Introduction
Aging-related changes in the immune system occur at two stages of life. The first begins at a young age in humans and mice, when the thymus begins to involute as the thymic micro-environment deteriorates, followed by reduction in the number of developing thymocytes. Additionally, and the bone marrow [BM] shows changes including an increase in the frequency of hematopoietic stem cells [HSCs] that provide short term re-population of immune cells and a decrease in the frequency of HSCs that provide long-term re-constitution [1, 2]. The second phase of immune system decline takes place in much older humans and mice and correlates with decreased ability to fight infections [3,4], inadequate response to vaccination [5–7] and decreased protection from cancer [8–10]. Despite thymic involution and BM changes, the peripheral immune system in adult mice remains reasonably stable until age-related decline is noted at a much older age. Stability of the immune system in adults is indispensable as immune cells play a critical role in tissue preservation and protection from infections and cancer [11]. However, mechanisms that regulate the maintenance of immune system during adult life remain to be adequately elucidated.
Klotho, a type I single-pass transmembrane protein with beta-glucuronidase activity, has been implicated in influencing the aging process [12]. While, the transmembrane protein is predominantly expressed in the kidney and choroid plexus of the brain, and not in immune cells [13–16], a soluble form of the Klotho peptide is found in blood, urine and cerebrospinal fluid and possesses hormone-like activity that can affect aging of tissues in which Klotho is not expressed [17]. Importantly, the levels of Klotho hormone decline with normal aging in mice and in humans [18], suggesting a role in immune system maintenance. A mutant Klotho allele [kl/kl], which reduces Klotho expression, was isolated in an unbiased mutagenesis scheme using C3HxC57BL/6 mice. The mutant mice displayed traits of premature aging and were short lived [19]. In contrast, overexpression of the gene extended the lifespan of mice, implicating the protein in aging-related functions [20]. Klotho deficiency in C3HxC57BL/6, BALB/c and 129 mice resulted in detrimental phenotypes in the immune system, including severe thymic atrophy and defective B cell lymphopoiesis [19,21,22]. In 129-Klotho-deficient mice, a significant increase in erythropoietin production by the kidney resulted in increased erythropoiesis in the BM and spleen and impaired hematopoiesis [23]. These studies show that Klotho-deficiency can affect the immune system, but the role of Klotho in the maintenance of immune system was difficult to assess in these systems due to dramatically accelerated aging and premature death at a young age.
Expression of kl/kl (Klotho- Klchr5:150,950,607-150,995,817) in C3HxC57BL/6 mice leads to higher levels of minerals and active vitamin D in the serum, which correlates with susceptibility to age-related phenotypes and pre-mature death [12]. Surprisingly, transfer of the kl/kl allele to the pure C57BL/6 genetic background [B6-kl/kl] restored lifespan comparable to C57BL/6 mice [24]. Amelioration of aging-related phenotypes were associated with comparable levels of serum phosphate, calcium and active vitamin D levels in B6-kl/kl and C57BL/6 mice [25]. Remarkably, it was demonstrated that genetic variations [deletions and substitutions] in super enhancer-like regulatory regions at the Cyp24a1 (Cyp24a1chr2:170,480,957-170,499,145) genetic locus in C3H, BALB/c and 129 mice resulted in lower basal levels of Cyp24a1 expression in kidney compared to C57BL/6 mice. These data suggested that higher basal expression of Cyp24a1 in C57BL/6 mice allowed proper metabolism of vitamin D in B6-kl/kl mice and rescued them from detrimental phenotypes that result from high levels of serum vitamin D [25]. Importantly, B6-kl/kl mice provide the opportunity to assess the effects of decreased expression of Klotho on immune cell development and maintenance of the immune system, without the confounding effects of high serum vitamin D and acute premature aging.
In this report, we demonstrate that immune cell development and age-dependent changes in the two primary immune compartments, bone marrow and the thymus, were comparable in age-matched B6-kl/kl and C57BL/6 mice. Remarkably, B6-kl/kl mice showed age-associated thymic involution and changes in the BM at a rate comparable to control C57BL/6 mice. Analysis of peripheral immune organs in adult mice showed that the frequency of immune cells in B6-kl/kl mice was comparable to age-matched C57BL/6 mice. Thus, these data reveal that all major immune cells developed normally and were maintained at similar levels in B6-kl/kl mice despite significantly low levels of Klotho expression. Overall, these data demonstrate a minor role of circulating Klotho in immune system development and maintenance in adult mice.
Results
The decrease in Klotho expression results in lower body weight in the B6-kl/kl mice
Klotho-hypormorphic C57BL/6xC3H-kl/kl mice [19] were backcrossed to C57BL/6J mice for 10 generations (B6-kl/kl). The purity of the genetic B6-kl/kl background was confirmed by typing ten mice for 96 C57BL/6 microsatellite markers using DNA extracted from the tails. All mice were homozygous for at least 94 out of the 96 markers analyzed (background ‘purity’ >98.96%) (Table 1). These data demonstrate that B6-kl/kl mice were syngenic with C57BL/6 mice.
Table 1. Genotyping of 96 microsatellite markers specific for a C57BL/6 background.
| # (Sex) | 1 (F) | 2 (F) | 3 (F) | 4 (F) | 5 (F) | 6 (M) | 7 (M) | 8 (F) | 9 (F) | 10 (M) | (mean ± SEM) |
| non-C57BL/6 markers | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0.1 ± 0.10 |
| C57BL/6 heterozygous makers | 0 | 1 | 1 | 1 | 2 | 0 | 0 | 2 | 1 | 1 | 0.9 ± 0.23 |
| C57BL/6 homozygous makers | 96 | 95 | 95 | 95 | 94 | 95 | 96 | 94 | 95 | 95 | 95 ± 0.21 |
| C57BL/6 background purity (%) | 100.00 | 99.48 | 99.48 | 99.48 | 98.96 | 98.96 | 100.00 | 98.96 | 99.48 | 99.48 | 99.42 ± 0.12 |
In the kidney of mutant C57BL/6xC3H-kl/kl mice the level of kl mRNA expression was shown to be significantly decreased [19]. To confirm that our mice retained this feature on the C57BL/6 background, expression of secreted and membrane kl mRNAs in kidney were analyzed by qPCR (Figure 1A). The data show that B6-kl/kl mice express significantly lower levels of secreted and membrane forms of kl mRNA (Figure 1B and C). The immune system is likely to be most affected by the expression of the soluble form, which was also expressed at significantly lower levels in B6-kl/kl mice (Figure 1B). These data show that the hypomorphic allele [kl/kl] was appropriately transferred to the C57BL/6 background in B6-kl/kl mice.
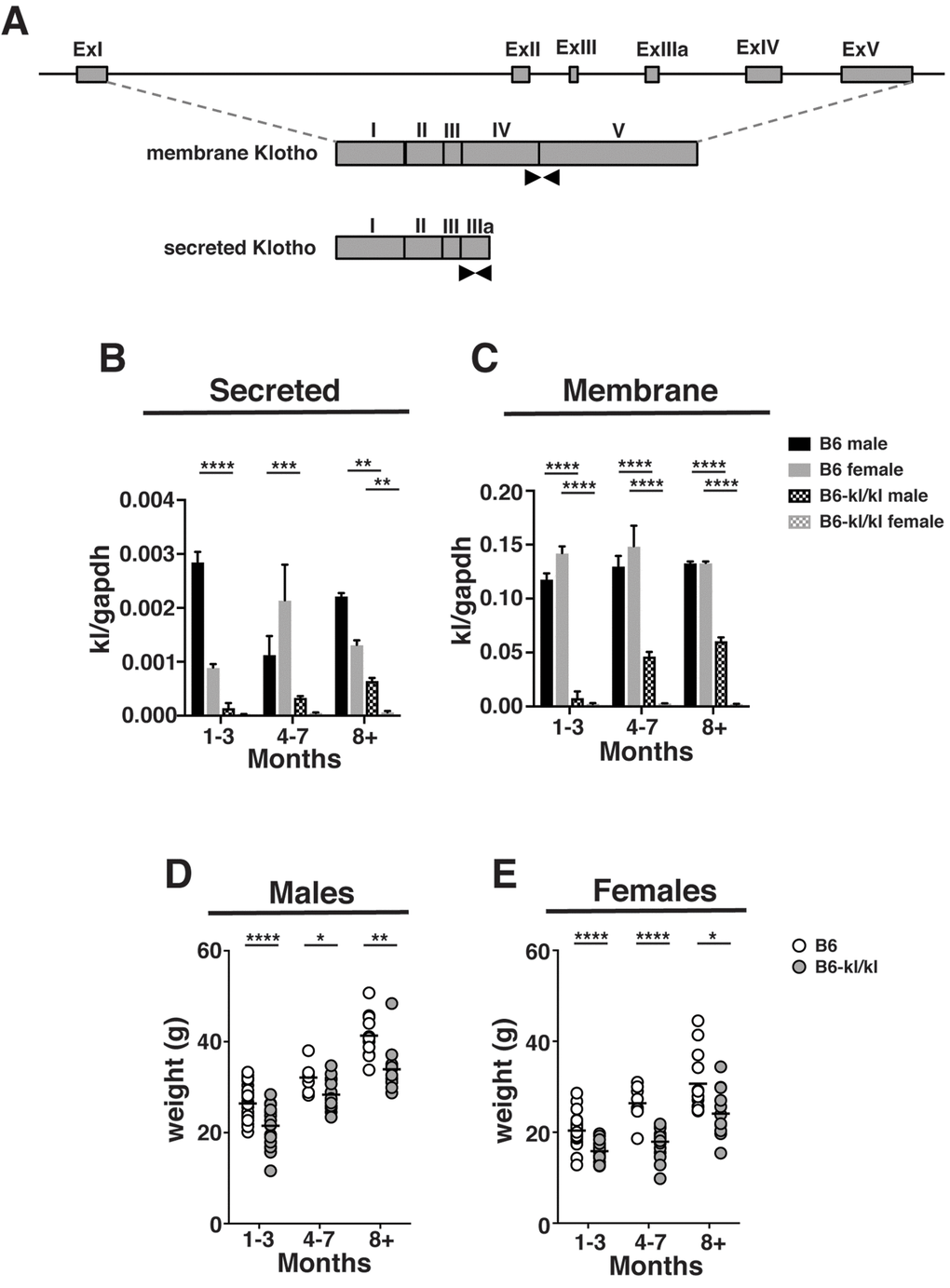
Figure 1. B6-kl/kl mice maintain an overall lower body weight than C57BL/6 as they age. (A) A graphical representation of the kl gene. Black arrows indicate the primer sets used to amplify the secreted and membrane forms by quantitative PCR. (B) Secreted kl mRNA and (C) membrane kl mRNA expression in kidney normalized to gapdh at 1-3 months, 4-7 months, and 8+ months of age (n=2 per group). Bars represent standard error mean. Statistical significance determined by 2way ANOVA and Tukey’s multiple comparison test: ** p ≤ 0.01, *** p ≤ 0.001, and *** p ≤ 0.0001. Weights of C57BL/6 and B6-kl/kl (D) male and (E) female mice at 1-3 months, 4-7 months, and 8+ months of age. For male mice C57BL/6 1-3 months n=34, 4-7 months n=8, and 8+ months n=11 and B6-kl/kl 1-3 months n=21, 4-7 months n=14, and 8+ months n=11. For female mice C57BL/6 1-3 months n=38, 4-7 months n=8, and 8+ months n=13 and B6-kl/kl 1-3months n=17, 4-7 months n=19, and 8+ months n=19. Statistical significance determined by multiple t tests: * p ≤ 0.05, ** p ≤ 0.001, and **** p ≤ 0.0001.
The C57BL/6xC3H-kl/kl mice lived a short lifespan and had reproductive organ defects [19]. In this study, B6-kl/kl mice lived a lifespan that was indistinguishable from littermates that were not mutant, and showed no abnormality in reproductive organs or impaired reproductive capability (data not shown). The C57BL/6xC3H-kl/kl mice were significantly smaller in size [19]. Likewise, both male and female B6-kl/kl mice in this study showed significantly lower body weight (Figure 1D and E) as was previously reported [24]. These results show that the effects of decreased Klotho expression on lifespan and the premature aging-related phenotypes were largely alleviated on a C57BL/6 background.
Frequency of immune cells in the Bone Marrow (BM) of B6-kl/kl mice was comparable to age-matched C57BL/6 mice
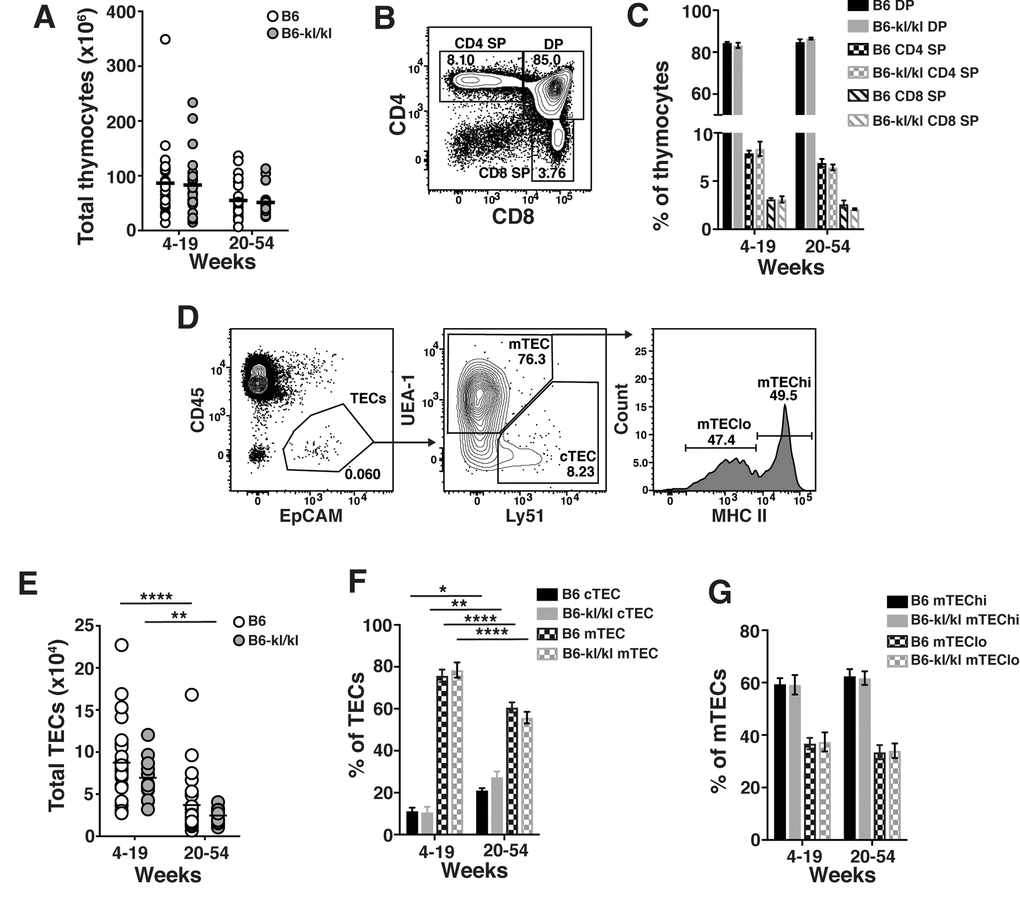
We sought to determine the effect of Klotho on immune cell development and maintenance in adult mice. We divided adult mice into young-adult (young-4-19 weeks) and older-adult (old-20-54 weeks) group. Total number of BM cells recovered from B6-kl/kl mice were significantly reduced in young-adult and older-adult B6-kl/kl mice compared to age-matched C57BL/6 mice (Figure 2A). This reduction in cell number is most likely due to the overall smaller size of the B6-kl/kl mice (Figure 1B and C). As mice age the frequency of multi-potential progenitors (MPP) and short-term HSCs increase [2]. These age-related changes were not affected by decreased Klotho expression as B6-kl/kl mice did not show differences in the frequency of MPPs or HSCs compared to age-matched C57Bl/6 mice (Figure 2B, C and D). Importantly, the comparable age-associated loss of MPPs and gain of HSCs in both C57Bl/6 and B6-kl/kl mice indicate normal maintenance with age in adult mice. Common lymphoid progenitors (CLPs) were also present in similar frequencies in the B6-kl/kl mice compared to the C57Bl/6 mice and showed an analogous and significant loss with age (Figure 2E and F). B6-kl/kl mice showed comparable myeloid cells in the bone marrow compared to C57Bl/6 mice (Figure 2G and H). There was a reduction in the frequency of pre-B cells in the young B6-kl/kl mice, but this did not affect B cell development as B cells, pro B cells and new B cell frequencies were not reduced (Figure 2I and J). Overall, in the B6-kl/kl BM, immune cells developed and were maintained at comparable frequencies to age-matched C57BL/6 mice.
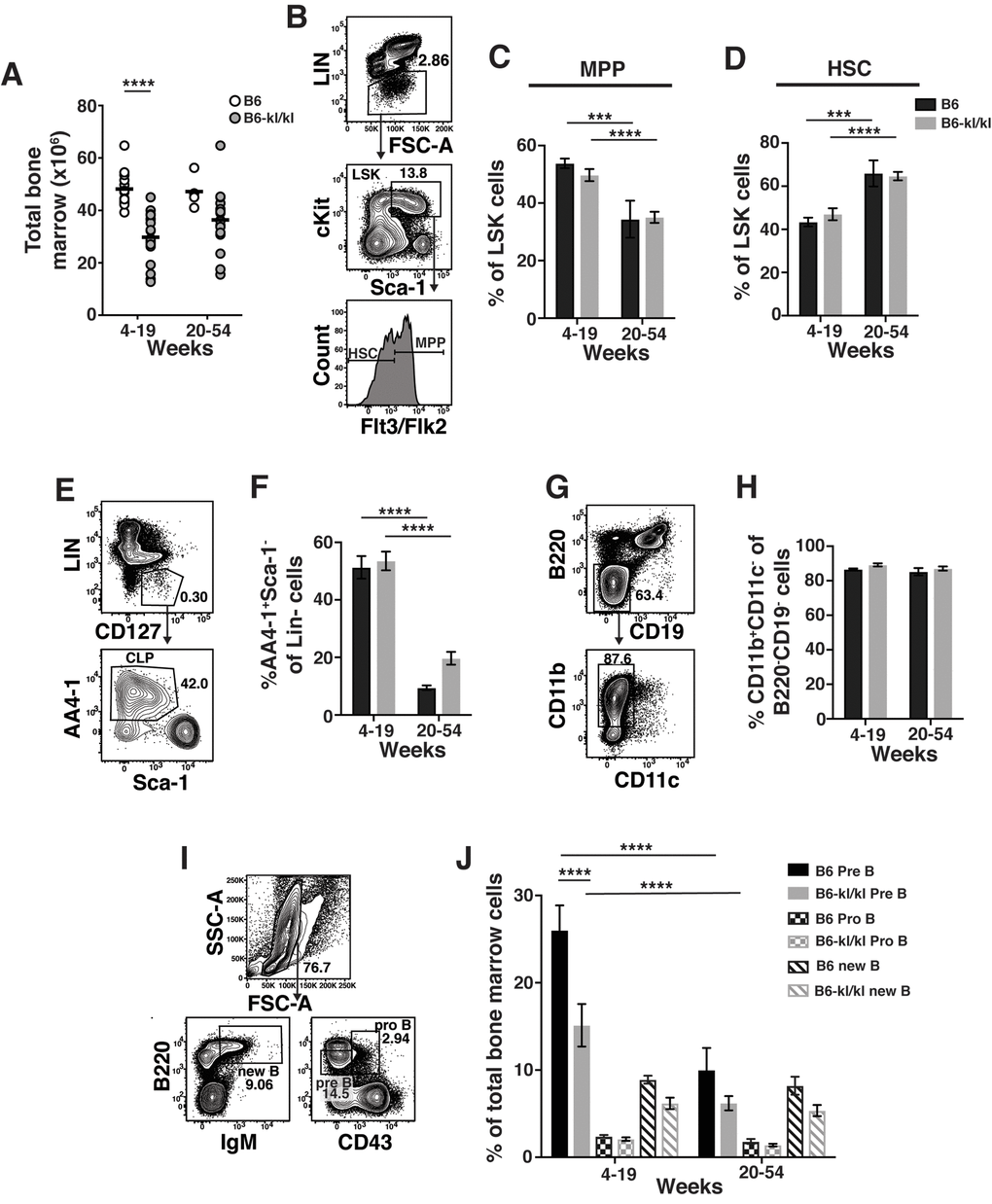
Figure 2. Immune cells in the bone marrow have a similar composition in C57BL/6 and B6-kl/kl mice. (A) Total bone marrow (BM) cells in C57BL/6 and B6-kl/kl at 4-19 weeks (C57BL/6 n=15 and B6-kl/kl n=18) of age or 20+ weeks (C57BL/6 n=4 and B6-kl/kl n=18) from pooled male and female mice. Statistical significance determined by multiple t tests: **** p ≤ 0.0001. (B) Representative flow cytometry plot and (C) frequency of MPP and (D) HSC of the LSK (Lin-cKit+Sca1+) cells. C57BL/6 n=15, 4-19 weeks; n=4, 20+ weeks. B6-kl/kl n=18, 4-19 weeks; n=18, 20+ weeks. Statistical significance determined by 2way ANOVA and Tukey’s multiple comparison test: *** p≤ 0.001 and **** p≤ 0.0001. (E) Representative flow cytometry plot of CLP (Lin-CD127+AA4.1+Sca1low), and (F) frequency of CLP in of Lin-CD127+ cells. C57BL/6 n=15, 4-19 weeks; n=4, 20+ weeks. B6-kl/kl n=18, 4-19 weeks; n=18, 20+ weeks. Statistical significance determined by 2way ANOVA and Tukey’s multiple comparison test: **** p ≤ 0.0001. (G) Representative flow cytometry plot of Myeloid cells (CD11b+CD11c-) and (H) frequency of CD11b+CD11c- cells of B220-CD19- cells. C57BL/6 n=10, 4-19 weeks; n=4, 20+ weeks. B6-kl/kl n=15, 4-19 weeks; n=15, 20+ weeks. (I) Representative flow cytometry plot for Pre B cells (B220lowCD43-), Pro B cells (B220+CD43+) and new B cells (B220+IgM+) and (J) frequency of B cell subsets from BM. C57BL/6 n=15, 4-19 weeks; n=4, 20+ weeks. B6-kl/kl n=18, 4-19 weeks; n=16, 20+ weeks. Statistical significance determined by 2way ANOVA and Tukey’s multiple comparison test: **** p ≤ 0.0001.
Innate and adaptive immune cell maintenance in the spleen of B6-kl/kl mice
The spleens from B6-kl/kl mice were neither visibly abnormal nor hypercellular and demonstrated no enhanced erythropoiesis as noted in Klotho-deficient 129 mice [23] (Figure 4A). In the young B6-kl/kl there was a small reduction in the number of splenocytes compared to the C57Bl/6 mice, which was ameliorated with age and therefore did not represent a defect in maintenance of immune cells (Figure 4A). The overall assessment of the innate immune cells in the spleen included natural killer T cells (NKT), neutrophils (Neut), natural killer cells (NK), dendritic cells (DC), and macrophages (Mac) (Figure 4B). The frequency of these cell populations was comparable in the young-adult and older-adult B6-kl/kl mice compared to C57BL/6 mice (Figure 4C-G). There was a small but significant reduction in the frequency of B cells in the spleens of older-adult B6-kl/kl mice compared to C57BL/6 mice (Figure 5A and B), but no differences in frequency of IgD+ or IgM+ B cells (Figure 5C). B6-kl/kl had comparable frequency of naïve (Tn), effector-phenotype (Teff), and memory-phenotype (Tmem) T cells in both CD4 and CD8 T cell compartments compared to C57BL/6 mice (Figure 5D-G). Together these data demonstrate that adaptive and innate immune systems remained unaffected by decreased Klotho expression in the spleen of B6-kl/kl mice.
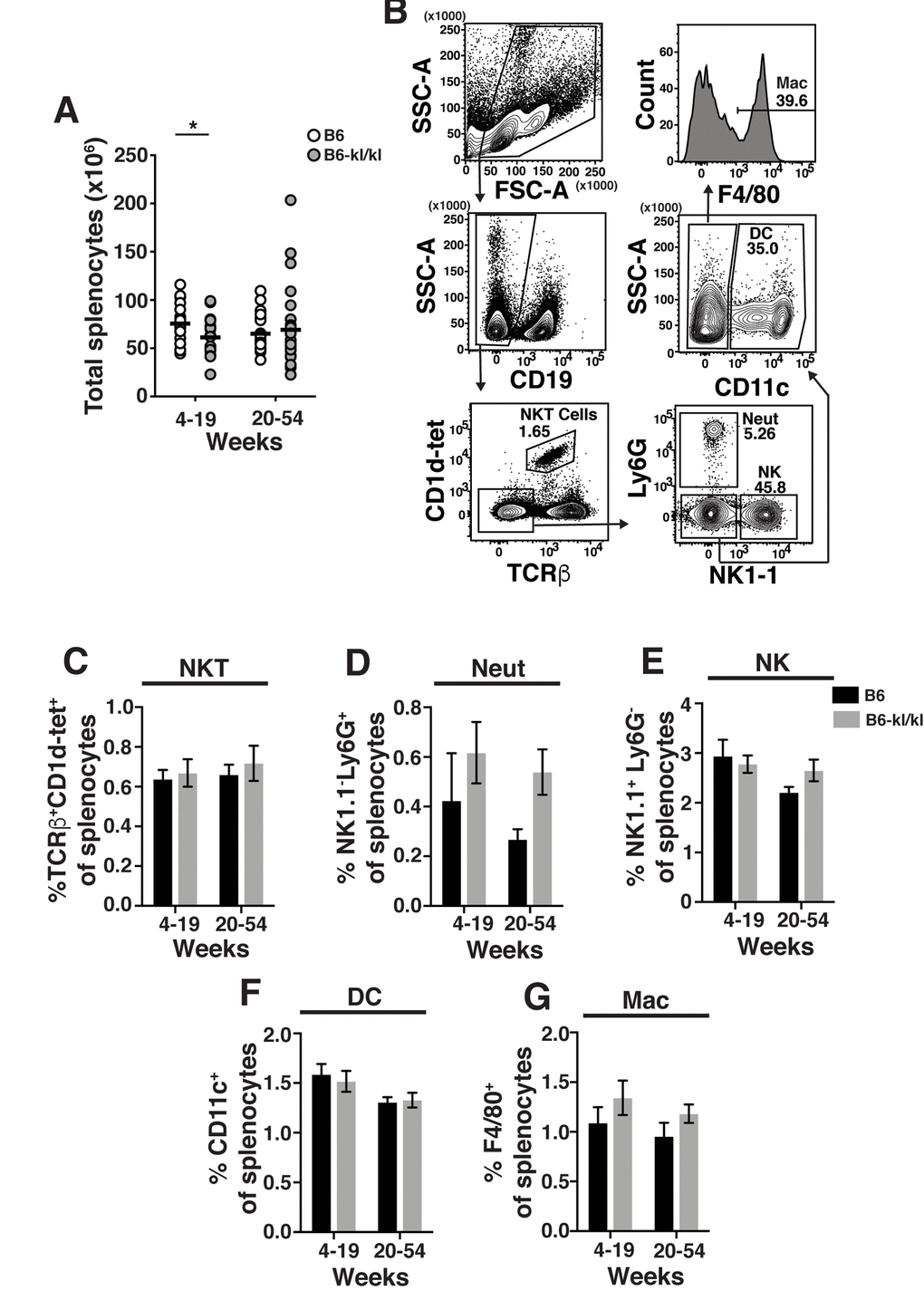
Figure 4. Innate immune cell composition in the spleen is similar in C57BL/6 and B6-kl/kl mice. (A) Total splenocytes in C57BL/6 and B6-kl/kl mice at 4-19 weeks (C57BL/6 n=25 and B6-kl/kl n=19) and 20+ weeks (C57BL/6 n=25 and B6-kl/kl n=27) of age from pooled male and female mice. Statistical significance determined by multiple t tests: * p ≤ 0.05. (B) Representative flow cytometry plots of NKT cells (CD19-CD1d+TCRβ+), Neutrophils (Neut) (CD19-CD1d-TCRβ--NK1.1-Ly6G+), NK cells (CD19-CD1d-TCRβ-NK1.1+Ly6G-), dendritic cells (DC) (CD19-CD1d-TCRβ-NK1.1-Ly6G-CD11c+), and macrophages (Mac) (CD19-CD1d-TCRβ-NK1.1-Ly6G-CD11c-F4/80+). Frequency of (C) NKT, (D) neutrophils, (E) NK cells, (F) dendritic cells, and (G) macrophages. C57BL/6 n=13, 4-19 weeks; n=19, 20+ weeks. B6-kl/kl n=11, 4-19 weeks; n=16, 20+ weeks. Bars represent the standard error mean.
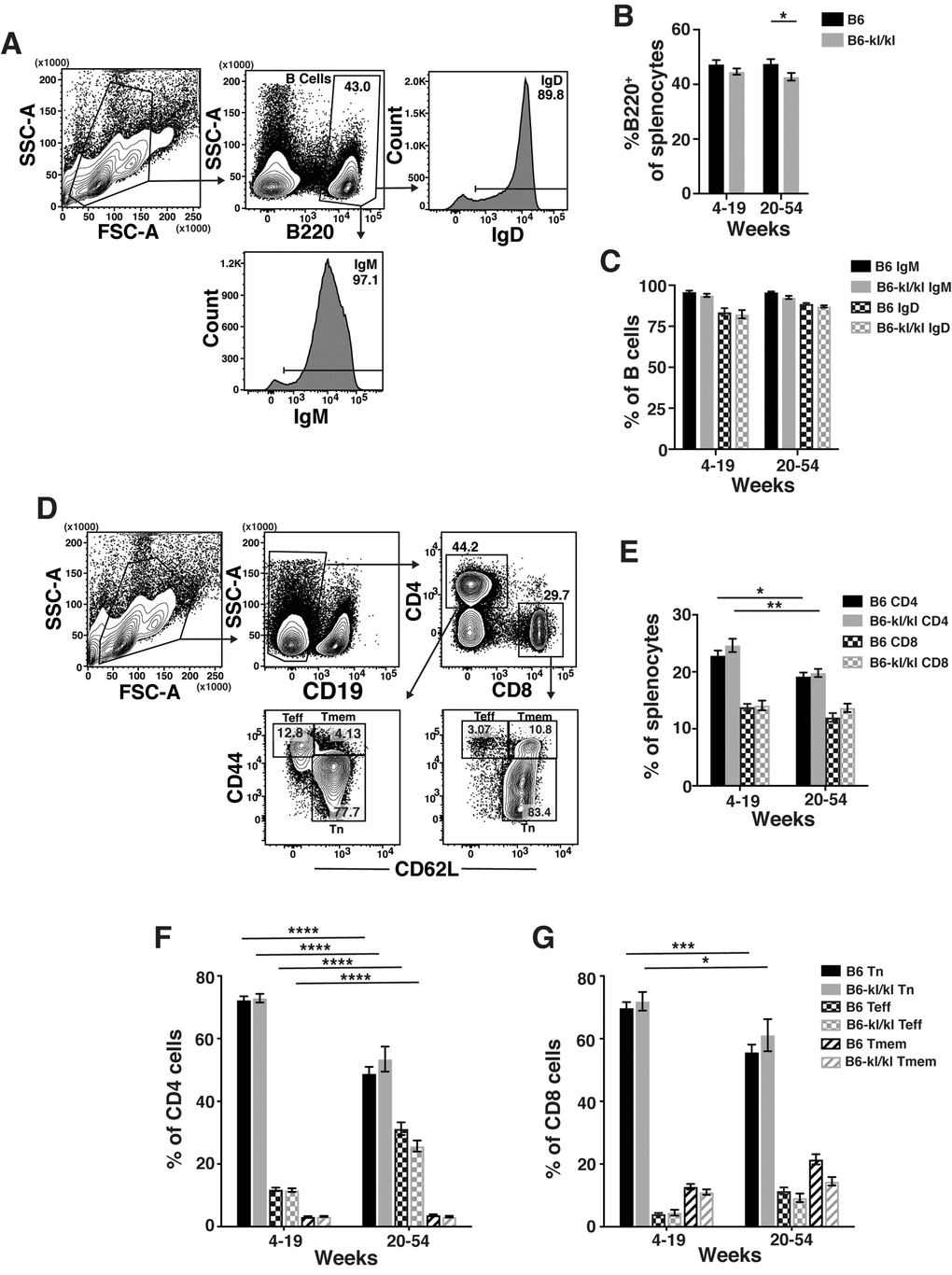
Figure 5. Adaptive immune cell composition in the spleen is similar in C57BL/6 and B6-kl/kl mice. (A) Representative flow cytometry plots of splenic B cells. Frequency of (B) total B cells and (C) IgM+ or IgD+ B cells in the spleen of C57BL/6 and B6-kl/kl mice at 4-19 weeks (C57BL/6 n=18 and B6-kl/kl n=18) and 20+ weeks (C57BL/6 n=16 and B6-kl/kl n=23) of age from pooled male and female mice. Bars represent the standard error mean. Statistical significance determined by 2way ANOVA and Tukey’s multiple comparison test: * p ≤ 0.05. (D) Representative flow cytometry plots of splenic naïve (Tn; CD62L+CD44lo), effector-phenotype (Teff; CD44hiCD62lo), and memory-phenotype (Tmem; CD44hiCD62+) CD4 T cells (CD19-CD4+) and CD8 T cells (CD19-CD8+). (E) Frequency of CD4 and CD8 T cells. C57BL/6 n=24, 4-19 weeks; n=25, 20+ weeks. B6-kl/kl n=19, 4-19 weeks; n=19, 20+ weeks. Statistical significance determined by 2way ANOVA and Tukey’s multiple comparison test: * p ≤ 0.05 and ** p ≤ 0.001. Bars represent the standard error mean. Frequency of naïve, effector-phenotype, and memory-phenotype of (F) CD4 T cells and (G) CD8 T cells. C57BL/6 n=24, 4-19 weeks; n=25, 20+ weeks. B6-kl/kl n=19, 4-19 weeks; n=19, 20+ weeks. Statistical significance determined by 2way ANOVA and Tukey’s multiple comparison test: *** p ≤ 0.001 and **** p ≤ 0.0001. Bars represent the standard error mean.
Discussion
In this report we demonstrate that the hormone Klotho does not control the development and maintenance of the immune system in adult mice. Specifically, kl/kl allele on a pure C57BL/6J background, after ten rounds of breeding with C57BL/6J mice, (B6-kl/kl) showed decreased expression of Klotho, but no age-related phenotypes or pre-mature death. Mutant animals showed no defects in fertility or lifespan but were smaller than age-matched C57BL/6 mice. Furthermore, the data overall demonstrate that in the absence of systemic stress from increased serum vitamin D [25], decreased expression of Klotho alone was not detrimental to immune cell development or maintenance of the immune system.
Aging-related effects of declining Klotho expression have been extensively documented. Since the identification of Klotho [19], scientists in several laboratories have defined alterations in mineral and vitamin D metabolic pathways through the interaction with FGF-23 that result in deleterious increase in serum phosphate, calcium and active vitamin D [27–30]. Accordingly, dietary and genetic manipulations that lowered serum levels of minerals and active vitamin D correlated with abrogation of aging-related phenotypes [31,32]. With respect to the regulation of serum vitamin D, recent analysis showed that higher basal expression of Cyp24a1 in C57BL/6 genetic background normalized active vitamin D levels in the serum of B6-kl/kl mice [25]. In the absence of increased vitamin-dependent systemic stress, B6-kl/kl mice showed no overt signs of distress and lived a life-span that was generally comparable to control C57BL/6 mice.
kl/kl allele on susceptible strains [C3H, BALB/c and 129], results in several acute effects on the immune system. The thymus is essentially absent in C3HxC57BL/6-kl/kl mice within a few weeks of birth [19] and deficits in B cell lymphopoieisis are noted after 2-weeks of age [22], when the detrimental effects of increased vitamin D levels in serum are the highest [33]. This suggests that the decrease in thymus size and B cell phenotype could be secondary to high vitamin D levels in the serum. Germline deletion of Klotho [on 129 genetic background] results in induction of erythropoietin (Epo) production in the kidney, which in turn induces abnormal generation of erythrocytes in the BM and subsequently in the spleen [23]. These phenotypes were not observed in B6-kl/kl mice. Unlike in the recently reported 129xC57BL/6-kl/kl mice [21], TEC populations and T cell development in B6-kl/kl mice thymus were also unaffected. These observations support the notion that the effects of Klotho-deficiency on the immune system could be attributed to systemic stress resulting from altered metabolic events, especially serum vitamin D levels. The observation that thymic involution and changes in the BM proceeded comparably in young-adult B6 and B6-kl/kl mice, taken with comparable presence of immune cells in the peripheral organs in young-adult and older-adult B6 and B6-kl/kl mice, suggest that immune system development and maintenance was normal in the absence of Klotho expression.
Materials and Methods
Mice
Klotho-hypormorphic kl/kl mice on C3H/Jx C57BL/6J mixed background were provided by Dr. Makoto Kuro-o [19] and backcrossed for 10 generations to C57BL/6J mice obtained from The Jackson Laboratory to generate B6-kl/kl mice. B6-kl/kl mice were screened by conventional PCR immediately after weaning using the primer forward: 5’-tggagattggaagtggac-3’ together with the primers reverse: 5’-caaggaccagttcatcatcg-3’ and reverse: 5’-ttaaggactcctgcatctgc-3’ for the Klotho hypomorphic and for the WT genotype, respectively. All mice were bred and maintained in an animal facility at the National Institute of Aging (NIA) in a 12-hour light-dark cycle and fed ad libitum with a diet containing 0.7% phosphorus (0.4% non-phytate phosphorus). The studies were carried out in accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals (NRC, 2010). The protocol was approved by the Animal Care and Use Committee of the NIA Intramural Research Program, NIH.
C57BL/6 background genotyping for B6-kl/kl mice
Tail samples from B6-kl/kl mice were sent to the Animal Molecular Diagnostics Laboratory at Frederick National Laboratory for Cancer Research to verify congenicity with C57BL/6 strain. Briefly, 1cm tails from 10 mutant mice were digested and DNA extracted using DNeasy® Blood & Tissue Kit (Qiagen, Venlo, Netherlands). DNA samples were then quantified using a nanodrop (BMG Labtech, Offenberg, Germany) and screened for 96 C57BL/6 microsatellite markers distributed over 19 chromosomes using an ABI 3130 Genetic Analyzer.
Tissue collection
Thymus, spleen and bone marrow were harvested and processed into single cell suspensions for flow cytometry analysis by following standard protocols. Thymic epithelial cells were obtained by gently teasing thymii to release the thymocytes and subsequently digested with 5mg/mL Liberase TH (Roche, Basel, Switerland) and 50mg/mL DNase I (Roche) at 37 °C, as previously described [34,35]. Bone marrow cells were harvested from the femur and tibia of the mice and processed accordingly to standard protocols.
Flow cytometry analysis
Cells were incubated with Fc Block (CD16/32, BD Biosciences, San Jose, CA), stained with antibodies and then fixed with 2% paraformaldehyde. Samples were acquired on the FACS Canto II (BD Biosciences) and analyzed using FlowJo (TreeStar, Ashland, OR). Dead cells were excluded using the eBioscience Fixable Viability Dye eFluor® 506 (ThermoFisher Scientific, Waltham, MA). The following antibodies conjugated to biotin, FITC, PE, PerCP-Cy5.5, PE-Cy5, PE-Cy7, APC, APC-Cy7 or Pacific Blue were used (ThermoFisher Scientific, BD Biosciences, BioLegend, San Diego, CA): anti-CD4 (GK1.5), anti-CD8 (53-6.7), anti-TCR-β (H57-597), anti-CD19 (6D5), anti-CD44 (IM7), anti-B220 (RA3-6B2), anti-IgM (RMM-1), anti-IgD (11-26c.2a), anti-NK1.1 (PK136), anti-F4/80 (BM8), anti-Ly6G (1A8), anti-CD11c (HL3), anti-CD62L (MEL-14), anti-EpCAM (G8.8), anti-CD45.2 (104), anti-Ly51 (BP-1), anti-MHCII (M5/114.15.2), anti-UEA-1 (B-1065, Vector Labs), anti-Sca-1 (E13-161.7), anti-cKit (2B8), anti-Flt3/Flk2 (A2F10), anti-CD127 (A7R34), anti-AA4.1 (AA4.1), anti-CD43 (S7), anti-CD11b (M1/70), and Streptavidin. The LIN cocktail contained anti-NK1.1, anti-CD11b, anti-GR1 (RB6-8C5), anti-TER119 (TER-119), anti-CD3e (145-2C11), anti-CD19, and anti-B220. APC- conjugated mouse CD1d tetramers loaded with glycolipid PBS-57 (CD1d-tet) and an unloaded tetramer comprised of only the glycolipid PBS57 were obtained from the tetramer facility of the US National Institutes of Health.
Gene expression analysis
Kidneys from C57BL/6 and B6-kl/kl mice were collected for RNA extraction and qPCR analysis. Briefly, kidneys were disrupted and homogenized using a TissueRuptor II, and RNA was extracted from lysates using the RNeasy mini kit (Qiagen). DNA was eliminated from the samples by incubating with DNase (Qiagen). First strand cDNA synthesis was performed by using 1μg of total RNA together with oligo(dT)12–18 and the Invitrogen SuperScript II Reverse Transcriptase (Thermo Fisher Scientific), accordingly to the manufacturer's instructions. Quantification of klotho mRNA expression was conducted using real-time qPCR performed on an Applied Biosystems ViiA™ 7 Real-Time PCR System (Thermo Fisher Scientific). Primers were designed to amplify specific amplicons of the membrane kl (F:5’-ggctctgaaagcctacgtgttg-3’; R:5’-gggagctgagcgatcactaagt-3’), secreted kl (F:5’- tgctggctttcctctaggtcat-3’; R:5’-ttaggcgttctgatgctgtca-3’) and gapdh (F:5’-gtcgtggagtctactggtgtc; R:5’cagaaggggcggagatgatg-3’) genes. Each cDNA sample was diluted 5-fold and then 5μL of dilutions added to 5pmol of each primer and SYBR Green Master (Roche). The cycling parameters were: 10 min at 95 °C, followed by 40 cycles of 15 s at 95 °C and 1 min at 60 °C. Quantification of gene expression was performed by the E^− ΔCt method using gapdh as the normalizer gene (where E stands for primer amplification efficiency). Each sample was quantified in triplicate and primer amplification efficiencies were calculated and validated with the standard curves obtained through the amplification of cDNA serial dilution.
Statistical analysis
Data is presented as mean ± standard error mean. Significance was determined using 2way ANOVA, Tukey’s multiple comparison tests, and multiple t-tests with GraphPad Prism 6.0 (GraphPad, San Diego, CA) software.
Acknowledgements
We thank NIA animal facility and genotyping facility for animal husbandry and genotyping, the Tetramer Facility of the US National Institutes of Health for providing PE- and APC-conjugated mouse CD1d tetramers loaded with glycolipid PBS-57 and the Animal Molecular Diagnostics Laboratory from NCI for the C57BL/6 background genotyping. We thank Dr. Kuro-o for providing C3HxC57BL/6-kl/kl mice and for comments on the work.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
This research was supported by the Intramural Research Program of the National Institute on Aging at the National Institutes of Health.
References
- 1. Nikolich-Žugich J. The twilight of immunity: emerging concepts in aging of the immune system. Nat Immunol. 2018; 19:10–19. https://doi.org/10.1038/s41590-017-0006-x [PubMed]
- 2. Säwen P, Eldeeb M, Erlandsson E, Kristiansen TA, Laterza C, Kokaia Z, Karlsson G, Yuan J, Soneji S, Mandal PK, Rossi DJ, Bryder D. Murine HSCs contribute actively to native hematopoiesis but with reduced differentiation capacity upon aging. eLife. 2018; 7:e41258. https://doi.org/10.7554/eLife.41258 [PubMed]
- 3. Yager EJ, Ahmed M, Lanzer K, Randall TD, Woodland DL, Blackman MA. Age-associated decline in T cell repertoire diversity leads to holes in the repertoire and impaired immunity to influenza virus. J Exp Med. 2008; 205:711–23. https://doi.org/10.1084/jem.20071140 [PubMed]
- 4. Nikolich-Žugich J. Ageing and life-long maintenance of T-cell subsets in the face of latent persistent infections. Nat Rev Immunol. 2008; 8:512–22. https://doi.org/10.1038/nri2318 [PubMed]
- 5. Haynes L, Swain SL. Why aging T cells fail: implications for vaccination. Immunity. 2006; 24:663–66. https://doi.org/10.1016/j.immuni.2006.06.003 [PubMed]
- 6. Aspinall R, Del Giudice G, Effros RB, Grubeck-Loebenstein B, Sambhara S. Challenges for vaccination in the elderly. Immun Ageing. 2007; 4:9. https://doi.org/10.1186/1742-4933-4-9 [PubMed]
- 7. Čičin-Šain L, Smyk-Pearson S, Currier N, Byrd L, Koudelka C, Robinson T, Swarbrick G, Tackitt S, Legasse A, Fischer M, Nikolich-Žugich D, Park B, Hobbs T, et al. Loss of naive T cells and repertoire constriction predict poor response to vaccination in old primates. J Immunol. 2010; 184:6739–45. https://doi.org/10.4049/jimmunol.0904193 [PubMed]
- 8. Prelog M. Aging of the immune system: a risk factor for autoimmunity? Autoimmun Rev. 2006; 5:136–39. https://doi.org/10.1016/j.autrev.2005.09.008 [PubMed]
- 9. Fulop T, Kotb R, Fortin CF, Pawelec G, de Angelis F, Larbi A. Potential role of immunosenescence in cancer development. Ann N Y Acad Sci. 2010; 1197:158–65. https://doi.org/10.1111/j.1749-6632.2009.05370.x [PubMed]
- 10. Foster AD, Sivarapatna A, Gress RE. The aging immune system and its relationship with cancer. Aging Health. 2011; 7:707–18. https://doi.org/10.2217/ahe.11.56 [PubMed]
- 11. Rankin LC, Artis D. Beyond Host Defense: Emerging Functions of the Immune System in Regulating Complex Tissue Physiology. Cell. 2018; 173:554–67. https://doi.org/10.1016/j.cell.2018.03.013 [PubMed]
- 12. Tohyama O, Imura A, Iwano A, Freund JN, Henrissat B, Fujimori T, Nabeshima Y. Klotho is a novel β-glucuronidase capable of hydrolyzing steroid β-glucuronides. J Biol Chem. 2004; 279:9777–84. https://doi.org/10.1074/jbc.M312392200 [PubMed]
- 13. Li SA, Watanabe M, Yamada H, Nagai A, Kinuta M, Takei K. Immunohistochemical localization of Klotho protein in brain, kidney, and reproductive organs of mice. Cell Struct Funct. 2004; 29:91–99. https://doi.org/10.1247/csf.29.91 [PubMed]
- 14. Hu MC, Shi M, Zhang J, Pastor J, Nakatani T, Lanske B, Razzaque MS, Rosenblatt KP, Baum MG, Kuro-o M, Moe OW. Klotho: a novel phosphaturic substance acting as an autocrine enzyme in the renal proximal tubule. FASEB J. 2010; 24:3438–50. https://doi.org/10.1096/fj.10-154765 [PubMed]
- 15. German DC, Khobahy I, Pastor J, Kuro-O M, Liu X. Nuclear localization of Klotho in brain: an anti-aging protein. Neurobiol Aging. 2012; 33:1483.e25–30. https://doi.org/10.1016/j.neurobiolaging.2011.12.018 [PubMed]
- 16. Lim K, Groen A, Molostvov G, Lu T, Lilley KS, Snead D, James S, Wilkinson IB, Ting S, Hsiao LL, Hiemstra TF, Zehnder D. α-Klotho Expression in Human Tissues. J Clin Endocrinol Metab. 2015; 100:E1308–18. https://doi.org/10.1210/jc.2015-1800 [PubMed]
- 17. Dalton GD, Xie J, An SW, Huang CL. New Insights into the Mechanism of Action of Soluble Klotho. Front Endocrinol (Lausanne). 2017; 8:323. https://doi.org/10.3389/fendo.2017.00323 [PubMed]
- 18. Arking DE, Krebsova A, Macek M
Sr , Macek MJr , Arking A, Mian IS, Fried L, Hamosh A, Dey S, McIntosh I, Dietz HC. Association of human aging with a functional variant of klotho. Proc Natl Acad Sci USA. 2002; 99:856–61. https://doi.org/10.1073/pnas.022484299 [PubMed] - 19. Kuro-o M, Matsumura Y, Aizawa H, Kawaguchi H, Suga T, Utsugi T, Ohyama Y, Kurabayashi M, Kaname T, Kume E, Iwasaki H, Iida A, Shiraki-Iida T, et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature. 1997; 390:45–51. https://doi.org/10.1038/36285 [PubMed]
- 20. Kurosu H, Yamamoto M, Clark JD, Pastor JV, Nandi A, Gurnani P, McGuinness OP, Chikuda H, Yamaguchi M, Kawaguchi H, Shimomura I, Takayama Y, Herz J, et al. Suppression of aging in mice by the hormone Klotho. Science. 2005; 309:1829–33. https://doi.org/10.1126/science.1112766 [PubMed]
- 21. Xing Y, Smith MJ, Goetz CA, McElmurry RT, Parker SL, Min D, Hollander GA, Weinberg KI, Tolar J, Stefanski HE, Blazar BR. Thymic Epithelial Cell Support of Thymopoiesis Does Not Require Klotho. J Immunol. 2018; 201:1829–33. https://doi.org/10.4049/jimmunol.1800670 [PubMed]
- 22. Okada S, Yoshida T, Hong Z, Ishii G, Hatano M, Kuro-O M, Nabeshima Y, Nabeshima Y, Tokuhisa T. Impairment of B lymphopoiesis in precocious aging (klotho) mice. Int Immunol. 2000; 12:861–71. https://doi.org/10.1093/intimm/12.6.861 [PubMed]
- 23. Vadakke Madathil S, Coe LM, Casu C, Sitara D. Klotho deficiency disrupts hematopoietic stem cell development and erythropoiesis. Am J Pathol. 2014; 184:827–41. https://doi.org/10.1016/j.ajpath.2013.11.016 [PubMed]
- 24. Phelps M, Pettan-Brewer C, Ladiges W, Yablonka-Reuveni Z. Decline in muscle strength and running endurance in klotho deficient C57BL/6 mice. Biogerontology. 2013; 14:729–39. https://doi.org/10.1007/s10522-013-9447-2 [PubMed]
- 25. Singh A, Verma A, Sallin MA, Lang F, Sen R, Sen JM. Noncoding variations in Cyp24a1 gene are associated with Klotho-mediated aging phenotypes in different strains of mice. Aging Cell. 2019; 18:e12949. https://doi.org/10.1111/acel.12949 [PubMed]
- 26. Palmer DB. The effect of age on thymic function. Front Immunol. 2013; 4:316. https://doi.org/10.3389/fimmu.2013.00316 [PubMed]
- 27. Kurosu H, Ogawa Y, Miyoshi M, Yamamoto M, Nandi A, Rosenblatt KP, Baum MG, Schiavi S, Hu MC, Moe OW, Kuro-o M. Regulation of fibroblast growth factor-23 signaling by klotho. J Biol Chem. 2006; 281:6120–23. https://doi.org/10.1074/jbc.C500457200 [PubMed]
- 28. Urakawa I, Yamazaki Y, Shimada T, Iijima K, Hasegawa H, Okawa K, Fujita T, Fukumoto S, Yamashita T. Klotho converts canonical FGF receptor into a specific receptor for FGF23. Nature. 2006; 444:770–74. https://doi.org/10.1038/nature05315 [PubMed]
- 29. Imura A, Tsuji Y, Murata M, Maeda R, Kubota K, Iwano A, Obuse C, Togashi K, Tominaga M, Kita N, Tomiyama K, Iijima J, Nabeshima Y, et al. α-Klotho as a regulator of calcium homeostasis. Science. 2007; 316:1615–18. https://doi.org/10.1126/science.1135901 [PubMed]
- 30. Nakatani T, Sarraj B, Ohnishi M, Densmore MJ, Taguchi T, Goetz R, Mohammadi M, Lanske B, Razzaque MS. In vivo genetic evidence for klotho-dependent, fibroblast growth factor 23 (Fgf23) -mediated regulation of systemic phosphate homeostasis. FASEB J. 2009; 23:433–41. https://doi.org/10.1096/fj.08-114397 [PubMed]
- 31. Ohnishi M, Nakatani T, Lanske B, Razzaque MS. Reversal of mineral ion homeostasis and soft-tissue calcification of klotho knockout mice by deletion of vitamin D 1α-hydroxylase. Kidney Int. 2009; 75:1166–72. https://doi.org/10.1038/ki.2009.24 [PubMed]
- 32. Tsujikawa H, Kurotaki Y, Fujimori T, Fukuda K, Nabeshima Y. Klotho, a gene related to a syndrome resembling human premature aging, functions in a negative regulatory circuit of vitamin D endocrine system. Mol Endocrinol. 2003; 17:2393–403. https://doi.org/10.1210/me.2003-0048 [PubMed]
- 33. Yoshida T, Fujimori T, Nabeshima Y. Mediation of unusually high concentrations of 1,25-dihydroxyvitamin D in homozygous klotho mutant mice by increased expression of renal 1α-hydroxylase gene. Endocrinology. 2002; 143:683–89. https://doi.org/10.1210/endo.143.2.8657 [PubMed]
- 34. Gray DH, Chidgey AP, Boyd RL. Analysis of thymic stromal cell populations using flow cytometry. J Immunol Methods. 2002; 260:15–28. https://doi.org/10.1016/S0022-1759(01)00493-8 [PubMed]
- 35. Hikosaka Y, Nitta T, Ohigashi I, Yano K, Ishimaru N, Hayashi Y, Matsumoto M, Matsuo K, Penninger JM, Takayanagi H, Yokota Y, Yamada H, Yoshikai Y, et al. The cytokine RANKL produced by positively selected thymocytes fosters medullary thymic epithelial cells that express autoimmune regulator. Immunity. 2008; 29:438–50. https://doi.org/10.1016/j.immuni.2008.06.018 [PubMed]