Introduction
HCC is one of the most common malignant tumors of the digestive system [1]. According to the latest global cancer statistics, both the morbidity and mortality of liver cancer rank third among all tumors [2, 3]. However, most HCC patients, in fact, cannot be confirmed until tumors at various clinical stages metastasize to distinct organs, including intrahepatic and extrahepatic metastasis, posing recurrence and mortality risks to postoperative patients [4–6]. Therefore, improving the early diagnosis of HCC followed by timely targeted treatments is conducive to reducing the likelihood of metastasis in the patients. The first priority is the improvement of early detection biomarkers, so the survival of HCC patients can be extended with improved prognosis [7].
The CTA family is a group of antigens with strict tissue specificity, which is only expressed in the germinal epithelium of testis and some cancerous tissues in humans [8, 9]. It can be used as a putative biomarker for tumor diagnosis and the efficacy evaluation of immunotherapy [10–12]. LDH-C4, also known as LDHC or LDHX and encoded by LDHC mRNA, is the initially identified LDH isoenzyme only expressed in testis and tumors [13–15]. Human LDHC is located in chromosome 11p15.3-15.5, of which the full-length mRNA is 1179 bp with an ORF of 999 bp that encodes a 35 kDa C subunit. The polymerization of a homologous series of four identical C subunits can form the tetrameric protein, LDH-C4 zymoprotein, with catalytic activity [16]. LDH-C4 as a member of the CTA family features tissue-specific expressions only in the mature testis and sperm of healthy human males, which provides sufficient energy for sperm activities and is closely related to sperm development and motility physiologically [13–15].
Currently, few studies report biofunctions of LDH-C4 in tumor cells, and some find that LDHC mRNA is expressed in various tumors with positive rates of expressions of its spliceosomes up to 47% in lung cancer, 44% in melanoma, 35% in breast cancer and 15% in colon cancer [17]. Therefore, it is speculated that LDHC can be involved in the occurrence and development of these tumors. Other studies show that the positive rate of LDHC mRNA expression is 25% in non-small cell lung cancer (NSCLC) tissues, which is similar to the expression levels in adenocarcinoma and squamous cell carcinoma [18]. Wang et al. find that LDHC mRNA and its protein levels are up-regulated in renal cell carcinoma (RCC), and patients with positive LDHC expressions have a worse prognosis [19].
Exosome as a kind of extracellular vesicle (EV) with a diameter of 30-150 nm can be released by all living cells and also be detected in a tumor microenvironment [20, 21]. The latest evidences show that exosomes participate in the tumorigenesis by triggering angiogenesis and metastasis and suppressing anti-tumor immunity [22, 23]. Circulating exosomes in liquid biopsies can be used as non-invasive biomarkers for early detection, diagnosis and efficacy evaluation for cancer patients [24, 25]. In our previous work, IHC staining has been carried out to determine LDH-C4 expressions in 145 breast cancer tissue microarrays, and expression levels of peripheral serum and exosomal LDHC mRNAs in 75 breast cancer patients have been quantitated using RT-PCR analysis. The results preliminarily show that serum and exosomal LDHC mRNA can be utilized as an effective biomarker of breast cancer [26]. In this study, expressions of serum and exosomal LDHC mRNAs in stages I-II HCC were explored using qRT-PCR, and levels of LDH-C4 in HCC tissues were quantitated by high-throughput tissue microarray and IHC analyses, and correlations between LDH-C4 expression and clinical pathological characteristics and between the expression and the prognosis of HCC patients were analyzed.
Results
Identification of isolated vesicles
The extraction process was summarized in Figure 1. TEM showed that the size of exosomes derived from serum ranged from 30 to 150 nm in diameter and featured small vesicles with membrane structures, which were consistent with the morphological characteristics of exosomes (Figure 2A). To confirm whether exosomes were successfully isolated and purified, expressions of exosome marker proteins CD9 and CD63 were determined by western blot. The results showed that CD9 and CD63 could be detected in the eluate containing serum exosomes of both HCC patients and healthy controls (Figure 2B). This suggested that exosomes extraction was successful.
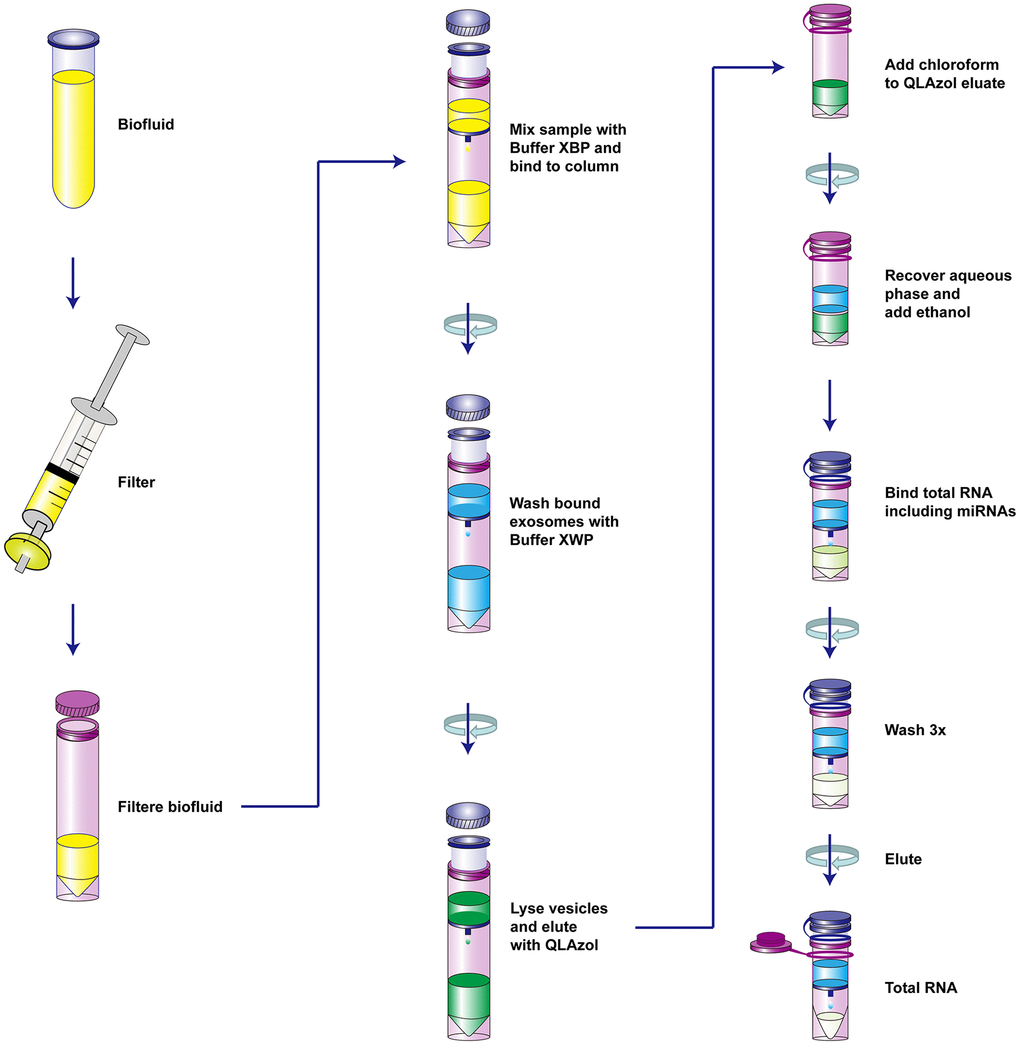
Figure 1. The schematic diagram of the isolation of serum vesicles and RNA extraction.
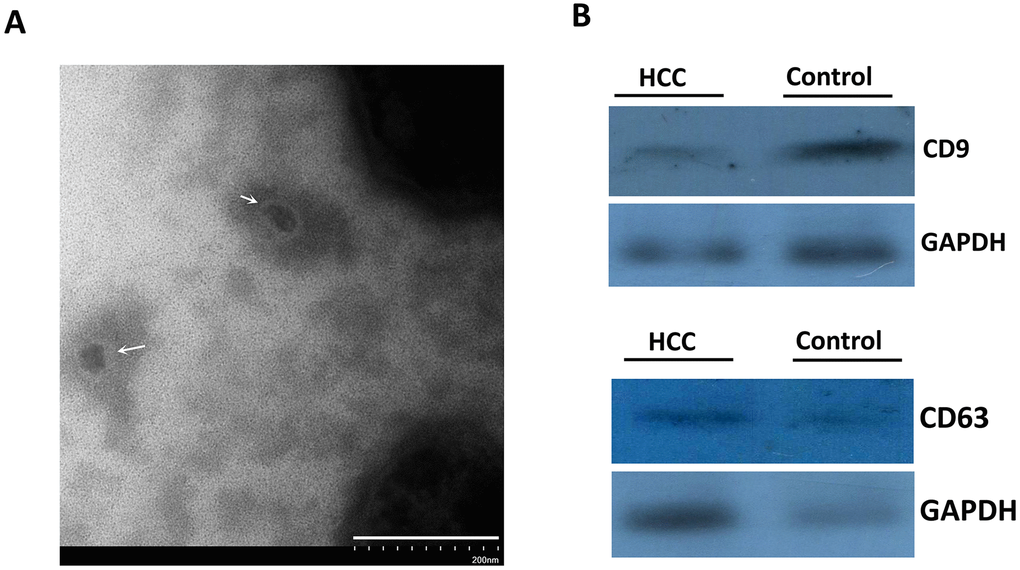
Figure 2. The identification of isolated vesicles in the serum using TEM and immunoblotting. (A) The morphology of serum exosomes under TEM. The white arrows indicated the isolated vesicles. (B) CD9 and CD63 proteins representing exosome markers were detected using western blot.
Serum and exosomal LDHC acted as an early diagnostic biomarker for HCC
The qRT-PCR analysis showed that positive rates of LDHC mRNA expressions in serum and exosomes were 68% (34 / 50) and 60% (30 / 50) in early-stage HCC patients, and 18% (18 / 100) and 17% (17 / 100) in healthy controls, respectively. The results also showed that the average expression levels of serum and exosomal LDHC mRNAs in the preliminary diagnosis group were 4.64- and 14.28-fold higher than those in the healthy control group, respectively (Figure 3A, 3B, 3D, 3E). ROC curve analysis showed that the sensitivity, specificity and AUC of serum LDHC mRNA in distinguishing early-stage HCC patients from healthy controls were 83.3%, 76.5%, and 0.8382 (youden index = 0.598), respectively (Figure 3C); and the AUC of exosomal LDHC in distinguishing early-stage HCC patients from healthy controls reached 0.9451, under which the sensitivity and specificity were estimated to be 88.2% and 93.3%, respectively (youden index = 0.816) (Figure 3F). The above results suggested that serum and exosomal LDHC could be considered as a promising biomarker for early diagnosis of HCC.
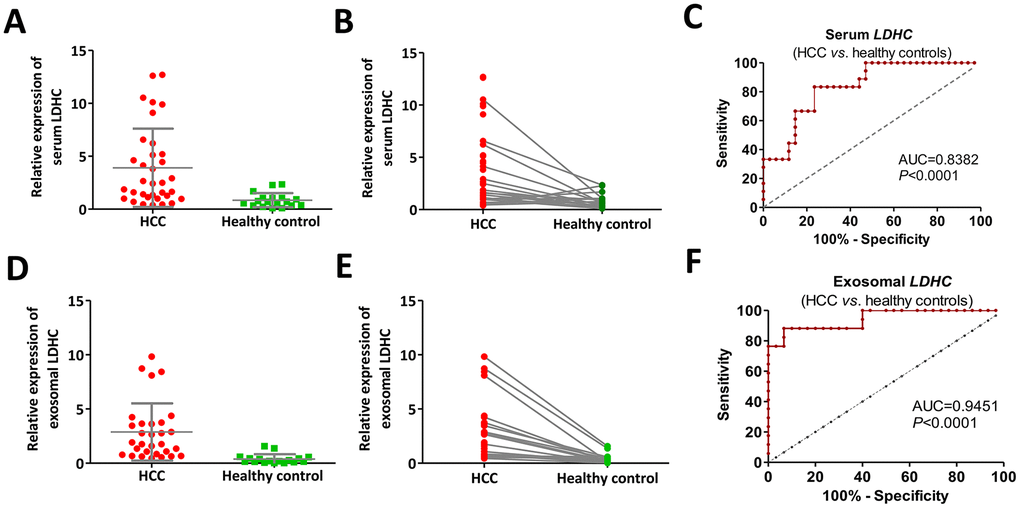
Figure 3. Cancer-specific pattern of serum and exosomal LDHC expressions in early-stage HCC determined its diagnostic implication. (A, B) Serum LDHC mRNA levels were significantly elevated in early-stage HCC serum samples compared with healthy individuals. (C) The ROC curve of serum LDHC mRNA for the identification of early-stage HCC. (D, E) Expressions of serum exosomal LDHC were elevated in early-stage HCC cases. (F) The ROC curve of serum exosomal LDHC in the diagnosis of early-stage HCC.
The correlations between the expression of LDHC in serum and exosomes and the clinicopathological characteristics of HCC patients
The correlation analysis showed that serum LDHC levels were positively correlated with the levels of AFP (r2 = 0.8295, P < 0.0001) and PIVKA-II (r2 = 0.3639,P = 0.0011) (Figure 4A, 4B). Exosomal LDHC levels were correlated with the levels of AFP (r2 = 0.7455, P < 0.0001) and PIVKA-II (r2 = 0.2665, P < 0.0001) in HCC cases (Figure 4F, 4G). However, there were nonsignificant correlations between serum and exosomal LDHC levels and clinicopathological indicators such as gender, age, tumor size (all P > 0.05, data not shown), as well as serum levels of carcino-embryonic antigen (CEA), Golgi protein 73 (GP-73), and glycoprotein antigen 199 (CA19-9) (Figure 4C–4E, 4H–4J).
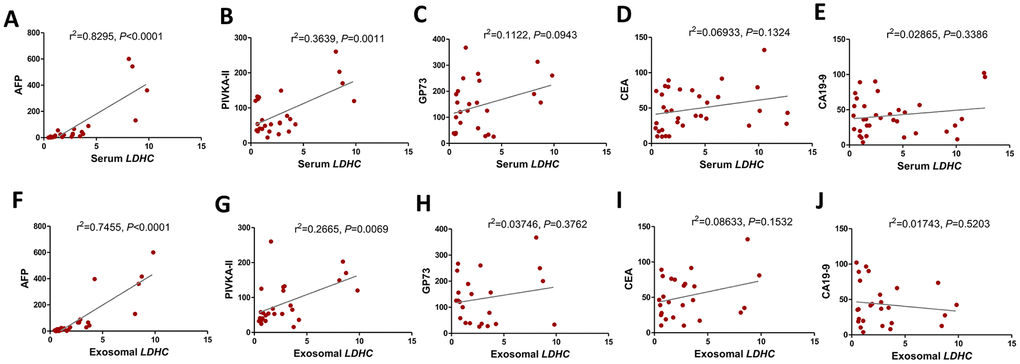
Figure 4. Serum and exosomal LDHC expressions were correlated with clinicopathological characteristics in HCC. Correlations between serum LDHC expressions and clinical indices encompassing (A) AFP, (B) PIVKA-II, (C) GP-73, (D) CEA, and (E) CA19-9 were analyzed. Serum exosomal LDHC expressions were associated with (F) AFP, and (G) PIVKA-II, but not correlated with the levels of (H) GP-73, (I) CEA, and (J) CA19-9 in patients with HCC.
Values of serum and exosomal LDHC in efficacy evaluation and recurrence monitoring
To explore the effects of LDHC mRNA on efficacy evaluation and recurrence monitoring, pre- and post-treatment LDHC mRNA expressions in the serum and exosomes of HCC patients were quantitated. It was found that the expression level of serum LDHC in the treatment group was significantly lower than that in the preliminary diagnosis group (AUC = 0.7036), while the average expression level of serum LDHC in the recurrence group was significantly higher than that in the treatment group (AUC = 0.9839) (Figure 5A–5D). Similarly, serum exosomal LDHC levels in the preliminary diagnosis group were significantly higher than the levels in the treatment group (AUC = 0.7113), and serum exosomal LDHC levels in the recurrence group were significantly higher than those in the treatment group (AUC = 0.9538) (Figure 5E–5H). These results suggested serum and exosomal LDHC could be used as an effective indicator for the evaluation of the efficacy of HCC-related treatments and recurrence prediction of patients.
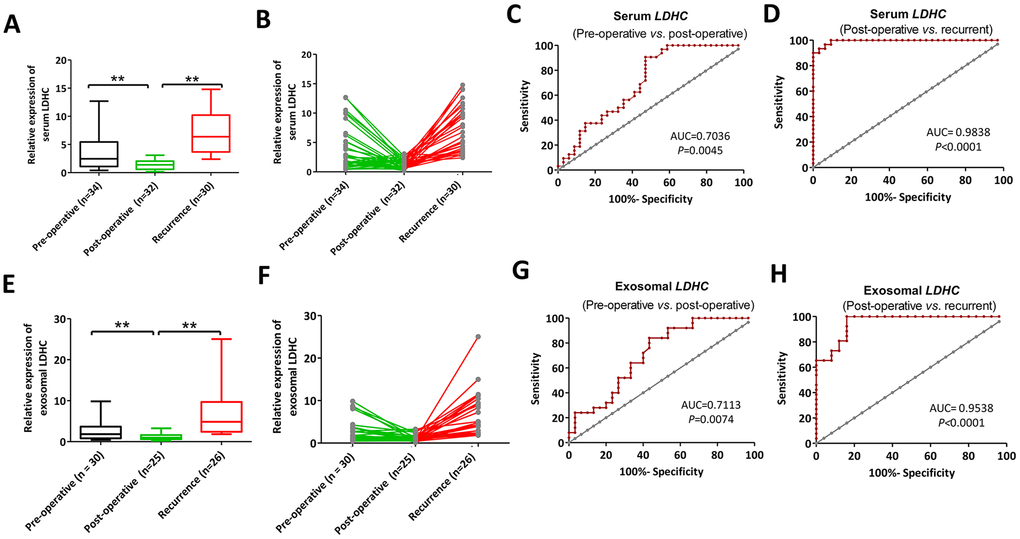
Figure 5. Serum (A, B) and exosomal (C, D) LDHC expressions in pre-operative, post-operative and recurrent patients with HCC. ROC curves of serum and exosomal LDHC mRNA expressions in the distinction between (E–H) pre-operative, post-operative, and recurrent cases. **P<0.01 vs. post-operative.
LDH-C4 as a prognostic marker of HCC
It was found that LDH-C4 was mainly localized in the cytoplasm of HCC cells, which was significantly up-regulated in HCC tissues (Figure 6A). The proportion of LDH-C4 high expression (+ + / + + +) in HCC tissues accounted for 55.84% (43 / 77), which was significantly higher than that in para-carcinoma tissues (26.32%, 20/76) (P < 0.0001). The correlation analysis showed that LDH-C4 levels were positively correlated with clinical stage (P = 0.002) and mass size (P = 0.023) of HCC (Table 1), but not related to KI-67, p53 and CD34 levels (Figure 6B). Moreover, LDH-C4 staining score was higher in stage 0-II HCC patients than that in stage III-IV HCC patients (Figure 6C). The Kaplan-Meier analysis showed that the overall survival of patients in the LDH-C4 low expression group was significantly prolonged compared with the LDH-C4 high expression group, while the prognosis of patients in the LDH-C4 high expression group was worse than that in the low expression group (P < 0.001) (Figure 6D). The COX regression model analysis showed that LDH-C4 was an independent risk factor for HCC prognosis (P = 0.016) (Table 2).
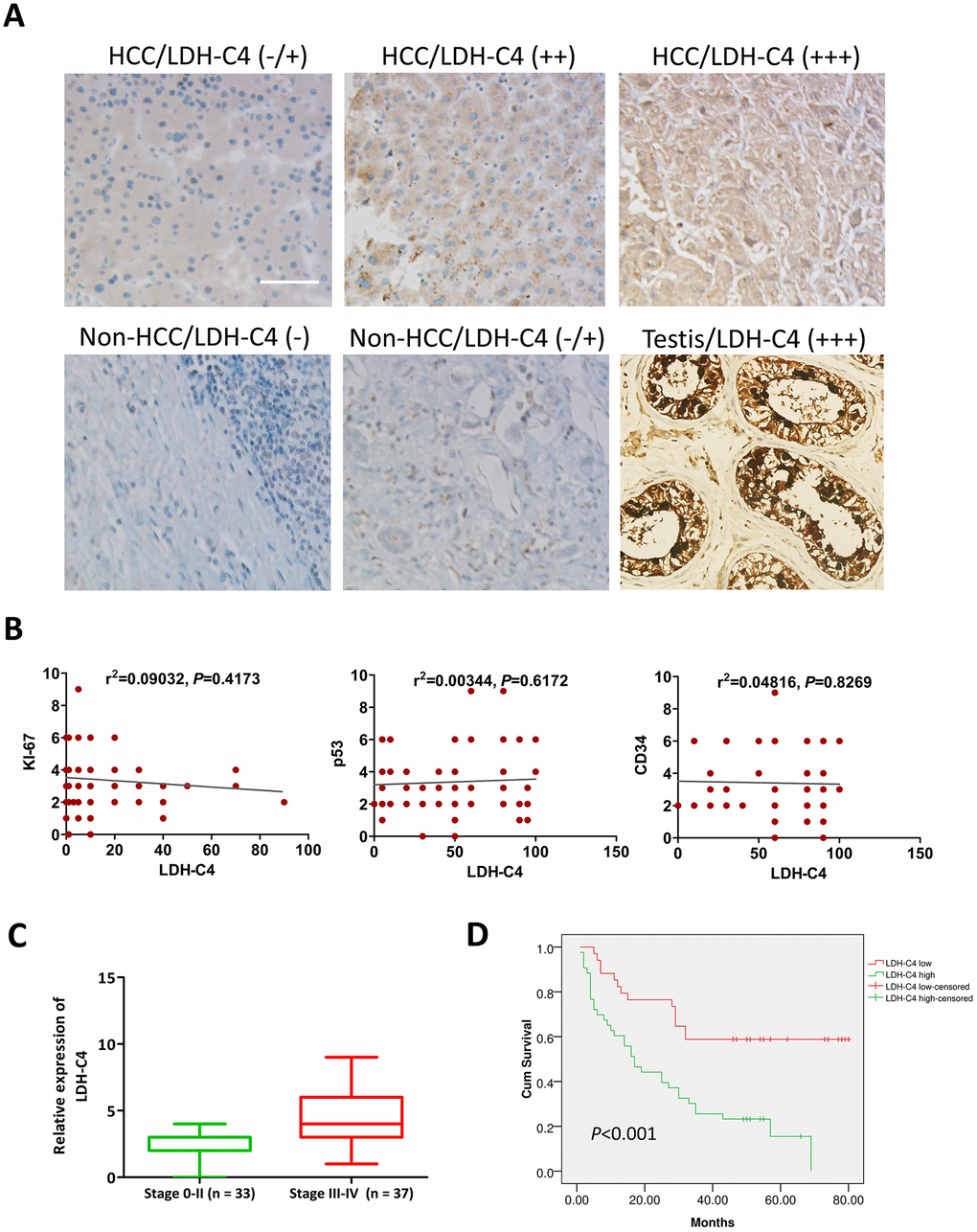
Figure 6. LDH-C4 expressions in HCC tissues using IHC analysis. (A) LDH-C4 levels were higher in HCC cancerous tissues than those in noncancerous tissues, and testis tissues were utilized as LDH-C4 positive controls (×200). (B) LDH-C4 levels were not significantly correlated with KI-67, p53 and CD34 expressions in HCC tissues. (C) LDH-C4 levels (rating scores) were higher in stage 0-II HCC patients than those in stage III-IV HCC patients. (D) The Kaplan-Meier analysis showed that LDH-C4 was negatively correlated with the prognosis of HCC patients, and higher levels of LDH-C4 implied worse prognosis.
Table 1. Correlations between LDH-C4 expressions and clinical characteristics in HCC patients.
| Clinicopathological features | Total case size | LDH-C4 expression (-/+) | LDH-C4 expression (++/+++) | P value |
| Age (years) | 0.355 | |||
| ≤50 | 27 | 10 | 17 | |
| >50 | 50 | 24 | 26 | |
| Gender | 0.468 | |||
| Male | 70 | 30 | 40 | |
| Female | 7 | 4 | 3 | |
| Tumor size (cm) | 0.023 | |||
| ≤5 | 32 | 19 | 13 | |
| >5 | 45 | 15 | 30 | |
| Clinical stage | 0.002 | |||
| Stage I+II | 33 | 21 | 12 | |
| Stage III+IV | 37 | 10 | 27 |
Table 2. The Cox regression analysis revealed independent risk factors for the prognosis of HCC patients.
| Risk factors | B value | SE | Wald | P value | Exp(β) | Exp(β) 95%CI | |
| Low | High | ||||||
| Clinical stage | -1.159 | 0.721 | 2.584 | 0.108 | 0.314 | 0.076 | 1.289 |
| T stage | -0.276 | 0.673 | 0.168 | 0.682 | 0.759 | 0.203 | 2.838 |
| Tumor size | -0.198 | 0.426 | 0.216 | 0.642 | 0.820 | 0.356 | 1.890 |
| LDH-C4 level | -0.926 | 0.383 | 5.857 | 0.016 | 0.396 | 0.187 | 0.839 |
Expressions of LDHC in HCC tissues analyzed based on GEPIA data
We also assessed the clinical significance of LDHC expressions in HCC based on GEPIA data that from TCGA. We found that LDHC levels were elevated in HCC tissues compared with non-cancer controls (Figure 7A), and also highly expressed in stage I-IV HCC patients (Figure 7B). The correlation analysis showed that LDHC expressions were positively associated with AFP levels (P = 0.048, Figure 7C), but not correlated with EGFR expressions in HCC patients (Figure 7D). The survival analysis showed that LDHC in HCC tissues was a nonsignificant predictor for OS and DFS of HCC patients (Figure 7E, 7F).
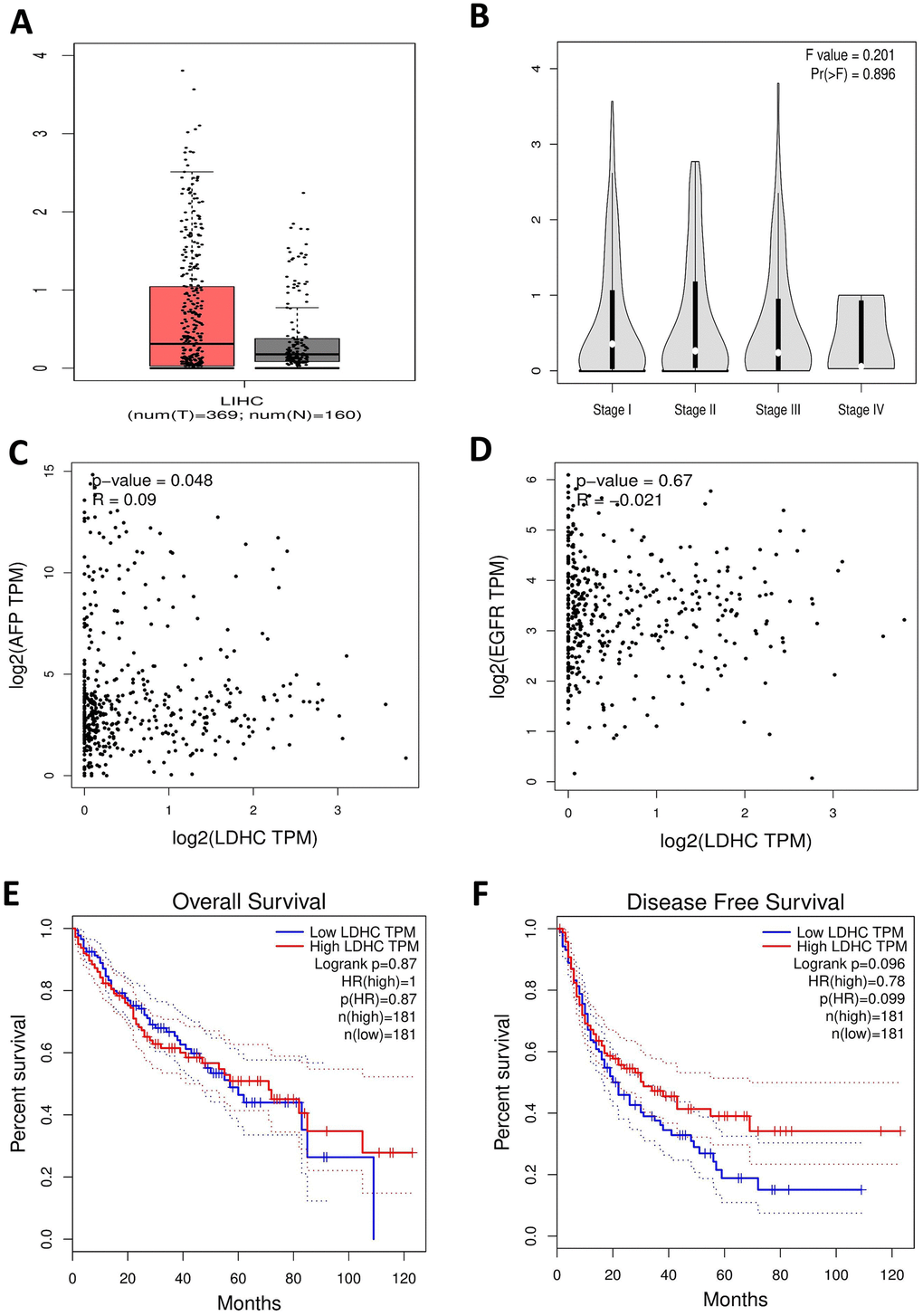
Figure 7. LDHC mRNA expressions in HCC tissues using TCGA data in the GEPIA database. (A) The box plot of LDHC expression in HCC specimens and non-cancerous tissues. (B) LDHC expressions in cases of stages I-IV HCC. (C) LDHC expressions were positively correlated with AFP levels in HCC tissues. (D) LDHC levels were not correlated with EGFR expressions in HCC tissues. LDHC expressions were not correlated with the (E) OS and (F) DFS of HCC patients.
Discussion
Tumor cell growth is an energy-intensive process [27]. It has been proven that cancer cells including HCC cell lines exhibit disordered metabolism accompanied by releases of a substantial amount of energy through aerobic glycolysis even in the presence of adequate oxygen [28–31]. LDH-C4 is an enzyme specifically expressed in the testis and spermatozoa of birds and mammals, catalyzing the transformation of lactic acid to pyruvate via pathways of glucose metabolism [13–16]. As a member of the CTA family, LDH-C4 is also highly expressed in various malignant tumors such as lung cancer, renal cancer, and melanoma [18, 19, 32]. As LDH-C4 has a preference for lactate as a substrate [14], it is conceivable that LDH-C4 activation in cancers may rely on lactate for ATP production. This may partially explain the aberrant high expression level of LDHC in high ATP-consumption tumors. However, studies on roles of LDH-C4 in liver tumors have not been fully elucidated.
Exosomes are 30-150 nm diameter membrane vesicles released into the extracellular matrix upon fusion of intracellular multivesicular bodies with the cell membrane [20, 21]. It functions as a molecular warehouse carrying proteins, DNAs and RNAs and prevents their degradation during the cell cycle [33, 34]. Exosomes have been detected in the tumor microenvironment, and new evidence shows that their involvement in the angiogenesis, immune activity and metastasis of tumors results in the acceleration of tumor development [22, 23]. Therefore, circulating exosomes from liquid biopsies can be used as non-invasive biomarkers for early detection, diagnosis and efficacy monitoring of cancers [24, 25]. In this study, we for the first time explore serum and exosomal LDHC mRNA expressions and LDH-C4 protein expressions in HCC tissues using qRT-PCR, high-throughput tissue microarrays and IHC analyses, as well as their clinical implications in early diagnosis and prognosis prediction of the patients.
Previous studies confirm that an elevated concentration of free nucleic acids in the blood of tumor patients, compared with healthy people, can be explained by the delivery of nucleic acids by exosomes [33, 34]. This has been verified in our previous study which has showed that exosomes carry and release LDH-C4 molecules to the peripheral blood of breast cancer patients [26]. The present study for the first time elucidates that LDHC mRNA exists in the serum and exosomes of stages I-II HCC patients with a positive rate of 68% and 60% and the diagnostic sensitivity and specificity of higher than 80%. Our results have shown that serum and exosomal LDHC levels are positively associated with the recurrence of HCC and negatively correlated with treatments. All this indicates that serum and exosomal LDHC mRNA has high application values in early diagnosis, efficacy evaluation and recurrence monitoring of HCC. We have previously reported that the positive rates of LDH-C4, serum LDHC and exosomal LDHC are 91.55% (130 / 145), 91.66% (22 / 24) and 87.50% (21 / 24) in breast cancer tissues using a microarray analysis [26], which are consistent with the results of this study.
Currently, other members of the CTA family including MAGEC2 and KK-LC-1 have been reported to be prognostic indicators for HCC [35, 36]. To investigate the prognostic value of LDH-C4, we further analyze the relationship between the results of microarray analysis and clinical data of the patients, and find that LDH-C4 protein expressions in HCC tissues are correlated with clinical stage and tumor size of HCC. This suggests that LDH-C4 may participate in HCC progression. On top of that, the survival analysis shows that the OS of HCC patients who present high expression levels of LDH-C4 is shortened and their prognosis is poor, compared with the patients whose LDH-C4 levels are low. The Cox regression analysis has further confirmed that LDH-C4 is an independent risk factor for prognosis: patients with a high LDH-C4 level have a risk ratio of 2.5252 (1 / 0.396) times higher than the low LDH-C4 expression HCC cases. A latest research reports that expressions of LDHC mRNA and LDH-C4 protein are significantly up-regulated in RCC, and the prognosis of patients showing a positive LDH-C4 expression is worse [19], which is consistent with our current results and previous findings that LDH-C4 is a good indicator for prognosis monitoring of breast cancer patients [26]. However, the analysis based on GEPIA database (including the correlations between clinical stage and the survival) does not fully in line with our results of high-throughput tissue microarray analysis. The explanations can be as follows. Firstly, the survival analysis has been conducted based on LDH-C4 protein expressions determined by immunohistochemical analysis using rating scores. Secondly, due to protein post-translation modifications, LDHC mRNA expression levels using mRNA datasets in GEPIA can be inconsistent with its protein levels. On the other hand, LDHC expressions quantitated using the mRNA data in GEPIA database are positively associated with AFP levels of HCC patients (P = 0.048, R = 0.09), which is seemingly consistent with our findings in the serum and exosomes. However, as the R2 value in the correlation analysis is very small, the correlation between AFP and LDHC needs to be further verified.
To sum up, this study demonstrates serum and exosomal LDH-C4 expressions in cancerous tissues of HCC patients and its clinical implications. It can be deemed as a promising biomarker for early diagnosis, efficacy evaluation, recurrence monitoring and prognosis prediction of HCC. It is also expected to be a potential target for immunotherapy of HCC. However, the small sample size included in our study inevitably have somewhat biased the results. So our further study series are in need of more data from large-sample clinical trials for the verification of current findings.
Materials and Methods
Clinical data
A total of 112 serum samples from HCC patients—including 50 cases of preliminarily diagnosed HCC (stages I-II), 32 treated cases and 30 recurrent ones—who were admitted to Fujian Cancer Hospital from December 2018 to December 2019 were collected. Another 100 serum samples from healthy volunteers were enrolled in the control group. All serum samples were collected with the approval of the ethics committee at our hospital (ethical grant No. SQ2015-049-01). Commercial high-throughput HCC microarray (HLivH180Su10, contained 93 HCC cases and 87 adjacent non-cancerous tissues) was provided by Shanghai Outdo Biotech Co., Ltd. (Shanghai, China), and only 77 samples from HCC cancer tissues and 76 from adjacent non-cancerous tissues were used. Among the 77 cancer patients consisting of 70 males and 7 females aged from 21 to 75 years with an average age of 45.32 ± 5.18 years, 33 cases were diagnosed as stage T1 + T2 (or stage I + II) HCC and 37 as stage T3 + T4 (or stage III + IV) HCC (unavailable data from 7 cases were not included). The tumor stage classification was guided by the TNM stage criteria of primary hepatocellular carcinoma (7th edition) in the American Joint Committee on Cancer (AJCC) / the Union for International Cancer Control (UICC) staging in 2010. All cancer tissues were confirmed by pathological examination. The enrolled patients had complete clinical data and follow-up information. The operation time ranged from January 2007 to November 2009, and the follow-up period varied between 4 and 7 years from September 2013.
Vesicle isolation and identification by western blot
Serum exosomes were extracted using an exoRNeasy Serum/Plasma Midi Kit (QIAGEN, Catalog No.77044) (Part I: vesicle isolation). The extracted exosomes were identified by transmission electron microscope (TEM). The detailed processes were described in our previous study [26]. Briefly, after total proteins extracted from the exosome eluate were lysed using RIPA lysis buffer, they were separated by gel electrophoresis (SDS-PAGE, 12% gels) at 80 V constant pressure and transferred to PVDF membranes at 250 mA constant current. TBST containing 5% skimmed milk powder was used for blocking the PVDF membranes overnight. Rabbit anti human CD9 monoclonal primary antibody (Abcam, Catalog No.ab92726; 1 : 300), rabbit anti human CD63 monoclonal primary antibody (Abcam, Catalog No.ab217345; 1 : 100), and mouse anti human GAPDH monoclonal primary antibody (Beyotime, Catalog No. AF0006; 1 : 1000) were added for incubation for 4 h at room temperature. Goat anti mouse / rabbit IgG (1 : 10000) with horseradish peroxidase (HRP) was added before a 2-h incubation at room temperature. ECL luminescent solution was employed for visualizing the protein bands which were subsequently photographed. Each experiment was repeated three times.
Real-time qRT-PCR
Total RNAs were extracted from serum and serum-derived exosomes respectively using the MiRNeasy Kit (QIAGEN, Catalog No.217184) and exoRNeasy Serum/Plasma Midi Kit (Part II: RNA isolation, Figure 1), according to the manufacturer’s instruction. The serum and exosomal RNAs were reversely transcripted into cDNAs using the Transcriptor first strand cDNA synthesis Kit (Roche) according to the manufacturer’s instruction. The primer sequences of LDHC and GAPDH were previously described [26]: LDHC-F: 5'-TCATTCCTGCCATAGTCCA-3', LDHC-R: 5'-CAATTACACGAGTTACAGGTA-3'; GAPDH-F: 5'-TCGACAGTCAGCCGCATCTTCTTT-3'; and GAPDH-R: 5'-ACCAAATCCGTTGACTCCGACCTT-3'. Levels of LDHC mRNAs were determined using the ABI7500 fluorescence quantitative PCR detector and SYBR green real time PCR master mix (ROX) kit under the following conditions: denaturation at 95 °C for 10 min, then 95 °C for 15 s, 40 cycles, and finally 60 °C for 1 min. Subsequently, relative expressions of LDHC mRNAs were calculated using the 2−ΔΔCt method normalized to GAPDH.
Immunohistochemical analysis
The immunohistochemical analysis was performed using an EliVisionTM Plus Two-Step Test Kit. The process was described in our previous study [37]. Briefly, wax-embedded tissues were dewaxed in xylene and rehydrated in a series of ethanol solutions. They were washed and treated with H2O2 for 10 min. T The antigen was extracted by heating citric acid in a boiling-water bath at 100-120 °C and high pressure for 3 min. Sections were incubated in the diluted (1 : 50) rabbit anti human LDHC monoclonal antibody (Abcam, Cat.no.ab52747) at 37 °C for 90 min, washed with PBS, and incubated with goat anti-rabbit antibody for 30 min. The sections were stained with DAB for 2 min, and hematoxylin was used for contrast staining.
The proportion of LDH-C4-positive cells in each sample was scored as follows. A positive rate of less than 5% was scored as 0, the rate of more than 5% and less than 25% was scored as 1, the rate of more than 25% and less than 50% as 2, and that of more than 50% as 3. The staining severity was graded according to the final score for LDH-C4 expressions based on the positive rates: 0 point was rated as “−”, 1-2 points as “+”, 3-5 points as “+ +”, and 6-9 points as “+ + +”. Among them, “− / +” was defined as negative or low expression and “+ + ~ + + +” as high expression. All sections were judged by two experienced pathologists using a double-blind method.
Bioinformatics analysis of available databases
LDHC mRNA expressions in HCC were also analyzed using available TCGA data in GEPIA (http://gepia.cancer-pku.cn/). A Log2FC cutoff value (Log2FC = 2, P = 0.01) was used to distinguish a high LDHC expression group from a low expression group. For the survival analysis, high and low LDHC expression groups were identified using a group cutoff of the median.
Statistical analyses
Data were analyzed using SPSS 16.0 software. Measurement data were presented as mean ± standard deviation (SD). We studied the fulfillment of the normality and homogeneity of variance assumption prior to the application of Student's t-test for comparisons between groups. Kaplan-Meier survival curve was used to analyze the overall survival of HCC patients. The correlations between LDH-C4 expressions and clinicopathological features and between the expression and the prognosis of HCC patients were analyzed using chi-square test and Spearman’s rho test. A Cox proportional hazard regression model was established to analyze independent prognostic risk factors of HCC. A P value of less than 0.05 was considered statistically significant.
Author Contributions
Zhaolei Cui: conceptualization, funding acquisition, writing – original draft. Yun Li: investigation, data curation, formal analysis. Yanni Gao and Lingying Kong: Data curation, formal analysis, methodology. Yingfeng Lin: software. Yan Chen: conceptualization, supervision, writing - review & editing.
Conflicts of Interest
The authors have declared that no conflicts of interest exists.
Funding
This study was sponsored by the National Natural Science Foundation of China (Grant number: 81802631), Fujian Provincial Health Technology Project (Grant number: 2016-CX-9), and Joint Funds for the Innovation of Science and Technology, Fujian province (Grant number: 2017Y9073), and Science and Technology Program of Fujian Province, China (Grant number: 2018Y2003).
References
- 1. Villanueva A. Hepatocellular carcinoma. N Engl J Med. 2019; 380:1450–62. https://doi.org/10.1056/NEJMra1713263 [PubMed]
- 2. Clark T, Maximin S, Meier J, Pokharel S, Bhargava P. Hepatocellular carcinoma: review of epidemiology, screening, imaging diagnosis, response assessment, and treatment. Curr Probl Diagn Radiol. 2015; 44:479–86. https://doi.org/10.1067/j.cpradiol.2015.04.004 [PubMed]
- 3. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019; 69:7–34. https://doi.org/10.3322/caac.21551 [PubMed]
- 4. Grandhi MS, Kim AK, Ronnekleiv-Kelly SM, Kamel IR, Ghasebeh MA, Pawlik TM. Hepatocellular carcinoma: from diagnosis to treatment. Surg Oncol. 2016; 25:74–85. https://doi.org/10.1016/j.suronc.2016.03.002 [PubMed]
- 5. Hartke J, Johnson M, Ghabril M. The diagnosis and treatment of hepatocellular carcinoma. Semin Diagn Pathol. 2017; 34:153–59. https://doi.org/10.1053/j.semdp.2016.12.011 [PubMed]
- 6. Poddar N, Ramlal R, Ravulapati S, Devlin SM, Gadani S, Vidal CI, Cao D, Befeler AS, Lai J. Extrahepatic metastasis of hepatocellular carcinoma arising from a hepatic adenoma without concurrent intrahepatic recurrence. Curr Oncol. 2017; 24:e333–36. https://doi.org/10.3747/co.24.3494 [PubMed]
- 7. Bruix J, Reig M, Sherman M. Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma. Gastroenterology. 2016; 150:835–53. https://doi.org/10.1053/j.gastro.2015.12.041 [PubMed]
- 8. Gjerstorff MF, Andersen MH, Ditzel HJ. Oncogenic cancer/testis antigens: prime candidates for immunotherapy. Oncotarget. 2015; 6:15772–87. https://doi.org/10.18632/oncotarget.4694 [PubMed]
- 9. Salmaninejad A, Zamani MR, Pourvahedi M, Golchehre Z, Hosseini Bereshneh A, Rezaei N. Cancer/testis antigens: expression, regulation, tumor invasion, and use in immunotherapy of cancers. Immunol Invest. 2016; 45:619–40. https://doi.org/10.1080/08820139.2016.1197241 [PubMed]
- 10. Al-Khadairi G, Decock J. Cancer testis antigens and immunotherapy: where do we stand in the targeting of PRAME? Cancers (Basel). 2019; 11:984. https://doi.org/10.3390/cancers11070984 [PubMed]
- 11. Gordeeva O. Cancer-testis antigens: unique cancer stem cell biomarkers and targets for cancer therapy. Semin Cancer Biol. 2018; 53:75–89. https://doi.org/10.1016/j.semcancer.2018.08.006 [PubMed]
- 12. Wei X, Chen F, Xin K, Wang Q, Yu L, Liu B, Liu Q. Cancer-testis antigen peptide vaccine for cancer immunotherapy: progress and prospects. Transl Oncol. 2019; 12:733–38. https://doi.org/10.1016/j.tranon.2019.02.008 [PubMed]
- 13. Goldberg E. Lactic and Malic dehydrogenases in human spermatozoa. Science. 1963; 139:602–03. https://doi.org/10.1126/science.139.3555.602 [PubMed]
- 14. Goldberg E, Eddy EM, Duan C, Odet F. LDHC: the ultimate testis-specific gene. J Androl. 2010; 31:86–94. https://doi.org/10.2164/jandrol.109.008367 [PubMed]
- 15. Gupta GS. LDH-C4: a target with therapeutic potential for cancer and contraception. Mol Cell Biochem. 2012; 371:115–27. https://doi.org/10.1007/s11010-012-1428-2 [PubMed]
- 16. Hawtrey CO, Goldberg E. Some kinetic aspects of sperm specific lactate dehydrogenase in mice. J Exp Zool. 1970; 174:451–61. https://doi.org/10.1002/jez.1401740408 [PubMed]
- 17. Koslowski M, Türeci O, Bell C, Krause P, Lehr HA, Brunner J, Seitz G, Nestle FO, Huber C, Sahin U. Multiple splice variants of lactate dehydrogenase C selectively expressed in human cancer. Cancer Res. 2002; 62:6750–55. [PubMed]
- 18. Grunwald C, Koslowski M, Arsiray T, Dhaene K, Praet M, Victor A, Morresi-Hauf A, Lindner M, Passlick B, Lehr HA, Schäfer SC, Seitz G, Huber C, et al. Expression of multiple epigenetically regulated cancer/germline genes in nonsmall cell lung cancer. Int J Cancer. 2006; 118:2522–28. https://doi.org/10.1002/ijc.21669 [PubMed]
- 19. Wang Y, Li G, Wan F, Dai B, Ye D. Prognostic value of d-lactate dehydrogenase in patients with clear cell renal cell carcinoma. Oncol Lett. 2018; 16:866–74. https://doi.org/10.3892/ol.2018.8782 [PubMed]
- 20. Boyiadzis M, Whiteside TL. The emerging roles of tumor-derived exosomes in hematological Malignancies. Leukemia. 2017; 31:1259–68. https://doi.org/10.1038/leu.2017.91 [PubMed]
- 21. Hesari A, Golrokh Moghadam SA, Siasi A, Rahmani M, Behboodi N, Rastgar-Moghadam A, Ferns GA, Ghasemi F, Avan A. Tumor-derived exosomes: potential biomarker or therapeutic target in breast cancer? J Cell Biochem. 2018; 119:4236–40. https://doi.org/10.1002/jcb.26364 [PubMed]
- 22. Saadatpour L, Fadaee E, Fadaei S, Nassiri Mansour R, Mohammadi M, Mousavi SM, Goodarzi M, Verdi J, Mirzaei H. Glioblastoma: exosome and microRNA as novel diagnosis biomarkers. Cancer Gene Ther. 2016; 23:415–18. https://doi.org/10.1038/cgt.2016.48 [PubMed]
- 23. Zhang J, Li S, Li L, Li M, Guo C, Yao J, Mi S. Exosome and exosomal microRNA: trafficking, sorting, and function. Genomics Proteomics Bioinformatics. 2015; 13:17–24. https://doi.org/10.1016/j.gpb.2015.02.001 [PubMed]
- 24. Kalluri R. The biology and function of exosomes in cancer. J Clin Invest. 2016; 126:1208–15. https://doi.org/10.1172/JCI81135 [PubMed]
- 25. Nedaeinia R, Manian M, Jazayeri MH, Ranjbar M, Salehi R, Sharifi M, Mohaghegh F, Goli M, Jahednia SH, Avan A, Ghayour-Mobarhan M. Circulating exosomes and exosomal microRNAs as biomarkers in gastrointestinal cancer. Cancer Gene Ther. 2017; 24:48–56. https://doi.org/10.1038/cgt.2016.77 [PubMed]
- 26. Cui Z, Chen Y, Hu M, Lin Y, Zhang S, Kong L, Chen Y. Diagnostic and prognostic value of the cancer-testis antigen lactate dehydrogenase C4 in breast cancer. Clin Chim Acta. 2020; 503:203–09. https://doi.org/10.1016/j.cca.2019.11.032 [PubMed]
- 27. Kroemer G, Pouyssegur J. Tumor cell metabolism: cancer’s achilles’ heel. Cancer Cell. 2008; 13:472–82. https://doi.org/10.1016/j.ccr.2008.05.005 [PubMed]
- 28. Cassim S, Vučetić M, Ždralević M, Pouyssegur J. Warburg and beyond: the power of mitochondrial metabolism to collaborate or replace fermentative glycolysis in cancer. Cancers (Basel). 2020; 12:1119. https://doi.org/10.3390/cancers12051119 [PubMed]
- 29. Smith RJ. Nutrition and metabolism in hepatocellular carcinoma. Hepatobiliary Surg Nutr. 2013; 2:89–96. https://doi.org/10.3978/j.issn.2304-3881.2012.11.02 [PubMed]
- 30. Cassim S, Raymond VA, Dehbidi-Assadzadeh L, Lapierre P, Bilodeau M. Metabolic reprogramming enables hepatocarcinoma cells to efficiently adapt and survive to a nutrient-restricted microenvironment. Cell Cycle. 2018; 17:903–16. https://doi.org/10.1080/15384101.2018.1460023 [PubMed]
- 31. Cassim S, Raymond VA, Lacoste B, Lapierre P, Bilodeau M. Metabolite profiling identifies a signature of tumorigenicity in hepatocellular carcinoma. Oncotarget. 2018; 9:26868–83. https://doi.org/10.18632/oncotarget.25525 [PubMed]
- 32. Tang H, Goldberg E. Homo sapiens lactate dehydrogenase c (ldhc) gene expression in cancer cells is regulated by transcription factor Sp1, CREB, and CpG island methylation. J Androl. 2009; 30:157–67. https://doi.org/10.2164/jandrol.108.005785 [PubMed]
- 33. Sasaki R, Kanda T, Yokosuka O, Kato N, Matsuoka S, Moriyama M. Exosomes and hepatocellular carcinoma: from bench to bedside. Int J Mol Sci. 2019; 20:1406. https://doi.org/10.3390/ijms20061406 [PubMed]
- 34. Tavakolizadeh J, Roshanaei K, Salmaninejad A, Yari R, Nahand JS, Sarkarizi HK, Mousavi SM, Salarinia R, Rahmati M, Mousavi SF, Mokhtari R, Mirzaei H. MicroRNAs and exosomes in depression: potential diagnostic biomarkers. J Cell Biochem. 2018; 119:3783–97. https://doi.org/10.1002/jcb.26599 [PubMed]
- 35. Chen Z, Zuo X, Pu L, Zhang Y, Han G, Zhang L, Wu Z, You W, Qin J, Dai X, Shen H, Wang X, Wu J. Hypomethylation-mediated activation of cancer/testis antigen KK-LC-1 facilitates hepatocellular carcinoma progression through activating the Notch1/Hes1 signalling. Cell Prolif. 2019; 52:e12581. https://doi.org/10.1111/cpr.12581 [PubMed]
- 36. Song X, Song W, Wang Y, Wang J, Li Y, Qian X, Pang X, Zhang Y, Yin Y. MicroRNA-874 functions as a tumor suppressor by targeting cancer/testis antigen HCA587/MAGE-C2. J Cancer. 2016; 7:656–63. https://doi.org/10.7150/jca.13674 [PubMed]
- 37. Kong L, Du W, Cui Z, Wang L, Yang Z, Zhang H, Lin D. Expression of lactate dehydrogenase C in MDA-MB-231 cells and its role in tumor invasion and migration. Mol Med Rep. 2016; 13:3533–38. https://doi.org/10.3892/mmr.2016.4963 [PubMed]


