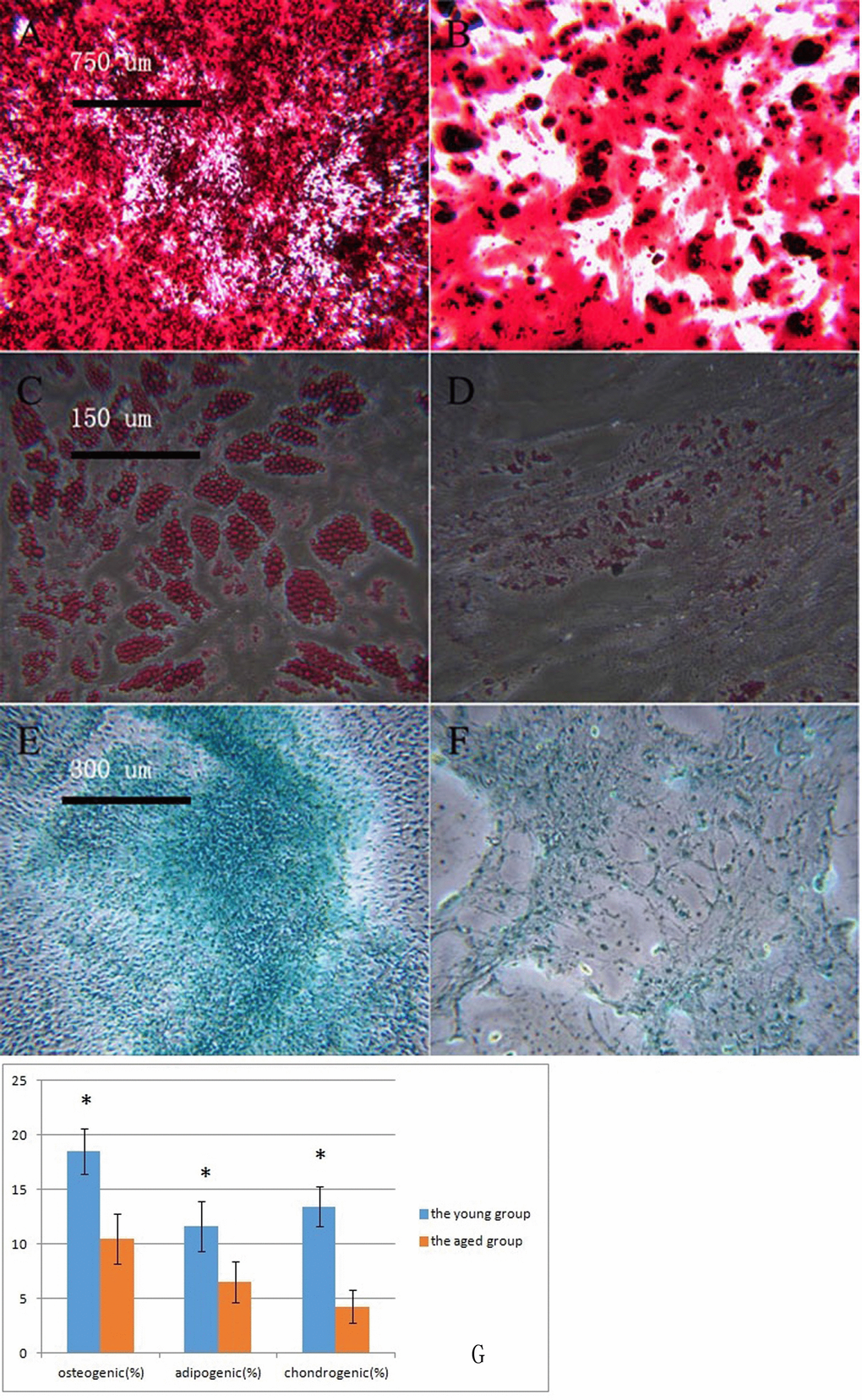
Figure 2. Macaque BMSCs differentiated into multiple cells (A, B) show the osteogenic induction of young and aged macaque BMSCs, respectively (×40); (C, D) show the adipogenic induction of young and aged macaque BMSCs, respectively (×200); (E, F) show the cartilage induction of young and aged macaque BMSCs, respectively. (×100); (G) We used ImageJ 1.37c software to quantitate the osteogenic, adipogenic and chondrogenic results. In the aged group, less osteogenic, adipogenic and chondrogenic markers were produced than in the young group (*p<0.05, n=3).
Proliferative ability of macaque P3 BMSCs
It was observed that compared with that in the young group, the expansion rate of P3 BMSCs in the aged group was significantly lower, and the proliferative ability was significantly reduced (Figure 3, Table 2, P < 0.05).
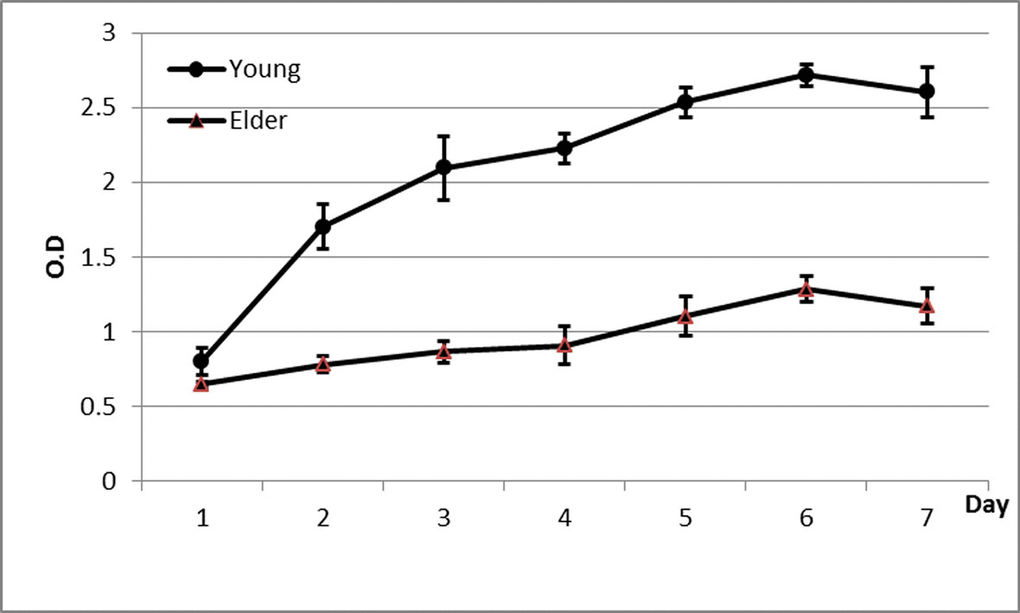
Figure 3. Proliferation curve of BMSCs from aged and young macaques as determined by the CCK-8 method It was observed that compared with that in the young group, the expansion rate of P3 BMSCs in the aged group was significantly lower, and the proliferative ability was significantly reduced.
Table 2. Proliferation of BMSCs from aged and young macaques by the CCK-8 assay
| n | Aged group | Young group | P-value | |
|---|---|---|---|---|
| Day 1 | 3 | 0.65 ± 0.02* | 0.80 ± 0.09 | 0.004 |
| Day 2 | 3 | 0.78 ± 0.06* | 1.70 ± 0.15 | <0.001 |
| Day 3 | 3 | 0.87 ± 0.07* | 2.10 ± 0.22 | <0.001 |
| Day 4 | 3 | 0.91 ± 0.12* | 2.23 ± 0.10 | <0.001 |
| Day 5 | 3 | 1.10 ± 0.13* | 2.53 ± 0.10 | <0.001 |
| Day 6 | 3 | 1.28 ± 0.08* | 2.72 ± 0.07 | <0.001 |
| Day 7 | 3 | 1.17 ± 0.12* | 2.60 ± 0.17 | <0.001 |
Antiaging role of macaque BMSCs in cocultures
Results of the induction of senescence in 293T cells
After induction by different concentrations of t-BHP, cells showed different apoptosis rates, and some cells showed senescence after induction (Figure 5A–5G). Induction with 200 μmol/L t-BHP for 6 h was performed to establish an aging model with macaque BMSCs.
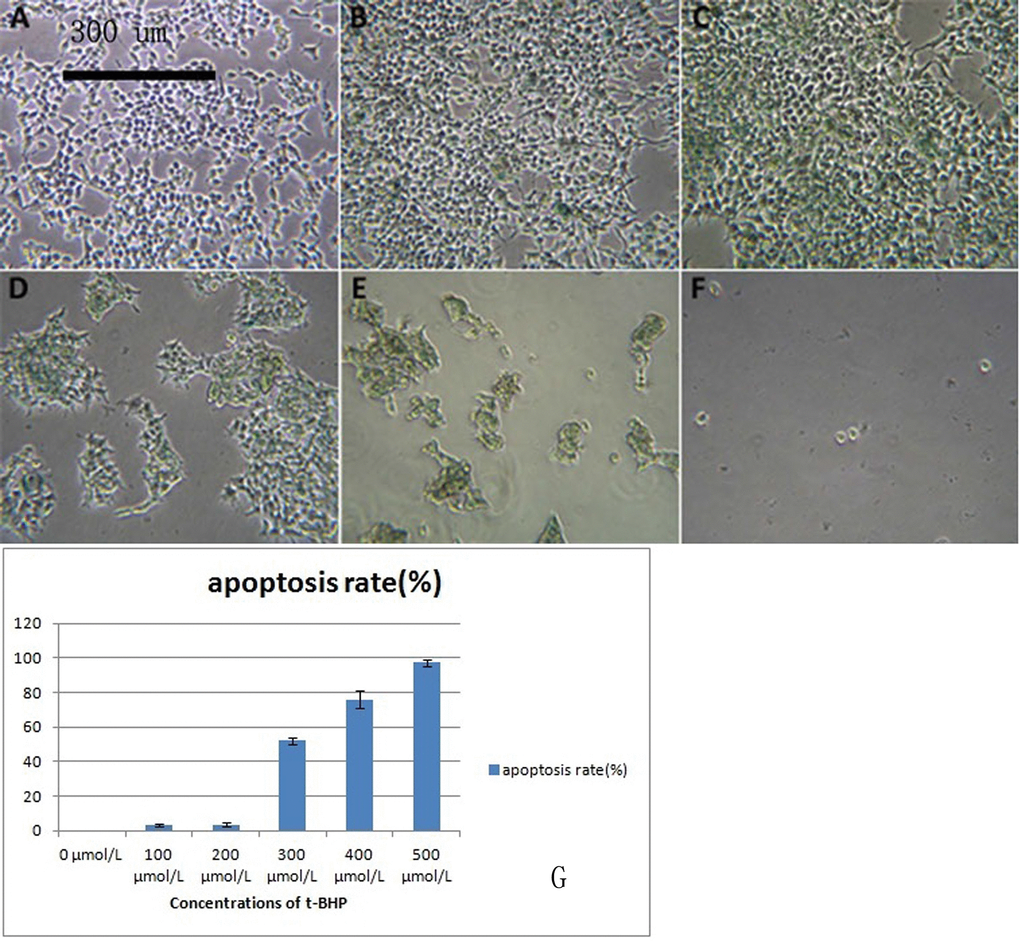
Figure 5. 293T cells treated with different concentrations of t-BHP and stained with SA-β-gal (×100) BMSCs were treated with t-BHP at concentrations of 0 μmol/L (A), 100 μmol/L (B), 200 μmol/L (C), 300 μmol/L (D), 400 μmol/L (E), and 500 μmol/L (F). We have quantified the apoptosis rate in (G). Induction with 200 μmol/L t-BHP for 6 h was performed to establish the aging macaque BMSC model.
Flow cytometry analysis of 293T cells before and after the induction of senescence showed that the detection rates of the surface antigens CD29, CD73, CD105 and HLA-ABC in senescent 293T cells were significantly decreased (Table 3, P < 0.05).
Table 3. Flow cytometry analysis of surface antigens in 293T cells before and after senescence induction (
| Surface antigens | Groups | Positive cell rate(%) | P-value |
|---|---|---|---|
| CD14 | Normal group | 78.8 ± 0.8 | 0.089 |
| Induction group | 76.3 ± 1.7 | ||
| CD29 | Normal group | 88.3 ± 1.1 | 0.009 |
| Induction group** | 84.5 ± 0.9 | ||
| CD90 | Normal group | 75.5 ± 1.0 | 0.078 |
| Induction group | 72.7 ± 1.8 | ||
| CD73 | Normal group | 4.55 ± 0.53 | 0.010 |
| Induction group* | 2.77 ± 0.42 | ||
| CD105 | Normal group | 83.2 ± 2.1 | 0.008 |
| Induction group** | 68.4 ± 4.8 | ||
| HLA-ABC | Normal group | 1.12 ± 0.04 | <0.001 |
| Induction group** | 0.19 ± 0.02 |
Telomerase and TCAB1 mRNA expression levels in 293T cells
After the 293T cells were cocultured with BMSCs for 5 d, the mRNA levels of telomeres in the control group and the induced senescence group were significantly higher than those measured before coculture (P < 0.05, Figure 6A), and the mRNA levels of TCAB1 were also significantly increased (P < 0.05, Figure 6B).
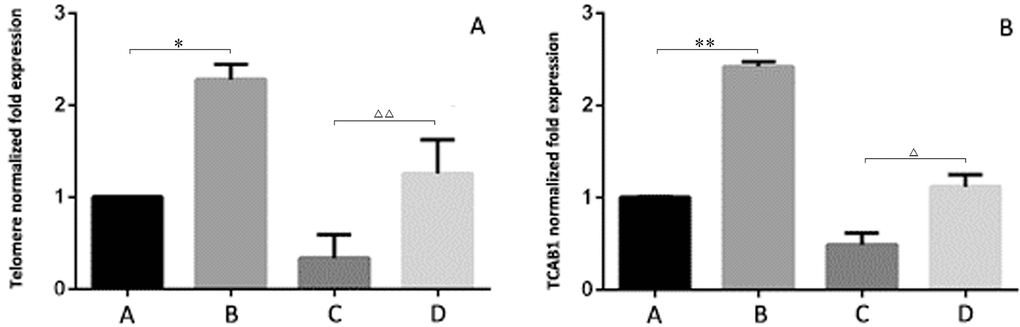
Figure 6. Relative expression levels of telomeres and TCAB1 in 293T cells by RT-PCR (A) shows the senescence-induced 293T cells cocultured with BMSCs. (B) shows the control 293T cells cocultured with BMSCs. (C) shows the senescence-induced 293T cells. (D) shows the control 293T cells. After the 293T cells were cocultured with BMSCs for 5 d, the mRNA levels of telomeres in the control group and the induced senescence group were significantly higher than those measured before coculture (P < 0.05, Figure 6A), and the mRNA levels of TCAB1 were also significantly increased (P < 0.05, Figure 6B).
TCAB1 protein expression levels in 293T cells
TCAB1 is expressed in 293T cells. After coculture with BMSCs, the expression level of TCAB1 was significantly higher than that before coculture. The TCAB1 expression level was weakly positive before induction and positive after induction (Figure 7).
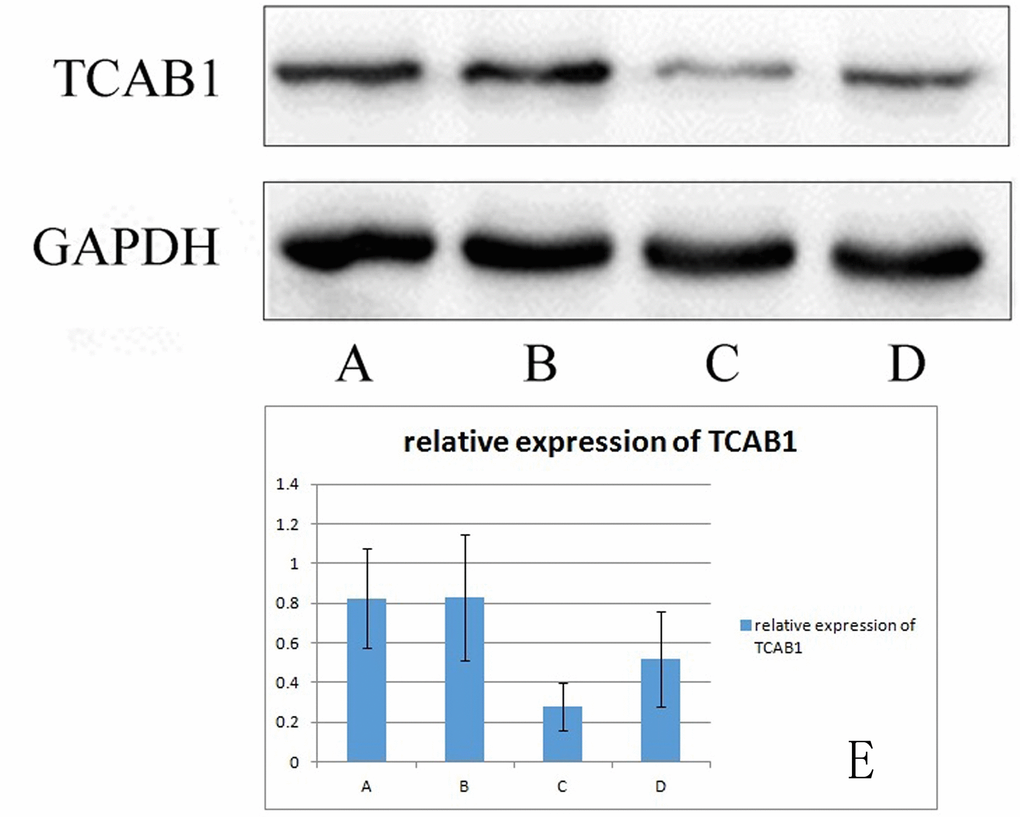
Figure 7. Relative expression of TCAB1 in 293T cells (A) shows TCAB1 expression in senescence-induced 293T cells cocultured with BMSCs. (B) shows TCAB1 expression in control 293T cells cocultured with BMSCs. (C) shows TCAB1 expression in senescence-induced 293T cells, and (D) shows TCAB1 expression in control 293T cells. (E) shows the Western blots with the quantitated results. TCAB1 expression was weakly positive before induction and positive after induction.
Results of induced aging in macaque BMSCs
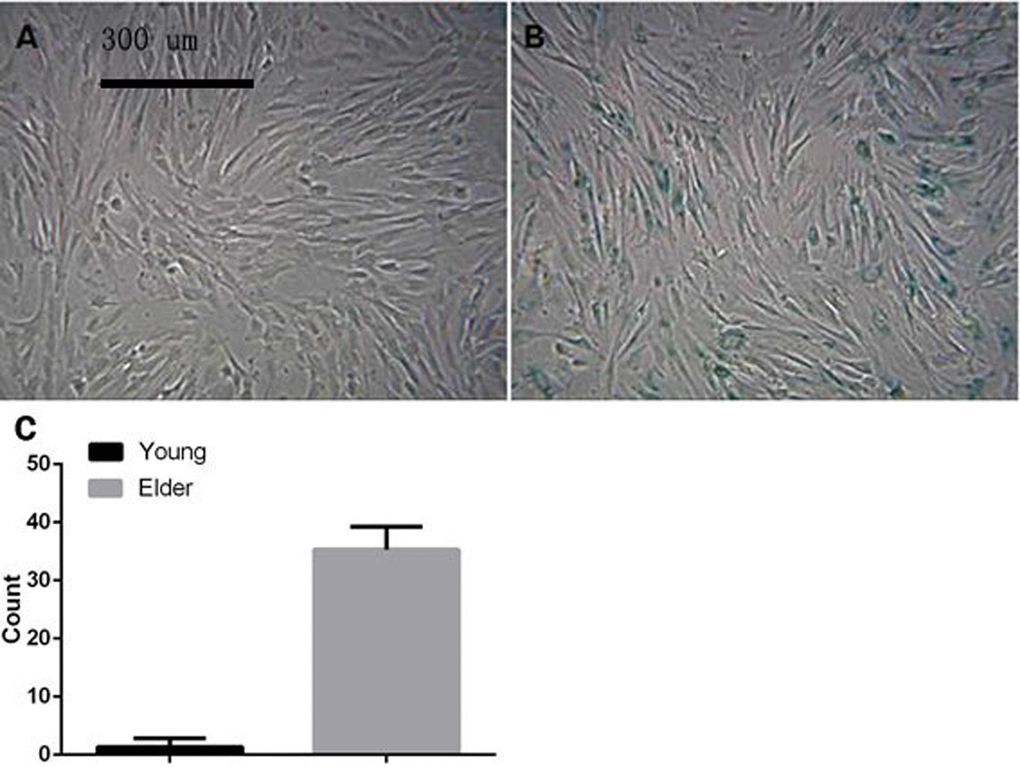
After induction with different concentrations of t-BHP for 6 h, SA-β-gal staining showed that some cells were senescent at a concentration of 200 μmol/L; the rate of positive staining was 34.0 ± 4.0%, and the number of SA-β-gal-positive cells was significantly higher than that in the other groups (P < 0.05). No cell death was observed. Therefore, the aging macaque BMSC model was established by exposure to 200 μmol/L t-BHP for 6 h (Figure 8).
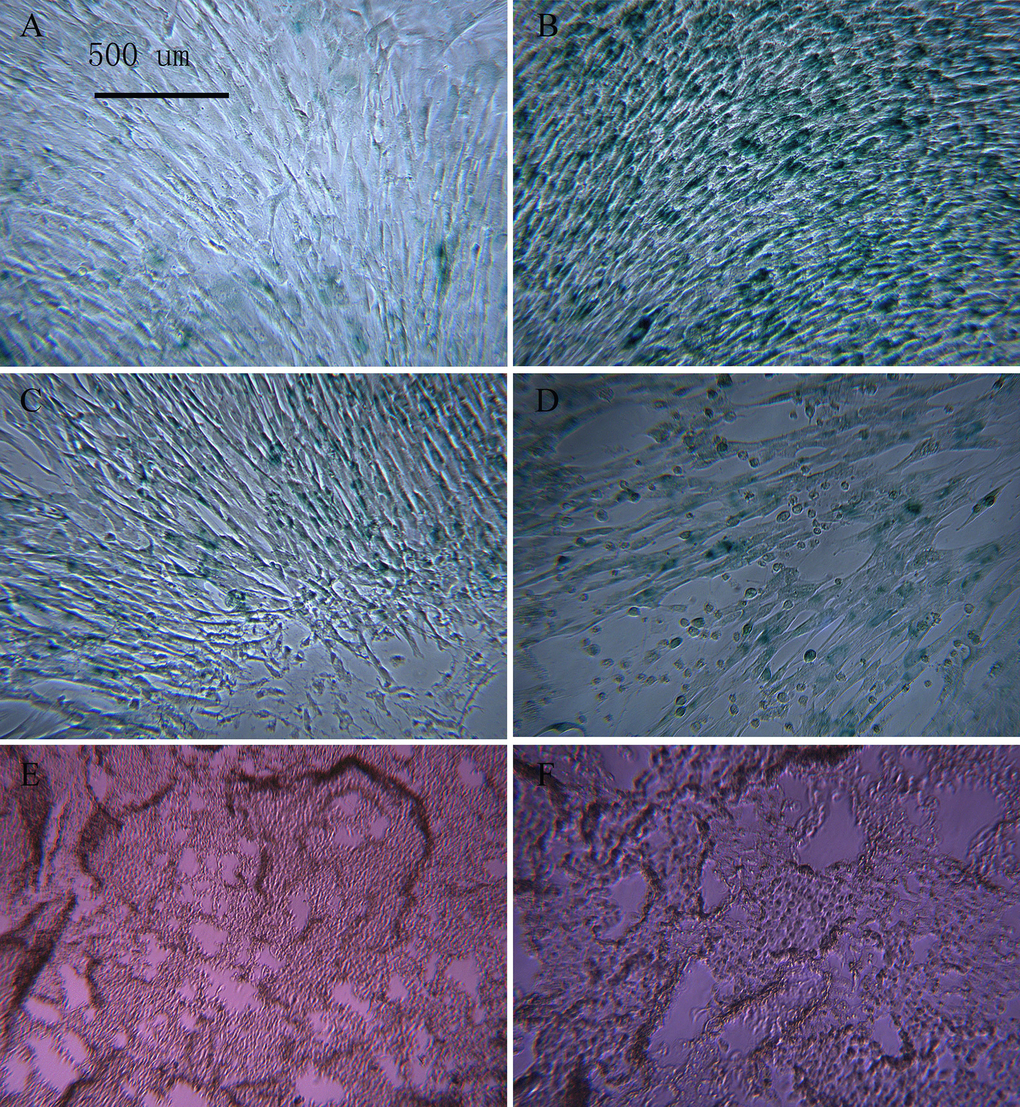
Figure 8. BMSCs treated with different concentrations of t-BHP and stained with SA-β-gal (×100) The concentration of t-BHP used to treat BMSCs is 100 μmol/L (A), 200 μmol/L (B), 300 μmol/L (C), 400 μmol/L (D), and 500 μmol/L (E), and 600 μmol/L (F). The aging macaque BMSC model was established by inducing cells with 200 μmol/L t-BHP for 6 h.
Detection of cytokine content in the culture supernatant of macaque BMSCs
The BMSC culture supernatants from the juvenile cell group, old cell group and induced aging group were analyzed by ELISA. IL-6, IL-11 and GM-CSF levels were significantly decreased in the elderly group and the induced aging group compared with the juvenile group (P < 0.01). IL-6, IL-11 and GM-CSF levels were also significantly lower in the induced aging group than in the elderly group (P < 0.01, Figure 9).
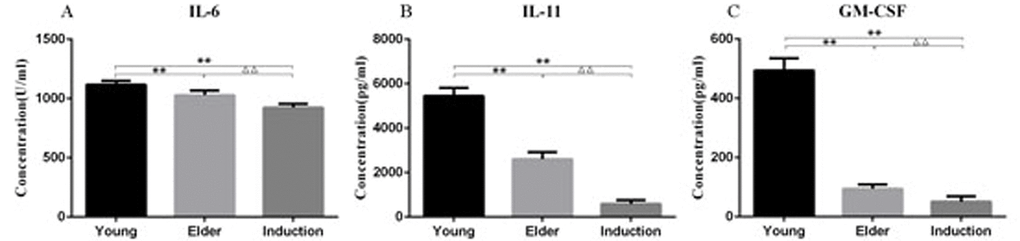
Figure 9. Cytokine levels in the culture supernatant of the young, aged and induced aged macaque BMSCs IL-6, IL-11 and GM-CSF levels were significantly decreased in the elderly group and the induced aging group compared with the juvenile group (P < 0.01). IL-6, IL-11 and GM-CSF levels were also significantly lower in the induced aging group than in the elderly group (P < 0.01).
RT-PCR detection of senescence-associated mRNA expression levels in BMSCs
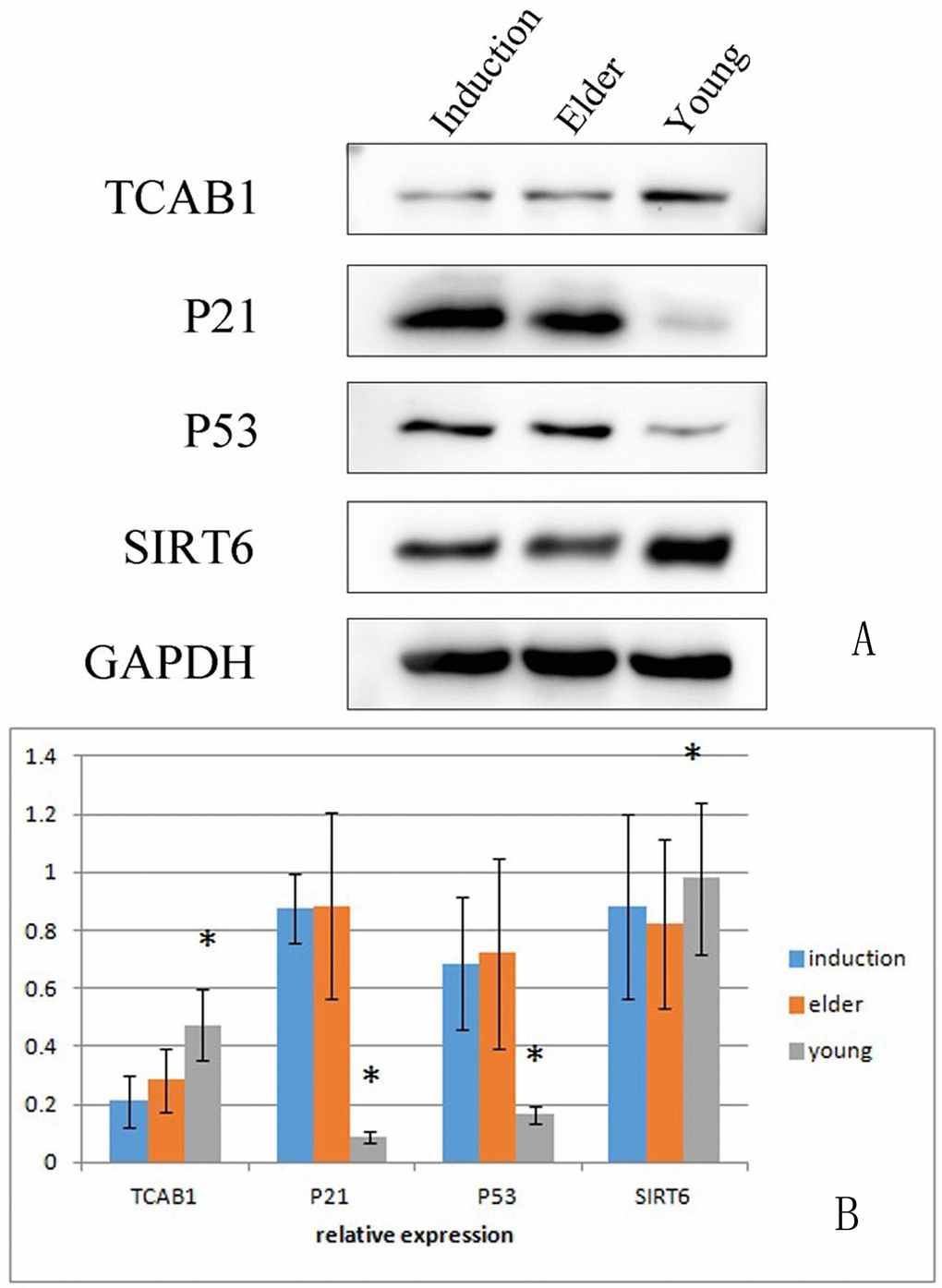
RT-PCR was used to detect the expression levels of senescence-associated mRNA in the juvenile cell group, old cell group and induced senescence group (Table 4). The results showed that TERT, SIRT1 and SIRT6 levels were significantly lower in the aged cell group than in the juvenile cell group (P < 0.01, Figure 10A, 10D, 10E). The level of P21 was significantly increased (P < 0.01, Figure 10C), and the level of TCAB1 was significantly decreased (P < 0.05, Figure 10B). Compared with those in the juvenile cell group, TERT and SIRT6 levels were significantly decreased (P < 0.01, Figure 10A) and P21 levels were significantly increased (P < 0.01, Figure 10C) in the induced senescence group. The level of TERT was significantly decreased in the induced aging group compared with the aged cell group (P < 0.01, Figure 10A), and that of SIRT6 was significantly decreased (P < 0.05, Figure 10E).
Table 4. Relative expression levels of senescence-associated mRNA in each group of BMSCs (
| n | TERT | TCAB1 | P21 | SIRT1 | SIRT6 | |
|---|---|---|---|---|---|---|
| Young cell group | 3 | 1.00 ± 0.04 | 1.01 ± 0.02 | 1.03 ± 0.03 | 1.00 ± 0.04 | 1.00 ± 0.09 |
| Aged cell group | 3 | 0.70 ± 0.03** | 0.18 ± 0.01* | 3.45 ± 0.48** | 0.82 ± 0.02** | 0.17 ± 0.02** |
| Induced aged group | 3 | 0.05 ± 0.00**△△ | 0.35 ± 0.22 | 2.31 ± 0.09** | 0.78 ± 0.12 | 0.43 ± 0.07**△△ |
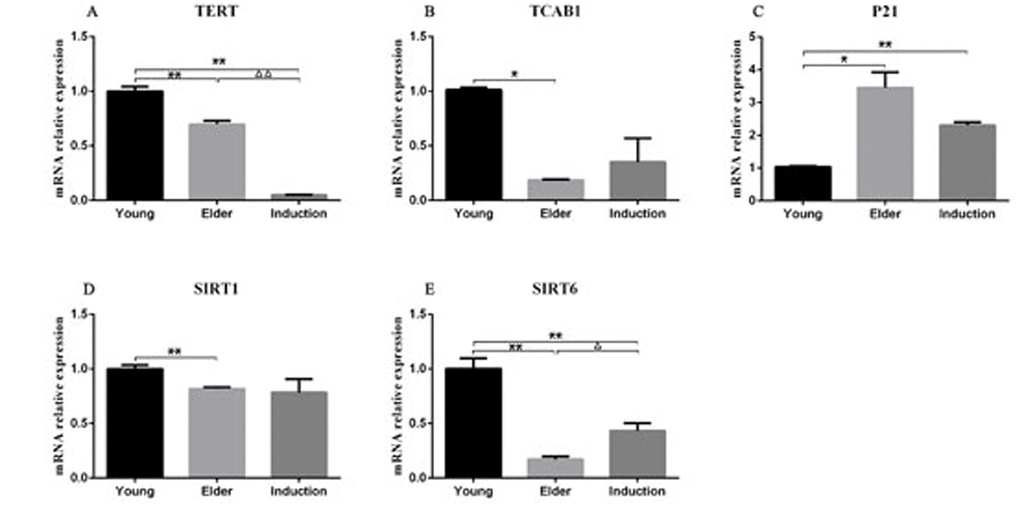
Figure 10. RT-PCR detection of the relative expression of TERT, TCAB1, P21, SIRT1 and SIRT6 in BMSCs The results showed that TERT, SIRT1 and SIRT6 expression levels were significantly lower in the aged cell group than in the juvenile cell group (P < 0.01). P21 expression was significantly increased (P < 0.01), and TCAB1 expression was significantly decreased (P < 0.05). Compared with those in the juvenile cell group, TERT and SIRT6 levels were significantly decreased (P < 0.01) and P21 levels were significantly increased (P < 0.01) in the induced senescence group. TERT expression was significantly decreased in the induced aging group compared with the aged cell group (P < 0.01), and SIRT6 expression was significantly decreased (P < 0.05).
Angiogenic factors in macaques
There were significant differences in angiogenesis-related factors between the juvenile group and the old group of macaques (P < 0.05). The levels of HB-EGF and PIGF in the juvenile group were lower than those in the old group, while the levels of VEGF in the juvenile group were higher than those in the old group (Table 5).
Table 5-1. Quantification of angiogenesis-related factors in juvenile and aged macaque (
| n | Angiogenin | ANG-2 | EGF | bFGF | HB-EGF | |
|---|---|---|---|---|---|---|
| Young group | 3 | 0.29 ± 0.50 | 112.22 ± 95.59 | 0.02 ± 0.02 | 17.22 ± 29.83 | 4.74 ± 0.88 |
| Aged group | 3 | 27.70 ± 38.30 | 138.10 ± 55.38 | 0.28 ± 0.24 | 191.90 ± 177.63 | 8.99 ± 0.86* |
Table 5-2. Quantification of angiogenesis-related factors in juvenile and aged macaques (
| n | HGF | Leptin | PDGF-BB | PIGF | VEGF | |
|---|---|---|---|---|---|---|
| Young group | 3 | 17.62 ± 25.21 | 60.46 ± 41.09 | 7.50 ± 7.02 | 0.18 ± 0.13 | 4.03 ± 1.20 |
| Aged group | 3 | 0.70 ± 1.18 | 337.42 ± 529.09 | 11.46 ± 13.29 | 3.24 ± 0.72* | 0.00 ± 0.00* |
Peripheral blood cell analysis
The number of white blood cells and lymphocytes isolated from the peripheral blood of old macaques was significantly increased (P < 0.05) and higher than the normal reference values. The other indicators showed no significant changes (Table 6). The results suggest that elderly macaques may be in a state of chronic inflammation.
Table 6-1. Analysis of peripheral blood cells from young and aged macaques (
| n | Leukocyte | Neutrophil granulocyte | Lymphocyte | Monocyte | |
|---|---|---|---|---|---|
| Young group | 3 | 8.1 ± 1.5 | 3.5 ± 0.7 | 4.3 ± 1.5 | 0.17 ± 0.05 |
| Aged group | 3 | 12.4 ± 1.6* | 3.9 ± 1.3 | 8.2 ± 1.5* | 0.24 ± 0.05 |
Table 6-2. Analysis of peripheral blood cells from young and aged macaques (
| n | Red blood cell | Blood platelets | |
|---|---|---|---|
| Young group | 3 | (6.2 ± 0.9) × 1012/L | (359.3 ± 12.5) × 109/L |
| Aged group | 3 | (5.7 ± 0.1) × 1012/L | (353.0 ± 60.5) × 109/L |
Transcriptome sequencing results of macaque peripheral blood mononuclear cells
Differentially expressed mRNA was analyzed by sequencing peripheral blood mononuclear cells from the juvenile and the aged groups. The clustered genes, which may have similar biological functions, are displayed in a heat map (Figure 12). According to the analysis, a total of 5,711 differentially expressed GO terms were detected, of which 2,636 were downregulated and 3,075 were upregulated. The GO terms of the three groups with corresponding gene numbers greater than two were screened. Each of the 10 items was ranked by the associated -log10 (P-value) value. The top 30 bar graphs of the GO enrichment analysis are shown in Figure 13A and 13B. The top 30 downregulated and upregulated GO terms are listed in Table 7 according to their P-value and sorted from small to large. The gene functions involved in the downregulated GO terms mainly included regulation of the IFN-γ response, defense against viruses, the type I IFN signaling pathway, replication of the negative regulatory viral genome, response to viruses, innate immune response, the IFN-γ signaling pathway, the response of IFN-α, the response of IFN-β, and the negative regulation of type I IFN product. The gene functions involved in the upregulated GO terms mainly included the transport of mevalonate and the behavioral response to nutrition, spectrin-related cytoskeleton, organic cyclic compound binding, directed red blood cell differentiation, hemidesmosome, regulation of DNA replication, hemidesmosome assembly, other microorganisms that kill cells, and regulation of cell migration.
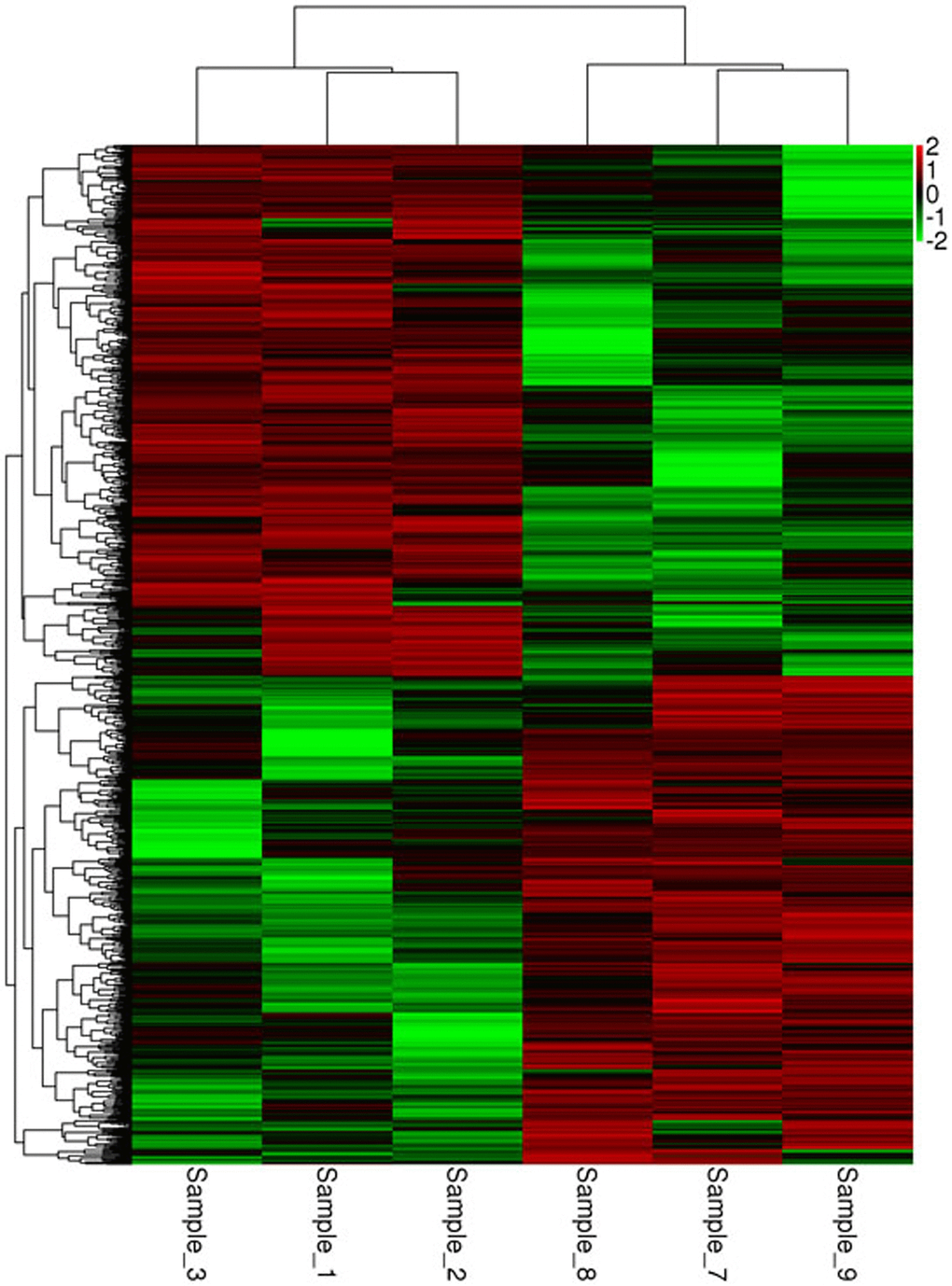
Figure 12. Heat map of the mRNA clustering analysis of macaque mononuclear cells from juvenile and old groups Samples_1, 2, and 3 correspond to the elderly group, and samples_7, 8, and 9 correspond to the young group. According to the analysis, a total of 5,711 differentially expressed GO terms were identified, of which 2,636 were downregulated and 3,075 were upregulated.
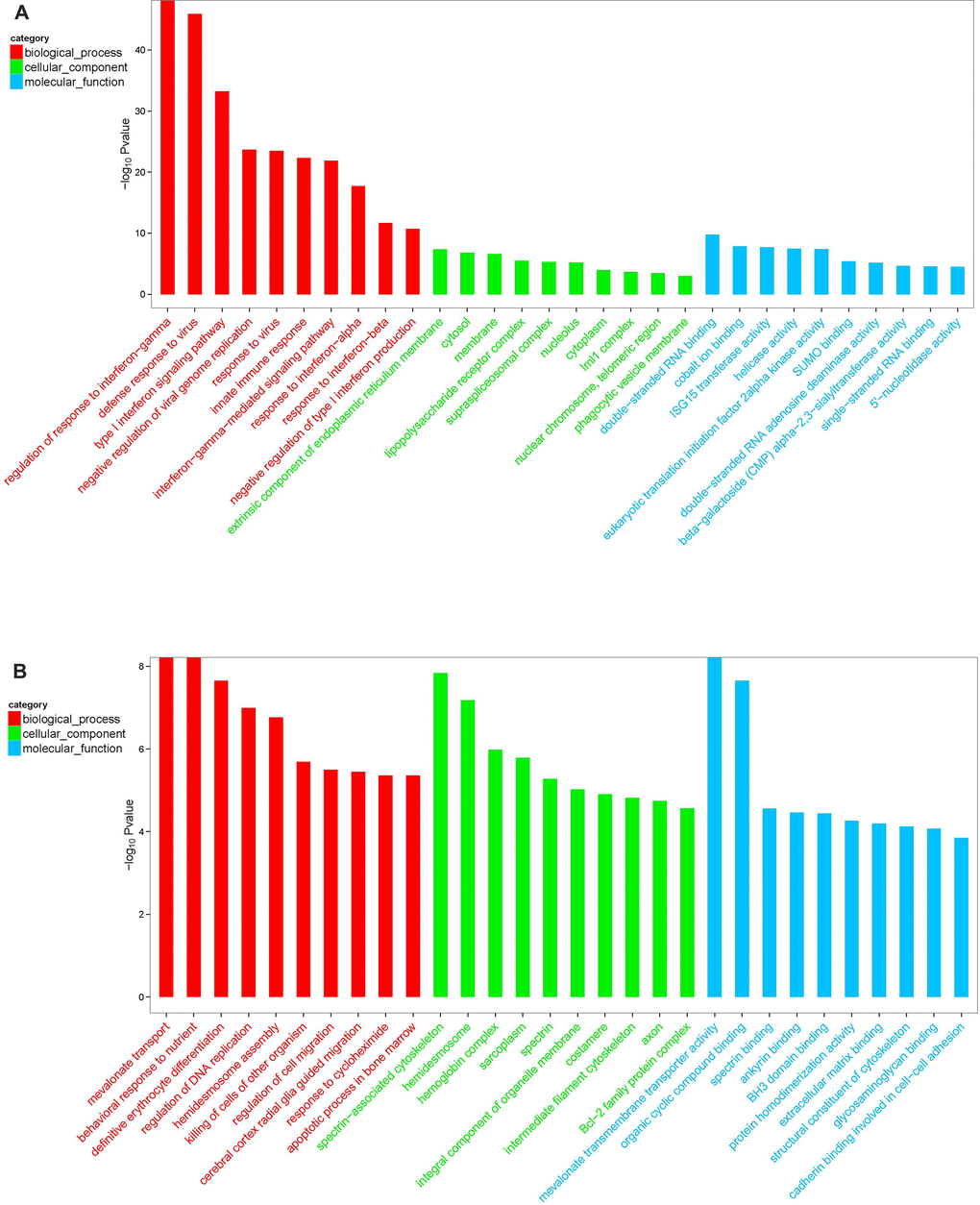
Figure 13. GO enrichment analysis of differentially expressed mRNA in macaque mononuclear cells from the juvenile and old age groups (A), Downregulated GO terms. (B), Upregulated GO terms. Red indicates the biological process-related GO term; green indicates the cell component-related GO term; and blue indicates the molecular function-related GO term. The top 30 bar graphs of the GO enrichment analysis are shown in Figure 13A and 13B. The top 30 downregulated and upregulated GO terms are listed in Table 7 according to their P-value and sorted from small to large.
Table 7. GO analysis of aging-related differentially expressed genes
| Expression | Term | Gene function | Gene count | Enrichment score | P -value |
|---|---|---|---|---|---|
| Down | GO:0060330 | Regulation of response to interferon-gamma | 3 | 89.23 | 0 |
| Down | GO:0051607 | Defense response to virus | 54 | 14.34 | 1.3E-46 |
| Down | GO:0060337 | Type I interferon signaling pathway | 33 | 19.25 | 6.2E-34 |
| Down | GO:0045071 | Negative regulation of viral genome replication | 22 | 20.88 | 2.1E-24 |
| Down | GO:0009615 | Response to virus | 30 | 11.74 | 3.4E-24 |
| Down | GO:0045087 | Innate immune response | 50 | 5.49 | 4.8E-23 |
| Down | GO:0060333 | Interferon-gamma-mediated signaling pathway | 27 | 12.29 | 1.4E-22 |
| Down | GO:0035455 | Response to interferon-alpha | 13 | 31.35 | 2.0E-18 |
| Down | GO:0035456 | Response to interferon-beta | 9 | 24.34 | 2.1E-12 |
| Down | GO:0032480 | Negative regulation of type I interferon production | 11 | 14.02 | 2.1E-11 |
| Down | GO:0003725 | Double-stranded RNA binding | 15 | 7.65 | 1.8E-10 |
| Down | GO:0050897 | Cobalt ion binding | 5 | 27.88 | 1.4E-08 |
| Down | GO:0042296 | ISG15 transferase activity | 4 | 39.66 | 2.1E-08 |
| Down | GO:0004386 | Helicase activity | 14 | 5.68 | 3.6E-08 |
| Down | GO:0004694 | Eukaryotic translation initiation factor 2alpha kinase activity | 4 | 35.69 | 4.2E-08 |
| Down | GO:0042406 | Extrinsic component of endoplasmic reticulum membrane | 5 | 23.48 | 4.6E-08 |
| Down | GO:0005829 | Cytosol | 147 | 1.48 | 1.6E-07 |
| Down | GO:0016020 | Membrane | 96 | 1.66 | 2.5E-07 |
| Down | GO:0046696 | Lipopolysaccharide receptor complex | 3 | 26.77 | 3.1E-06 |
| Down | GO:0032183 | SUMO binding | 5 | 11.74 | 3.9E-06 |
| Down | GO:0044530 | Supraspliceosomal complex | 3 | 24.33 | 4.8E-06 |
| Down | GO:0005730 | Nucleolus | 45 | 1.98 | 6.1 E-06 |
| Down | GO:0003726 | Double-stranded RNA adenosine deaminase activity | 3 | 22.31 | 7.2E-06 |
| Down | GO:0003836 | Beta-galactoside (CMP) alpha-2,3-sialyltransferase activity | 4 | 11.51 | 2.3E-05 |
| Down | GO:0003727 | Single-stranded RNA binding | 6 | 6.86 | 2.8E-05 |
| Down | GO:0008253 | 5'-Nucleotidase activity | 4 | 10.82 | 3.2E-05 |
| Down | GO:0005737 | Cytoplasm | 207 | 1.24 | 0.0001 |
| Down | GO:1990130 | Iml1 complex | 3 | 9.91 | 0.0002 |
| Down | GO:0000784 | Nuclear chromosome, telomeric region | 9 | 3.40 | 0.0004 |
| Down | GO:0030670 | Phagocytic vesicle membrane | 6 | 3.88 | 0.0010 |
| Up | GO:0015728 | Mevalonate transport | 3 | 81.66 | 0 |
| Up | GO:0051780 | Behavioral response to nutrient | 3 | 81.66 | 0 |
| Up | GO:0015130 | Mevalonate transmembrane transporter activity | 3 | 81.66 | 0 |
| Up | GO:0014731 | Spectrin-associated cytoskeleton | 10 | 9.07 | 1.5E-08 |
| Up | GO:0097159 | Organic cyclic compound binding | 3 | 61.24 | 2.2E-08 |
| Up | GO:0060318 | Definitive erythrocyte differentiation | 3 | 61.24 | 2.2E-08 |
| Up | GO:0030056 | Hemidesmosome | 8 | 10.54 | 6.7E-08 |
| Up | GO:0006275 | Regulation of DNA replication | 8 | 10.05 | 1.0E-07 |
| Up | GO:0031581 | Hemidesmosome assembly | 8 | 9.47 | 1.7E-07 |
| Up | GO:0031640 | Killing of cells of other organism | 4 | 18.15 | 2.0E-06 |
| Up | GO:0030334 | Regulation of cell migration | 12 | 4.54 | 3.2E-06 |
| Up | GO:0021801 | Cerebral cortex radial glia guided migration | 4 | 16.33 | 3.6E-06 |
| Up | GO:0005833 | Hemoglobin complex | 4 | 20.41 | 1.1E-06 |
| Up | GO:0016528 | Sarcoplasm | 6 | 10.42 | 1.6E-06 |
| Up | GO:0046898 | Response to cycloheximide | 3 | 24.50 | 4.4E-06 |
| Up | GO:0071839 | Apoptotic process in bone marrow | 3 | 24.50 | 4.4E-06 |
| Up | GO:0008091 | Spectrin | 7 | 7.33 | 5.3E-06 |
| Up | GO:0031301 | Integral component of organelle membrane | 4 | 13.61 | 9.5E-06 |
| Up | GO:0043034 | Costamere | 9 | 5.10 | 1.2E-05 |
| Up | GO:0045111 | Intermediate filament cytoskeleton | 11 | 4.20 | 1.5E-05 |
| Up | GO:0030424 | Axon | 24 | 2.49 | 1.8E-05 |
| Up | GO:0097136 | Bcl-2 family protein complex | 3 | 16.33 | 2.7E-05 |
| Up | GO:0030507 | Spectrin binding | 9 | 4.65 | 2.8E-05 |
| Up | GO:0030506 | Ankyrin binding | 8 | 5.03 | 3.5E-05 |
| Up | GO:0051434 | BH3 domain binding | 3 | 15.31 | 3.6E-05 |
| Up | GO:0042803 | Protein homodimerization activity | 37 | 1.94 | 5.5E-05 |
| Up | GO:0050840 | Extracellular matrix binding | 5 | 7.29 | 6.4E-05 |
| Up | GO:0005200 | Structural constituent of cytoskeleton | 14 | 3.04 | 7.5E-05 |
| Up | GO:0005539 | Glycosaminoglycan binding | 4 | 8.83 | 8.5E-05 |
| Up | GO:0098641 | Cadherin binding involved in cell-cell adhesion | 24 | 2.19 | 0.0001 |
KEGG analysis of the mRNA target genes mainly showed enrichment in 322 pathways, of which 170 were upregulated and 152 were downregulated, showing a top-enriched KEGG pathway map (Figure 14, Table 8). The enriched downregulated KEGG pathways mainly included the NOD-like receptor signaling pathway, the RIG-I-like receptor signaling pathway, the cytosolic DNA-sensing pathway, the Toll-like receptor signaling pathway, nucleotide metabolism-related pyrimidine metabolism, glycosphingolipid biosynthesis-ganglio series related to polysaccharide biosynthesis and metabolism, nicotinate and nicotinamide metabolism associated with cofactor and vitamin metabolism, one carbon pool by folate, growth-related osteoclast differentiation, environmental information processing, and the signal transduction-related NF-kappa B signaling pathways. The enriched upregulated KEGG pathways mainly included the carbon fixation pathways in prokaryotes related to energy metabolism, the mTOR signaling pathway related to environmental information processing and signal transduction, cell growth and apoptosis, antigen processing and presentation related to the immune system, hematopoietic cell lineage, the Fanconi anemia pathway, homologous recombination, growth-related axon guidance, digestion-related digestion and absorption, bile secretion associated with genetic information processing, and replication and repair.
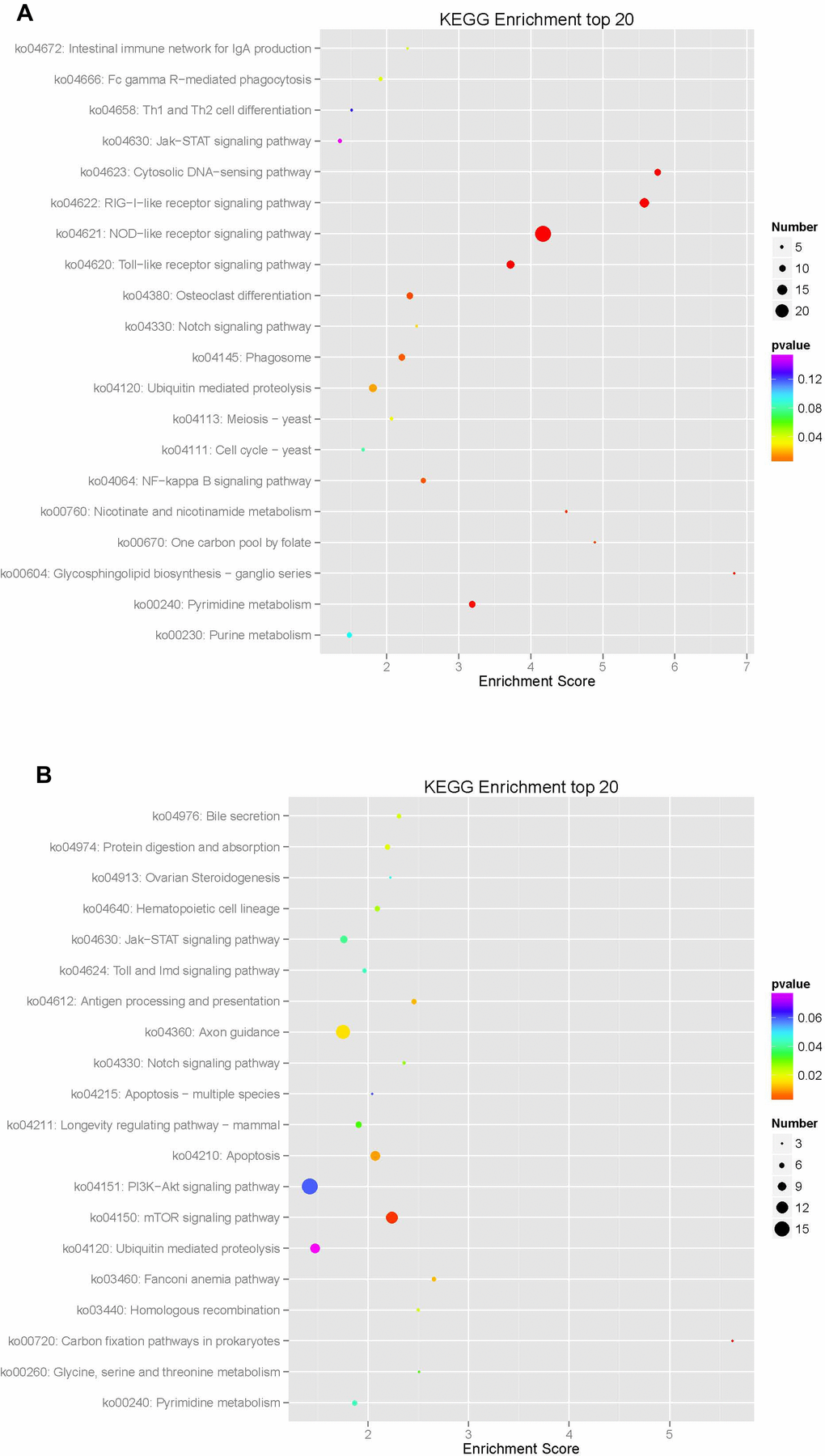
Figure 14. Enrichment analysis of KEGG pathways in differentially expressed mRNA in macaque mononuclear cells from the juvenile and old age groups The enrichment analysis showed that the target gene was mainly enriched in 322 pathways, of which 170 were upregulated and 152 were downregulated, resulting in a top-enriched KEGG pathway map (A), downregulated KEGG pathways; and (B), upregulated KEGG pathways.
Table 8. KEGG pathway analysis of differentially expressed aging-related pathways
| Expression | Pathway ID | Pathway name | Gene count | Enrichment score | P-value |
|---|---|---|---|---|---|
| Down | ko04621 | NOD-like receptor signaling pathway | 24 | 4.17 | 7.2E-10 |
| Down | ko04622 | RIG-I-like receptor signaling pathway | 14 | 5.58 | 3.6E-08 |
| Down | ko04623 | Cytosolic DNA-sensing pathway | 10 | 5.76 | 1.4E-06 |
| Down | ko04620 | Toll-like receptor signaling pathway | 12 | 3.72 | 2.3E-05 |
| Down | ko00240 | Pyrimidine metabolism | 10 | 3.19 | 0.0003 |
| Down | ko00604 | Glycosphingolipid biosynthesis-ganglio series | 3 | 6.82 | 0.0009 |
| Down | ko00760 | Nicotinate and nicotinamide metabolism | 4 | 4.49 | 0.0020 |
| Down | ko00670 | One carbon pool by folate | 3 | 4.89 | 0.0033 |
| Down | ko04380 | Osteoclast differentiation | 10 | 2.32 | 0.0042 |
| Down | ko04064 | NF-kappa B signaling pathway | 8 | 2.50 | 0.0050 |
| Down | ko04145 | Phagosome | 10 | 2.21 | 0.0060 |
| Down | ko04120 | Ubiquitin mediated proteolysis | 12 | 1.81 | 0.0162 |
| Down | ko04330 | Notch signaling pathway | 4 | 2.42 | 0.0255 |
| Down | ko04113 | Meiosis - yeast | 5 | 2.07 | 0.0350 |
| Down | ko04666 | Fc gamma R-mediated phagocytosis | 6 | 1.91 | 0.0387 |
| Down | ko04672 | Intestinal immune network for IgA production | 3 | 2.29 | 0.0423 |
| Down | ko04111 | Cell cycle - yeast | 5 | 1.67 | 0.0798 |
| Down | ko00230 | Purine metabolism | 8 | 1.48 | 0.0937 |
| Down | ko04658 | Th1 and Th2 cell differentiation | 4 | 1.5096 | 0.1273 |
| Down | ko04630 | Jak-STAT signaling pathway | 6 | 1.35 | 0.1585 |
| Up | ko00720 | Carbon fixation pathways in prokaryotes | 3 | 5.63 | 0.0020 |
| Up | ko04150 | mTOR signaling pathway | 12 | 2.24 | 0.0030 |
| Up | ko04210 | Apoptosis | 10 | 2.07 | 0.0096 |
| Up | ko04612 | Antigen processing and presentation | 6 | 2.46 | 0.0116 |
| Up | ko03460 | Fanconi anemia pathway | 5 | 2.65 | 0.0118 |
| Up | ko04360 | Axon guidance | 14 | 1.75 | 0.0150 |
| Up | ko04974 | Protein digestion and absorption | 6 | 2.19 | 0.0204 |
| Up | ko04976 | Bile secretion | 5 | 2.31 | 0.0220 |
| Up | ko03440 | Homologous recombination | 4 | 2.50 | 0.0224 |
| Up | ko04640 | Hematopoietic cell lineage | 6 | 2.09 | 0.0255 |
| Up | ko04330 | Notch signaling pathway | 4 | 2.36 | 0.0278 |
| Up | ko04211 | Longevity regulating pathway - mammal | 7 | 1.91 | 0.0318 |
| Up | ko00260 | Glycine, serine and threonine metabolism | 3 | 2.51 | 0.0321 |
| Up | ko04630 | Jak-STAT signaling pathway | 8 | 1.76 | 0.0398 |
| Up | ko00240 | Pyrimidine metabolism | 6 | 1.87 | 0.0430 |
| Up | ko04624 | Toll and Imd signaling pathway | 5 | 1.96 | 0.0432 |
| Up | ko04913 | Ovarian Steroidogenesis | 3 | 2.22 | 0.0467 |
| Up | ko04151 | PI3K-Akt signaling pathway | 16 | 1.42 | 0.0599 |
| Up | ko04215 | Apoptosis - multiple species | 3 | 2.04 | 0.0600 |
| Up | ko04120 | Ubiquitin mediated proteolysis | 10 | 1.47 | 0.0796 |
Discussion
Impact of aging on BMSCs
The life expectancy of macaques is approximately 25-30 years and may be longer under artificial breeding conditions. Female macaques are approximately 3 years old and males are 4 years old when they begin to mature. Therefore, macaques are considered young when they are within 3 years of age and considered of old age when they exceed 13 years of age; these guidelines were applied for macaque grouping.
Senescence-associated SA-β-gal is a hydrolase that catalyzes the hydrolysis of β-galactoside to monosaccharides in senescent cells. This hydrolase is therefore considered a biomarker of senescent cells, as is p16INK4A [20, 21]. In this study, SA-β-gal staining was used to quantify the number of senescent cells; and the conditions suitable for inducing cell senescence were evaluated. The aging macaque BMSC model was constructed by treating BMSC with 200 μmol/L t-BHP for 6 h. The overall deterioration of cell stemness and the decrease in MSC activity leading to insufficient functional cell renewal in tissues and organs may be an important mechanism of aging.
BMSCs can secrete a variety of biologically active molecules, including cytokines, chemokines and growth factors, which can promote the repair and regeneration of damaged tissue and organs and maintain many physiological functions, such as homeostasis. In this study, 293T cells were cocultured with BMSCs for 5 d, and the expression of telomeres and the telomerase core protein component TCAB1 was detected. The expression of telomeres and TCAB1 mRNA was significantly increased after coculture of 293T cells with BMSCs. After coculture with BMSCs, it was possible to reverse telomere shortening and improve telomerase activity due to the secretion of various biologically active substances, thereby improving the aging state of the cells.
Evaluation of the aging macaque BMSC model
In this study, expression of the senescence-associated gene p21 in BMSCs was detected by RT-PCR. The aged group and the induced senescence group had significantly increased levels of p21 compared with the young group. Western blot analysis showed that both p21 and p53 were highly expressed in the aged group and the induced aging group. The results indicated that both groups of cells had entered the aging state.
Sirtuin (SIRT) is a family of proteins homologous to silent information regulator 2 (Sir2), which are also known as antiaging enzymes. The positive role of SIRT1 in combating aging in MSCs has been confirmed. SIRT1 protects cells from aging-related DNA damage, induces the expression of TERT, and enhances telomerase activity [22, 23]. There is a close correlation among SIRT1, telomere length and human life [24]. SIRT1 plays an important role in maintaining the young state of MSCs [25].
SIRT6 is another member of the Sirtuin family with antiaging effects. In this study, SIRT6 was detected by RT-PCR and Western blotting. The expression of SIRT6 in the aged and the induced senescence BMSC groups was significantly lower than that in the juvenile cell group. BMSCs in the aged cell group and the induced senescence group became aged, and the ability of cells to become activated decreased, while accelerated senescence was observed.
TERT is an important substance for stabilizing the length and structure of telomeres. In this study, the expression of TERT and TCAB1 in the aged cell group was significantly lower than that in the juvenile cell group, as observed by RT-PCR analysis of TERT and TCAB1. The telomerase activity of BMSCs was decreased, the telomere length was shortened, and the proliferation potential of cells in vitro was reduced.
BMSCs can secrete a variety of cytokines and play an immunomodulatory role. In this study, the cytokine levels in the cell culture supernatants of each group were detected by ELISA. The cytokine levels in the aged cell group and the induced aging group were significantly lower than those in the juvenile cell group. The results indicated that the aging of cells is accompanied by a decline in paracrine ability, which affects the immune regulation function.
Some authors [26] have found that bone marrow-derived multipotent MSCs are the most frequently investigated cell type for potential regenerative strategies because they are relatively easy to isolate and are able to differentiate into several mesenchymal lineages. These authors concluded that antioxidant supplementation during MSC expansion reduces the DNA damage load and increases the MSC yield.
Effects of aging on macaques
Increased risk of bacterial infection and chronic inflammation often accompany aging. The proportion of white blood cells and lymphocytes in the peripheral blood of macaques in the elderly group was elevated, suggesting that the old macaques may be in a chronic inflammatory state. The bone marrow supernatant of macaques was analyzed using an angiogenic protein-antibody microarray. The levels of HB-EGF and PIGF in aged macaques were significantly increased, suggesting that the risk of tumors in elderly macaques is increased or that tumors may be present in aged macaques.
Some authors [27] have found that the regeneration potential of MSCs diminishes with advanced age, and this diminished potential is associated with changes in cellular functions. These changes represent novel insights into the aging process and could have implications regarding the potential for autologous stem cell therapy in older patients. In our current manuscript, an aging macaque BMSC model was successfully developed. Both the aged cell group and the induced aging group showed aging morphology; in addition, the expression of aging genes was increased, and the expression of aging-related proteins was increased. The expression trends of the aged cell group and the induced aging group were largely similar. Transcriptome sequencing of macaque mononuclear cells showed that senescence in macaques was related to multiple biological processes, multiple cellular components and multiple molecular functions. Therefore, the novelty of the current manuscript is straightforward.
Macaque mononuclear cell transcriptome sequencing analysis
Regarding the genomic sequence, the similarity between macaque and human is approximately 93%, and the miRNAs are relatively conserved during evolution; these miRNAs can inhibit gene expression and can have biological significance for tumorigenesis, development, metabolism and viral diseases [28].
Peripheral blood mononuclear cells were sequenced to analyze the differentially expressed mRNA. Through GO enrichment analyses, it was found that the GO terms of the functions of downregulated genes mainly included regulation of the IFN-γ response, defense against virus, the type I interferon signaling pathway, negative regulation of viral genome replication, the innate immune response IFN-γ mediated signaling pathway, IFN-α response, IFN-β response, and negative regulation of type I IFN products. The gene functions involved in the downregulated GO terms are primarily related to the immune system. The immune system is an important defense mechanism of the body against the stress of pathogens. Age-related immune system dysfunction is also called immune aging, which is characterized by increased susceptibility to infectious diseases, risk of developing autoimmune diseases, and risk of tumor occurrence [29]. In general, the downregulated GO terms are closely related to the immune system. Immunosuppression leads to decreased secretion of related cytokines and weakened immune function. The data suggest that the ability of the old macaques to defend against viruses is reduced.
The gene functions involved in the upregulated GO terms mainly included the transport of mevalonate, the behavioral response to nutrition, the spectrin-associated cytoskeleton, the binding of organic cyclic compounds, and the differentiation of erythrocytes. The relationship between these gene functions and aging is still unclear and warrants further investigation. Overall, the downregulation of immune function-related pathways suggests a decrease in immune function in aged macaques, similar to the results of the GO enrichment analysis.
The upregulated KEGG pathways involved the carbon fixation pathways in prokaryotes, the mTOR signaling pathways, and the cell apoptosis pathways. In this study, sequencing analysis showed that the expression of the mTOR signaling pathway was upregulated, suggesting an increased risk of tumors in elderly macaques. Cell apoptosis involves the removal of damaged or redundant cells by activating cysteine-containing aspartate-specific cysteine proteases, and the results suggest that there is an increase in apoptosis in aged macaques.
Materials and Methods
Detection of the proliferation, senescence, phenotype and differentiation ability of macaque BMSCs
Three macaques with an average age of 2.7 years (2.7 ± 0.58) and three macaques with an average age of 20.7 years (20.7 ± 0.58) were selected for the young and old groups. Bone marrow was collected using the aseptic technique. BMSCs were isolated and cultured by adherent culture screening. The structure and morphological changes were observed under the microscope. The CCK-8 method was used after 7 d of dynamic monitoring to study the old and young macaque P3 BMSCs. The β-galactosidase reagent detected the proportion of senescent cells. Flow cytometry was used to detect the cell surface antigens CD29, CD45, CD73, CD90, CD105, CD184 and HLA-DR. BMSCs were induced for differentiation into bone, fat and chondrocytes. Experimental protocols were approved by the Experimental Animal Ethics Committee of Kunming General Hospital of The People’s Liberation Army.
Construction and evaluation of the aging macaque BMSC model
Young cynomolgus BMSCs were treated with t-BHP at a concentration of 100-600 μmol/L for 6 h. The senescence ratio of the cells was measured using the β-galactosidase reagent. ELISA was used to detect the levels of IL-6, IL-11 and GM-CSF in young, aged and induced aged BMSC culture supernatants. RT-PCR was used to detect the mRNA expression levels of TERT, TCAB1, P21, SIRT1 and SIRT6 in BMSCs. The expression levels of the aging-associated proteins TCAB1, P21, P53 and SIRT6 in BMSCs were detected by Western blotting.
Anti-293T cell aging effect of young macaque BMSCs
For this experiment, 293T cells were treated with Tert-butyl hydroperoxide (t-BHP) at a concentration of 0-500 μmol/L for 2 h, and the senescence ratio of the cells was measured using the β-galactosidase reagent. The senescent 293T cells were cultured in the Transwell coculture system together with young macaque BMSCs. The relative expression of the telomere- and telomerase-related gene TBAB1 was detected by RT-PCR. The expression changes of the TCAB1 protein were detected by Western blotting.
Detection of cytokines in macaque bone marrow tissue and sequencing of the mononuclear cell transcriptome
A protein-antibody microarray was used to detect the levels of angiogenesis-related factors in the supernatant of bone marrow from macaques in the aging and young groups. The mRNA of mononuclear cells in the peripheral blood from macaques in the old and young groups was sequenced by second-generation gene sequencing technology. The sequencing data were analyzed by bioinformatics methods to reveal the differences in transcriptome levels in the young and old groups.
Statistical analysis
All statistical analyses were performed using SPSS 21.0 statistical software. The measurement results were expressed as the mean ± standard deviation (
Conclusions
1. The aged cell group is more prone to morphological changes associated with aging when cultured in vitro, and the proliferative ability of the cells in this group is decreased. The number of aged cells in this group is increased, while the multidirectional differentiation ability and the paracrine ability are decreased. MSC stemness is comprehensively decreased, resulting in insufficient functional cell turnover in organs, which may be an important mechanism for aging in the body. Cocultures of aging 293T cells with young macaque BMSCs showed that BMSCs have antiaging effects.
2. An aging macaque BMSC model was successfully developed. Both the aged cell group and the induced aging group showed aging morphology; the expression of aging genes was increased, and the expression of aging-related proteins was increased. The expression trends of the aged cell group and the induced aging group were largely similar.
3. The results of the protein chip experiments suggest that the risk of tumors in old macaques is increased. The peripheral blood cell analysis suggests that the old macaques may be in a chronic inflammatory state and that BMSCs may interact with the microenvironment in vivo.
4. Transcriptome sequencing of macaque mononuclear cells showed that senescence in macaques is related to multiple biological processes, multiple cellular components and multiple molecular functions. The GO terms and KEGG pathways involved in mRNA target gene alteration suggested that the immune functions of old macaques were decreased, the tumor risk was increased, apoptosis was increased, and the involved genes may be related to the aging of macaques. Further research is needed to confirm these results.
Supplementary Materials
Acknowledgements
We thank American Journal Experts for assisting with the preparation of this manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This work was supported by grants from the Yunnan Science and Technology Plan Project Major Science and Technology Project (2018ZF007) and the Yunnan Province Applied Basic Research Program Key Project (2018FA041, 2017FA040 and 2015FA039) and Applied Basic Research of Yunnan Province (2017FB042).
References
- 1. Mooney KM, Morgan AE, Mc Auley MT. Aging and computational systems biology.Wiley Interdiscip Rev Syst Biol Med.2016;8:123-139. https://doi.org/10.1002/wsbm.1328 [PubMed]
- 2. Hayflick L. The cell biology of aging.J Invest Dermatol.1979;73:8-14. https://doi.org/10.1111/1523-1747.ep12532752 [PubMed]
- 3. Sahin E and Depinho RA. Linking functional decline of telomeres, mitochondria and stem cells during ageing.Nature.2010;464:520-528. https://doi.org/10.1038/nature08982 [PubMed]
- 4. Schultz MB and Sinclair DA. When stem cells grow old: phenotypes and mechanisms of stem cell aging.Development.2016;143:3-14. https://doi.org/10.1242/dev.130633 [PubMed]
- 5. Scudellari M. Ageing research: Blood to blood.Nature.2015;517:426-429. https://doi.org/10.1038/517426a [PubMed]
- 6. Bianco P, Barker R, Brustle O, Cattaneo E, Clevers H, Daley GQ, De Luca M, Goldstein L, Lindvall O, Mummery C, Robey PG, Sattler de Sousa EBC, Smith A. Regulation of stem cell therapies under attack in Europe: for whom the bell tolls.EMBO J.2013;32:1489-1495. https://doi.org/10.1038/emboj.2013.114 [PubMed]
- 7. Kaltz N, Funari A, Hippauf S, Delorme B, Noel D, Riminucci M, Jacobs VR, Haupl T, Jorgensen C, Charbord P, Peschel C, Bianco P, Oostendorp RA. In vivo osteoprogenitor potency of human stromal cells from different tissues does not correlate with expression of POU5F1 or its pseudogenes.Stem Cells.2008;26:2419-2424. https://doi.org/10.1634/stemcells.2008-0304 [PubMed]
- 8. Bianco P, Robey PG, Simmons PJ. Mesenchymal stem cells: revisiting history, concepts, and assays.Cell Stem Cell.2008;2:313-319. https://doi.org/10.1016/j.stem.2008.03.002 [PubMed]
- 9. aKuznetsov SA, Cherman N, Riminucci M, Collins MT, Robey PG, Bianco P. Age-dependent demise of GNAS-mutated skeletal stem cells and “normalization” of fibrous dysplasia of bone.J Bone Miner Res.2008;23:1731-1740. https://doi.org/10.1359/jbmr.080609 [PubMed]
- 10. Riminucci M, Robey PG, Saggio I, Bianco P. Skeletal progenitors and the GNAS gene: fibrous dysplasia of bone read through stem cells.J Mol Endocrinol.2010;45:355-364. https://doi.org/10.1677/JME-10-0097 [PubMed]
- 11. Robey PG, Kuznetsov SA, Riminucci M, Bianco P. Bone marrow stromal cell assays: in vitro and in vivo.Methods Mol Biol.2014;1130:279-293. https://doi.org/10.1007/978-1-62703-989-5_21 [PubMed]
- 12. Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement.Cytotherapy.2006;8:315-317. https://doi.org/10.1080/14653240600855905 [PubMed]
- 13. Ghannam S, Bouffi C, Djouad F, Jorgensen C, Noel D. Immunosuppression by mesenchymal stem cells: mechanisms and clinical applications.Stem Cell Res Ther.2010;1:2 https://doi.org/10.1186/scrt2 [PubMed]
- 14. Djouad F, Bouffi C, Ghannam S, Noel D, Jorgensen C. Mesenchymal stem cells: innovative therapeutic tools for rheumatic diseases.Nat Rev Rheumatol.2009;5:392-399. https://doi.org/10.1038/nrrheum.2009.104 [PubMed]
- 15. Honczarenko M, Le Y, Swierkowski M, Ghiran I, Glodek AM, Silberstein LE. Human bone marrow stromal cells express a distinct set of biologically functional chemokine receptors.Stem Cells.2006;24:1030-1041. https://doi.org/10.1634/stemcells.2005-0319 [PubMed]
- 16. Sordi V, Malosio ML, Marchesi F, Mercalli A, Melzi R, Giordano T, Belmonte N, Ferrari G, Leone BE, Bertuzzi F, Zerbini G, Allavena P, Bonifacio E, et al. Bone marrow mesenchymal stem cells express a restricted set of functionally active chemokine receptors capable of promoting migration to pancreatic islets.Blood.2005;106:419-427. https://doi.org/10.1182/blood-2004-09-3507 [PubMed]
- 17. Introna M, Lucchini G, Dander E, Galimberti S, Rovelli A, Balduzzi A, Longoni D, Pavan F, Masciocchi F, Algarotti A, Mico C, Grassi A, Deola S, et al. Treatment of graft versus host disease with mesenchymal stromal cells: a phase I study on 40 adult and pediatric patients.Biol Blood Marrow Transplant.2014;20:375-381. https://doi.org/10.1016/j.bbmt.2013.11.033 [PubMed]
- 18. Zhang H, Fazel S, Tian H, Mickle DA, Weisel RD, Fujii T, Li RK. Increasing donor age adversely impacts beneficial effects of bone marrow but not smooth muscle myocardial cell therapy.Am J Physiol Heart Circ Physiol.2005;289:H2089-2096. https://doi.org/10.1152/ajpheart.00019.2005 [PubMed]
- 19. Gibbs RA, Rogers J, Katze MG, Bumgarner R, Weinstock GM, Mardis ER, Remington KA, Strausberg RL, Venter JC, Wilson RK, Batzer MA, Bustamante CD, Eichler EE, et al. Evolutionary and biomedical insights from the rhesus macaque genome.Science.2007;316:222-234. https://doi.org/10.1126/science.1139247 [PubMed]
- 20. Hall BM, Balan V, Gleiberman AS, Strom E, Krasnov P, Virtuoso LP, Rydkina E, Vujcic S, Balan K, Gitlin I, Leonova K, Polinsky A, Chernova OB, et al. Aging of mice is associated with p16(Ink4a)- and beta-galactosidase-positive macrophage accumulation that can be induced in young mice by senescent cells.Aging (Albany NY).2016;8:1294-1315. https://doi.org/10.18632/aging.100991 [PubMed]
- 21. Hall BM, Balan V, Gleiberman AS, Strom E, Krasnov P, Virtuoso LP, Rydkina E, Vujcic S, Balan K, Gitlin II, Leonova KI, Consiglio CR, Gollnick SO, et al. p16(Ink4a) and senescence-associated beta-galactosidase can be induced in macrophages as part of a reversible response to physiological stimuli.Aging (Albany NY).2017;9:1867-1884. https://doi.org/10.18632/aging.101268 [PubMed]
- 22. Choudhery MS, Khan M, Mahmood R, Mehmood A, Khan SN, Riazuddin S. Bone marrow derived mesenchymal stem cells from aged mice have reduced wound healing, angiogenesis, proliferation and anti-apoptosis capabilities.Cell Biol Int.2012;36:747-753. https://doi.org/10.1042/CBI20110183 [PubMed]
- 23. Chen H, Liu X, Zhu W, Hu X, Jiang Z, Xu Y, Wang L, Zhou Y, Chen P, Zhang N, Hu D, Zhang L, Wang Y, et al. SIRT1 ameliorates age-related senescence of mesenchymal stem cells via modulating telomere shelterin.Front Aging Neurosci.2014;6:103 https://doi.org/10.3389/fnagi.2014.00103 [PubMed]
- 24. Kim S, Bi X, Czarny-Ratajczak M, Dai J, Welsh DA, Myers L, Welsch MA, Cherry KE, Arnold J, Poon LW, Jazwinski SM. Telomere maintenance genes SIRT1 and XRCC6 impact age-related decline in telomere length but only SIRT1 is associated with human longevity.Biogerontology.2012;13:119-131. https://doi.org/10.1007/s10522-011-9360-5 [PubMed]
- 25. Yuan HF, Zhai C, Yan XL, Zhao DD, Wang JX, Zeng Q, Chen L, Nan X, He LJ, Li ST, Yue W, Pei XT. SIRT1 is required for long-term growth of human mesenchymal stem cells.J Mol Med (Berl).2012;90:389-400. https://doi.org/10.1007/s00109-011-0825-4 [PubMed]
- 26. Alves H, Mentink A, Le B, van Blitterswijk CA, de Boer J. Effect of antioxidant supplementation on the total yield, oxidative stress levels, and multipotency of bone marrow-derived human mesenchymal stromal cells.Tissue Eng Part A.2013;19:928-937. https://doi.org/10.1089/ten.tea.2011.0700 [PubMed]
- 27. Yu JM, Wu X, Gimble JM, Guan X, Freitas MA, Bunnell BA. Age-related changes in mesenchymal stem cells derived from rhesus macaque bone marrow.Aging Cell.2011;10:66-79. https://doi.org/10.1111/j.1474-9726.2010.00646.x [PubMed]
- 28. Yue J, Sheng Y, Orwig KE. Identification of novel homologous microRNA genes in the rhesus macaque genome.BMC Genomics.2008;9:8 https://doi.org/10.1186/1471-2164-9-8 [PubMed]
- 29. Ponnappan S and Ponnappan U. Aging and immune function: molecular mechanisms to interventions.Antioxid Redox Signal.2011;14:1551-1585. https://doi.org/10.1089/ars.2010.3228 [PubMed]