Introduction
Age-related female reproductive decline is associated with the neuroendocrine regulation of the hypothalamic-pituitary-ovarian axis (HPO), in which an attenuated luteinizing hormone (LH) surge is one of the earliest signs associated with reproductive senescence in female rats and mice [1, 2]. Previous research has documented that the activation of gonadotropin-releasing hormone (GnRH) neurons is remarkedly reduced in middle-aged mice during the GnRH/LH surge [3–5]. However, the underlying mechanisms of the hypothalamic regulation for the early reproductive decline remain obscure.
To date, a body of studies have documented that hypothalamic astrocytes are one of the key cells that constitute the neural circuits responsible for GnRH neuron activation and GnRH release. Studies in rodents have shown that ovarian estradiol (E2) stimulates progesterone synthesis in hypothalamic astrocytes, which is the key to the activation of the GnRH neurons [6–8]. Specifically, estradiol acts on membrane-associated estradiol receptor-alpha (ERα) in astrocytes to increase the release of intracellular store of Ca2+ via a phospholipase C (PLC)/ inositol trisphosphate (IP3) signaling pathway. The increased Ca2+ level in turn activates the protein kinase A (PKA), leading to protein phosphorylation and the activation of cholesterol-side-chain cleavage enzyme (P450scc) and 3β-hydroxysteroid dehydrogenase (3β-HSD), which are responsible for limiting the rate of progesterone synthesis. Then, the newly synthesized progesterone then diffuses out of the astrocytes and activates estradiol-induced progesterone receptors in proximal neurons, triggering the neural cascades and resulting in the GnRH/LH surge [6].
Available studies have also demonstrated that astrocytes are essential for the GnRH neuronal activity and GnRH release through multiple pathways. In rodents and rhesus monkeys, the remarkable structural plasticity of astrocytes has been confirmed to ensheath GnRH cell bodies and/or to oppose the GnRH processes. These structural relationships are dynamically regulated by ovarian estradiol and are crucial for normal female reproductive function [9–11]. In addition, astrocytes also synthesize and release cytokines that regulate GnRH secretion, including prostaglandin E and a lot of growth factors, such as transforming growth factors (TGFα and TGF-β1), basic fibroblast growth factor IGF1, and epidermal growth factor family, etc. [12–15].
Taken together, the existent literature shows that astrocytes play a crucial part in the puberty onset, estrus cycle, and fertility [7, 8, 16]. But what changes take place in the hypothalamic astrocytes during aging remains blurred. Therefore, we hypothesized that when the repeated cycles of ovarian estradiol induce the synthesis of neuroprogesterone in hypothalamic astrocytes, the metabolic pathway of estradiol also simultaneously accelerates the senescence of astrocytes, which would weaken or change the role of astrocytes in regulating the female reproductive function. We tested this hypothesis with a mouse model receiving bilateral ovariectomy and an estradiol replacement and in vitro primary cultured hypothalamic astrocytes treated with estradiol.
Results
Estradiol induces the senescence of astrocytes in the hypothalamus
As astrocytes in the hypothalamic RP3V play a crucial part in the estradiol positive feedback [16, 22, 23], it follows that the senescence of astrocytes in RP3V may be associated with ovarian estradiol. To confirm this hypothesis, a mouse model with ovarian resection was adopted in the study. A total of 45 female mice (aged 3 months old) with regular estrus cycles determined by daily vaginal smears were selected and randomly placed into 3 groups receiving bilateral ovariectomy (OVX group), bilateral ovariectomy plus estradiol replacement (OVX+E2 group) or sham-operation (control group). The OVX+E2 group received twice a 0.25mg dose of a 90-day sustained release pellet of 17β-estradiol placed subcutaneously. The dose of the 17β-estradiol in mice has been found to correspond to the estradiol levels in a pro-estrus phase [24, 25]. Brain tissue samples were obtained from the three groups when they were raised to 9 months old (Figure 2A). Compared with those of the sham group and OVX+E2 group, the number of SA-β-gal positive astrocytes in the RP3V of the OVX group was significantly reduced, and the mean fluorescence intensity of astrocytes expressing γ-H2AX and p16 was also obviously attenuated. Meanwhile, no significant difference was evident between the OVX+E2 and the sham group (one-way ANOVA effect of treatment p=0.0944, df=2,10, F=3.895; p=0.5026, df=2,81, F=0.4641; p=0.6268, df=2,42, F=1.614; respectively) (Figure 2B). The results show that the sustaining effect of estradiol is involved in the senescence of astrocytes in the hypothalamic RP3V.
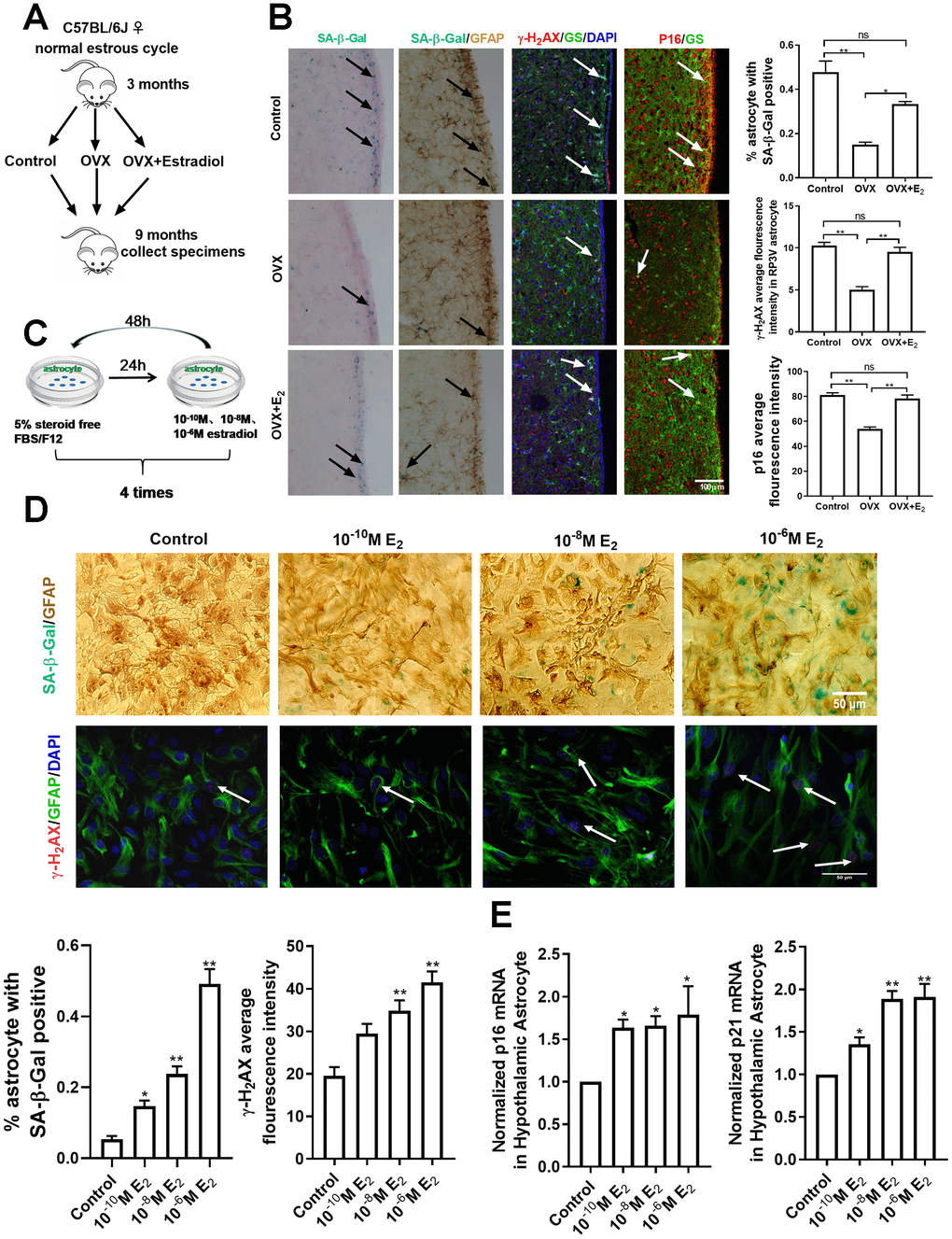
Figure 2. Estradiol induces senescence of astrocytes in the hypothalamus. (A) The flow chart of mouse castration and estradiol intervention. (B) Representative microscopies showing SA-β-gal staining in the control (n=5), OVX (n=5) and OVX+E2 groups (n=5), black arrows representing SA-β-Gal–positive cells (left one). Dual-label immunohistochemistry showing astrocytes by GFAP staining (brown) and by SA-β-Gal staining (blue), black arrows representing SA-β-Gal–positive astrocytes (left two). Dual-label immunofluorescence showing astrocytes (green) with γ-H2AX (red), white arrows representing γ-H2AX–positive astrocytes (right two). Dual-label immunofluorescence showing astrocytes (green) with p16 (red), white arrows representing p16–positive astrocytes (right one). Scale bar= 100μm. (C) The flow chart of estradiol intervention in primary cultured astrocytes. (D) Dual-label immunohistochemistry showing astrocytes by GFAP staining (brown) and by SA-β-Gal staining (blue) with three different estradiol concentrations (10-10M, 10-8M, 10-6M) (upper), black arrows representing SA-β-Gal–positive astrocytes. Scale bar= 100 μm. Dual-label immunofluorescence showing astrocytes (green) and γ-H2AX (red) with different estradiol concentrations, white arrows representing γ-H2AX–positive astrocytes. Scale bar=50μm. (E) Detection of p16 and p21 mRNA levels under different estradiol concentrations. Estradiol increased the expression of p16 and p21 with different estradiol concentrations in hypothalamic astrocytes (n = 3-4). The experiments used two-way analysis of variance. The p-value was determined by One-way ANOVA: *p<0.05, ** p< 0.01.OVX, i.e. ovariectomy, OVX+E2, i.e. ovariectomy plus estradiol replacement, 10-6M E2, i.e.10-6M estradiol concentrations.
To further confirm the above results, primary cultured astrocytes from the neonatal mouse hypothalamus were directly treated with 17β-estradiol. The intervention concentration of estradiol was 10-6M, 10-8M, and 10-10M, according to the experimental results of CCK8 (Supplementary Figure 1B). The astrocytes were first cultured with a carbon-adsorbed serum, which did not contain steroid hormones, for 24h before the 17β-estradiol intervention, and then incubated with different estradiol concentrations for 48 h. The process was repeated 4 times to induce cell senescence [6] (Figure 2C). The analysis showed that compared with the solvent-treated astrocytes, estradiol augmented the number of SA-β-Gal positive astrocytes in the hypothalamus in a concentration-dependent manner. The number of γ-H2AX positive astrocytes and the mean fluorescence intensity of γ-H2AX expression in astrocytes were significantly enhanced in the estradiol-treated group (10-8M and 10-6M) (one-way ANOVA effect of treatment p<0.05, df=3,11, F=1.219;Kruskal-Wallis test, p<0.01, respectively) (Figure 2D). In addition, the mRNA levels of p16 and p21 in the estradiol-treated group were significantly higher than those in the control sample (one-way ANOVA effect of treatment p<0.05, df=3,10, F=2.391;p<0.05, df=3,9, F=1.525, respectively) (Figure 2E). However, estradiol treatment did not induce a similar phenomenon in the cortex-derived astrocytes but displayed an opposite change instead, as shown in the supplemental materials (Supplementary Figure 1C–1E). Altogether, the results of in vivo and in vitro analyses show that estradiol induces the senescence of hypothalamic astrocytes.
The estradiol metabolites 2-OHE2 and 4-OHE2 are closely associated with astrocyte senescence
To understand the mechanism of estradiol-induced cellular senescence, we investigated into the intracellular metabolic pathway of estradiol [26]. Estradiol metabolism operates in two pathways (Figure 3A): a conjugative pathway, which forms sulfates and glucuronides, respectively catalyzed by sulfotransferases (SULTs) and UDP-glucuronosyl transferases (UGTs), and a hydroxylated pathway. In the latter, estradiol is hydroxylated by CYPs (cytochrome p450) isoforms, including CYP1A1, CYP1A2, and CYP1B1, to form catechol estradiols, such as 2-hydroxyestradiol (2-OHE2) and 4-hydroxyestradiol (4-OHE2), which have long been thought to be an intermediate metabolite responsible for DNA damage and oxidative stress [27, 28]. So targeted HPLC was proceeded on hypothalamic tissues to examine the level of 2-OHE2 and 4-OHE2 and the results indicated their existence in the hypothalamus of the 10-month-old mice (Figure 3B). Furthermore, the primary cultured astrocytes from the hypothalamus were respectively treated with 2-OHE2 (20nM, 24h) and 4-OHE2 (20nM, 24h). The mean fluorescence intensity of p16 and γ-H2AX in the astrocytes treated with hydroxyl estradiols significantly increased when compared with that of the control group (one-way ANOVA effect of treatment p<0.01, df=2,96, F=0.5358;Kruskal-Wallis test, p<0.01, respectively) (Figure 3C). Similarly, the mRNA level of p16 and p21 in the astrocytes treated with hydroxyl estradiols was obviously higher than that of the control group (one-way ANOVA effect of treatment p<0.01, df=2,6, F=1.982; p<0.01, df=2,6, F=1.33, respectively) (Figure 3D). These results suggest that the intracellular metabolites of estradiol, 2-OHE2 and 4-OHE2, facilitate the astrocyte senescence.
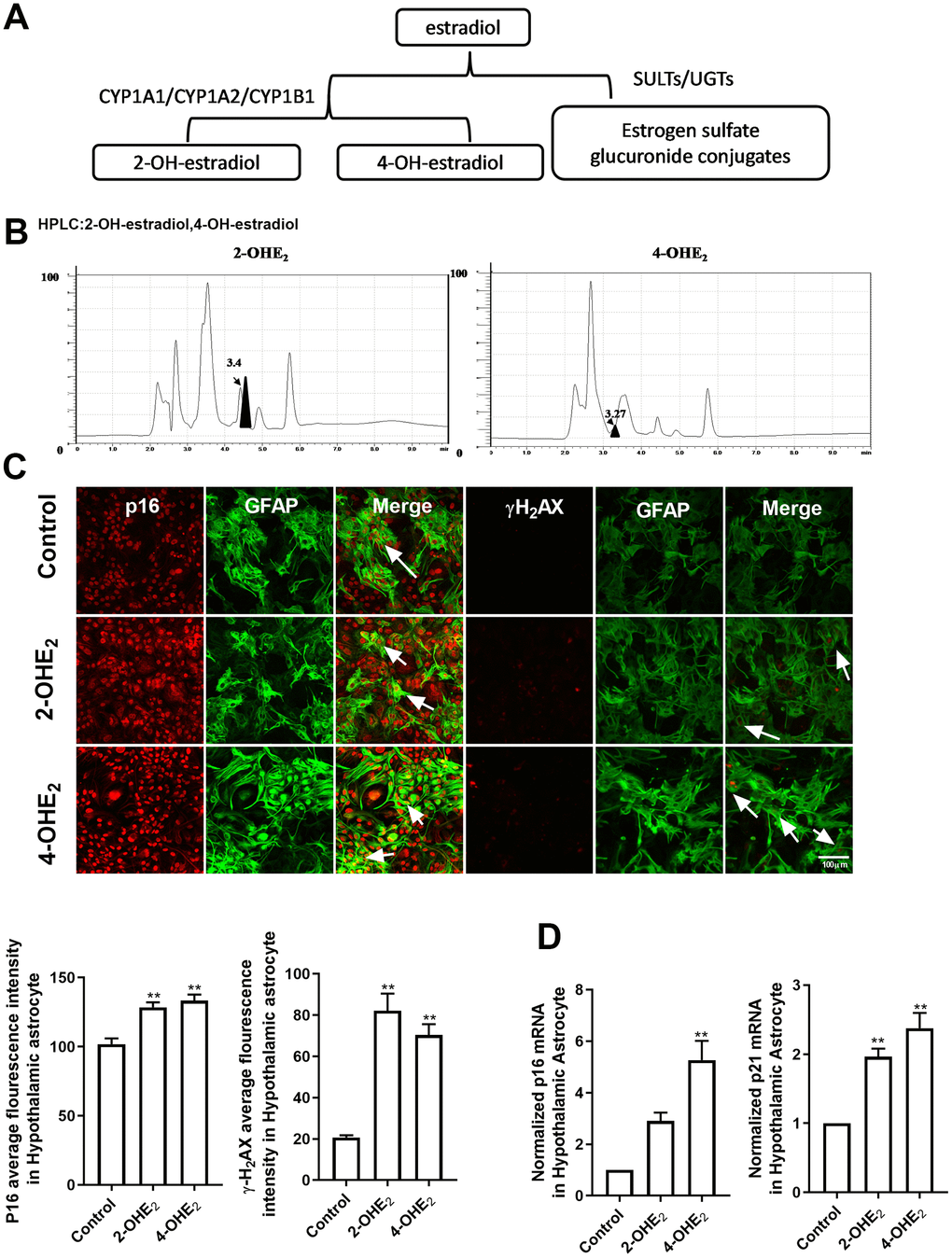
Figure 3. The estradiol metabolites 2-OHE2 and 4-OHE2 are closely associated with astrocyte senescence. (A) The flow chart of estradiol metabolism. (B) HPLC chromatogram of 2-OHE2 and 4-OHE2 in hypothalamic tissue of 10-month-old mice. (C) Dual-label immunofluorescence showing astrocytes (green) and p16 (red) with 2-OHE2 and 4-OHE2, white arrows representing γH2AX–positive astrocytes (left). Dual-label immunofluorescence showing astrocytes (green) and γ-H2AX (red) with 2-OHE2 and 4-OHE2, white arrows representing γ-H2AX–positive astrocytes (right). Scale bar= 100 μm. (D) Detection of p16 and p21 mRNA levels with 2-OHE2 and 4-OHE2 intervention. 2-OHE2 and 4-OHE2 respectively increased the expression of p16 and p21 in hypothalamic astrocytes (n=3). The p-value was determined by One-way ANOVA: ** p< 0.01.
PKA-CYP signaling mediates estradiol-induced senescence of hypothalamic astrocytes
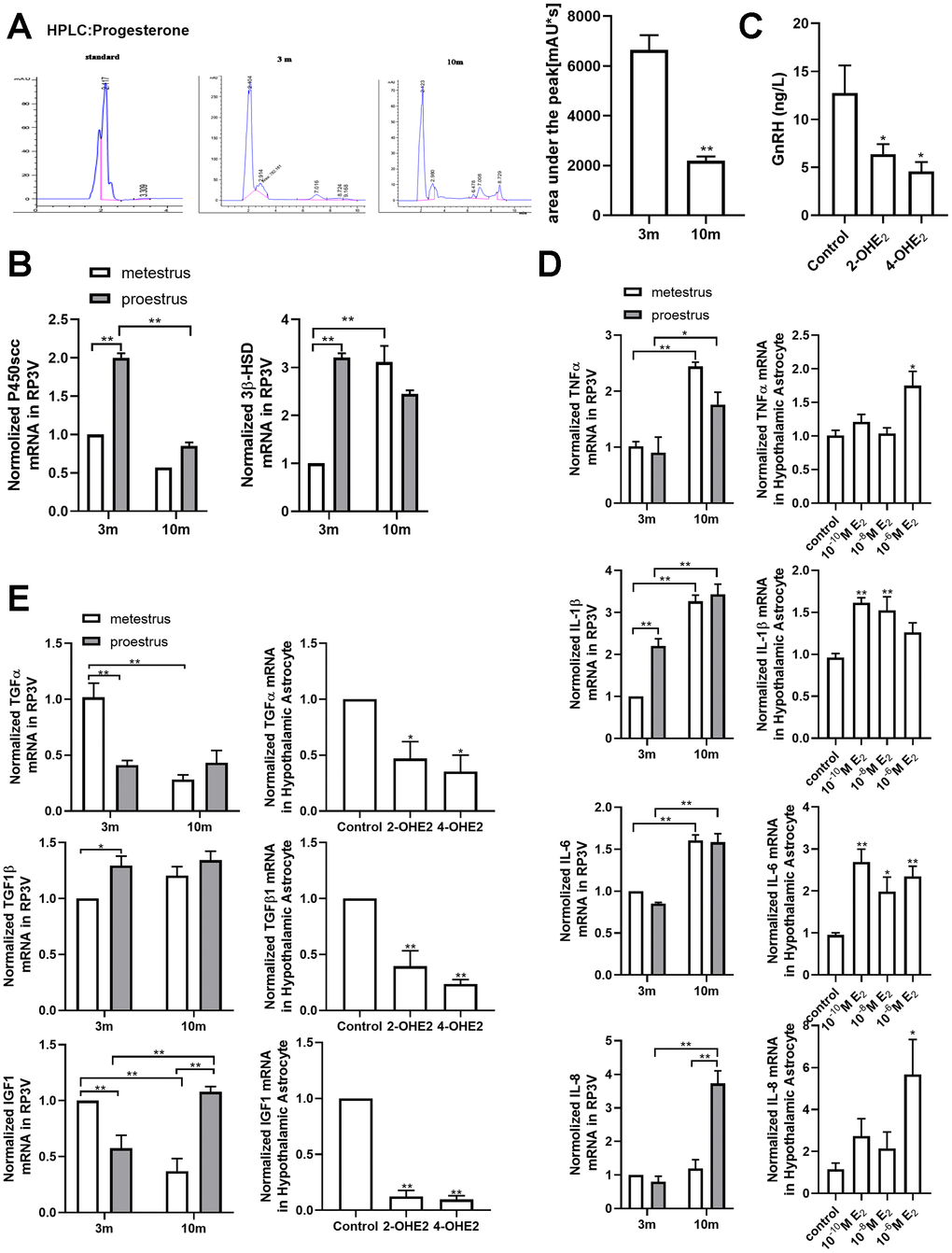
CYPs are the key enzymes in the metabolism of estradiol into hydroxyl estradiols. It is reported that estradiol activates the protein kinase A (PKA) pathway to promote neuroprogesterone synthesis in the hypothalamic astrocytes, and that PKA can enhance the expression of CYPs in tumor cell models [29, 30]. Therefore, the activation of the PKA-CYPs pathway was examined in both the in vivo animal model and the in vitro primary cultured hypothalamic astrocytes. In the young mice with regular estrous cycle, the phosphorylation of PKA in the hypothalamus was significantly higher during proestrus than during metestrus (Unpaired t test, df=10;t=10.04;p<0.01) (Figure 4A); Similarly, the mRNA level of the three subunits of the CYP gene (CYP1A1/CYP1A2/CYP1B1) during proestrus was also obviously higher than that during metestrus (Figure 4B). In the ovariectomized mice, the mRNA level of CYP1A1, CYP1A2, and CYP1B1 in the OVX group was significantly lower than that in the control and OVX+E2 groups, while no significant difference was evident between the control and OVX+E2 groups (Figure 4C). In primary cultured hypothalamic astrocytes, the estradiol treatment (10-6M) obviously improved the phosphorylation level of PKA (one-way ANOVA effect of treatment p<0.01, df=3,11, F=1.297) (Figure 4D) and the mRNA expression of CYP subunits (Figure 4E). In contrast, no similar phenomenon was observed in the primary cultured cortical astrocytes treated with estradiol (Supplementary Figure 2A). Moreover, the cultured hypothalamic astrocytes were further respectively treated with Forskolin, an agonist for PKA, and H-89, an inhibitor for PKA. A 24-hour treatment with H-89 (10μM) decreased the expression of CYPs; however, a 24-hour treatment with Forskolin (30μM) significantly upregulated CYPs level (Figure 4F). These data indicate that estradiol activates the PKA pathway and then up-regulates the expression of CYPs in hypothalamic astrocytes.
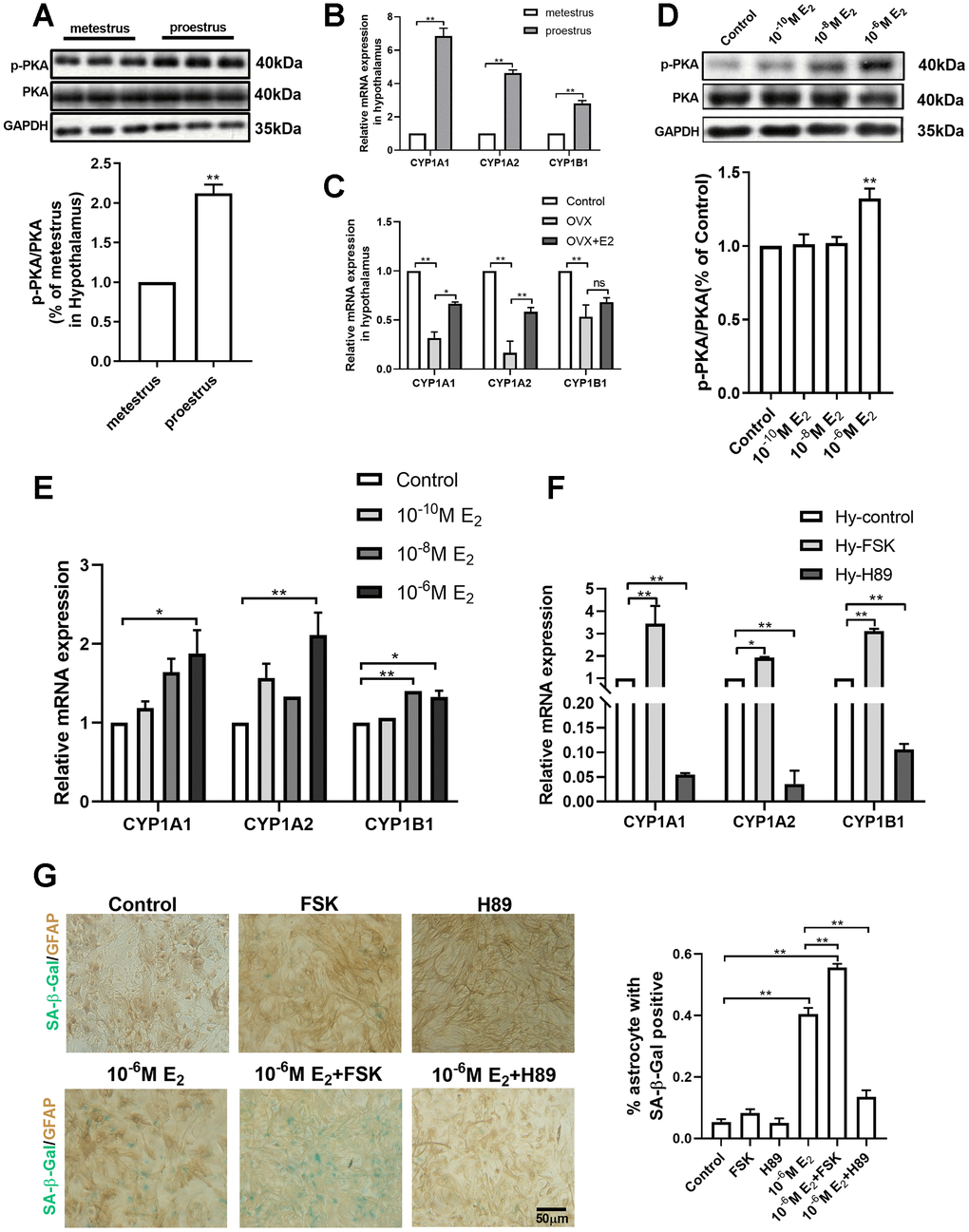
Figure 4. PKA-CYP signaling mediates estradiol-induced senescence of hypothalamic astrocytes. (A) The expression of PKA and p-PKA in hypothalamus during proestrus and metestrus at 3 months of age as determined by Western blotting (n = 6). (B) Effects of estrous cycle on the mRNA expression of CYP1A1, CYP1A2 and CYP1B1 gene in the hypothalamus as determined by qPCR. Metestrus vs. proestrus, n=3 (upper). (C) Effects of estradiol on the mRNA expression of CYP1A1, CYP1A2 and CYP1B1 gene in the hypothalamic tissue as determined by qPCR. OVX group vs. control group, OVX group vs. OVX+E2 group, n=3. The p-value of (A–C) was determined by Student’s t test,*p<0.05,** p< 0.01. (D) Expression of PKA and p-PKA in primary cultured hypothalamic astrocytes with the intervention of different estradiol concentrations as determined by Western blotting (n = 5). (E) Effects of estradiol on the mRNA expression of CYP1A1, CYP1A2 and CYP1B1 gene in primary cultured hypothalamic astrocytes with the intervention of different estradiol concentrations, as compared with the control, n= 3. (F) Effects of PKA activator (Forskolin, 10μM, 24h) and inhibitor (H89, 30μM, 24h) on the mRNA expression of CYP1A1, CYP1A2 and CYP1B1 gene in the primary cultured hypothalamic astrocytes, as compared with the control, n=3. (G) Dual-label immunohistochemistry showing astrocytes (brown) and SA-β-Gal staining (blue) with the effects of 10-6M estradiol, together with Forskolin and H89, respectively. Black arrows represent SA-β-Gal –positive astrocytes. n=3, scale bar=50μm. The p-value was determined by One-way ANOVA:*p<0.05, ** p< 0.01. FSK, i.e. forskolin.
To clarify the role of the PKA pathway in activating CYPs in the estradiol-induced senescence of hypothalamic astrocytes, the primary cultured hypothalamic astrocytes were respectively treated with the PKA inhibitor and agonist alone and with estradiol (10-6M). The interventions were divided into the following six groups: the vehicle group, 10-6M E2 group, H89 group (10μM, 24h), H89+10-6M E2 group, Forskolin group (30μM, 24h), and Forskolin+10-6M E2 group. After four consecutive 17β-estradiol interventions, no significant difference in the percentage of SA-β-Gal positive astrocytes was found among the vehicle group, H89 group and Forskolin group. Of note, the percentage of senescent astrocytes was prominently higher in Forskolin+10-6M E2 group than in Forskolin group and 10-6M E2 group. On the contrary, H89 treatment obviously decreased the E2-induced senescent astrocytes (one-way ANOVA effect of treatment p<0.01, df=5,23, F=1.668) (Figure 4G). Taken together, these results suggest that the activation of CYPs by PKA is a key pathway for the estradiol-induced senescence of hypothalamic astrocytes.
Discussion
This research found that astrocytes within the hypothalamic RP3V, not in the cortex and hippocampus, accumulate senescence-related markers with increasing age; that this cellular senescence is closely related to ovarian estradiol and compromises the regulation of progesterone synthesis and GnRH secretion. The molecular mechanism revealed that the PKA-CYPs signaling metabolized intracellular estradiol into 2-OHE2 and 4-OHE2, which were involved in the astrocyte senescence within RP3V.
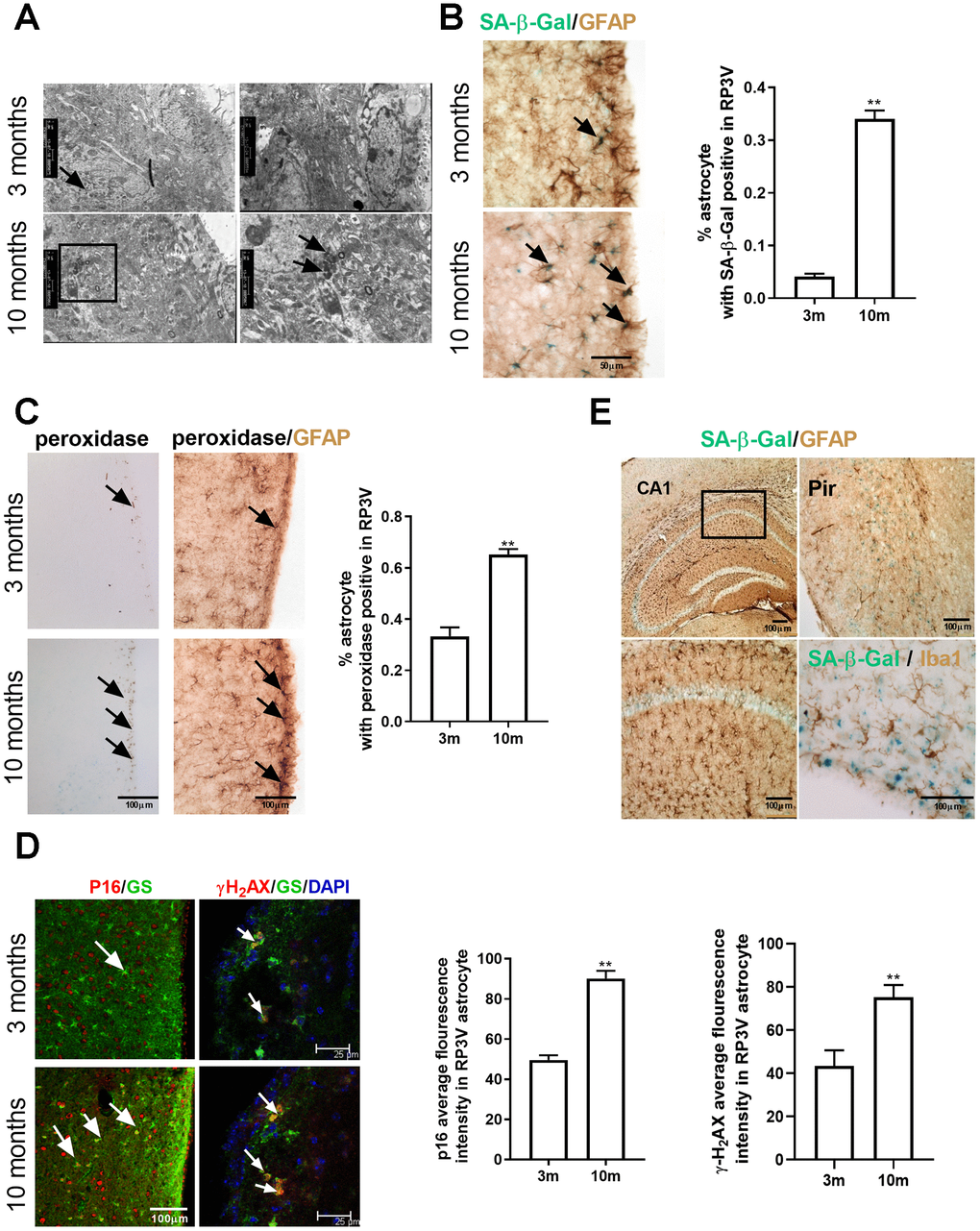
It has been reported that hypothalamic astrocytes facilitate the estrogen positive feedback [7, 8, 16, 33]. The current study confirms the relationship between the estrogen positive feedback and the aging of hypothalamic astrocytes from the following three aspects. The age-dependent changes of the senescent markers of astrocytes within RP3V in intact female mice were investigated (Supplementary Figure 1A). We found that the number of peroxidase staining and SA-β-Gal-positive astrocytes increased gradually from 3 months to 9 months of age, while almost no senescent astrocytes were observed at 1 month and 2 months of age. In rodents, reproductive function is fully developed at 3 months old, accompanied by regular estrous cycles. Therefore, we also tested the effect of ovarian-derived estradiol on the senescence of hypothalamic astrocytes. In the current study, the senescent characteristics of astrocytes in RP3V of the ovariectomized 3-month-old female mice were significantly reduced (Figure 2A, 2B). In addition, to mimic the positive feedback effect of estradiol in the body, the primary cultured hypothalamic astrocytes were repeatedly interfered with different concentrations of estradiol (10-10M, 10-8M, and 10-6M), in which 10-6M estradiol for 48h has been demonstrated to induce progesterone secretion in hypothalamic astrocytes [6] and 10-10M estradiol is equivalent to the basic estradiol level in the normal cycle [34, 35]. As we expected, four consecutive estradiol interventions induced the senescence of hypothalamic astrocytes (Figure 2C–2E) but the phenomenon was not found in the cultured astrocytes from the cortex (Supplementary Figure 1C–1D). These results suggest that ovarian estradiol is closely related to the senescence of hypothalamic astrocytes, which is consistent with previous reports [36, 37].
Estradiol promotes the synthesis of progesterone in astrocytes by activating the cAMP/PKA pathway. Consistent with literature reports [6, 8, 38], compared with that during metestrus, the phosphorylation of PKA and mRNA level of P450scc and 3β-HSD during proestrus significantly increased in the 3-month-old mice (Figures 4A and 5B). Therefore, we speculate that the PKA-CYPs pathway is involved in estradiol-induced senescence of hypothalamic astrocytes and put it to verification. In further experiments, estradiol significantly upregulated the phosphorylation of PKA and mRNA level of CYPs subunits in scenarios both in vivo (Figure 4A–4C) and in vitro (Figure 4D–4E). Moreover, the metabolization of estradiol into catechol estradiols by CYPs promoted oxidative damage to the hypothalamus. We found that 2-OHE2 and 4-OHE2 were present in the hypothalamus of the 10-month-old mice and that a low dose of 2-OHE2 or 4-OHE2 induced the senescence of cultured hypothalamic astrocytes (Figure 3). These findings are in line with the conclusion of the damage of catechol estradiols to DNA [27, 28]. Still, the regulation of PKA activity affected the estradiol-induced senescence of hypothalamic astrocytes (Figure 4G). In this study, we tried to use CYPs inhibitors to investigate whether it can inhibit the estradiol-induced astrocyte senescence. Unfortunately, CYPs are composed of multiple subtypes, and commercial CYPs inhibitors are currently not effective in inhibiting all subtypes. We expect to use CYPs blockers to prevent the senescence of hypothalamic astrocytes in the future.
Within more than two decades, many studies have shown that estradiol has a clear neuroprotective effect on AD, PD and stroke et al [39–42]. Our current conclusions concerning estradiol in the cortex do not contradict previous reports. In our study, ovarian estradiol selectively induced the senescence of hypothalamic astrocytes, not those in the hippocampus or cortex. The activation of the PKA pathway by estradiol not only mediates the process of progesterone synthesis but also up-regulates the activation of the CYPs pathway, promoting the catechin metabolism of estradiol. Of note, the activation of the PKA-CYPs pathway has not been found in the cortex during positive feedback effect of estradiol during metestrus and proestrus period (Supplementary Figure 2B, 2C). Further experimentation is needed to explore the underlying reasons for the differential activation of the PKA pathway in astrocytes from distinct brain regions.
What interests us most is the functional changes that may result from the senescent astrocytes. As expected, in the middle-aged mice, not only the level of progesterone but also the key enzyme of progesterone synthesis (P450scc) were significantly lower than those of the young mice. Another enzyme of progesterone synthesis (3β-HSD) was up-regulated instead, which may be a consequence of estradiol treatment [43]. Unfortunately, we have not been able to detect directly the progesterone level in the estradiol-induced senescent astrocytes, due to technical limitations, and used HPLC instead. When GT1-7 was incubated with the conditional culture medium of astrocytes (CCMA) that were pretreated by catechin estradiols, the level of GnRH secreted by GT1-7 was obviously reduced (Figure 5C). However, the pretreatment of CCMAs with the different concentrations of estradiol did not cut down the GnRH level (data not shown). We speculate that the degree of estradiol-induced senescence in astrocytes is not as serious as that by catechin estradiols (Supplementary Figure 3A). Similarly, we only detected a decrease of growth factors, TNF-α and IGF1, both in animal models of different ages and the cellular model receiving 2-OHE2 and 4-OHE2 interventions, but not in the cellular model receiving estradiol intervention. Studies have shown that a high expression of IL-6 and IL-1β can inhibit the secretion of GnRH in the hypothalamus, which in turn affects the reproductive function [32, 44]. Our study found that, regardless of proestrus or metestrus, the transcription levels of IL-1β, TNF-α, and IL-6 were significantly higher in 10-month-old mice than in their 3-month-old counterparts, respectively. These data suggest that the aging of astrocytes in the hypothalamus is implicated in the decline of GnRH neuronal activation and GnRH release.
In summary, this study shows that astrocytes within the hypothalamic RP3V accumulate senescence-related markers with increasing age, accompanied by a decreased progesterone production, reduced ability of GnRH neurons to secret GnRH, decreased production of growth factors, and increased levels of pro-inflammatory cytokines. The underlying mechanism involves the activation of PKA-CYPs pathway by ovarian estradiol, promoting the metabolization of estradiol into 2-OHE2 and 4-OHE2 and accelerating the astrocyte senescence within RP3V (Figure 6). The findings confirm that ovarian estradiol induces the senescence of hypothalamic astrocytes. The senescent astrocytes compromise the regulation of progesterone synthesis and GnRH secretion, which may contribute to aging-related declines in female reproductive function.
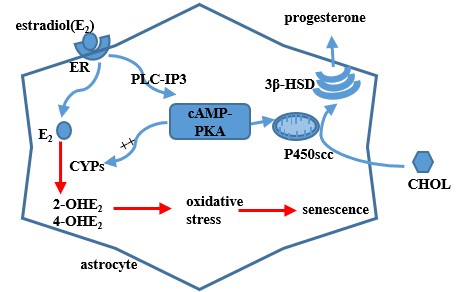
Figure 6. A model of estradiol action on hypothalamic astrocytes involving the progesterone production on the basis of present results and previous findings. Circulating estradiol acts on astrocytes that regulate progesterone (PROG) synthesis to activate the cAMP-PKA pathway. PKA can phosphorylate cytochrome P450scc and 3β-HSD, which regulate PROG production. The activated PKA pathway can also increase the expression of CYPs genes, which participate in the process of estradiol metabolism. Metabolic products of estradiol, 2-OHE2 and 4-OHE2, exert strong oxidative stress on astrocytes. ER, i.e. estrogen receptor; CHOL, i.e.cholesterol.
Materials and Methods
Animals and treatment protocols
All C57BL/6J female mice were purchased from Slack Animal Co (Shanghai, China). Experimental mice were housed (≤ 5 animals/cage) in a pathogen-free colony (IVC system, Tecniplast, Italy) and allowed free access to food and water. The colony was maintained under a 12-h light/12-h dark cycle with the temperature set at 24 °C. All protocols and procedures used in these studies were approved by the Institutional Animal Care and Use Committee of Fujian Medical University and in compliance with NIH’s Guidelines for the Care and Use of Laboratory Animals (NIH Publication No. 85-23 Rev. 1985). Estrous cycles of the young-aged mice (3–4 months old) and middle-aged mice (9–10 months old) were monitored by daily vaginal smears. In this study only mice having at least two consecutive cycles of a regular 4–5 day estrous period were included. The selected mice received, under anesthesia, a bilateral ovariectomy (OVX) or OVX plus a pellet containing 0.25mg of 17β-estradiol (OVX+E2, Innovative Research of America, #NE-121), which undergoes a 90-day continual release and was subcutaneously implanted in the posterior neck. The OVX+E2 group received another embedded 17β-estradiol sustained-release tablets 90 days later. Animals were transported to the operating room for tissue harvest at 9 months old.
Transmission electron microscopy
The mice were anesthetized with 10% chloral hydrate and injected with normal saline, and their brains were immediately removed. The hypothalamic area was dissected (1×1×1 mm), immersed in 2.5% glutaraldehyde buffer, fixed in 1% tetroxide, dehydrated in the gradient concentration ethanol, and finally embedded in Epon812. The targeted subregion was observed under a transmission electron microscope (EM208, Royal Philips Electronics, Netherlands).
Hypothalamus dissection and qPCR
The experiments were performed as to previously reported [4]. Briefly, animals were anaesthetized and brains were collected. The preoptic area anterior hypothalamus (POA-AH) for biochemical experiment. For POA-AH, the caudal border was made by a coronal cut just posterior to the entry point of the optic chiasm; the rostral border was exactly 3mm anterior. This coronal section (3 mm thick) was laid rostral side up on a chilled glass plate, then an isosceles triangle-shaped cut was made with its apex just under the midline of the corpus callosum and the 2 legs passing through the anterior commissure. Total RNA was extracted from the dissected hypothalamus and reversely transcribed into cDNA. The primer sets were presented in the Supplementary Table 1. Each reaction was performed in triplicate. Fold changes were calculated by 2-ΔΔt, where ΔCt =Ct (target gene) - Ct (actin/GAPDH) and Δ(ΔCt) =ΔCt (experimental groups) –mean ΔCt.
Senescence-associated β-galactosidase (SA-β-Gal) assay combined with immunohistochemistry
The experiments were performed as to previously reported [4]. Briefly, the dissected brains were post-fixed in 4 % paraformaldehyde, dehydrolyzed in 30 % sucrose, and then sliced into sections (40μm). For SA-β-Gal staining, sections were immersed in a SA-β-Gal staining solution. After incubation at 37 °C in the dark overnight, sections were washed and treated with 3 % H2O2 for 10 min, and then washed. They were next blocked in a blocking buffer for 60 min, then incubated with Anti-Iba1or anti-GFAP (Supplementary Table 2) in TBS for 24 h. They were then incubated with biotinylated second antibody at RT for 90 min. After being washed in TBS, these sections were further incubated in Vector Elite avid in peroxidase at RT for 1h. Sections were stained with DAB for 1–10 min, then cover-slipped with a permanent mounting medium, and detected under a microscope.
Peroxidase assay combined with immunohisto-chemistry
The sections were washed with TBS and then developed directly with DAB for 10 min. After another TBS wash, the brain sections were examined by immunohistochemistry.
Immunofluorescence
The experiments were performed as to previously reported [4]. Briefly, sections were washed and then blocked at RT for 1h. To detect astrocytes, the sections were incubated at 4 °C for 24 h with primary antibodies in TBS: polyclonal chicken anti-GFAP, polyclonal rabbit anti-GS, polyclonal rabbit anti-P16, and monoclonal mouse anti-γH2AX (Supplementary Table 2). The sections were then washed and incubated at RT for 1 h in a mixture of secondary antibodies. Afterwards, the sections were washed, mounted on glass slides, and then cover-slipped with prolong Gold anti-fade reagent.
Hypothalamic primary astrocyte cultures and conditioned medium
The astrocytes were obtained from the brains of neonatal mice (d1-2, C57BL/6J). Hypothalamus and cortices were dissected, freed of meninges in an ice-cold salt ion buffer. Digestive enzyme was compounded using Papain Suspension (Worthington, Lakewood, NJ) and salt ion buffer. The separated brain tissue was placed in the filtered digestive enzyme and dissolved in an incubator with 5% CO2 at 37°C for 20min. Then, enzyme activity was blocked by an excess of fetal bovine serum (FBS, HyClone, Utah, USA). All of the enzymes were removed and tissues were mechanically dissociated in a DME/F12 medium (HyClone, Utah, USA) containing 10% FBS and 1% penicillin-streptomycin (HyClone, Utah, USA). After mechanical dissociation, cells were seeded and maintained in an incubator with 5% CO2 at 37°C for 2 weeks in DME/F-12 containing 10% FBS and 1% penicillin-streptomycin in poly-L-lysine coated 6-well or 24-well plates. The culture medium was refreshed twice a week. The purity of astrocytes was about 95% of GFAP-positive cells.
Prior to drug treatment, all astrocytes were subjected to steroid starvation for 24 hours in DME/F12 (supplemented with 5% of fetal bovine serum separated by charcoal; Sigma-Aldrich, F6765, St. Louis, MO). After steroid-starvation, some cultures were respectively treated for 48h with 17β-estradiol (10-10M, 10-8M, 10-6M; Sigma-Aldrich, E2758, St. Louis, MO), Forskolin (10μM, 24h), H89 (30μM, 24h), 17β-estradiol (10-6M) + Forskolin, 17β-estradiol (10-6M) + H89, or estradiol-free DME/F12 culture media. The process of steroid starvation and drug treatment was repeated 4 times. Other cultures were respectively treated with 2-OHE2 (20nM) and 4-OHE2 (20nM) for 24h before the medium refreshment. After 36h, the collected media were cultured with the GT1-7 lines for 48h.
Sample preparation and HPLC analysis
The weighed sample was ground and suspended in a mixture of water and methanol (1:1). The sample was homogenized and then centrifuged by high-speed centrifugation in a vacuum. The residue was dissolved in water and methanol (1:1), filtered through a 0.2μ ultracentrifuge filter (Merck Millipore, MA, USA) and prepared for HPLC analysis. The process was repeated in duplicate during HPLC analysis. Pure standards were used to optimize the HPLC conditions prior to sample analysis. HPLC was performed by Shanghai Institute of Organic Chemistry, Chinese Academy of Science.
Statistical analysis
Data were presented as means ± standard error (SEM) of a percent relative ratio. Statistical comparisons between 2 independent groups were analyzed by the unpaired Student’s t-test; comparisons among 3 or more independent groups, were by a one-way analysis of variance (ANOVA), together with Kruskal-Wallis test where appropriate. Data were processed using GraphPad Software. p < 0.05 was considered statistically significant.
Author Contributions
Xiaoman Dai executed immunohistochemistry and molecular experiments, and wrote the manuscript; Luyan Hong performed the cell experiments and analyzed all the data; Hui Shen contributed to the animal maintenance; Qiang Du preformed the animal surgery; Qinyong Ye provided substantial contributions to development of methodology; Jing Zhang and Xiaochun Chen conceived and designed the project, and prepared and revised the manuscript; and all authors read and commented on the manuscript.
Conflicts of Interest
All authors have no potential conflicts of interests or financial issues to declare.
Funding
The study was funded by National Natural Science Foundation of China to Jing Zhang (No.81671400) and Fujian Province Health and Family Planning Youth Research Project to Xiaoman Dai (No.2018-1-45).
References
- 1. Cooper RL, Conn PM, Walker RF. Characterization of the LH surge in middle-aged female rats. Biol Reprod. 1980; 23:611–15. https://doi.org/10.1095/biolreprod23.3.611 [PubMed]
- 2. Scarbrough K, Wise PM. Age-related changes in pulsatile luteinizing hormone release precede the transition to estrous acyclicity and depend upon estrous cycle history. Endocrinology. 1990; 126:884–90. https://doi.org/10.1210/endo-126-2-884 [PubMed]
- 3. Zhang J, Yang LM, Pan XD, Lin N, Chen XC. Increased vesicular γ-GABA transporter and decreased phosphorylation of synapsin I in the rostral preoptic area is associated with decreased gonadotrophin-releasing hormone and c-Fos coexpression in middle-aged female mice. J Neuroendocrinol. 2013; 25:753–61. https://doi.org/10.1111/jne.12050 [PubMed]
- 4. Zhang J, Yang L, Lin N, Pan X, Zhu Y, Chen X. Aging-related changes in RP3V kisspeptin neurons predate the reduced activation of GnRH neurons during the early reproductive decline in female mice. Neurobiol Aging. 2014; 35:655–68. https://doi.org/10.1016/j.neurobiolaging.2013.08.038 [PubMed]
- 5. Lloyd JM, Hoffman GE, Wise PM. Decline in immediate early gene expression in gonadotropin-releasing hormone neurons during proestrus in regularly cycling, middle-aged rats. Endocrinology. 1994; 134:1800–05. https://doi.org/10.1210/endo.134.4.8137745 [PubMed]
- 6. Micevych PE, Chaban V, Ogi J, Dewing P, Lu JK, Sinchak K. Estradiol stimulates progesterone synthesis in hypothalamic astrocyte cultures. Endocrinology. 2007; 148:782–89. https://doi.org/10.1210/en.2006-0774 [PubMed]
- 7. Mohr MA, Wong AM, Tomm RJ, Soma KK, Micevych PE. Pubertal development of estradiol-induced hypothalamic progesterone synthesis. Horm Behav. 2019; 111:110–13. https://doi.org/10.1016/j.yhbeh.2018.12.007 [PubMed]
- 8. Micevych P, Sinchak K. Estradiol regulation of progesterone synthesis in the brain. Mol Cell Endocrinol. 2008; 290:44–50. https://doi.org/10.1016/j.mce.2008.04.016 [PubMed]
- 9. Prevot V, Croix D, Bouret S, Dutoit S, Tramu G, Stefano GB, Beauvillain JC. Definitive evidence for the existence of morphological plasticity in the external zone of the median eminence during the rat estrous cycle: implication of neuro-glio-endothelial interactions in gonadotropin-releasing hormone release. Neuroscience. 1999; 94:809–19. https://doi.org/10.1016/S0306-4522(99)00383-8 [PubMed]
- 10. Baroncini M, Allet C, Leroy D, Beauvillain JC, Francke JP, Prevot V. Morphological evidence for direct interaction between gonadotrophin-releasing hormone neurones and astroglial cells in the human hypothalamus. J Neuroendocrinol. 2007; 19:691–702. https://doi.org/10.1111/j.1365-2826.2007.01576.x [PubMed]
- 11. Prevot V, Dehouck B, Poulain P, Beauvillain JC, Buée-Scherrer V, Bouret S. Neuronal-glial-endothelial interactions and cell plasticity in the postnatal hypothalamus: implications for the neuroendocrine control of reproduction. Psychoneuroendocrinology. 2007 (Suppl 1); 32:S46–51. https://doi.org/10.1016/j.psyneuen.2007.03.018 [PubMed]
- 12. Moeller-Gnangra H, Ernst J, Pfeifer M, Heger S. ErbB4 point mutation in CU3 inbred rats affects gonadotropin-releasing-hormone neuronal function via compromised neuregulin-stimulated prostaglandin E2 release from astrocytes. Glia. 2019; 67:309–20. https://doi.org/10.1002/glia.23541 [PubMed]
- 13. Gallo F, Morale MC, Farinella Z, Avola R, Marchetti B. Growth factors released from astroglial cells in primary culture participate in the cross talk between luteinizing hormone-releasing hormone (LHRH) neurons and astrocytes. Effects on LHRH neuronal proliferation and secretion. Ann N Y Acad Sci. 1996; 784:513–16. https://doi.org/10.1111/j.1749-6632.1996.tb16272.x [PubMed]
- 14. Galbiati M, Zanisi M, Messi E, Cavarretta I, Martini L, Melcangi RC. Transforming growth factor-beta and astrocytic conditioned medium influence luteinizing hormone-releasing hormone gene expression in the hypothalamic cell line GT1. Endocrinology. 1996; 137:5605–09. https://doi.org/10.1210/endo.137.12.8940390 [PubMed]
- 15. Buchanan CD, Mahesh VB, Brann DW. Estrogen-astrocyte-luteinizing hormone-releasing hormone signaling: a role for transforming growth factor-beta(1). Biol Reprod. 2000; 62:1710–21. https://doi.org/10.1095/biolreprod62.6.1710 [PubMed]
- 16. Micevych P, Soma KK, Sinchak K. Neuroprogesterone: key to estrogen positive feedback? Brain Res Brain Res Rev. 2008; 57:470–80. https://doi.org/10.1016/j.brainresrev.2007.06.009 [PubMed]
- 17. Sohal RS, Brunk UT. Lipofuscin as an indicator of oxidative stress and aging. Adv Exp Med Biol. 1989; 266:17–26. https://doi.org/10.1007/978-1-4899-5339-1_2 [PubMed]
- 18. Geng YQ, Guan JT, Xu XH, Fu YC. Senescence-associated beta-galactosidase activity expression in aging hippocampal neurons. Biochem Biophys Res Commun. 2010; 396:866–69. https://doi.org/10.1016/j.bbrc.2010.05.011 [PubMed]
- 19. Krishnamurthy J, Torrice C, Ramsey MR, Kovalev GI, Al-Regaiey K, Su L, Sharpless NE. Ink4a/Arf expression is a biomarker of aging. J Clin Invest. 2004; 114:1299–307. https://doi.org/10.1172/JCI22475 [PubMed]
- 20. Zwart SR, Jessup JM, Ji J, Smith SM. Saturation diving alters folate status and biomarkers of DNA damage and repair. PLoS One. 2012; 7:e31058. https://doi.org/10.1371/journal.pone.0031058 [PubMed]
- 21. Scarpato R, Verola C, Fabiani B, Bianchi V, Saggese G, Federico G. Nuclear damage in peripheral lymphocytes of obese and overweight Italian children as evaluated by the gamma-H2AX focus assay and micronucleus test. FASEB J. 2011; 25:685–93. https://doi.org/10.1096/fj.10-168427 [PubMed]
- 22. Kuo J, Micevych P. Neurosteroids, trigger of the LH surge. J Steroid Biochem Mol Biol. 2012; 131:57–65. https://doi.org/10.1016/j.jsbmb.2012.01.008 [PubMed]
- 23. Micevych P, Bondar G, Kuo J. Estrogen actions on neuroendocrine glia. Neuroendocrinology. 2010; 91:211–22. https://doi.org/10.1159/000289568 [PubMed]
- 24. Nequin LG, Alvarez J, Schwartz NB. Measurement of serum steroid and gonadotropin levels and uterine and ovarian variables throughout 4 day and 5 day estrous cycles in the rat. Biol Reprod. 1979; 20:659–70. https://doi.org/10.1095/biolreprod20.3.659 [PubMed]
- 25. Kunzler J, Youmans KL, Yu C, Ladu MJ, Tai LM. APOE modulates the effect of estrogen therapy on Aβ accumulation EFAD-Tg mice. Neurosci Lett. 2014; 560:131–36. https://doi.org/10.1016/j.neulet.2013.12.032 [PubMed]
- 26. Xing Y, Jia JP, Ji XJ, Tian T. Estrogen associated gene polymorphisms and their interactions in the progress of Alzheimer’s disease. Prog Neurobiol. 2013; 111:53–74. https://doi.org/10.1016/j.pneurobio.2013.09.006 [PubMed]
- 27. Cavalieri E, Chakravarti D, Guttenplan J, Hart E, Ingle J, Jankowiak R, Muti P, Rogan E, Russo J, Santen R, Sutter T. Catechol estrogen quinones as initiators of breast and other human cancers: implications for biomarkers of susceptibility and cancer prevention. Biochim Biophys Acta. 2006; 1766:63–78. https://doi.org/10.1016/j.bbcan.2006.03.001 [PubMed]
- 28. Li KM, Todorovic R, Devanesan P, Higginbotham S, Köfeler H, Ramanathan R, Gross ML, Rogan EG, Cavalieri EL. Metabolism and DNA binding studies of 4-hydroxyestradiol and estradiol-3,4-quinone in vitro and in female ACI rat mammary gland in vivo. Carcinogenesis. 2004; 25:289–97. https://doi.org/10.1093/carcin/bgg191 [PubMed]
- 29. Tsuchiya Y, Nakajima M, Takagi S, Katoh M, Zheng W, Jefcoate CR, Yokoi T. Binding of steroidogenic factor-1 to the regulatory region might not be critical for transcriptional regulation of the human CYP1B1 gene. J Biochem. 2006; 139:527–34. https://doi.org/10.1093/jb/mvj055 [PubMed]
- 30. Oesch-Bartlomowicz B, Oesch F. Cytochrome-P450 phosphorylation as a functional switch. Arch Biochem Biophys. 2003; 409:228–34. https://doi.org/10.1016/S0003-9861(02)00558-1 [PubMed]
- 31. Sarchielli E, Comeglio P, Squecco R, Ballerini L, Mello T, Guarnieri G, Idrizaj E, Mazzanti B, Vignozzi L, Gallina P, Maggi M, Vannelli GB, Morelli A. Tumor Necrosis Factor-α Impairs Kisspeptin Signaling in Human Gonadotropin-Releasing Hormone Primary Neurons. J Clin Endocrinol Metab. 2017; 102:46–56. https://doi.org/10.1210/jc.2016-2115 [PubMed]
- 32. Igaz P, Salvi R, Rey JP, Glauser M, Pralong FP, Gaillard RC. Effects of cytokines on gonadotropin-releasing hormone (GnRH) gene expression in primary hypothalamic neurons and in GnRH neurons immortalized conditionally. Endocrinology. 2006; 147:1037–43. https://doi.org/10.1210/en.2005-0729 [PubMed]
- 33. Stephens SB, Tolson KP, Rouse ML
Jr , Poling MC, Hashimoto-Partyka MK, Mellon PL, Kauffman AS. Absent Progesterone Signaling in Kisspeptin Neurons Disrupts the LH Surge and Impairs Fertility in Female Mice. Endocrinology. 2015; 156:3091–97. https://doi.org/10.1210/en.2015-1300 [PubMed] - 34. Preuss TG, Gurer-Orhan H, Meerman J, Ratte HT. Some nonylphenol isomers show antiestrogenic potency in the MVLN cell assay. Toxicol In Vitro. 2010; 24:129–34. https://doi.org/10.1016/j.tiv.2009.08.017 [PubMed]
- 35. Ropero AB, Pang Y, Alonso-Magdalena P, Thomas P, Nadal A. Role of ERβ and GPR30 in the endocrine pancreas: A matter of estrogen dose. Steroids. 2012; 77:951–58. https://doi.org/10.1016/j.steroids.2012.01.015 [PubMed]
- 36. Mydlarski MB, Liberman A, Schipper HM. Estrogen induction of glial heat shock proteins: implications for hypothalamic aging. Neurobiol Aging. 1995; 16:977–81. https://doi.org/10.1016/0197-4580(95)02018-7 [PubMed]
- 37. Swartz WJ, Eroschenko VP. Neonatal exposure to technical methoxychlor alters pregnancy outcome in female mice. Reprod Toxicol. 1998; 12:565–73. https://doi.org/10.1016/S0890-6238(98)00041-0 [PubMed]
- 38. Chen C, Kuo J, Wong A, Micevych P. Estradiol modulates translocator protein (TSPO) and steroid acute regulatory protein (StAR) via protein kinase A (PKA) signaling in hypothalamic astrocytes. Endocrinology. 2014; 155:2976–85. https://doi.org/10.1210/en.2013-1844 [PubMed]
- 39. Lan YL, Zhao J, Li S. Update on the neuroprotective effect of estrogen receptor alpha against Alzheimer’s disease. J Alzheimers Dis. 2015; 43:1137–48. https://doi.org/10.3233/JAD-141875 [PubMed]
- 40. Morale MC, Serra PA, L’episcopo F, Tirolo C, Caniglia S, Testa N, Gennuso F, Giaquinta G, Rocchitta G, Desole MS, Miele E, Marchetti B. Estrogen, neuroinflammation and neuroprotection in Parkinson’s disease: glia dictates resistance versus vulnerability to neurodegeneration. Neuroscience. 2006; 138:869–78. https://doi.org/10.1016/j.neuroscience.2005.07.060 [PubMed]
- 41. Liu R, Yang SH. Window of opportunity: estrogen as a treatment for ischemic stroke. Brain Res. 2013; 1514:83–90. https://doi.org/10.1016/j.brainres.2013.01.023 [PubMed]
- 42. Engler-Chiurazzi EB, Brown CM, Povroznik JM, Simpkins JW. Estrogens as neuroprotectants: estrogenic actions in the context of cognitive aging and brain injury. Prog Neurobiol. 2017; 157:188–211. https://doi.org/10.1016/j.pneurobio.2015.12.008 [PubMed]
- 43. Goodson JL, Saldanha CJ, Hahn TP, Soma KK. Recent advances in behavioral neuroendocrinology: insights from studies on birds. Horm Behav. 2005; 48:461–73. https://doi.org/10.1016/j.yhbeh.2005.04.005 [PubMed]
- 44. Kang SS, Kim SR, Leonhardt S, Jarry H, Wuttke W, Kim K. Effect of interleukin-1beta on gonadotropin-releasing hormone (GnRH) and GnRH receptor gene expression in castrated male rats. J Neuroendocrinol. 2000; 12:421–29. https://doi.org/10.1046/j.1365-2826.2000.00466.x [PubMed]