Introduction
Skin aging is a biological process consisting in two types: intrinsic or chronological aging, and extrinsic aging or photoaging [1]. The latter is mainly due to the progressive and cumulative exposure to ultraviolet radiation [2]. Although pathogenetically distinct, several studies revealed that chronological aging and photoaging share some crucial molecular pathways, particularly in subjects aged over 70 years [3]. Intrinsic aged and photoaged skin are characterized from epidermal atrophy and dermal thinning for the progressive decrease of cellularity, collagen synthesis and the increased collagen degradation from upregulation of dermal activity of matrix metalloproteinases (MMPs) and the reduction of TGF-β signaling [4,5]. Dermal fibroblasts greatly contribute to the production, organization and homeostasis of extracellular matrix (ECM) [6]. Age-related reduced proliferation of fibroblasts further contributes to impair dermal homeostasis and favors age-related skin alterations [7,8]. The critical role of fibroblasts in skin integrity is indirectly confirmed by the promotion of skin regeneration and rejuvenation obtained by experimental autologous injection of cultured dermal fibroblasts [9]. The impairment of TGF-β signaling-related genes is a major contributor for the reduction of ECM protein synthesis in aged skin [10,11]. Human skin converts vitamin A (retinol) to its biologically active metabolite all-trans retinoic acid (atRA) [12]. The latter contributes to control cell growth and differentiation as well as to maintain skin homeostasis [13]. In particular, atRA is known to have a strong effect on growth and differentiation of human epidermal cells in vivo and in vitro [14-16]. Topical atRA administration, used for the therapy of precancerous actinic keratosis, is also applied to counteract skin aging, and likely favors dermal collagen accumulation [17,18]. Efficacy of retinoid therapy was documented in both intrinsic aging and photoaging of skin [19]. Sun-exposed and sun-protected adult skin received equal beneficial effect from atRA therapy, with beneficial effects particularly on keratinocyte proliferation and dermal collagen production, whereas neonatal skin was unresponsive [3]. These findings suggest the selective efficacy of retinoid treatment in aged skin [16,19]. Two classes of receptors mediate the biological effects of atRA. Classically, retinoids exert their pleiotropic and transcriptional effects through the binding to nuclear receptors, namely the family of retinoic acid receptors (RARα, β, and γ) and retinoid X receptors (RXRα, β, and γ), with specific functions [20,21]. Recent studies documented that also cytoplasmic cellular retinol banding proteins (CRBP-I and CRBP-II) and cellular retinoic acid binding proteins (CRABP-I and CRABP-II) play a crucial role in skin homeostasis and carcinogenesis, likely by contributing to regulate intracytoplasmatic retinoid trafficking [22,23]. CRABP-I is present in many tissues and cells, whereas CRABP-II is recognized as the predominant form of human epidermis [15,24]. In particular, CRABP-II is expressed by fibroblasts and suprabasal keratinocytes, whereas CRABP-I mostly from epidermal melanocytes [24,25]. CRABP-II selectively binds atRA and its expression increased after atRA treatment, suggesting a sort of intracellular regulatory feedback [26]. Although CRABP-II involvement in skin carcinogenesis is well documented [27], no evidence of its potential role in aging process exists. To this aim, we investigated CRABP-II expression in the skin of aged donors and the effects of CRABP-II loss on skin aging in mice.
Results
CRABP-II expression is reduced in human epidermis with aging
To investigate the potential role of CRABP-II in skin aging, immunostaining was performed on paraffin sections of human skin samples from donors of different age. As shown in Fig. 1A, immunohistochemistry documented in young/adult human epidermis a faint cytoplasmic CRABP-II expression in basal and malpighian keratinocytes, a more intense cytoplasmic immunoreaction, also nuclear, in the granular layer, and its absence in the stratum corneum, according to the expression pattern described by others [28–30]. CRABP-II was also strongly expressed from dermal fibroblasts and cutaneous adnexal structures. Semiquantitative evaluation (Figure 1B) of immunohistochemistry documented that CRABP-II expression in old human epidermis and dermis was reduced compared with that of young donors (p<0.01 and p<0.0001, respectively).
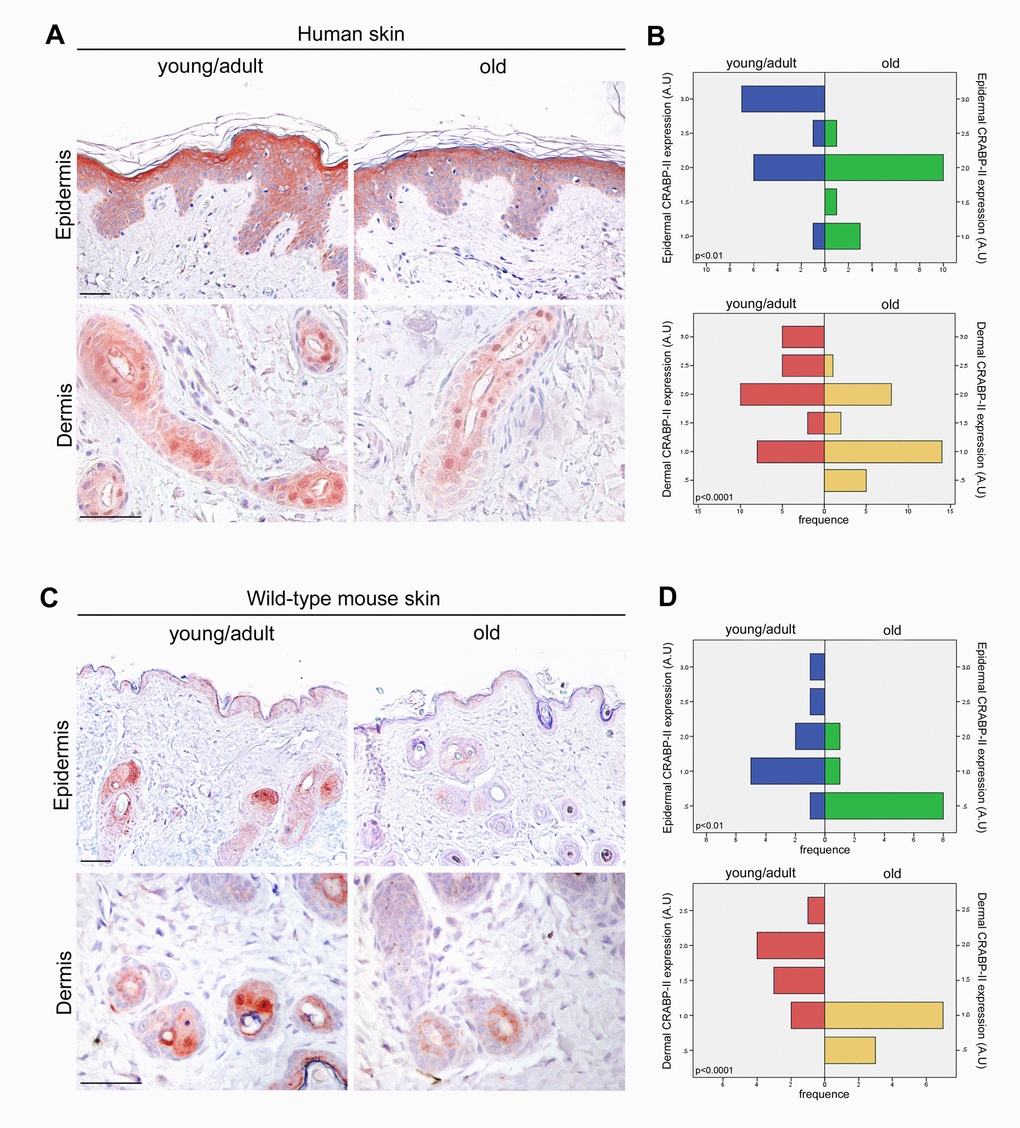
Figure 1. CRABP-II expression is reduced in aged human and mouse skin. (A) Representative images of CRABP-II immunostaining of human normal skin from young/adult and old donors. (B) Bar graphs show semiquantitative evaluation of human epidermal and dermal CRABP-II immunostaining (n=15 young/adult and n=15 old), respectively. (C) Representative images of CRABP-II immunostaining of young/adult and old wild-type mouse skin. (D) Bar graphs show semiquantitative evaluation of mouse epidermal and dermal CRABP-II immunostaining (n=10 young/adult and n=10 old WT). Scale bar: 50µm. Mann-Whitney’s U-test. Abbreviation: AU, arbitrary units.
CRABP-II gene deletion accelerates skin aging
The above data highlight an age-related decrease of CRABP-II expression in human skin with aging. To verify if that decrease was not species-related, we investigated CRABP-II also in mouse skin. Similarly to humans, epidermal and dermal CRABP-II expression was reduced in old compared with young/adult wild-type mice (Figure 1C and D; p<0.01 and p<0.0001). Microscopic and morphometric examination of H&E-stained skin sections (Figure 2A and B) also revealed a decreased number of epidermal layers and a reduction of dermal thickness in young/adult CRABP-II knock-out compared with wild-type mice (p<0.05), that became more pronounced comparing old groups (p<0.01). A hypodermic thinning (p<0.01) and a decrease of pilosebaceous units (p<0.05) were also observed in CRABP-II knock-out compared with wild-type young/adult mice. The reduction of hypodermis thickness and pilosebaceous units was even more marked in old CRABP-II knock-out compared with wild-type mice (p<0.001 and p<0.01, respectively). Masson’s trichrome staining showed thickened collagen bundles in wild-type young/adult mice, instead in wild-type old and CRABP-II knock-out mice collagen bundles decreased and became loose (Figure 2A). CD31 immunostaining (Figure 3A and 3B) showed also a reduction in dermal blood vessels in CRABP-II knock-out mice compared with both young/adult and old with wild-type groups (p<0.05).
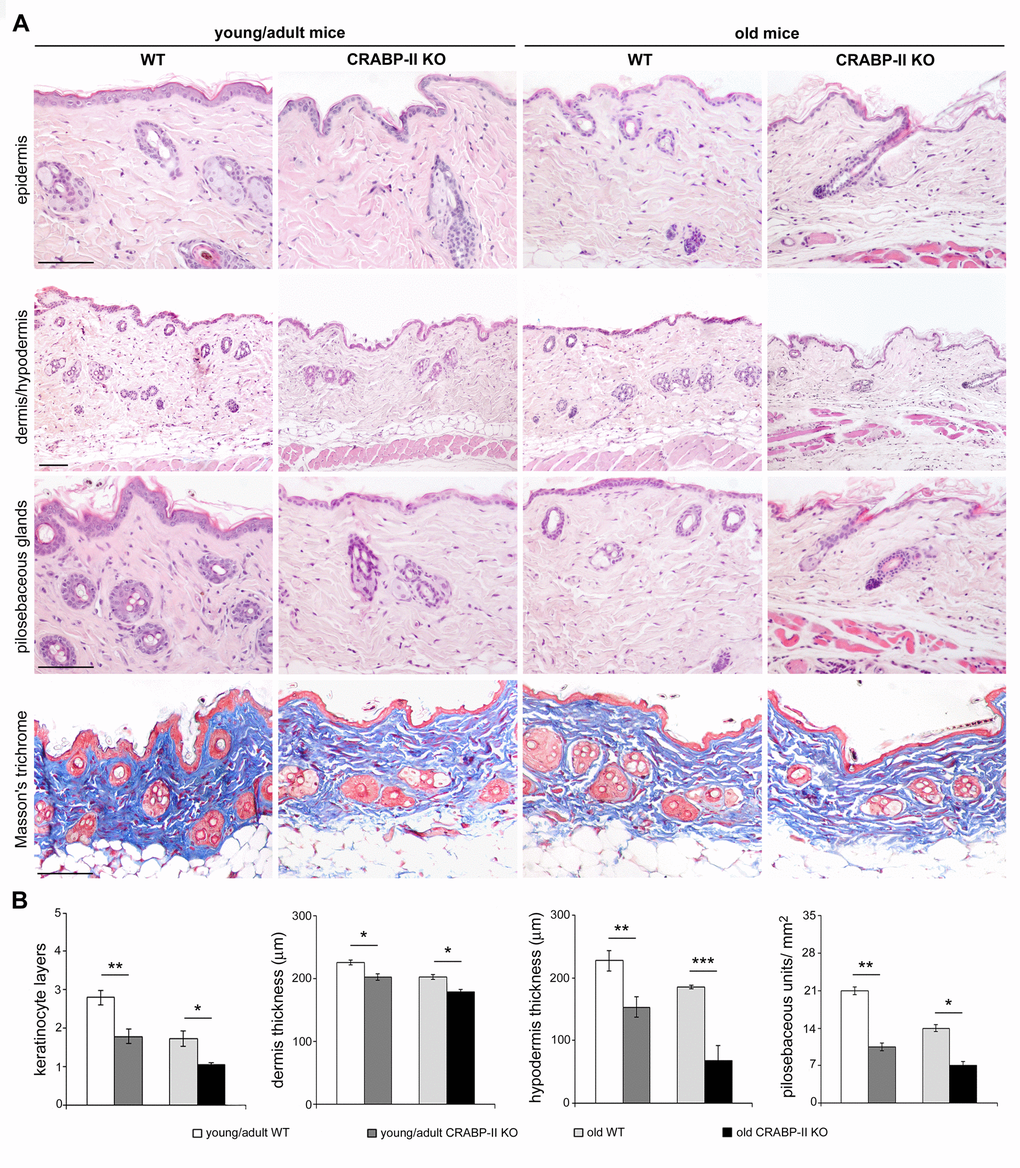
Figure 2. Skin aging histological features compare earlier and more pronounced in CRABP-IIknock-out mice. (A) Representative Haematoxylin&Eosin and Masson's trichrome-stained skin sections of young/adult and old wild-type (WT) and CRABP-II knock-out (KO) mice. (B) Bar graphs show semiquantitative evaluation of skin aging parameters (n=10 young/adult and n=10 old WT, n=10 young/adult and n=10 old CRABP-II KO). Scale bar: 100µm. Values are group mean ± SEM. t-Test: *, ** and *** indicate p<0.05, p <0.01 and p< 0.001, respectively.
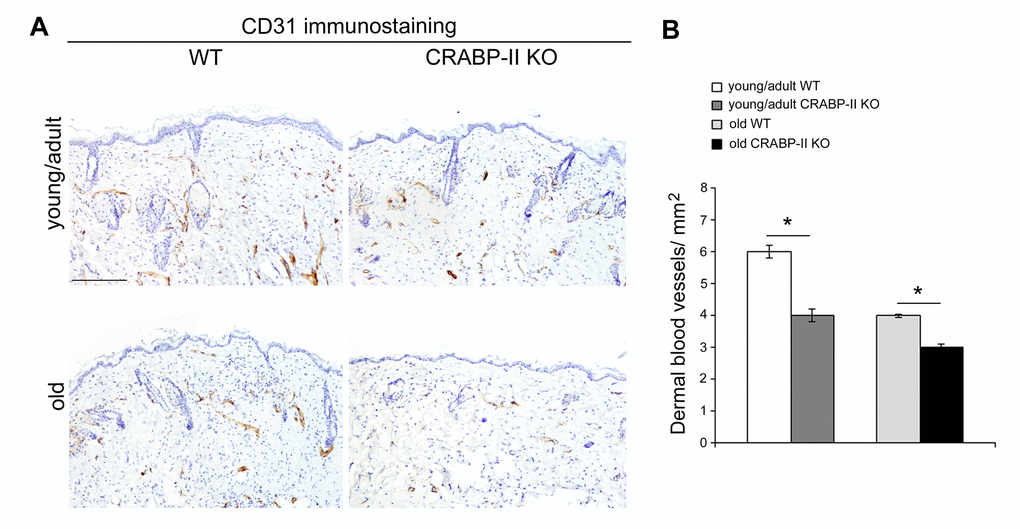
Figure 3. Dermal blood vessels are reduced in CRABP-IIknock-out mice. (A) Representative images and (B) bar graph showing semiquantitative evaluation of CD31-positive dermal small vessels in young/adult and old wild-type (WT) and CRABP-II knock-out (KO) mice (n=10 young/adult and n=10 old WT, n=10 young/adult and n=10 old CRABP-II KO). Scale bar: 100µm. Values are group mean ± SEM. t-Test: * indicates p<0.05.
Epidermal proliferation and differentiation are reduced in old CRABP-II knock-out mice
In order to define if an impaired proliferation contributed to the severity of age-related alterations observed in CRABP-II knock-out mice, we evaluated keratinocyte and dermal cell proliferation in vivo by Ki67+ immunohistochemistry. Both young/adult and old CRABP-II knock-out showed a reduction of epidermal Ki67+ basal keratinocytes compared to age-matched wild-type mice (Figure 4A and 4B; p<0.01 and p<0.05, respectively). Also the percentage of Ki67+ dermal cells of young/adult and old CRABP-II knock-out was reduced compared to age-matched wild-type mice (p<0.001 and p<0.05, respectively).
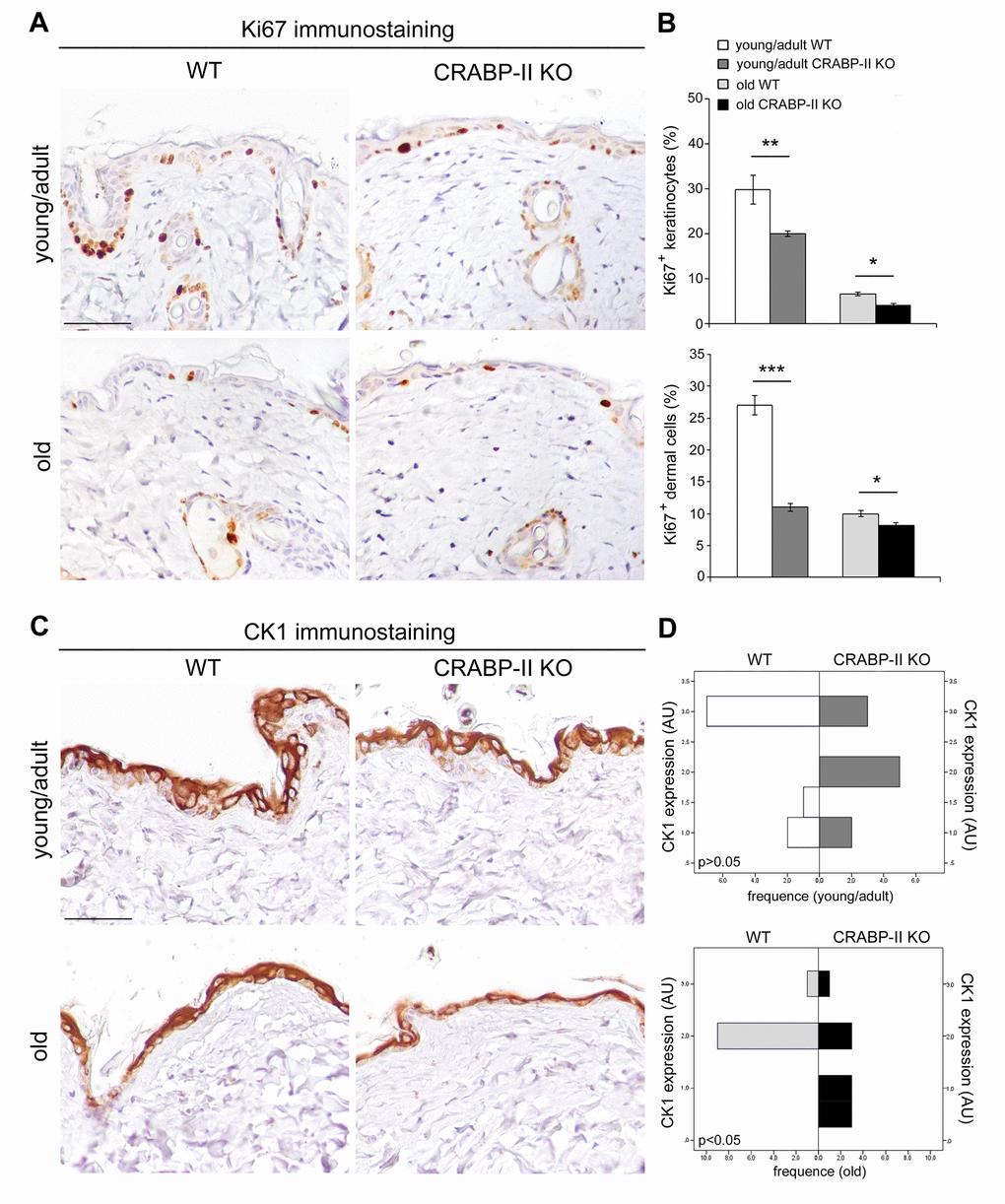
Figure 4. Epidermal proliferation and differentiation are reduced in CRABP-IIknock-out mice. (A) Representative images of Ki67 immunostaining of young/adult and old wild-type (WT) and CRABP-II knock-out (KO) skin. Scale bar: 50µm. (B) Bar graphs show semiquantitative evaluation of Ki67 positive epidermal keratinocytes and dermal cells (n=10 young/adult and n=10 old WT, n=10 young/adult and n=10 old CRABP-II KO). Values are mean ± SEM. t-Test: *, ** and *** indicate p< 0.05, p< 0.01 and p< 0.001, respectively. (C) Representative images of cytokeratin 1 (CK1) immunostaining of young/adult and old WT and CRABP-II KO skin. Scale bar: 50µm. (D) Bar graphs showing the semiquantitative CK1 evaluation (n=10 young/adult and n=10 old WT, n=10 young/adult and n=10 old CRABP-II KO). Mann-Whitney’s U-test. Abbreviation: AU, arbitrary units.
To verify if an impaired differentiation process associated to the reduced keratinocyte proliferation in CRABP-II knock-out mice, we analyzed cytokeratin 1 (CK1) expression, a marker of epidermal differentiation, by immunohistochemistry. As reported in Figure 4C and 4D, epidermal CK1 expression was significantly reduced in old but not in young/adult CRABP-II knock-out compared to wild-type mice counterpart (p<0.05). Altogether, those data indicated that reduced cell proliferation is an early pathophysiological sign of aging, whereas impaired keratinocyte differentiation is a later event in CRABP-II knock-out mice skin.
CRABP-II loss influences proliferation and transcriptional profile of cultured dermal fibroblasts
To verify if the age-related reduction of dermal fibroblast proliferation associated to changes of transcriptional profile, we cultured dermal fibroblasts in a two-dimensional system. As shown in Figure 6A, dermal fibroblasts of CRABP-II knock-out proliferated less compared with those from wild-type mice, in particular at 4 and 6 days after seeding (p<0.001), confirming the in vivo results. Then, we investigated the effects of CRABP-II loss on the transcriptional profile of dermal fibroblasts (Figure 6B). We documented a reduction of Col1A1 and Col1A2 (p<0.01), TGFβ1, TGFβRI and TGFβRII (p<0.05), and an increase of MMP2 (p<0.01) transcript levels in cultured dermal fibroblasts from young/adult CRABP-II knock-out compared with those from wild-type mice. These data strongly suggest that age-related effects are, at least in part, mediated by an early alteration of TGFβ-related pathway, a reduction of collagen synthesis and an increased MMP-mediated ECM remodeling.
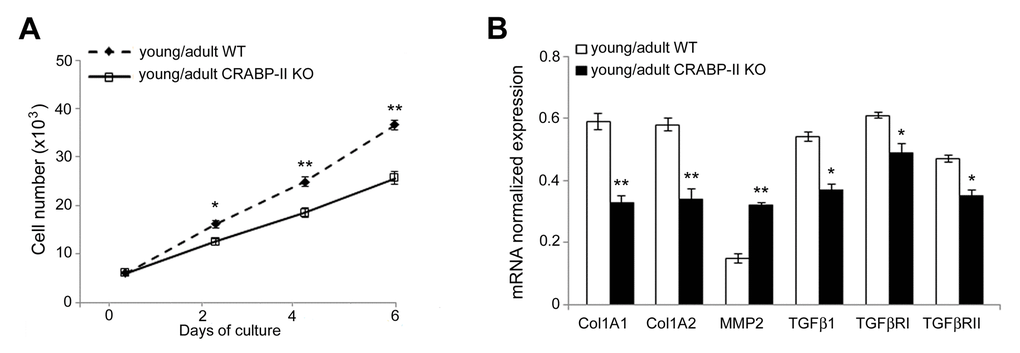
Figure 6. Proliferation rate and transcriptional profiling are altered in cultured dermal fibroblasts from CRABP-IIknock-out mice. (A) Growth curve of cultured dermal fibroblasts from young/adult wild-type (WT, n=3) and CRABP-II knock-out mice (KO, n=3). (B) Col1A1, Col1A2, MMP2, TGFβ1, TGFβ RI and TGFβ RII mRNA normalized expression in dermal fibroblasts from young/adult WT and CRABP-II KO. Values are group mean ± SEM. t-Test: * and ** indicate p< 0.05 and p< 0.01, respectively.
Discussion
In this study, we investigated for the first time the role of CRABP-II expression in skin aging. We documented that reduced CRABP-II expression characterizes human and mouse aged skin and loss of CRABP-II gene leads to a premature and a more severe skin aging involving both dermal and hypodermal compartments. CRABPs are a family of proteins that specifically bind atRA facilitating its transport into the nucleus where atRA binds its receptors (RARs). CRABP-II, first detected in embryonic and neonatal skin of rats and chicks, is also now recognized as the predominant form in human epidermis [24,25]. atRA is known to have a profound effect on growth and differentiation of human epidermal cells both in vivo and in vitro [15]. CRABP-II has been suggested to be a key marker of retinoid-linked activity in the skin, although its exact function is still partially unknown [16]. Some studies reported that CRABP-II is induced by atRA in full-thickness skin, in epidermis and cultured fibroblasts [2,31,32]. In particular, epidermal differentiation, stimulated by culture at the liquid-air interface, tended to enhance CRABP II expression [25]. According to previous studies [28–30], we documented CRABP-II expression in differentiating suprabasal keratinocytes, in particular the granular layer, with a cytoplasmic and nuclear distribution. Since we documented a reduction of CRABP-II expression in aged human and mouse skin, to better clarify its role in skin aging, we investigated the onset and the severity of age-related skin changes in CRABP-II knock-out C57bl/6 mice. We documented an early reduction of epidermal keratinocyte layers, a dermal and hypodermal thinning and a decreased number of pilosebaceous units in young/adult CRABP-II knock-out. This reduction was further enhanced in old CRABP-II knock-out skin when compared with wild-type mice. In addition, Masson’s trichrome staining revealed a better quality of dermal collagen in wild-type young/adult mice that showed collagen bundles more thickened than wild-type old and CRABP-II knock-out mice, whose collagen bundles decreased and became loose. This finding confirmed the decreased collagen content and its degradation observed in aged skin [33].
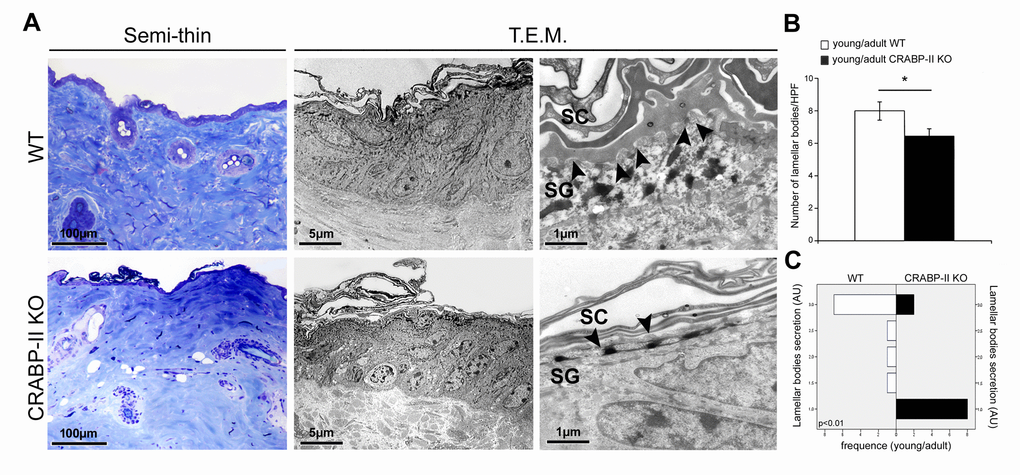
Since age-related epidermal thinning and dermal atrophy are due to the decrease of keratinocyte and dermal fibroblast turnover rate [16], we analyzed if CRABP-II gene loss influences cutaneous cell proliferation and differentiation in vivo by immunohistochemistry. We documented that keratinocytes and dermal fibroblasts in CRABP-II knock-out proliferated less than wild-type starting from young/adult mice, and this reduction got worse comparing old group. Similarly, CK1 expression slightly decreased in young/adult and more markedly in old CRABP-II knock-out mice when compared with wild-type group. Reduced CK1 expression is reported to be a characteristic of skin aging [34,35]. The senescence of transit-amplifying suprabasal cells (CK1 positive) is considered to be responsible for epidermal hypoplasia and epidermal thinning during the late phase of skin aging [36]. Our data support that the age-related changes appear earlier in CRABP-II knock-out old mice with a reduced epidermal and dermal proliferation and successively become more severe, with an impaired keratinocyte differentiation. It has been reported that retinol treatment increased dermal vascularity by stimulating endothelial cell proliferation [37]. The decrease of dermal blood vessels in CRABP-II knock-out compared with wild-type mice strongly suggests that a reduced vascular supply contributes to the age-related epidermal and dermal atrophy [38]. Aged skin shows alterations of permeability and severe dermal sclerosis, resulting an age-related impairment of epidermal barrier [39]. The stratum corneum is the outermost skin layer and provides a highly efficient barrier for water and a protection against infections [40]. Epidermal lamellar bodies are vesicles containing lipids and enzymes present in keratinocytes of stratum spinosum in high number at the interface of the stratum granulosum and stratum corneum [40]. Before extracellular exocytosis, lipids from lamellar bodies are enzymatically metabolized to free ceramides and fatty acids that contribute to the formation of a protective barrier in the stratum corneum [40]. Ultrastructural analysis documented a reduced number of lamellar bodies as well as their secretion in CRABP-II knock-out mice starting from young/adult group, suggesting that the loss of CRABP-II expression favors an early impairment of epidermal barrier function.
Aging promotes aberrant collagen homeostasis by down-regulating type I collagen accumulation, the major structural protein in skin, and promoting collagen degradation [14]. In particular, the alteration of organization, structure and content of dermal collagen derives from the imbalance of its homeostasis, likely due to the elevated activity of matrix metalloproteinases (MMPs) [41]. Imbalance of collagen homeostasis is considered responsible for the winkled appearance and atrophy of aged skin [41]. Vitamin A and its metabolites have been shown to promote new deposition of collagen and prevent its degradation by increasing type I procollagen and reducing MMP-1 activity [10]. In order to clarify if CRABP-II loss also influences dermal collagen homeostasis, we cultured dermal fibroblasts from CRABP-II knock-out and wild-type young/adult mice. Our data confirmed the in vivo observation of a reduced proliferation rate in the dermis of CRABP-II knock-out mice. It has been hypothesized that retinoids increase collagen synthesis either by restoring procollagen expression, inhibited by c-Jun signaling triggered by UV irradiation, or by activating TGF-β-mediated signal transduction [8,42], which may in turn induce collagen synthesis in dermal fibroblasts by a paracrine action [42]. In fact, CRABP-II knock-out-derived dermal fibroblasts displayed a different modulation of TGFβ-related pathway, in particular a reduced expression of Col1A1, Col1A2, TGFβ1, TGFβRII and an increase of MMP2 transcripts compared with wild-type mice. Those data strongly suggest that age-related changes in CRABP-II knock-out mice are at least in part mediated by an alteration of TGFβ-related pathway signaling, with a consequent reduction of collagen synthesis and an increased MMP-mediated extracellular matrix degradation.
In conclusion, our data strongly support the involvement of CRABP-II in skin aging, since CRABP-II gene loss accelerated age-related skin changes. However, these findings suggest the need of further human studies on chronological aging and photoaged skin to establish the specific role of CRABP-II and CRABP-II-related retinoid signaling and to develop new targeted therapeutic interventions to counteract skin aging.
Materials and Methods
Human skin
Anonymous sections of human normal skin were obtained from the paraffin block archive of the Anatomic Pathology, Tor Vergata University of Rome, from patients undergoing skin surgery for non-malignant lesions. Before surgical procedures, written consent was obtained and the study were in accordance with Declaration of Helsinki and local Ethical Committee guidelines. Patients were arbitrary divided in two groups: young/adult, from 18 to 45 years (n=15; mean age 31.1) and old, from 60 to 75 years (n=15; mean age 68.7).
Generation of CRABP-II null mice and mutant genotyping
Transgenic C57Bl/6 mice heterozygous for CRABPII gene were kindly gifted by Dr. V. Giguère (McGill University/Animal Resources Centre, Montreal, Canada). CRABP-II gene knock-out mice were obtained, as reported [27]. Mice were housed in a temperature-controlled facility with a 12 light/dark cycle, according to the EU guide for the use of laboratory animals and the study approved by Institutional Animal Care and Use Committee, IACUC, according to the Dlgs 116/92. To confirm the mice progenies, DNA was obtained from the terminal portion of the tails by using the manufacture’s extraction protocol (Qiagen, Hilden-Germany, QIAamp-kit-tissue). The genotype was confirmed by reverse transcription PCR analysis, using specific primers [27].
Microscopic investigation of mice skin
Mice were enrolled in two age groups, 5-10 months (young/adult, n=10) and 18-28 months (old, n=10) for both wild-type and CRABP-II knock-out. Dorsal skin was excised after killing by cervical dislocation and harvested for histological examination. Skin samples were fixed in 10% buffered formalin. Paraffin sections were stained with Haematoxylin&Eosin for morphometric quantification of keratinocytes layers, dermis and hypothermis thickness and pilosebaceous units [43]. The quality and distribution of collagen was evaluated after tissue staining with the Masson’s trichrome technique [33].
Immunohistochemical study
For immunohistochemistry, tissue samples (n=10 young/adult and n=10 old WT, n=10 young/adult and n=10 old CRABP-II KO) were placed in 10% buffered formalin or zinc fixative for 24 h, dehydrated and embedded in paraffin [27]. Serial 4-μm thick sections were deparaffinized, rehydrated and after antigen retrieval and nonspecific peroxidase blocking, incubated with rabbit polyclonal anti-CRABP-II (A300-809A, 1:300, Bethyl, Texas, USA), rabbit monoclonal anti-Ki-67 (NB500-170, 1:300, Novus Biological, USA), anti-cytokeratin 1 (ab24643, 1:2500, Abcam, UK), rat monoclonal CD31 antibody (5573550, 1:100, BD Pharmingen, San Diego, CA, USA). Positive controls (tissues expressing the antigens) and negative controls (tissues not expressing the antigens and immunoglobulin isotype-matched negative controls) were included. Semiquantitative evaluation of CRABP-II and CK1 staining intensity was performed under a light microscope by two researches, independently, by using a grading system in arbitrary units as follows: absence of positivity (0), focal presence (0.5), weak (1), moderate (2), and strong (3) in at least ten randomly selected fields/mouse [27]. The mean scores from each animal were calculated and analyzed using a non-parametric statistical analysis (see Statistical analysis). Ki67 positivity, as the percentage of positive nuclei/total nuclei/field (at 400X magnification) in basal epidermal layer and dermal fibroblasts, was evaluated in at least ten randomly selected fields/mouse [44]. The number of skin dermal blood vessels (CD31 positive) per mm2 was evaluated in at least ten randomly selected fields/mouse at 400X magnification [45,46]. The mean score for every sample was calculated and analyzed (see Statistical analysis). The inter-observer reproducibility was > 95%.
Ultrastructural study
For transmission electron microscopy, small mouse skin samples (n=10 young/adult and n=10 old WT, n=10 young/adult and n=10 old CRABP-II KO) were fixed in Karnovsky’s solution, processed and embedded in EPON 812 [47]. Thin sections were stained with 0.1% toluidine blue, and ultrathin sections, counterstained with uranyl acetate and lead citrate, and photographed with H-7100FA Hitachi transmission electron microscope (Japan). We took ten images (magnification X 15,000) from each specimen to calculate the number of lamellar bodies using an 8×4 cm sized rectangular grid just below the stratum corneum and counted the lamellar bodies within it; the degree of secretion of lamellar bodies was estimated using a semiquantitative scale (+ = 1, ++ = 2, +++ = 3), as reported [48]. The mean score for every sample was calculated and analyzed (see Statistical analysis).
Isolation of mouse dermal fibroblasts
For isolation of fibroblasts, wild-type (n=3) and CRABP-II knock-out mice (n=3) were killed by cervical dislocation, soaked in betadine and placed in ice, then washed twice with 70% ethanol. The dermis was stretched in 0.25% trypsin solution for epidermis separation, then dermal samples were cut into small pieces were placed in Dulbecco's modified Eagle’s medium (DMEM) plus 2% HEPES and digested with 0.1% of Collagenase type 1 (Sigma-Aldrich, St. Louis, MO, USA). Dermal fibroblasts were used for experiments between passage 1 and 3.
Cell growth assay
Dermal fibroblasts were seeded at density of 3x103 cells/cm2 into 24-well plates and allowed to attach for 24 hours, starved in DMEM containing 0.1% FBS for 24 hours prior to be cultured in complete growth medium. Then, cells were trypsinized and counted, as reported [49].
Reverse transcriptase and Real-Time polymerase chain reaction
Total RNA was isolated using the TRIZOL reagent, reverse transcribed and Real-Time polymerase chain reaction performed using iQTM SYBR® Green Supermix (Bio-Rad, CA, USA) in an iQ5 Multicolour Real-time PCR Detection System (Bio-Rad) [50] using primer sequences for collagen 1 alpha 1 (Col1A1), collagen 1 alpha 2 (Col1A2), transforming growth factor 1 (TGFβ1), TGFRI, TGFRII, matrix metalloproteinase-2 (MMP-2) and CRABP-II listed in Supplementary Table 1. Experiments were performed in triplicate and expression values were normalized to GAPDH levels using the following formula: 2− (ΔCT) [51].
Statistical analysis
For all analyses, p value < 0.05 was considered significant. For categorical variables (semiquantitative evaluation of CRABP2 and CK1 immunohistochemistry as well as lamellar bodies’ secretion), the Mann-Whitney’s U-test was performed. For the remaining quantitative variables, differences were analyzed by one-way analysis of variance (ANOVA) followed from a Bonferroni post hoc test and using the Student t-test. Results were presented as the mean ± SEM (standard error of mean). All quantitative data sets presented here passed the normality tests. Homogeneous variance of the input data was confirmed using an F-test. Statistical analyses were performed with SPSS software (IBM, SPSS Statistics, US. version 23).
Supplementary Materials
Acknowledgements
We thank for the kind gift of CRABP-II transgenic mice Dr. V. Giguère (McGill University/Animal Resources Centre, Montreal, Canada). We thank also Dr. S. Cappelli and Dr. Volpe for their technical collaboration.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
The authors received no specific funding for this work.
References
- 1. Jenkins G. Molecular mechanisms of skin ageing. Mech Ageing Dev. 2002; 123:801–10. https://doi.org/10.1016/S0047-6374(01)00425-0 [PubMed]
- 2. Mukherjee S, Date A, Patravale V, Korting HC, Roeder A, Weindl G. Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety. Clin Interv Aging. 2006; 1:327–48. https://doi.org/10.2147/ciia.2006.1.4.327 [PubMed]
- 3. Varani J, Fisher GJ, Kang S, Voorhees JJ. Molecular mechanisms of intrinsic skin aging and retinoid-induced repair and reversal. J Investig Dermatol Symp Proc. 1998; 3:57–60. https://doi.org/10.1038/jidsymp.1998.14 [PubMed]
- 4. Quan T, Fisher GJ. Role of Age-Associated Alterations of the Dermal Extracellular Matrix Microenvironment in Human Skin Aging: A Mini-Review. Gerontology. 2015; 61:427–34. https://doi.org/10.1159/000371708 [PubMed]
- 5. Makrantonaki E, Zouboulis CC. Molecular mechanisms of skin aging: state of the art. Ann N Y Acad Sci. 2007; 1119:40–50. https://doi.org/10.1196/annals.1404.027 [PubMed]
- 6. Tracy LE, Minasian RA, Caterson EJ. Extracellular Matrix and Dermal Fibroblast Function in the Healing Wound. Adv Wound Care (New Rochelle). 2016; 5:119–36. https://doi.org/10.1089/wound.2014.0561 [PubMed]
- 7. Varani J, Schuger L, Dame MK, Leonard C, Fligiel SE, Kang S, Fisher GJ, Voorhees JJ. Reduced fibroblast interaction with intact collagen as a mechanism for depressed collagen synthesis in photodamaged skin. J Invest Dermatol. 2004; 122:1471–79. https://doi.org/10.1111/j.0022-202X.2004.22614.x [PubMed]
- 8. Fisher GJ, Shao Y, He T, Qin Z, Perry D, Voorhees JJ, Quan T. Reduction of fibroblast size/mechanical force down-regulates TGF-β type II receptor: implications for human skin aging. Aging Cell. 2016; 15:67–76. https://doi.org/10.1111/acel.12410 [PubMed]
- 9. Munavalli GS, Smith S, Maslowski JM, Weiss RA. Successful treatment of depressed, distensible acne scars using autologous fibroblasts: a multi-site, prospective, double blind, placebo-controlled clinical trial. Dermatol Surg. 2013; 39:1226–36. https://doi.org/10.1111/dsu.12204 [PubMed]
- 10. Quan T, Shao Y, He T, Voorhees JJ, Fisher GJ. Reduced expression of connective tissue growth factor (CTGF/CCN2) mediates collagen loss in chronologically aged human skin. J Invest Dermatol. 2010; 130:415–24. https://doi.org/10.1038/jid.2009.224 [PubMed]
- 11. Verrecchia F, Mauviel A. Transforming growth factor-beta signaling through the Smad pathway: role in extracellular matrix gene expression and regulation. J Invest Dermatol. 2002; 118:211–15. https://doi.org/10.1046/j.1523-1747.2002.01641.x [PubMed]
- 12. Fisher C, Blumenberg M, Tomić-Canić M. Retinoid receptors and keratinocytes. Crit Rev Oral Biol Med. 1995; 6:284–301. https://doi.org/10.1177/10454411950060040201 [PubMed]
- 13. Smeland EB, Rusten L, Jacobsen SE, Skrede B, Blomhoff R, Wang MY, Funderud S, Kvalheim G, Blomhoff HK. All-trans retinoic acid directly inhibits granulocyte colony-stimulating factor-induced proliferation of CD34+ human hematopoietic progenitor cells. Blood. 1994; 84:2940–45. [PubMed]
- 14. Quan T, Qin Z, Shao Y, Xu Y, Voorhees JJ, Fisher GJ. Retinoids suppress cysteine-rich protein 61 (CCN1), a negative regulator of collagen homeostasis, in skin equivalent cultures and aged human skin in vivo. Exp Dermatol. 2011; 20:572–76. https://doi.org/10.1111/j.1600-0625.2011.01278.x [PubMed]
- 15. Eller MS, Oleksiak MF, McQuaid TJ, McAfee SG, Gilchrest BA. The molecular cloning and expression of two CRABP cDNAs from human skin. Exp Cell Res. 1992; 198:328–36. https://doi.org/10.1016/0014-4827(92)90387-N [PubMed]
- 16. Bellemère G, Stamatas GN, Bruère V, Bertin C, Issachar N, Oddos T. Antiaging action of retinol: from molecular to clinical. Skin Pharmacol Physiol. 2009; 22:200–09. https://doi.org/10.1159/000231525 [PubMed]
- 17. Cho S, Lowe L, Hamilton TA, Fisher GJ, Voorhees JJ, Kang S. Long-term treatment of photoaged human skin with topical retinoic acid improves epidermal cell atypia and thickens the collagen band in papillary dermis. J Am Acad Dermatol. 2005; 53:769–74. https://doi.org/10.1016/j.jaad.2005.06.052 [PubMed]
- 18. Varani J, Warner RL, Gharaee-Kermani M, Phan SH, Kang S, Chung JH, Wang ZQ, Datta SC, Fisher GJ, Voorhees JJ. Vitamin A antagonizes decreased cell growth and elevated collagen-degrading matrix metalloproteinases and stimulates collagen accumulation in naturally aged human skin. J Invest Dermatol. 2000; 114:480–86. https://doi.org/10.1046/j.1523-1747.2000.00902.x [PubMed]
- 19. Hernandez-Perez E, Khawaja HA, Alvarez TY. Oral isotretinoin as part of the treatment of cutaneous aging. Dermatol Surg. 2000; 26:649–52. https://doi.org/10.1046/j.1524-4725.2000.99210.x [PubMed]
- 20. Giguere V, Ong ES, Segui P, Evans RM. Identification of a receptor for the morphogen retinoic acid. Nature. 1987; 330:624–29. https://doi.org/10.1038/330624a0 [PubMed]
- 21. Xiao JH, Durand B, Chambon P, Voorhees JJ. Endogenous retinoic acid receptor (RAR)-retinoid X receptor (RXR) heterodimers are the major functional forms regulating retinoid-responsive elements in adult human keratinocytes. Binding of ligands to RAR only is sufficient for RAR-RXR heterodimers to confer ligand-dependent activation of hRAR beta 2/RARE (DR5). J Biol Chem. 1995; 270:3001–11. https://doi.org/10.1074/jbc.270.7.3001 [PubMed]
- 22. Bushue N, Wan YJ. Retinoid pathway and cancer therapeutics. Adv Drug Deliv Rev. 2010; 62:1285–98. https://doi.org/10.1016/j.addr.2010.07.003 [PubMed]
- 23. Doldo E, Costanza G, Agostinelli S, Tarquini C, Ferlosio A, Arcuri G, Passeri D, Scioli MG, Orlandi A. Vitamin A, cancer treatment and prevention: the new role of cellular retinol binding proteins. BioMed Res Int. 2015; 2015:624627. https://doi.org/10.1155/2015/624627 [PubMed]
- 24. Sanquer S, Gilchrest BA. Characterization of human cellular retinoic acid-binding proteins-I and -II: ligand binding affinities and distribution in skin. Arch Biochem Biophys. 1994; 311:86–94. https://doi.org/10.1006/abbi.1994.1212 [PubMed]
- 25. Sanquer S, Eller MS, Gilchrest BA. Retinoids and state of differentiation modulate CRABP II gene expression in a skin equivalent. J Invest Dermatol. 1993; 100:148–53. https://doi.org/10.1111/1523-1747.ep12462785 [PubMed]
- 26. Fisher GJ, Voorhees JJ. Molecular mechanisms of retinoid actions in skin. FASEB J. 1996; 10:1002–13. https://doi.org/10.1096/fasebj.10.9.8801161 [PubMed]
- 27. Passeri D, Doldo E, Tarquini C, Costanza G, Mazzaglia D, Agostinelli S, Campione E, Di Stefani A, Giunta A, Bianchi L, Orlandi A. Loss of CRABP-II Characterizes Human Skin Poorly Differentiated Squamous Cell Carcinomas and Favors DMBA/TPA-Induced Carcinogenesis. J Invest Dermatol. 2016; 136:1255–66. https://doi.org/10.1016/j.jid.2016.01.039 [PubMed]
- 28. Karlsson T, Virtanen M, Sirsjö A, Rollman O, Vahlquist A, Törmä H. Topical retinoic acid alters the expression of cellular retinoic acid-binding protein-I and cellular retinoic acid-binding protein-II in non-lesional but not lesional psoriatic skin. Exp Dermatol. 2002; 11:143–52. https://doi.org/10.1034/j.1600-0625.2002.110206.x [PubMed]
- 29. Kang S, Duell EA, Fisher GJ, Datta SC, Wang ZQ, Reddy AP, Tavakkol A, Yi JY, Griffiths CE, Elder JT, Voorhees JJ. Application of retinol to human skin in vivo induces epidermal hyperplasia and cellular retinoid binding proteins characteristic of retinoic acid but without measurable retinoic acid levels or irritation. J Invest Dermatol. 1995; 105:549–56. https://doi.org/10.1111/1523-1747.ep12323445 [PubMed]
- 30. Elder JT, Aström A, Pettersson U, Tavakkol A, Griffiths CE, Krust A, Kastner P, Chambon P, Voorhees JJ. Differential regulation of retinoic acid receptors and binding proteins in human skin. J Invest Dermatol. 1992; 98:673–79. https://doi.org/10.1111/1523-1747.ep12499896 [PubMed]
- 31. Shih MY, Kane MA, Zhou P, Yen CL, Streeper RS, Napoli JL, Farese RV
Jr . Retinol Esterification by DGAT1 Is Essential for Retinoid Homeostasis in Murine Skin. J Biol Chem. 2009; 284:4292–99. https://doi.org/10.1074/jbc.M807503200 [PubMed] - 32. Nelson CH, Buttrick BR, Isoherranen N. Therapeutic potential of the inhibition of the retinoic acid hydroxylases CYP26A1 and CYP26B1 by xenobiotics. Curr Top Med Chem. 2013; 13:1402–28. https://doi.org/10.2174/1568026611313120004 [PubMed]
- 33. Terao M, Tani M, Itoi S, Yoshimura T, Hamasaki T, Murota H, Katayama I. 11β-hydroxysteroid dehydrogenase 1 specific inhibitor increased dermal collagen content and promotes fibroblast proliferation. PLoS One. 2014; 9:e93051. https://doi.org/10.1371/journal.pone.0093051 [PubMed]
- 34. Oender K, Trost A, Lanschuetzer C, Laimer M, Emberger M, Breitenbach M, Richter K, Hintner H, Bauer JW. Cytokeratin-related loss of cellular integrity is not a major driving force of human intrinsic skin aging. Mech Ageing Dev. 2008; 129:563–71. https://doi.org/10.1016/j.mad.2008.05.008 [PubMed]
- 35. Salerno S, Morelli S, Giordano F, Gordano A, Bartolo LD. Polymeric membranes modulate human keratinocyte differentiation in specific epidermal layers. Colloids Surf B Biointerfaces. 2016; 146:352–62. https://doi.org/10.1016/j.colsurfb.2016.06.026 [PubMed]
- 36. Velarde MC, Flynn JM, Day NU, Melov S, Campisi J. Mitochondrial oxidative stress caused by Sod2 deficiency promotes cellular senescence and aging phenotypes in the skin. Aging (Albany NY). 2012; 4:3–12. https://doi.org/10.18632/aging.100423 [PubMed]
- 37. Shao Y, He T, Fisher GJ, Voorhees JJ, Quan T. Molecular basis of retinol anti-ageing properties in naturally aged human skin in vivo. Int J Cosmet Sci. 2017; 39:56–65. https://doi.org/10.1111/ics.12348 [PubMed]
- 38. Gunin AG, Kornilova NK, Petrov VV, Vasil’eva OV. Age-related changes in the number and proliferation of fibroblasts in the human skin. Adv Gerontol. 2011; 24:43–47. [PubMed]
- 39. Ghadially R, Brown BE, Sequeira-Martin SM, Feingold KR, Elias PM. The aged epidermal permeability barrier. Structural, functional, and lipid biochemical abnormalities in humans and a senescent murine model. J Clin Invest. 1995; 95:2281–90. https://doi.org/10.1172/JCI117919 [PubMed]
- 40. Barland CO, Zettersten E, Brown BS, Ye J, Elias PM, Ghadially R. Imiquimod-induced interleukin-1 alpha stimulation improves barrier homeostasis in aged murine epidermis. J Invest Dermatol. 2004; 122:330–36. https://doi.org/10.1046/j.0022-202X.2004.22203.x [PubMed]
- 41. Cole MA, Quan T, Voorhees JJ, Fisher GJ. Extracellular matrix regulation of fibroblast function: redefining our perspective on skin aging. J Cell Commun Signal. 2018; 12:35–43. https://doi.org/10.1007/s12079-018-0459-1 [PubMed]
- 42. Fisher GJ, Datta S, Wang Z, Li XY, Quan T, Chung JH, Kang S, Voorhees JJ. c-Jun-dependent inhibition of cutaneous procollagen transcription following ultraviolet irradiation is reversed by all-trans retinoic acid. J Clin Invest. 2000; 106:663–70. https://doi.org/10.1172/JCI9362 [PubMed]
- 43. Bhattacharyya TK, Thomas JR. Histomorphologic changes in aging skin: observations in the CBA mouse model. Arch Facial Plast Surg. 2004; 6:21–25. https://doi.org/10.1001/archfaci.6.1.21 [PubMed]
- 44. Gentile P, Garcovich S, Bielli A, Scioli MG, Orlandi A, Cervelli V. The Effect of Platelet-Rich Plasma in Hair Regrowth: A Randomized Placebo-Controlled Trial. Stem Cells Transl Med. 2015; 4:1317–23. https://doi.org/10.5966/sctm.2015-0107 [PubMed]
- 45. Orlandi A, Francesconi A, Marcellini M, Ferlosio A, Spagnoli LG. Role of ageing and coronary atherosclerosis in the development of cardiac fibrosis in the rabbit. Cardiovasc Res. 2004; 64:544–52. https://doi.org/10.1016/j.cardiores.2004.07.024 [PubMed]
- 46. Tudisco L, Orlandi A, Tarallo V, De Falco S. Hypoxia activates placental growth factor expression in lymphatic endothelial cells. Oncotarget. 2017; 8:32873–83. https://doi.org/10.18632/oncotarget.15861 [PubMed]
- 47. Orlandi A, Ferlosio A, Ciucci A, Francesconi A, Lifschitz-Mercer B, Gabbiani G, Spagnoli LG, Czernobilsky B. Cellular retinol binding protein-1 expression in endometrial hyperplasia and carcinoma: diagnostic and possible therapeutic implications. Mod Pathol 2006; 19:797–803. https://doi.org/10.1038/modpathol.3800586 [PubMed]
- 48. Kim TH, Choi EH, Kang YC, Lee SH, Ahn SK. The effects of topical alpha-hydroxyacids on the normal skin barrier of hairless mice. Br J Dermatol. 2001; 144:267–73. https://doi.org/10.1046/j.1365-2133.2001.04011.x [PubMed]
- 49. Cervelli V, Scioli MG, Gentile P, Doldo E, Bonanno E, Spagnoli LG, Orlandi A. Platelet-rich plasma greatly potentiates insulin-induced adipogenic differentiation of human adipose-derived stem cells through a serine/threonine kinase Akt-dependent mechanism and promotes clinical fat graft maintenance. Stem Cells Transl Med. 2012; 1:206–20. https://doi.org/10.5966/sctm.2011-0052 [PubMed]
- 50. Spitalieri P, Quitadamo MC, Orlandi A, Guerra L, Giardina E, Casavola V, Novelli G, Saltini C, Sangiuolo F. Rescue of murine silica-induced lung injury and fibrosis by human embryonic stem cells. Eur Respir J. 2012; 39:446–57. https://doi.org/10.1183/09031936.00005511 [PubMed]
- 51. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001; 25:402–08. https://doi.org/10.1006/meth.2001.1262 [PubMed]