Introduction
The lymphatic vascular system functions to regulate tissue fluid transport and facilitate macromolecular absorption [1]. Tissue fluid is collected from the interstitial space by lymphatic capillaries and then transported through collector lymphatic vessels back into the blood stream [1, 2]. The recirculation of fluid and cells through extensive lymph transport is required for the maintenance of homeostasis [3, 4]. Lymphatic vessels are also key routes for the trafficking of immune cells from tissues to lymph nodes during immune responses [5].
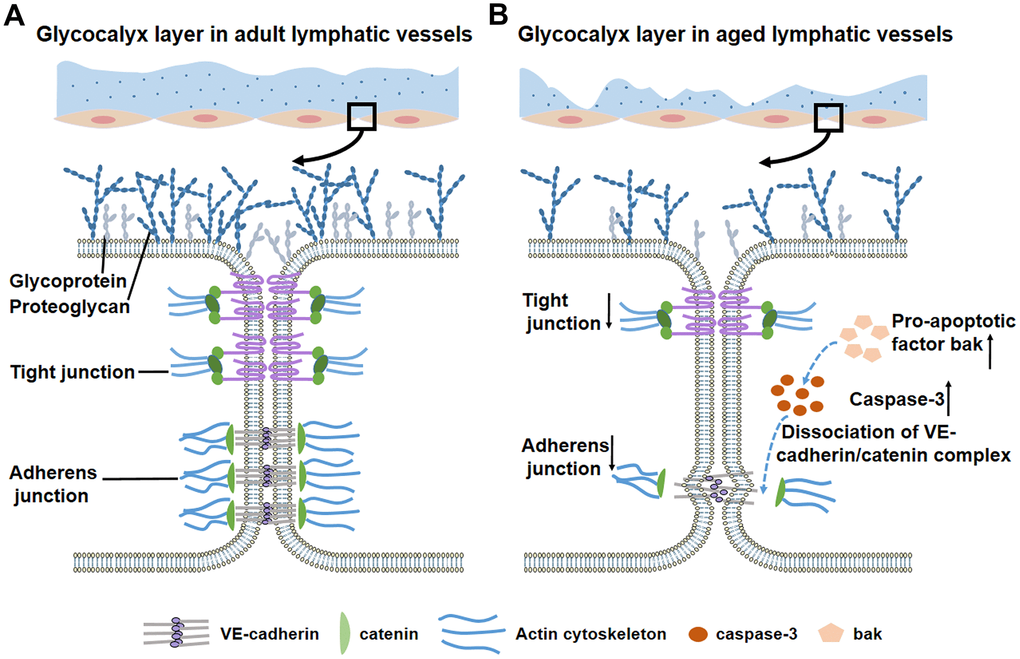
The aging process induces changes in structure and function of lymphatic networks [6]. Lymphatic-related diseases are prevalent in elderly, such as lymphedema [7]. In 1960s, the specific “varicose bulges” in muscular lymphatic vessels was observed and this bulges were increased with age [8, 9]. Muscle cell atrophy, elastic elements destruction, and aneurysm-like formations were also found in aged lymphatic vessels [10–12]. Aging associated alterations in lymphatic contractility decrease pump efficiency which result in excessive retention of tissue fluid within interstitial spaces [13, 14]. Reduced responsiveness to inflammatory stimuli in aged lymphatic vessels decreases the normal capacity to react against foreign organisms [15]. The occurrence of high permeability is caused by the loss of glycocalyx and the dysfunction of junctional proteins [6, 16, 17]. In addition, increased caspase-3 activity, the dissociation of the VE-cadherin/catenin complex and the low expression of actin cytoskeleton that occur in aged blood vessels may also be seen in aged lymphatic vessels [16, 18]. Knowledge of the regulatory mechanisms underlying in these disorders is critical to our understanding of the aging-related diseases of lymphatic vessels.
Structure of lymphatic vessels and their functions in lymph transport
The initial lymphatic vessels are dispersed in the interstitial space of parenchymal organs [6, 19]. These lymphatic capillaries are composed solely of a layer of lymphatic endothelial cells that are directly anchored to the extracellular matrix through filaments [2, 20–22]. The distinctive oak leaf-shaped endothelial cells of initial lymphatics are loosely apposed with overlapping borders and linked with each other by discontinuous, button-like junctions [4, 23]. Regions between buttons are open to allow the entry of fluid and cells without repetitive formation and dissolution of intercellular junctions [4]. These specific structures may function as lymphatic primary valves that prevent the tissue fluid taken up by lymphatic capillaries to be released back into the interstitial space [24] (Figure 1B).
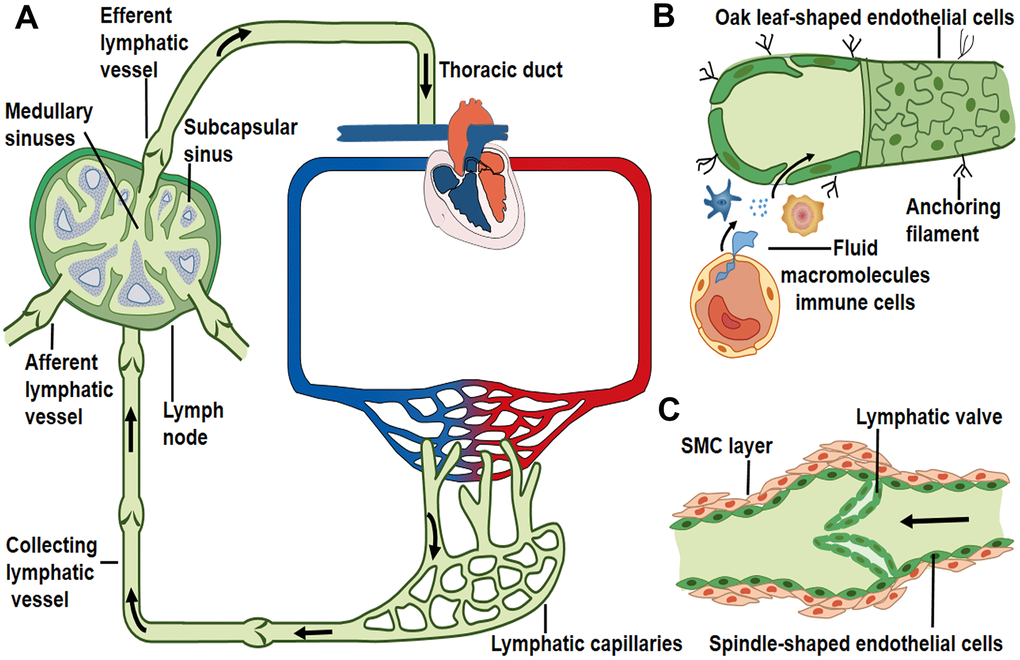
Figure 1. Lymph transport along lymphatic vessels. (A) Unidirectional lymph flow route: lymphatic capillaries collect peripheral tissue fluid and converge into larger collecting vessels, then lymph drains into the lymph node from the afferent lymphatic vessels and flows out from the efferent lymphatic vessel. Afterwards lymph fluid flows through the thoracic duct and the right lymphatic trunk, eventually enters into venous circulation. Arrows indicate the direction of lymph flow. (B) Interstitial fluid, macromolecules and immune cells which extravasate from blood vessels are collected by lymphatic capillaries. Initial lymphatics are composed of a layer of oak leaf-shaped endothelial cells and lack of muscle layers. (C) Lymphatic collectors contain intraluminal valve and SMC layers that enable the unidirectional lymph flow.
Blind-ended lymphatic capillaries converge into the larger collecting lymphatic vessels [4, 25]. Lymphatic collectors are comprised of spindle-shaped endothelial cells with a complete basement membrane covered by one or more muscle layers [4, 25] (Figure 1C). The secondary valve structures inside the collecting lymphatic vessels separate two adjacent lymphangions to prevent the back-flow of lymph and to overcome opposing pressure gradients [26–28]. Collecting lymphatic endothelial cells are connected to each other through continuous zipper-like junctions, similar to those in blood vessels [4]. The transition from the button-like junctions of initial lymphatics to zippers in collecting lymphatics is typically abrupt in individual vessels [4].
The constituents of lymph include extravasated fluid, macromolecules and immune cells [2]. These elements in peripheral tissues enter initial lymphatic vessels through the opening of primary valves or the vesicular transport of endothelial cells [4, 24, 29]. Lymphatic collectors gather initial lymphatic fluid and propel lymph forward by spontaneous contractions of the surrounding muscle layers [30]. This driving force promotes the unidirectional flow of lymph fluid into the afferent lymphatic vessels of lymph nodes [1, 31]. The afferent lymphatic vessels convey lymph into the sub-capsular sinus and along the lymph node sinus toward the efferent lymphatic vessels [1, 32]. The lymph then drains into the thoracic duct or the right lymphatic duct, and returns to venous circulation through left and right subclavian veins, respectively [1, 33, 34] (Figure 1A).
Aging-associated changes in collector lymphatic muscle cells investiture
The primary function of collector lymphatic vessels are to transport lymph back into the blood circulatory system [2]. The intrinsic contractile activities generated by lymphatic muscle cells determine the forward movement of lymph against an adverse pressure gradient [26, 27]. The lymphangion, the structural unit of lymphatic collectors, is divided into three parts: pre-valve zone, valve zone, and post-valve zone [35, 36]. Independent contraction of lymphangions propagates the peristalsis-like wave [37].
Studies on aged lymphatic vessels have demonstrated that the aging process changes lymphatic muscle cell composition [10]. As shown in images of mesenteric lymphatic vessels (MLV) immunohistochemically labeled for actin, zones located upstream (pre-valve zones) and above (valve zones) lymphatic valves exhibit significantly less muscle cell investiture with discontinuous and irregular muscle cell organization in aged groups [10]. These low muscle cell investiture zones consist of longitudinally-oriented muscle cells which connect adjacent lymphangions [10]. Muscle cells in these zones may have an impact on lymphatic valve gating and electrical coupling between lymphangions, while aging associated changes in longitudinally oriented muscle cells may alter these two functions [10, 38, 39]. In the elderly, decreased number of muscle cells surrounding lymphatic valve may limit the ability of these cells to mediate bio-directional propagation of contractile waves [10, 38]. In addition, loss of muscle cells may lead to decrease in lymphatic productivity (mainly through the reduction of contractile frequency), impaired lymphatic valve closure and subsequent reflux of lymph in aged lymphatic collector vessels [39]. Compromised pathogen transport by aged lymphatic collectors has been shown [6], and pathogens may spread in the opposite direction of normal lymph flow due to possible disruption of lymphatic valve gating [10]. Furthermore, the thin-walled low muscle cell investiture zones in aged lymphatic vessels may transform into aneurysm-like formations at high pressure [10]. The aneurysm-like formations are the ideal places for the development of low-velocity turbulent lymph flow, and the accumulation of various molecules, pathogens, and cancer cells [10]. These noxious substances may disseminate across the thin lymphatic wall, and decrease the ability of immune system to control infectious in aging. Further experimental work are needed to confirm these perspectives.
On the contrary, the downstream (post-valve zones) lymphatic valves surrounded by circularly-oriented muscle cells do not show any significant aging-associated difference in muscle cell investiture [10]. Muscle cells in these zones constitute 92–95% percent of total vessel length with a relatively consistent muscle investiture even in aged groups [10]. As the major cells to generate the contractile force, muscle cells in post-valve zones are necessary for lymph pump activity [40]. Since the high muscle cell investiture of post-valve zones was not affected by aging, the aging-associated inhibition of amplitude of lymphatic contractility is not as prominent as the aging-associated reduction of lymphatic contractile frequency under resting condition [41, 42].
The aging process reduces the levels of proteins that regulate muscle contraction [6]. Proteomic profiling of rat mesenteric lymphatic vessels was performed on 9-month-old (adult) and 24-month-old (aged) rats [6]. The muscle contractile proteins (troponin, and myosin), cytoskeleton-associated proteins (actin, gelsolin, and dynein), and myosin binding proteins are substantially reduced in the lymphatic collectors isolated from aged rats [6]. Na+, K+, and Ca++ channels, which are involved in generation of muscle cell action potential and induction of cell depolarization, are also decreased in lymph collectors of 24-month-old rats [6]. Down-regulation of muscle contraction proteins may mediate aging-inhibited lymphatic pump activity.
Aging-associated alteration of NO–dependent regulatory mechanisms
NO-dependent regulatory mechanisms control lymphatic contractility and lymph flow in lymphatic vessels [42]. Under normal conditions, there are multiple sources of nitric oxide (NO) in the lymphatic vasculature [26, 43–46]: 1) endothelial NO synthase (eNOS) from lymphatic endothelial cells; 2) inducible NO synthase (iNOS) from immune cells or lymphatic muscle cells; and 3) neuronal NO synthase (nNOS) from the perivascular lymphatic nerves. The role of nNOS in regulating lymphatic contractions still requires further exploration. The following section focuses on NO production from eNOS and iNOS.
eNOS in lymphatic endothelial cells is required for maintaining normal contractile events under physiological conditions [46]. During the contraction cycle, the intrinsic spontaneous pumping activities promote sustained forward flow which change pulsatile shear stress [47]. The lymphatic endothelium is highly sensitive to flow/stress and potentially generates NO [26]. The phasic generation of NO acts on lymphatic muscle layers concomitantly with the reduction of contractile frequency and tone [30, 48]. This spontaneous transient suppression of pump events is essential for increased diastolic filling of lympangions [49]. NO inhibits vasomotion primarily through the NO-induced production of cyclic GMP (cGMP) and the subsequent activation of both cGMP- and cAMP-dependent protein kinases (PKG and PKA) [49, 50]. The NO/cGMP regulatory pathway inhibits Ca++ release from intramyocellular stores and affects the pacemaker events of lymphatic muscle cells, leading to a decrease in contractile amplitude and frequency [49, 51, 52]. Gasheva et al compared the NO-dependent self-regulatory mechanism between an adult group and an aged group in the thoracic duct (TD) [13]. In adult rats, NO is produced from eNOS activity in the lymphatic endothelial cells in response to imposed flow [45], (Figure 2A). With an increase in imposed flow, the enhanced eNOS activity mediates the inhibition of the lymph pump in adult rats [48]. This inhibition reduces lymphatic pacemaker activity and contraction frequency [13]. This kind of spontaneous transient depolarization is essential for increased diastolic filling and the subsequent production of a larger contraction amplitude [45]. In contrast, the TD segment in old rats behaves differently in response to the imposed flow [13] (Figure 2B). No significant inhibition of lymph pump occurs in the aged group at high levels of imposed flow [13]. Furthermore, the contraction frequency and fractional lymph flow is unchanged in comparison with the adult group [13]. This aging-related alteration illustrates that the self-regulatory adjustment of lymphatic vessels is reduced to the changes in lymph flow [45]. Further experiments demonstrated that the reversal of eNOS/iNOS activity causes contractile functional impairment in the aged group [13].
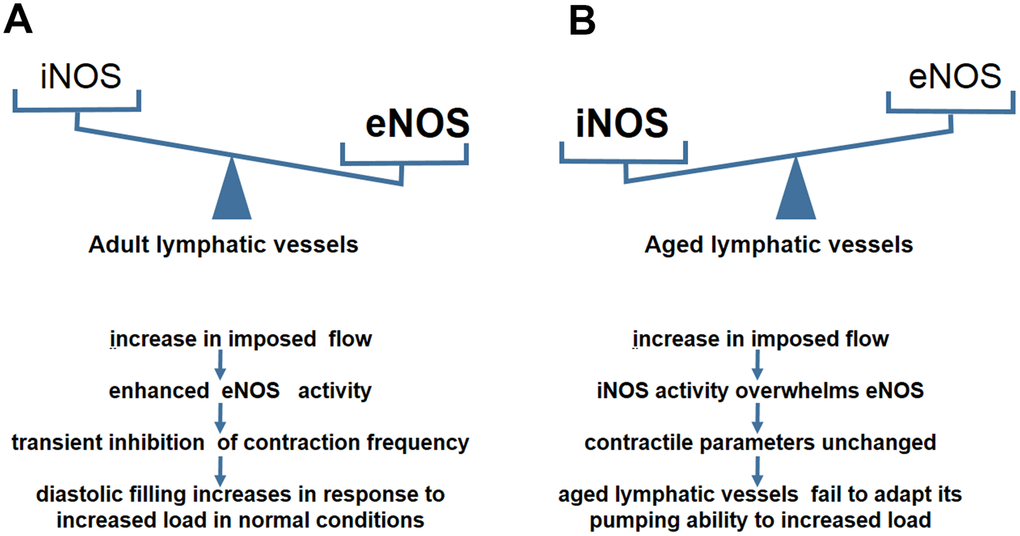
Figure 2. NO–dependent regulatory mechanisms in aged lymphatic vessels. (A) In adult lymphatic vessels, the enhanced eNOS activity mediates the transient inhibition of contraction frequency to adapt the load by the increase of imposed flow. (B) In aged lymphatic vessels, increased iNOS level renders the contractile parameters unchanged in response to increased imposed flow.
A chronic inflammatory environment often exists in the elderly [53, 54]. Excessive NO produced by CD45+CD11b+Gr-1+ myeloid cells overwhelms the spatial and temporal NO gradients produced by eNOS during inflammation [13, 43]. iNOS- derived NO may cause continuous relaxation of peri-lymphatic smooth muscle cells (SMCs), increase of vessel diameter, and decrease of inotropy, leading to reduction of contraction strength [43]. Therefore, we proposed that increased iNOS activity in the aged causes lymphatic vessels to be less responsive to imposed flow due to the presence of chronic inflammatory environments. In addition, NO is synthesized from L-arginine as a substrate for NO synthases, particularly for eNOS [55]. Ageing-induced up-regulation of arginase, the enzyme that degrades L-arginine, reduces L-arginine available for eNOS [56]. Thus, synthesis of NO is compromised in the circulation of the elderly [57]. Decreased eNOS activity leads to the loss of ability to regulate imposed flow, and consequently, the lymphatic vessels of aged rats are unable to adapt their pumping ability to transport the increasing level of lymph flow [13, 45]. Quantitative analyses also found an aging-related reversal in eNOS/iNOS expression in the TD segment [13]. The data showed a significant decrease in the relative levels of eNOS and a dramatic increase in the iNOS levels in old rats [13]. Lower sensitivity to the imposed flow induced by iNOS causes difficulties in the maintenance of the lymphatic contraction efficiency and adequate diastolic filling [11, 13]. Therefore, the lymphatic vessels of aged rats fail to appropriately adapt their contractility to various preload/ after load challenges.
Conclusions
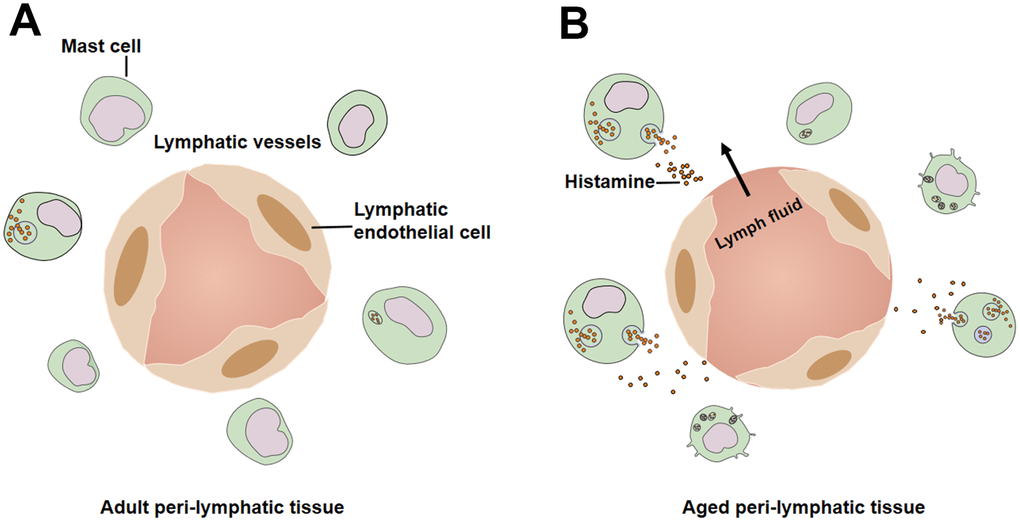
In this review, we summarize the anatomical and functional changes in aged lymphatic vessels. The aging-associated remodeling of the vascular wall is characterized by decreased muscle cells and enlarged lymphatic diameter, which lead to contractile dysfunction. In addition, eNOS/iNOS disturbances diminish contractile ability with increased extrinsic lymph flow. Aging-related hyperpermeability, resulting from decreased glycocalyx and intercellular junctions, contributes to bacterial escape in aged lymphatic vasculature. Meanwhile, aging induces the basal activation of peri-lymphatic mast cells, restricting the recruitment of immune cells and affecting the reactions to acute inflammation. Thus, aging is a major risk factor for decreased pump activity, increased permeability, and delayed immune response in lymphatic system. Understanding the mechanisms underlying lymphatic aging is crucial for the treatment of vascular diseases in the elderly.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
The work was supported by grants from National Natural Science Foundation of China (81873473 and 81570255), Key Science and Technology Research Plan of Shandong Province (2017GSF218037) and Shandong Taishan Scholarship.
References
- 1. Aspelund A, Robciuc MR, Karaman S, Makinen T, Alitalo K. Lymphatic System in Cardiovascular Medicine. Circ Res. 2016; 118:515–30. https://doi.org/10.1161/CIRCRESAHA.115.306544 [PubMed]
- 2. Pepper MS, Skobe M. Lymphatic endothelium: morphological, molecular and functional properties. J Cell Biol. 2003; 163:209–13. https://doi.org/10.1083/jcb.200308082 [PubMed]
- 3. Witte MH, Jones K, Wilting J, Dictor M, Selg M, McHale N, Gershenwald JE, Jackson DG. Structure function relationships in the lymphatic system and implications for cancer biology. Cancer Metastasis Rev. 2006; 25:159–84. https://doi.org/10.1007/s10555-006-8496-2 [PubMed]
- 4. Baluk P, Fuxe J, Hashizume H, Romano T, Lashnits E, Butz S, Vestweber D, Corada M, Molendini C, Dejana E, McDonald DM. Functionally specialized junctions between endothelial cells of lymphatic vessels. J Exp Med. 2007; 204:2349–62. https://doi.org/10.1084/jem.20062596 [PubMed]
- 5. Randolph GJ, Angeli V, Swartz MA. Dendritic-cell trafficking to lymph nodes through lymphatic vessels. Nat Rev Immunol. 2005; 5:617–28. https://doi.org/10.1038/nri1670 [PubMed]
- 6. Zolla V, Nizamutdinova IT, Scharf B, Clement CC, Maejima D, Akl T, Nagai T, Luciani P, Leroux JC, Halin C, Stukes S, Tiwari S, Casadevall A, et al. Aging-related anatomical and biochemical changes in lymphatic collectors impair lymph transport, fluid homeostasis, and pathogen clearance. Aging Cell. 2015; 14:582–94. https://doi.org/10.1111/acel.12330 [PubMed]
- 7. Karaman S, Buschle D, Luciani P, Leroux JC, Detmar M, Proulx ST. Decline of lymphatic vessel density and function in murine skin during aging. Angiogenesis. 2015; 18:489–98. https://doi.org/10.1007/s10456-015-9479-0 [PubMed]
- 8. Zhdanov DA. [Senile changes in the lymphatic capillaries and vessels]. Arkh Anat Gistol Embriol. 1960; 39:24–36. [PubMed]
- 9. Zerbino DD. [Senile changes in the outflow lymphatic vessels]. Arkh Anat Gistol Embriol. 1960; 39:37–42. [PubMed]
- 10. Bridenbaugh EA, Nizamutdinova IT, Jupiter D, Nagai T, Thangaswamy S, Chatterjee V, Gashev AA. Lymphatic muscle cells in rat mesenteric lymphatic vessels of various ages. Lymphat Res Biol. 2013; 11:35–42. https://doi.org/10.1089/lrb.2012.0025 [PubMed]
- 11. Gashev AA, Chatterjee V. Aged lymphatic contractility: recent answers and new questions. Lymphat Res Biol. 2013; 11:2–13. https://doi.org/10.1089/lrb.2013.0003 [PubMed]
- 12. Rabinovitz AJ, Saphir O. THE THORACIC DUCT; SIGNIFICANCE OF AGE-RELATED CHANGES AND OF LIPID IN THE WALL. Circulation. 1965; 31:899–905. https://doi.org/10.1161/01.CIR.31.6.899 [PubMed]
- 13. Gasheva OY, Knippa K, Nepiushchikh ZV, Muthuchamy M, Gashev AA. Age-related alterations of active pumping mechanisms in rat thoracic duct. Microcirculation. 2007; 14:827–39. https://doi.org/10.1080/10739680701444065 [PubMed]
- 14. Grada AA, Phillips TJ. Lymphedema: pathophysiology and clinical manifestations. J Am Acad Dermatol. 2017; 77:1009–20. https://doi.org/10.1016/j.jaad.2017.03.022 [PubMed]
- 15. Chatterjee V, Gashev AA. Aging-associated shifts in functional status of mast cells located by adult and aged mesenteric lymphatic vessels. Am J Physiol Heart Circ Physiol. 2012; 303:H693–702. https://doi.org/10.1152/ajpheart.00378.2012 [PubMed]
- 16. Tharakan B, Hellman J, Sawant DA, Tinsley JH, Parrish AR, Hunter FA, Smythe WR, Childs EW. β-Catenin dynamics in the regulation of microvascular endothelial cell hyperpermeability. Shock. 2012; 37:306–11. https://doi.org/10.1097/SHK.0b013e318240b564 [PubMed]
- 17. Sawant DA, Tharakan B, Adekanbi A, Hunter FA, Smythe WR, Childs EW. Inhibition of VE-cadherin proteasomal degradation attenuates microvascular hyperpermeability. Microcirculation. 2011; 18:46–55. https://doi.org/10.1111/j.1549-8719.2010.00067.x [PubMed]
- 18. Oakley R, Tharakan B. Vascular hyperpermeability and aging. Aging Dis. 2014; 5:114–25. [PubMed]
- 19. Oliver G, Alitalo K. The lymphatic vasculature: recent progress and paradigms. Annu Rev Cell Dev Biol. 2005; 21:457–83. https://doi.org/10.1146/annurev.cellbio.21.012704.132338 [PubMed]
- 20. Leak LV, Burke JF. Fine structure of the lymphatic capillary and the adjoining connective tissue area. Am J Anat. 1966; 118:785–809. https://doi.org/10.1002/aja.1001180308 [PubMed]
- 21. Gerli R, Ibba L, Fruschelli C. A fibrillar elastic apparatus around human lymph capillaries. Anat Embryol (Berl). 1990; 181:281–86. https://doi.org/10.1007/BF00174621 [PubMed]
- 22. Schmid-Schönbein GW. Microlymphatics and lymph flow. Physiol Rev. 1990; 70:987–1028. https://doi.org/10.1152/physrev.1990.70.4.987 [PubMed]
- 23. Zöltzer H. Initial lymphatics—morphology and function of the endothelial cells. Lymphology. 2003; 36:7–25. [PubMed]
- 24. Mendoza E, Schmid-Schönbein GW. A model for mechanics of primary lymphatic valves. J Biomech Eng. 2003; 125:407–14. https://doi.org/10.1115/1.1568128 [PubMed]
- 25. Mäkinen T, Norrmén C, Petrova TV. Molecular mechanisms of lymphatic vascular development. Cell Mol Life Sci. 2007; 64:1915–29. https://doi.org/10.1007/s00018-007-7040-z [PubMed]
- 26. Chakraborty S, Davis MJ, Muthuchamy M. Emerging trends in the pathophysiology of lymphatic contractile function. Semin Cell Dev Biol. 2015; 38:55–66. https://doi.org/10.1016/j.semcdb.2015.01.005 [PubMed]
- 27. Mislin H. Active contractility of the lymphangion and coordination of lymphangion chains. Experientia. 1976; 32:820–22. https://doi.org/10.1007/BF02003701 [PubMed]
- 28. Zawieja D. Lymphatic biology and the microcirculation: past, present and future. Microcirculation. 2005; 12:141–50. https://doi.org/10.1080/10739680590900003 [PubMed]
- 29. Trzewik J, Mallipattu SK, Artmann GM, Delano FA, Schmid-Schönbein GW. Evidence for a second valve system in lymphatics: endothelial microvalves. FASEB J. 2001; 15:1711–17. https://doi.org/10.1096/fj.01-0067com [PubMed]
- 30. Gashev AA, Zawieja DC. Hydrodynamic regulation of lymphatic transport and the impact of aging. Pathophysiology. 2010; 17:277–87. https://doi.org/10.1016/j.pathophys.2009.09.002 [PubMed]
- 31. Swartz MA. The physiology of the lymphatic system. Adv Drug Deliv Rev. 2001; 50:3–20. https://doi.org/10.1016/S0169-409X(01)00150-8 [PubMed]
- 32. Jeltsch M, Tammela T, Alitalo K, Wilting J. Genesis and pathogenesis of lymphatic vessels. Cell Tissue Res. 2003; 314:69–84. https://doi.org/10.1007/s00441-003-0777-2 [PubMed]
- 33. Geng X, Cha B, Mahamud MR, Lim KC, Silasi-Mansat R, Uddin MK, Miura N, Xia L, Simon AM, Engel JD, Chen H, Lupu F, Srinivasan RS. Multiple mouse models of primary lymphedema exhibit distinct defects in lymphovenous valve development. Dev Biol. 2016; 409:218–33. https://doi.org/10.1016/j.ydbio.2015.10.022 [PubMed]
- 34. Tammela T, Alitalo K. Lymphangiogenesis: molecular mechanisms and future promise. Cell. 2010; 140:460–76. https://doi.org/10.1016/j.cell.2010.01.045 [PubMed]
- 35. McHale NG, Roddie IC. Pumping activity in isolated segments of bovine mesenteric lymphatics. J Physiol. 1975; 244:70P–72P. [PubMed]
- 36. Gashev AA, Orlov RS, Borisov AV, Kliuchin’ski T, Andreevskaia MV, Bubnova NA, Borisova RP, Andreev IA, Erofeev NP, Priklonskaia EG. [The mechanisms of lymphangion interaction in the process of lymph movement]. Fiziol Zh SSSR Im I M Sechenova. 1990; 76:1489–508. [PubMed]
- 37. McHale NG, Roddie IC. The effect of transmural pressure on pumping activity in isolated bovine lymphatic vessels. J Physiol. 1976; 261:255–69. https://doi.org/10.1113/jphysiol.1976.sp011557 [PubMed]
- 38. Zawieja DC, Davis KL, Schuster R, Hinds WM, Granger HJ. Distribution, propagation, and coordination of contractile activity in lymphatics. Am J Physiol. 1993; 264:H1283–91. https://doi.org/10.1152/ajpheart.1993.264.4.h1283 [PubMed]
- 39. Davis MJ, Rahbar E, Gashev AA, Zawieja DC, Moore JE
Jr . Determinants of valve gating in collecting lymphatic vessels from rat mesentery. Am J Physiol Heart Circ Physiol. 2011; 301:H48–60. https://doi.org/10.1152/ajpheart.00133.2011 [PubMed] - 40. Gashev AA. Basic mechanisms controlling lymph transport in the mesenteric lymphatic net. Ann N Y Acad Sci. 2010 (Suppl 1); 1207:E16–20. https://doi.org/10.1111/j.1749-6632.2010.05710.x [PubMed]
- 41. Nagai T, Bridenbaugh EA, Gashev AA. Aging-associated alterations in contractility of rat mesenteric lymphatic vessels. Microcirculation. 2011; 18:463–73. https://doi.org/10.1111/j.1549-8719.2011.00107.x [PubMed]
- 42. Akl TJ, Nagai T, Coté GL, Gashev AA. Mesenteric lymph flow in adult and aged rats. Am J Physiol Heart Circ Physiol. 2011; 301:H1828–40. https://doi.org/10.1152/ajpheart.00538.2011 [PubMed]
- 43. Liao S, Cheng G, Conner DA, Huang Y, Kucherlapati RS, Munn LL, Ruddle NH, Jain RK, Fukumura D, Padera TP. Impaired lymphatic contraction associated with immunosuppression. Proc Natl Acad Sci USA. 2011; 108:18784–89. https://doi.org/10.1073/pnas.1116152108 [PubMed]
- 44. Shirasawa Y, Ikomi F, Ohhashi T. Physiological roles of endogenous nitric oxide in lymphatic pump activity of rat mesentery in vivo. Am J Physiol Gastrointest Liver Physiol. 2000; 278:G551–56. https://doi.org/10.1152/ajpgi.2000.278.4.G551 [PubMed]
- 45. Gasheva OY, Zawieja DC, Gashev AA. Contraction-initiated NO-dependent lymphatic relaxation: a self-regulatory mechanism in rat thoracic duct. J Physiol. 2006; 575:821–32. https://doi.org/10.1113/jphysiol.2006.115212 [PubMed]
- 46. Hagendoorn J, Padera TP, Kashiwagi S, Isaka N, Noda F, Lin MI, Huang PL, Sessa WC, Fukumura D, Jain RK. Endothelial nitric oxide synthase regulates microlymphatic flow via collecting lymphatics. Circ Res. 2004; 95:204–09. https://doi.org/10.1161/01.RES.0000135549.72828.24 [PubMed]
- 47. Dixon JB, Greiner ST, Gashev AA, Cote GL, Moore JE
Jr , Zawieja DC. Lymph flow, shear stress, and lymphocyte velocity in rat mesenteric prenodal lymphatics. Microcirculation. 2006; 13:597–610. https://doi.org/10.1080/10739680600893909 [PubMed] - 48. Gashev AA, Davis MJ, Zawieja DC. Inhibition of the active lymph pump by flow in rat mesenteric lymphatics and thoracic duct. J Physiol. 2002; 540:1023–37. https://doi.org/10.1113/jphysiol.2001.016642 [PubMed]
- 49. von der Weid PY, Zhao J, Van Helden DF. Nitric oxide decreases pacemaker activity in lymphatic vessels of guinea pig mesentery. Am J Physiol Heart Circ Physiol. 2001; 280:H2707–16. https://doi.org/10.1152/ajpheart.2001.280.6.H2707 [PubMed]
- 50. Lincoln TM, Cornwell TL, Komalavilas P, Boerth N. Cyclic GMP-dependent protein kinase in nitric oxide signaling. Methods Enzymol. 1996; 269:149–66. https://doi.org/10.1016/S0076-6879(96)69017-X [PubMed]
- 51. Murad F, Forstermann U, Nakane M, Schmidt H, Pollock J, Sheng H, Matsumoto T, Warner T, Mitchell J, Tracey R, et al. The nitric oxide-cyclic GMP signal transduction pathway in vascular smooth muscle preparations and other tissues. Jpn J Pharmacol. 1992 (Suppl 2); 58:150P–57P. https://doi.org/10.1007/978-1-4615-2437-3_6 [PubMed]
- 52. Warner TD, Mitchell JA, Sheng H, Murad F. Effects of cyclic GMP on smooth muscle relaxation. Adv Pharmacol. 1994; 26:171–94. https://doi.org/10.1016/S1054-3589(08)60054-X [PubMed]
- 53. Brüünsgaard H, Pedersen BK. Age-related inflammatory cytokines and disease. Immunol Allergy Clin North Am. 2003; 23:15–39. https://doi.org/10.1016/S0889-8561(02)00056-5 [PubMed]
- 54. Bruunsgaard H, Pedersen M, Pedersen BK. Aging and proinflammatory cytokines. Curr Opin Hematol. 2001; 8:131–36. https://doi.org/10.1097/00062752-200105000-00001 [PubMed]
- 55. Santhanam L, Christianson DW, Nyhan D, Berkowitz DE. Arginase and vascular aging. J Appl Physiol (1985). 2008; 105:1632–42. https://doi.org/10.1152/japplphysiol.90627.2008 [PubMed]
- 56. Holowatz LA, Thompson CS, Kenney WL. L-Arginine supplementation or arginase inhibition augments reflex cutaneous vasodilatation in aged human skin. J Physiol. 2006; 574:573–81. https://doi.org/10.1113/jphysiol.2006.108993 [PubMed]
- 57. Rubio-Ruiz ME, Pérez-Torres I, Soto ME, Pastelín G, Guarner-Lans V. Aging in blood vessels. Medicinal agents FOR systemic arterial hypertension in the elderly. Ageing Res Rev. 2014; 18:132–47. https://doi.org/10.1016/j.arr.2014.10.001 [PubMed]
- 58. Reitsma S, Slaaf DW, Vink H, van Zandvoort MA, oude Egbrink MG. The endothelial glycocalyx: composition, functions, and visualization. Pflugers Arch. 2007; 454:345–59. https://doi.org/10.1007/s00424-007-0212-8 [PubMed]
- 59. Iozzo RV. Perlecan: a gem of a proteoglycan. Matrix Biol. 1994; 14:203–08. https://doi.org/10.1016/0945-053X(94)90183-X [PubMed]
- 60. Perrimon N, Bernfield M. Specificities of heparan sulphate proteoglycans in developmental processes. Nature. 2000; 404:725–28. https://doi.org/10.1038/35008000 [PubMed]
- 61. Huxley VH, Curry FE. Differential actions of albumin and plasma on capillary solute permeability. Am J Physiol. 1991; 260:H1645–54. https://doi.org/10.1152/ajpheart.1991.260.5.H1645 [PubMed]
- 62. Henry CB, Duling BR. Permeation of the luminal capillary glycocalyx is determined by hyaluronan. Am J Physiol. 1999; 277:H508–14. https://doi.org/10.1152/ajpheart.1999.277.2.H508 [PubMed]
- 63. Vink H, Duling BR. Capillary endothelial surface layer selectively reduces plasma solute distribution volume. Am J Physiol Heart Circ Physiol. 2000; 278:H285–89. https://doi.org/10.1152/ajpheart.2000.278.1.H285 [PubMed]
- 64. van Haaren PM, VanBavel E, Vink H, Spaan JA. Localization of the permeability barrier to solutes in isolated arteries by confocal microscopy. Am J Physiol Heart Circ Physiol. 2003; 285:H2848–56. https://doi.org/10.1152/ajpheart.00117.2003 [PubMed]
- 65. van den Berg BM, Vink H, Spaan JA. The endothelial glycocalyx protects against myocardial edema. Circ Res. 2003; 92:592–94. https://doi.org/10.1161/01.RES.0000065917.53950.75 [PubMed]
- 66. Zhang C, Liu Q, Dong F, Li L, Du J, Xie Q, Hu H, Yan S, Zhou X, Li C, Lobe CG, Liu J. Catalpol downregulates vascular endothelial-cadherin expression and induces vascular hyperpermeability. Mol Med Rep. 2016; 13:373–78. https://doi.org/10.3892/mmr.2015.4522 [PubMed]
- 67. Falk MM. Adherens junctions remain dynamic. BMC Biol. 2010; 8:34. https://doi.org/10.1186/1741-7007-8-34 [PubMed]
- 68. Krouwer VJ, Hekking LH, Langelaar-Makkinje M, Regan-Klapisz E, Post JA. Endothelial cell senescence is associated with disrupted cell-cell junctions and increased monolayer permeability. Vasc Cell. 2012; 4:12. https://doi.org/10.1186/2045-824X-4-12 [PubMed]
- 69. Dejana E, Bazzoni G, Lampugnani MG. Vascular endothelial (VE)-cadherin: only an intercellular glue? Exp Cell Res. 1999; 252:13–19. https://doi.org/10.1006/excr.1999.4601 [PubMed]
- 70. Wu WB, Peng HC, Huang TF. Disintegrin causes proteolysis of beta-catenin and apoptosis of endothelial cells. Involvement of cell-cell and cell-ECM interactions in regulating cell viability. Exp Cell Res. 2003; 286:115–27. https://doi.org/10.1016/S0014-4827(03)00105-8 [PubMed]
- 71. Stagg HW, Bowen KA, Sawant DA, Rodriguez M, Tharakan B, Childs EW. Tumor necrosis factor-related apoptosis-inducing ligand promotes microvascular endothelial cell hyperpermeability through phosphatidylinositol 3-kinase pathway. Am J Surg. 2013; 205:419–25. https://doi.org/10.1016/j.amjsurg.2012.10.027 [PubMed]
- 72. Childs EW, Tharakan B, Hunter FA, Tinsley JH, Cao X. Apoptotic signaling induces hyperpermeability following hemorrhagic shock. Am J Physiol Heart Circ Physiol. 2007; 292:H3179–89. https://doi.org/10.1152/ajpheart.01337.2006 [PubMed]
- 73. Sawant DA, Tharakan B, Hunter FA, Smythe WR, Childs EW. Role of β-catenin in regulating microvascular endothelial cell hyperpermeability. J Trauma. 2011; 70:481–87. https://doi.org/10.1097/TA.0b013e31820b3ed7 [PubMed]
- 74. Du L, Dong F, Guo L, Hou Y, Yi F, Liu J, Xu D. Interleukin-1β increases permeability and upregulates the expression of vascular endothelial-cadherin in human renal glomerular endothelial cells. Mol Med Rep. 2015; 11:3708–14. https://doi.org/10.3892/mmr.2015.3172 [PubMed]
- 75. Bazzoni G, Dejana E. Endothelial cell-to-cell junctions: molecular organization and role in vascular homeostasis. Physiol Rev. 2004; 84:869–901. https://doi.org/10.1152/physrev.00035.2003 [PubMed]
- 76. Regan-Klapisz E, Krouwer V, Langelaar-Makkinje M, Nallan L, Gelb M, Gerritsen H, Verkleij AJ, Post JA. Golgi-associated cPLA2alpha regulates endothelial cell-cell junction integrity by controlling the trafficking of transmembrane junction proteins. Mol Biol Cell. 2009; 20:4225–34. https://doi.org/10.1091/mbc.e08-02-0210 [PubMed]
- 77. Borish L, Joseph BZ. Inflammation and the allergic response. Med Clin North Am. 1992; 76:765–87. https://doi.org/10.1016/S0025-7125(16)30325-X [PubMed]
- 78. Amin K. The role of mast cells in allergic inflammation. Respir Med. 2012; 106:9–14. https://doi.org/10.1016/j.rmed.2011.09.007 [PubMed]
- 79. Boe DM, Boule LA, Kovacs EJ. Innate immune responses in the ageing lung. Clin Exp Immunol. 2017; 187:16–25. https://doi.org/10.1111/cei.12881 [PubMed]
- 80. Gilmore TD. Introduction to NF-kappaB: players, pathways, perspectives. Oncogene. 2006; 25:6680–84. https://doi.org/10.1038/sj.onc.1209954 [PubMed]
- 81. Nizamutdinova IT, Maejima D, Nagai T, Meininger CJ, Gashev AA. Histamine as an Endothelium-Derived Relaxing Factor in Aged Mesenteric Lymphatic Vessels. Lymphat Res Biol. 2017; 15:136–45. https://doi.org/10.1089/lrb.2016.0062 [PubMed]
- 82. Nizamutdinova IT, Dusio GF, Gasheva OY, Skoog H, Tobin R, Peddaboina C, Meininger CJ, Zawieja DC, Newell-Rogers MK, Gashev AA. Mast cells and histamine are triggering the NF-κB-mediated reactions of adult and aged perilymphatic mesenteric tissues to acute inflammation. Aging (Albany NY). 2016; 8:3065–90. https://doi.org/10.18632/aging.101113 [PubMed]
- 83. Zhang H, Li L, Wang Y, Dong F, Chen X, Liu F, Xu D, Yi F, Kapron CM, Liu J. NF-κB signaling maintains the survival of cadmium-exposed human renal glomerular endothelial cells. Int J Mol Med. 2016; 38:417–22. https://doi.org/10.3892/ijmm.2016.2640 [PubMed]
- 84. de Martin R, Vanhove B, Cheng Q, Hofer E, Csizmadia V, Winkler H, Bach FH. Cytokine-inducible expression in endothelial cells of an I kappa B alpha-like gene is regulated by NF kappa B. EMBO J. 1993; 12:2773–79. https://doi.org/10.1002/j.1460-2075.1993.tb05938.x [PubMed]
- 85. Cromer WE, Zawieja SD, Tharakan B, Childs EW, Newell MK, Zawieja DC. The effects of inflammatory cytokines on lymphatic endothelial barrier function. Angiogenesis. 2014; 17:395–406. https://doi.org/10.1007/s10456-013-9393-2 [PubMed]
- 86. Kim CH, Lee KH, Lee CT, Kim YW, Han SK, Shim YS, Yoo CG. Aggregation of beta2 integrins activates human neutrophils through the IkappaB/NF-kappaB pathway. J Leukoc Biol. 2004; 75:286–92. https://doi.org/10.1189/jlb.0103038 [PubMed]
- 87. Kim KE, Koh YJ, Jeon BH, Jang C, Han J, Kataru RP, Schwendener RA, Kim JM, Koh GY. Role of CD11b+ macrophages in intraperitoneal lipopolysaccharide-induced aberrant lymphangiogenesis and lymphatic function in the diaphragm. Am J Pathol. 2009; 175:1733–45. https://doi.org/10.2353/ajpath.2009.090133 [PubMed]
- 88. Knudsen E, Iversen PO, Van Rooijen N, Benestad HB. Macrophage-dependent regulation of neutrophil mobilization and chemotaxis during development of sterile peritonitis in the rat. Eur J Haematol. 2002; 69:284–96. https://doi.org/10.1034/j.1600-0609.2002.02657.x [PubMed]
- 89. Chatterjee V, Gashev AA. Mast cell-directed recruitment of MHC class II positive cells and eosinophils towards mesenteric lymphatic vessels in adulthood and elderly. Lymphat Res Biol. 2014; 12:37–47. https://doi.org/10.1089/lrb.2013.0031 [PubMed]
- 90. Donato AJ, Black AD, Jablonski KL, Gano LB, Seals DR. Aging is associated with greater nuclear NF kappa B, reduced I kappa B alpha, and increased expression of proinflammatory cytokines in vascular endothelial cells of healthy humans. Aging Cell. 2008; 7:805–12. https://doi.org/10.1111/j.1474-9726.2008.00438.x [PubMed]