Introduction
Sepsis, a disease with a high and increased prevalence worldwide, is the major cause of critical illness resulting in admission to Intensive Care Unit. Although more patients survive sepsis and are increasingly discharged from the hospital, they often experience long-term cognitive and psychological impairment with significant socioeconomic impact [1–3]. Previous studies have demonstrated neurobehavioral abnormities after sepsis by lipopolysaccharide (LPS) challenge or cecal ligation and puncture (CLP) in rodent models of sepsis [4–6]. However, the reported studies primarily focused on the relatively short stage after sepsis development. Given the possibility that infections in early life may be associated with increased risk of Alzheimer’s Disease (AD) [7], understanding the long-term impact of sepsis on brain function and its pathophysiological mechanisms are urgently needed.
Proteomic approach has grown rapidly and is a powerful and promising tool in identifying disease phenotypes, drug targets, and clinical biomarkers [8]. With the development of proteomic techniques, such as isobaric tagging for relative and absolute quantitation (iTRAQ) with liquid chromatography-mass spectrometry (LC-MS) analyses, have greatly improved the detection ability and reproducibility. It has been widely used in exploring the molecular markers and mechanisms in various diseases, including cancer, cardiovascular diseases, and psychiatric illnesses [9–12]. Although various mechanisms that contribute to the pathogenesis of sepsis-induced neurobehavioral abnormities have been revealed by studies on individual genes or proteins, systematic analysis of the hippocampal proteomic profile is still lacking.
Therefore, the aim of the present study was to investigate the long-term neurobehavioral alterations one year after CLP or LPS challenge. Our study showed that LPS challenge but not CLP induced long-term neurobehavioral abnormities in sepsis survivors, we thus focused our research on LPS-induced animals. Given the key role of hippocampal oscillation network in cognitive function, we tested whether hippocampal oscillations would be affected by LPS exposure, and if so, whether that effect would be mediated by impaired synaptic plasticity.
Results
Survival rate
We observed 7-day survival rate after LPS injection or CLP, we showed the survival rate was 63% in the LPS group and 66% in the LPS + roscovitine group, which was significantly lower than that in the control group (P = 0.0066, Figure 1B). In the current study, many animals died during the observation period. Ultimately, 21 mice in the control group, 20 mice in the LPS group, and 18 mice in the LPS + roscovitine group survived before behavior tests. In addition, CLP induced significantly decreased survival rate (78.846%) compared with sham group (100%) (P = 0.0259, Figure 1C).
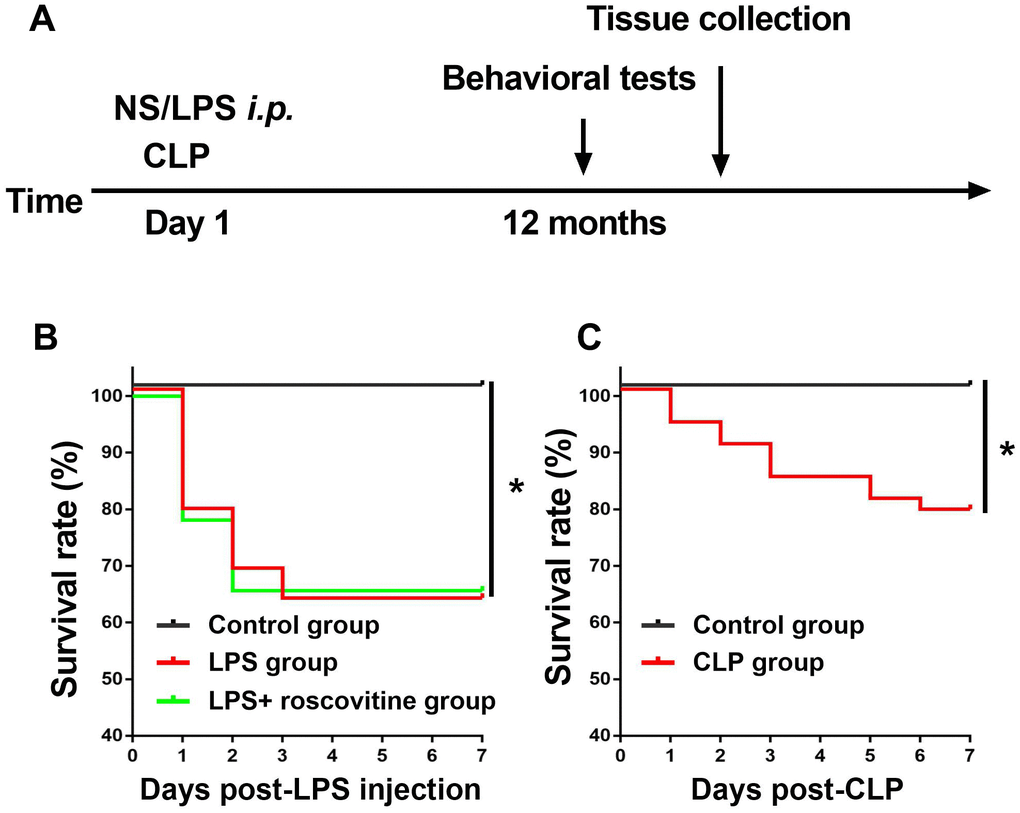
Figure 1. (A) Timeline of the experimental procedures of the present study. (B) Effects of LPS on survival rate, n = 25 for control group, n = 40 for LPS group, and n=33 for LPS + roscovitine group. (C) Effects of CLP on survival rate, n = 22 for control group, n = 52 for CLP group. LPS, lipopolysaccharide; NS, normal saline, *P < 0.05.
Identification of altered proteins in the hippocampus of LPS-exposed mice
Figure 2A shows the design for proteomic and phosphoproteomic analysis. We used the iTRAQ approach and performed large-scale quantitative analysis. We identified 25045 unique peptides and 4163 proteins from each sample. Significant differences in protein expressions were determined by the following threshold: “P ≤ 0.05 and fold change ≥ 1.2 or <0.83”, which has been adopted by previous study [9]. Accordingly, 16 proteins showing significant changes were observed in hippocampal samples between control and LPS-exposed mice (Figure 2B and Table 1). Briefly, Rars2, Clic6, Rttn, Gtpbp10, Itgb7, Hbb-bh0, Sept10, Mmp1a, Eml1, Krt8, Ubqln4, Serpini1, Ca5b, Elmo1, Gng10 were significantly upregulated, while Smad1 was downregulated. These differentially expressed proteins were displayed using a heatmap (Figure 2D). Gene ontology (GO) classification showed these differentially expressed proteins involved in different molecular function categories, and participated in many biological processes (Figure 4). The GO data provided an overview showing that abundant changes are exhibited by changes in structural protein, extracellular matrix, membrane protein composition, and enzyme catalytic activities following LPS challenge.
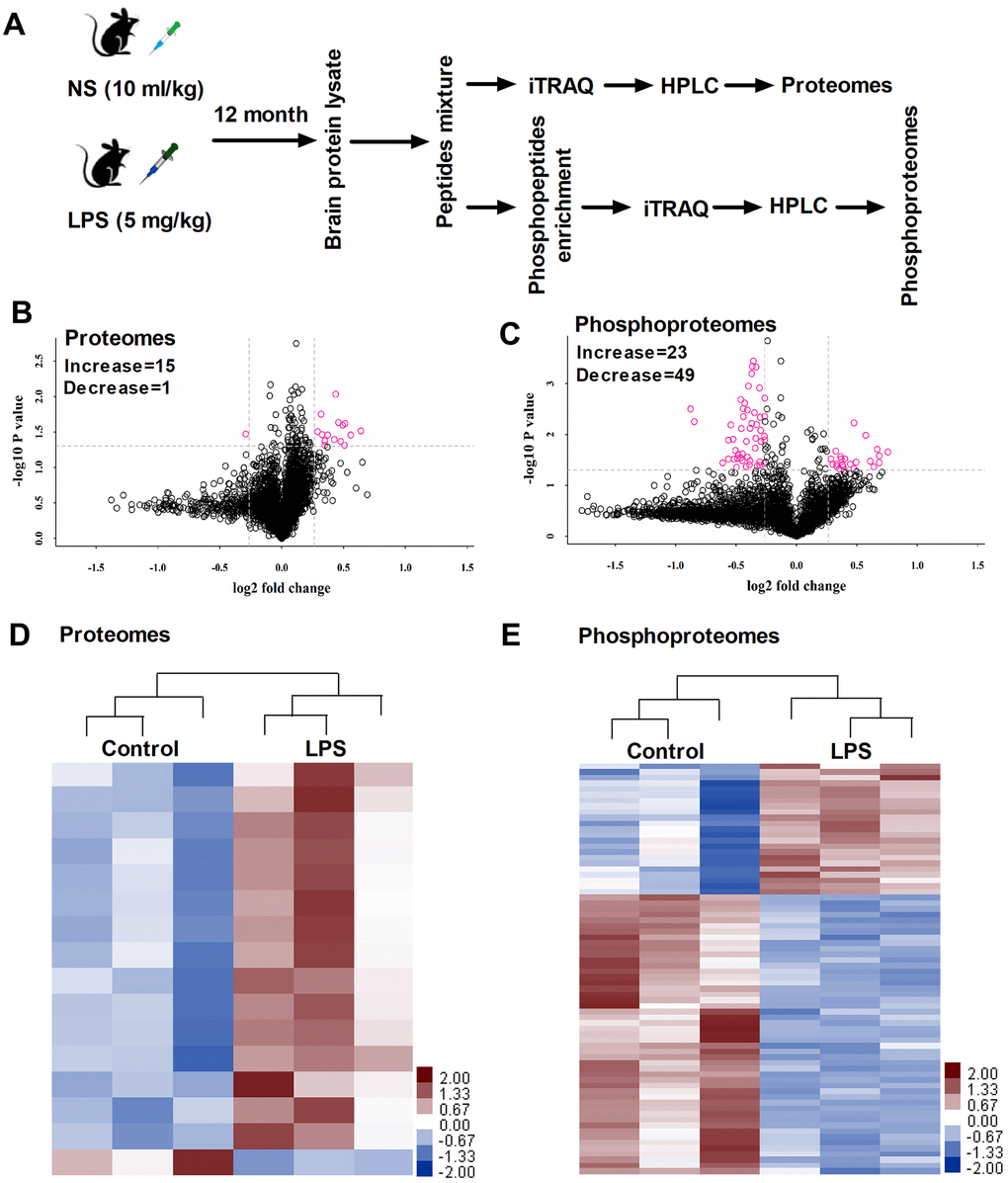
Figure 2. (A) Timeline of the proteins and phosphoproteins procedure. (B, C) Volcano plot indicating significantly altered proteins and phosphoproteins between control and LPS groups. (D, E) Heat map showing significantly altered proteins and phosphoproteins between control and LPS groups (n = 3). LPS, lipopolysaccharide; NS, normal saline; ITRAQ, isobaric tagging for relative and absolute quantitation; HPLC, High performance liquid chromatography.
Table 1. Differential proteomics in hippocampus by iTRAQ analysis from control and LPS mice.
| Accession numbers | Protein names | Gene names | Unique peptides | Peptides coverage | Fold change |
| Q3U186 | Rars2 | Rars2 | 1 | 2.7681661 | 1.55797 |
| Q8BHB9 | Clic6 | Clic6 | 1 | 2.1812081 | 1.47219 |
| Q8R4Y8 | Rttn | Rttn | 2 | 0.7637017 | 1.42522 |
| Q8K013 | Gtpbp10 | Gtpbp10 | 1 | 6.010929 | 1.42171 |
| P26011 | Itgb7 | Itgb7 | 1 | 1.1166253 | 1.4073 |
| P04443 | Hbb-bh0 | Hbb-bh0 | 1 | 6.8027211 | 1.39139 |
| Q8C650 | Sept10 | Sept10 | 1 | 3.7610619 | 1.37436 |
| Q9EPL5 | Mmp1a | Mmp1a | 1 | 3.6637931 | 1.35294 |
| Q05BC3 | Eml1 | Eml1 | 1 | 1.3513514 | 1.34336 |
| P11679 | Krt8 | Krt8 | 3 | 8.9795918 | 1.29396 |
| Q99NB8 | Ubqln4 | Ubqln4 | 1 | 7.7181208 | 1.27273 |
| O35684 | Serpini1 | Serpini1 | 1 | 5.6097561 | 1.27273 |
| Q9QZA0 | Ca5b | Ca5b | 1 | 5.362776 | 1.2531 |
| Q8BPU7 | Elmo1 | Elmo1 | 2 | 9.3535076 | 1.24682 |
| Q9CXP8 | Gng10 | Gng10 | 1 | 22.058824 | 1.22305 |
| P70340 | Smad1 | Smad1 | 1 | 3.0107527 | 0.81683 |
We further screened the phosphoproteins based on their fold-changes in expression level. We identified 72 proteins that showed at least a 20% increase or decrease in expression, of which 23 were upregulated and 49 were downregulated (Figure 2C and Table 2). These differentially expressed proteins were also displayed using a heatmap (Figure 2E). We noted a significantly increased expression of Cdk11b, indicating that the neurons could undergo apoptosis following LPS challenge. Also, LPS also increased microtubule-associated proteins, such as Map1b, Dctn1, Map2, and Ank2 relative to controls. In addition to neuronal cell signaling pathway, we have identified several biological functions altered in LPS-exposed mice, such as Syngap1, Bsn, Shisa6, Synpo, Pclo, Ppp1r9b, and Dlgap2 for synaptic related proteins, suggesting a potential mechanistic link between the synaptic dysfunction and LPS-induced neurobehavioral abnormities.
Table 2. Differential phosphoproteomics in hippocampus by iTRAQ analysis from control and LPS mice.
| Accession numbers | Gene names | Description | Fold change |
| P24788 | Cdk11b | Cyclin-dependent kinase 11B | 1.68937696 |
| Q9QYR6 | Map1a | Microtubule-associated protein 1A | 1.60799652 |
| P14869 | Rplp0 | 60S acidic ribosomal protein P0 | 1.60799652 |
| P14873 | Map1b | Microtubule-associated protein 1B | 1.58955546 |
| P47955 | Rplp1 | 60S acidic ribosomal protein P1 | 1.55754476 |
| Q9QYR6 | Map1a | Microtubule-associated protein 1A | 1.52737995 |
| P70704 | Atp8a1 | Phospholipid-transporting ATPase IA | 1.48797678 |
| O08788 | Dctn1 | Dynactin subunit 1 | 1.40730337 |
| Q91YM2 | Arhgap35 | Rho GTPase-activating protein 35 | 1.39043825 |
| P20357 | Map2 | Microtubule-associated protein 2 | 1.38893312 |
| Q9QWY8 | Asap1 | Arf-GAP with SH3 domain, ANK repeat and PH domain-containing protein 1 | 1.36127509 |
| A2ARP1 | Ppip5k1 | Inositol hexakisphosphate and diphosphoinositol-pentakisphosphate kinase 1 | 1.31979892 |
| Q9JIS5 | Sv2a | Synaptic vesicle glycoprotein 2A | 1.31570822 |
| Q80YE4 | Aatk | Serine/threonine-protein kinase LMTK1 | 1.30680507 |
| Q4QQM5 | Miga1 | Mitoguardin 1 | 1.30237913 |
| O54781 | Srpk2 | SRSF protein kinase 2 | 1.29231945 |
| Q9D7P6 | Iscu | Iron-sulfur cluster assembly enzyme ISCU, mitochondrial | 1.28783835 |
| Q8C8R3 | Ank2 | Ankyrin-2 | 1.27703985 |
| Q6PB44 | Ptpn23 | Tyrosine-protein phosphatase non-receptor type 23 | 1.25856229 |
| Q9JMH9 | Myo18a | Unconventional myosin-XVIIIa | 1.25818592 |
| Q8BP99 | UPF0500 protein C1orf216 homolog | 1.25309801 | |
| Q80TL4 | Phf24 | PHD finger protein 24 | 1.23094087 |
| Q8CC27 | Cacnb2 | Voltage-dependent L-type calcium channel subunit beta-2 | 1.21856509 |
| P56399 | Usp5 | Ubiquitin carboxyl-terminal hydrolase 5 | 0.83323152 |
| Q9Z2H5 | Epb41l1 | Band 4.1-like protein 1 | 0.83318057 |
| Q3UHJ0 | Aak1 | AP2-associated protein kinase 1 | 0.83262065 |
| Q01815 | Cacna1c | Voltage-dependent L-type calcium channel subunit alpha-1C | 0.83150183 |
| P14873 | Map1b | Microtubule-associated protein 1B | 0.83119658 |
| Q9WTX2 | Prkra | Interferon-inducible double-stranded RNA-dependent protein kinase activator A | 0.81518451 |
| Q8C8R3 | Ank2 | Ankyrin-2 | 0.81378476 |
| Q9EPJ9 | Arfgap1 | ADP-ribosylation factor GTPase-activating protein 1 | 0.8115942 |
| Q99JX3 | Gorasp2 | Golgi reassembly-stacking protein 2 | 0.80965309 |
| Q80TI0 | Gramd1b | GRAM domain-containing protein 1B | 0.80965309 |
| Q3V3V9 | Carmil2 | Capping protein, Arp2/3 and myosin-I linker protein 2 | 0.80856195 |
| Q9Z1B3 | Plcb1 | 1-phosphatidylinositol 4,5-bisphosphate phosphodiesterase beta-1 | 0.79802218 |
| Q9Z2H5 | Epb41l1 | Band 4.1-like protein 1 | 0.79533214 |
| Q9QWI6 | Srcin1 | SRC kinase signaling inhibitor 1 | 0.79265013 |
| F6SEU4 | Syngap1 | Ras/Rap GTPase-activating protein SynGAP | 0.79104478 |
| Q3TY60 | Fam131b | Protein FAM131B | 0.78890877 |
| Q7TME0 | Plppr4 | Phospholipid phosphatase-related protein type 4 | 0.78518298 |
| O88737 | Bsn | Protein bassoon | 0.78071217 |
| Q9JM52 | Mink1 | Misshapen-like kinase 1 | 0.77619893 |
| G3XA57 | Rab11fip2 | Rab11 family-interacting protein 2 | 0.77327816 |
| D3YVF0 | Akap5 | A-kinase anchor protein 5 | 0.77304965 |
| Q61097 | Ksr1 | Kinase suppressor of Ras 1 | 0.76626435 |
| P20357 | Map2 | Microtubule-associated protein 2 | 0.765 |
| Q3UHD9 | Agap2 | Arf-GAP with GTPase, ANK repeat and PH domain-containing protein 2 | 0.76315016 |
| Q68EF6 | Begain | Brain-enriched guanylate kinase-associated protein | 0.75901495 |
| P33173 | Kif1a | Kinesin-like protein KIF1A | 0.75416545 |
| Q9R0K7 | Atp2b2 | Plasma membrane calcium-transporting ATPase 2 | 0.75416545 |
| Q9WV92 | Epb41l3 | Band 4.1-like protein 3 | 0.75 |
| Q924A2 | Cic | Protein capicua homolog | 0.74876129 |
| Q3UH99 | Shisa6 | Protein shisa-6 | 0.73690304 |
| O54829 | Rgs7 | Regulator of G-protein signaling 7 | 0.73589818 |
| P48453 | Ppp3cb | Serine/threonine-protein phosphatase 2B catalytic subunit beta isoform | 0.73260179 |
| P97427 | Crmp1 | Dihydropyrimidinase-related protein 1 | 0.7323903 |
| Q9Z0P4 | Palm | Paralemmin-1 | 0.72860847 |
| Q8K2Y9 | Ccm2 | Cerebral cavernous malformations protein 2 homolog | 0.72612198 |
| Q9QYG0 | Ndrg2 | Protein NDRG2 | 0.72562554 |
| Q8CC35 | Synpo | Synaptopodin | 0.71604232 |
| Q9QYX7 | Pclo | Protein piccolo | 0.71253212 |
| Q3UHD9 | Agap2 | Arf-GAP with GTPase, ANK repeat and PH domain-containing protein 2 | 0.70842825 |
| P04370 | Mbp | Myelin basic protein | 0.70426136 |
| Q9CYZ2 | Tpd52l2 | Tumor protein D54 | 0.69875425 |
| P35803 | Gpm6b | Neuronal membrane glycoprotein M6-b | 0.69491525 |
| O88703 | Hcn2 | Potassium/sodium hyperpolarization-activated cyclic nucleotide-gated channel 2 | 0.68586682 |
| Q6R891 | Ppp1r9b | Neurabin-2 | 0.67691448 |
| Q8BJ42 | Dlgap2 | Disks large-associated protein 2 | 0.67597765 |
| Q80TJ1 | Cadps | Calcium-dependent secretion activator 1 | 0.65425972 |
| O55131 | Sept7 | Septin-7 | 0.5560166 |
| Q5FWK3 | Arhgap1 | Rho GTPase-activating protein 1 | 0.54400412 |
Next, we performed some of the functional changes with regarding to apoptosis, mitochondria dysfunction, and microtubule formation. As shown in Figure 3, we showed that LPS induced significantly increased expressions of cleaved caspase-3 (t = 3.413, P = 0.0143) and cytochrome C (t = 2.844, P = 0.0294) in the hippocampus as compared with the control group, two markers of apoptosis and mitochondria dysfunction, respectively. However, there was no difference in microtubule-associated protein such as microtubule-associated protein-2 (MAP-2) between the control and LPS groups (t = 1.241, P = 0.2609).
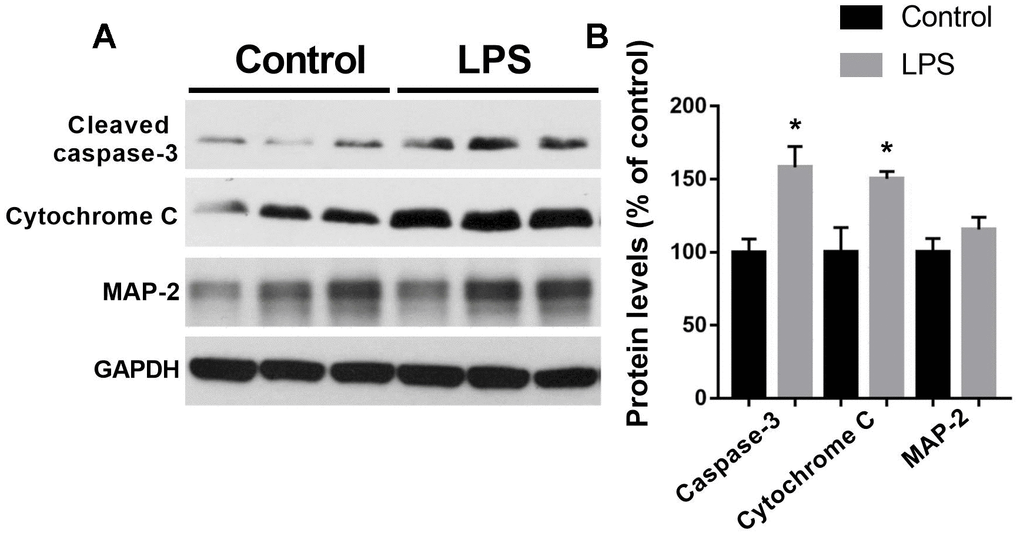
Figure 3. Expressions of cleaved caspase-3, cytochrome C, and MAP-2 in the hippocampus by western blotting analysis. (A) Representative Western blots bands of cleaved caspase-3, cytochrome C, and MAP-2 in the hippocampus; (B) Quantitative analysis of cleaved caspase-3, cytochrome C, and MAP-2. Data are presented as the mean ± SEM, n = 4, *P < 0.05.
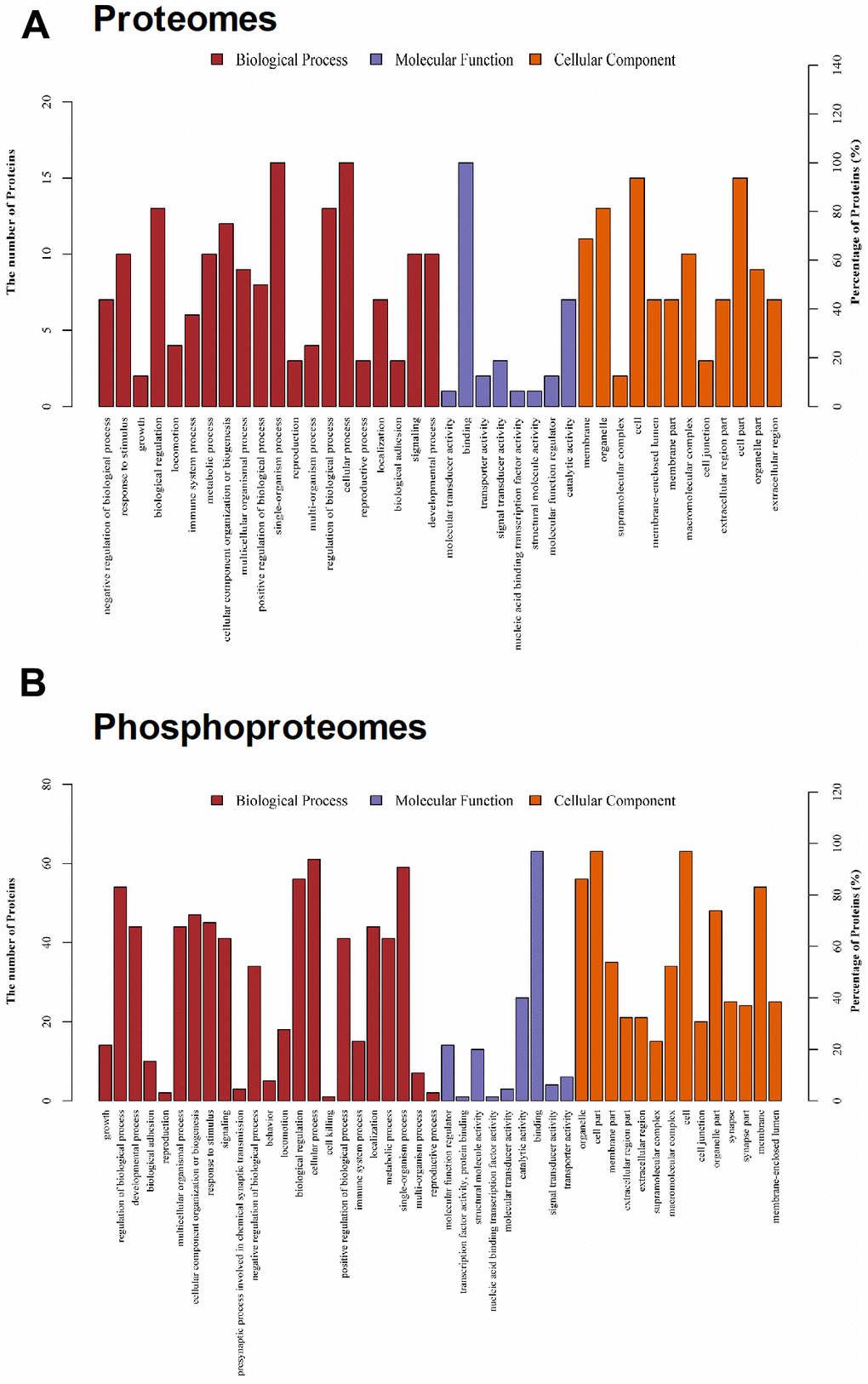
Figure 4. Gene ontology (GO) classification for differentially expressed proteins. (A) For proteomes, populations of proteins that showed alteredexpression are indicated based on their GO for molecular function, biological process, and cellular components. (B) For phosphoproteomes, populations of proteins that showed alteredexpression are indicated based on their GO for molecular function, biological process, and cellular components.
LPS induced significantly decreased pSynGAP1
Next, we constructed pathological protein–protein interaction (PPI) networks based on significantly changed phosphoproteins. As shown in Figure 5, nodes indicate proteins with increased or decreased phosphorylation and the proteins directly connected to the altered proteins in the PPI database (blue). In particular, we detected SynGAP1 is among the hub of synaptic plasticity. To further verify the reliability of the iTRAQ results, we examined SynGAP1 and pSynGAP1 levels using the western blot approach (Figure 6). Although SynGAP1 was not affected (t = 0.7615, P = 0.4725), we showed that pSynGAP1 in the hippocampus was significantly decreased in LPS-exposed mice (t = 2.632, P = 0.039), suggesting the iTRAQ results in the present study were reliable.
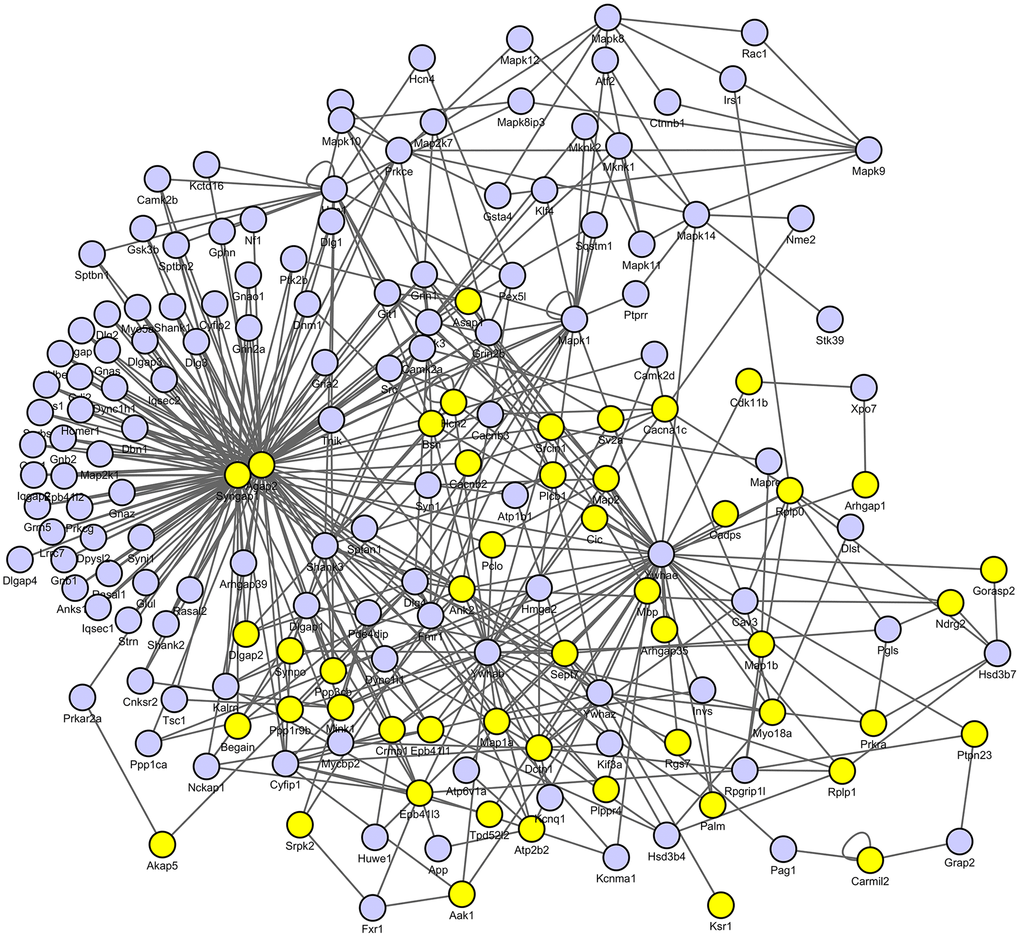
Figure 5. Protein–protein interaction (PPI) networks of differential proteins between control and LPS groups. The PPI analysis was based on fold change of protein–protein interaction, which showed SynGAP1 was identified as the hub of synaptic plasticity.
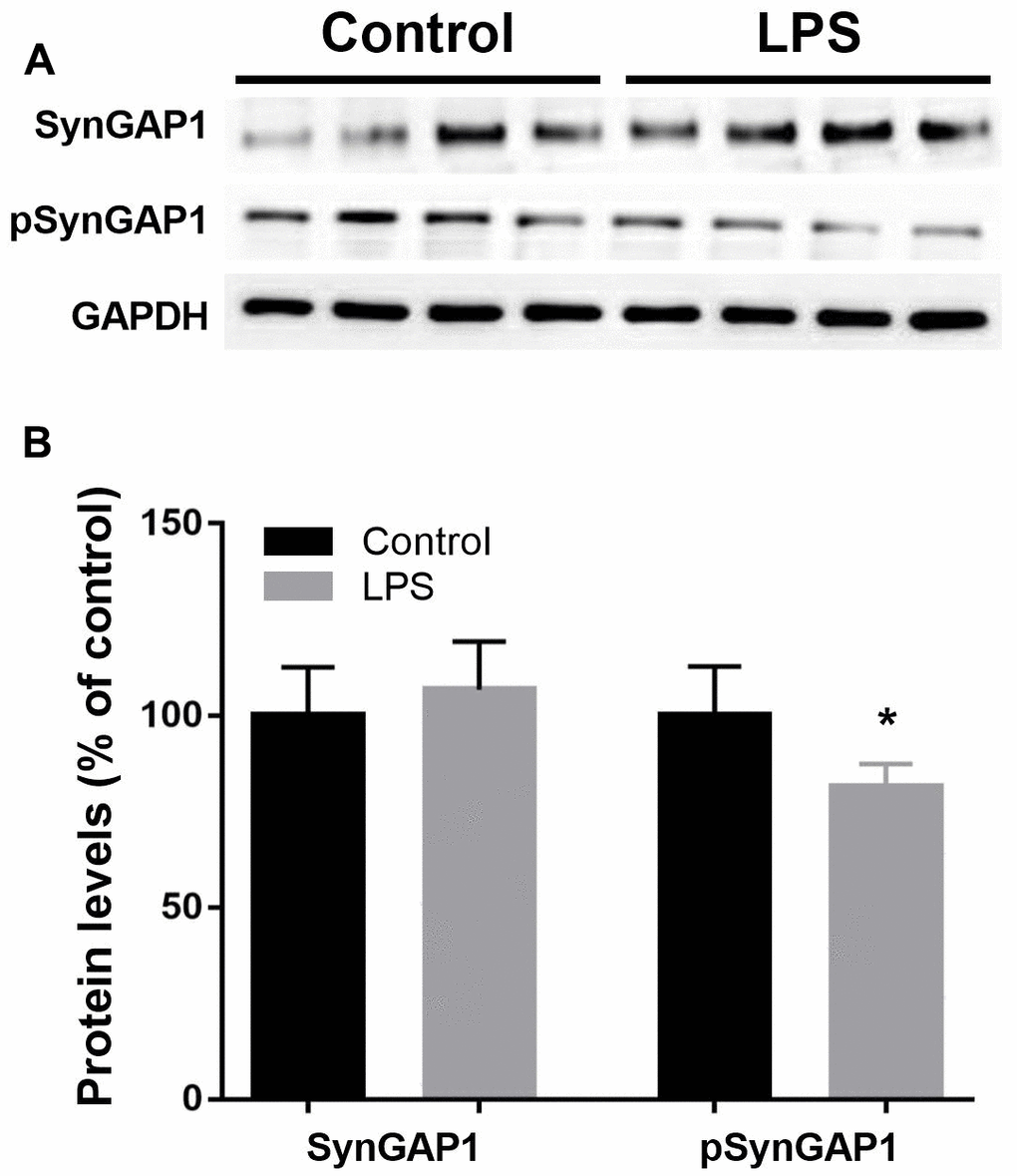
Figure 6. Validation of Syngap1 and pSyngap1 in hippocampus by western blotting analysis. (A) Representative Western blots bands of Syngap1 and pSyngap1 in the hippocampus; (B) Quantitative analysis of Syngap1 and pSyngap1 levels between groups. Data are presented as the mean ± SEM, n = 4, *P < 0.05.
Decreased hippocampal pCamKII, pSynGAP levels, and dendritic spine density following LPS challenge were rescued by roscovitine
It has been suggested that Cdk5 inhibition increased pCamKII, which further can increase pSynGAP levels [13]. For this reason, we determined whether Cdk5 inhibition by roscovitine can increase hippocampal pCamKII and pSynGAP levels. As shown in Figure 7A, 7B, roscovitine administration increased hippocampal pCamKII (F2,9 = 18.56, P = 0.0007) and pSynGAP (F2,9 = 11.19, P = 0.0035) levels in LPS + roscovitine group compared with LPS group. In addition, LPS significantly reduced dendritic spine density, while roscovitine administration attenuated LPS-induced dendritic spine loss (F2,9 = 6.182, P = 0.011; Figure 7C, 7D).
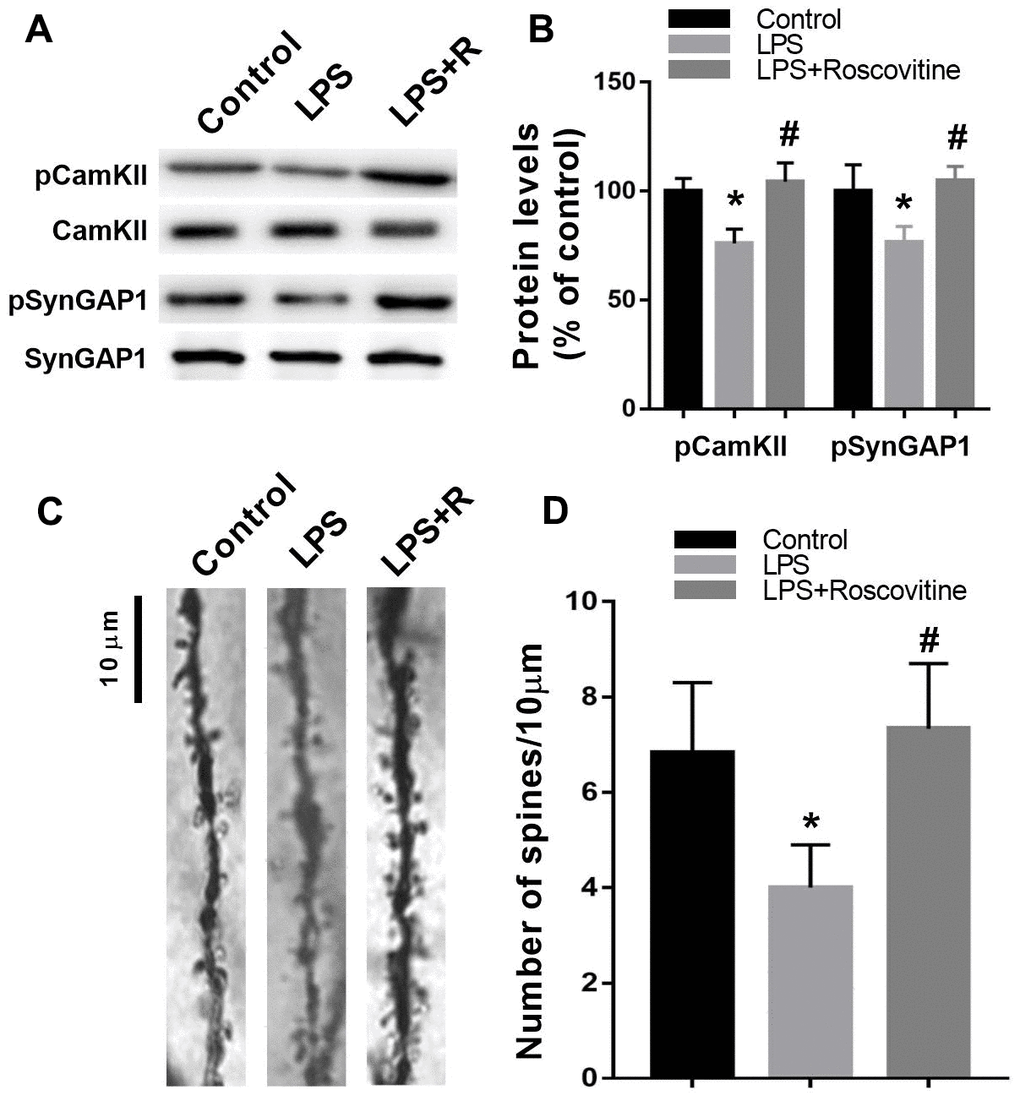
Figure 7. Decreased hippocampal pCamKII, pSynGAP levels, and dendritic spine density following LPS challenge were rescued by roscovitine. (A, B) LPS induced significantly decreased hippocampal pCamKII and pSynGAP levels, which were prevented by roscovitine treatment. (C, D) LPS induced significantly increased hippocampal dendritic spine loss, which was reversed by roscovitine treatment. Data are presented as the mean ± SEM, n = 4, *P < 0.05 vs control group; #P < 0.05 vs LPS group. LPS, lipopolysaccharide.
Decreased theta and gamma oscillations in the CA1 of the hippocampus following LPS challenge were prevented by roscovitine
It has been shown that Syngap1 plays a critical role in network function [14], we further examined whether decreased pSynGAP1 affected brain oscillations in the CA1 of the hippocampus following LPS challenge. As shown in Figure 8, we found that theta and gamma oscillation power were significantly reduced in the LPS group, which were rescued by roscovitine (theta oscillation: F2,9 = 8.799, P = 0.0076; gamma oscillation: F2,9 = 7.391, P = 0.0126). However, there was no difference in α and β oscillation power among groups (alpha oscillation: F2,9 = 2.697, P = 0.1209; beta oscillation: F2,9 = 4.13, P = 0.0534).
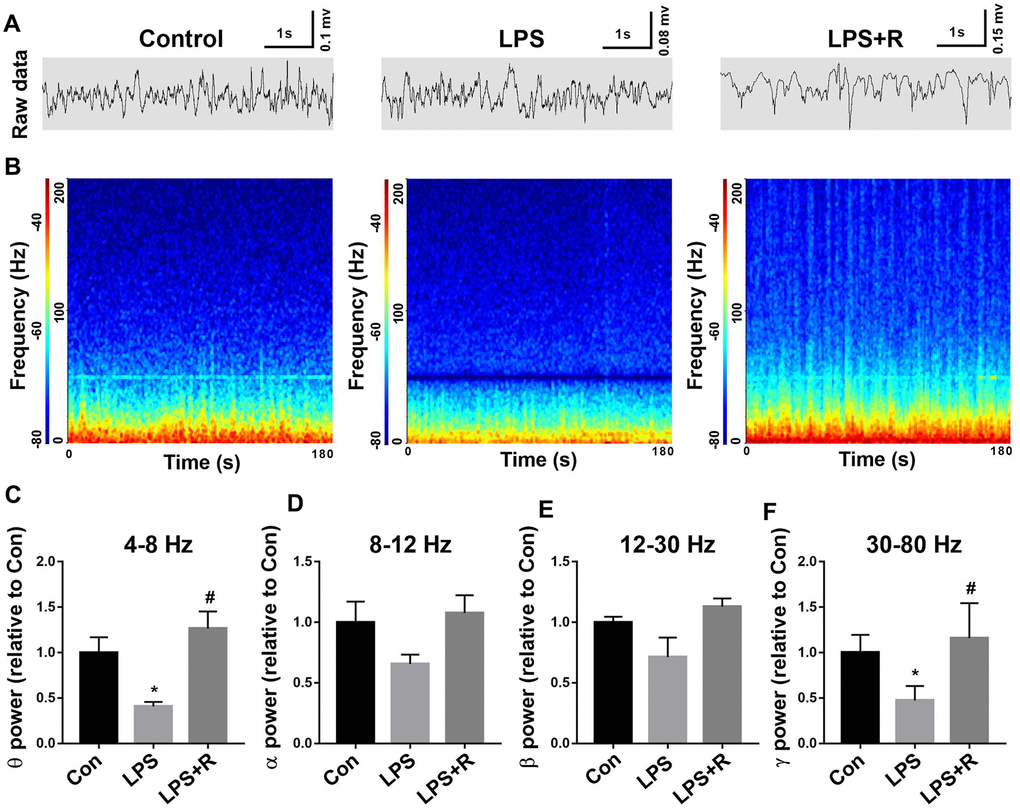
Figure 8. Decreased gamma oscillation in the CA1 of the hippocampus following LPS challenge was prevented by roscovitine. (A, B) Example recordings and example power spectra in the hippocampal. (C–F) Summary of LFP power, including θ, α, β, and γ oscillation. The theta and gamma oscillation powers were significantly lower in LPS group when compared with control group, which were prevented by roscovitine. Data are shown as mean ± SEM, n = 4, *P < 0.05 vs control group; #P < 0.05 vs LPS group. LPS, lipopolysaccharide; R, roscovitine.
LPS-induced neurobehavioral abnormities were attenuated by roscovitine
The open field test was performed to investigate whether LPS influences locomotor activity and anxiety-like behavior. As shown in Figure 9A, 9B, LPS had no effect on the total distance traveled. However, roscovitine treatment significantly increased total distance traveled in the LPS + roscovitine group compared with LPS group (F2,31 = 4.054, P = 0.0273). There was no difference in time spent in the center of the open arena among groups (F2,31 = 0.7745, P = 0.4696). In the elevated plus maze, there was a trend toward a decreased time in the open arms (F2,31 = 3.264, P = 0.0517, Figure 9C). In addition, no difference in time in the closed arms was observed between groups (F2,31 = 1.593, P = 0.2194, Figure 9D). However, CLP did not significantly affect total distance traveled (t = 0.1962, P = 0.8462, Figure 10A) or time spent in the center (t = 0.2069, P = 0.838, Figure 10B) when compared with the sham group. In addition, there was no difference in the time in the open (t = 0.2585, P = 0.7985, Figure 10C) or closed arms (t = 0.6287, P = 0.536, Figure 10D) between the two groups.
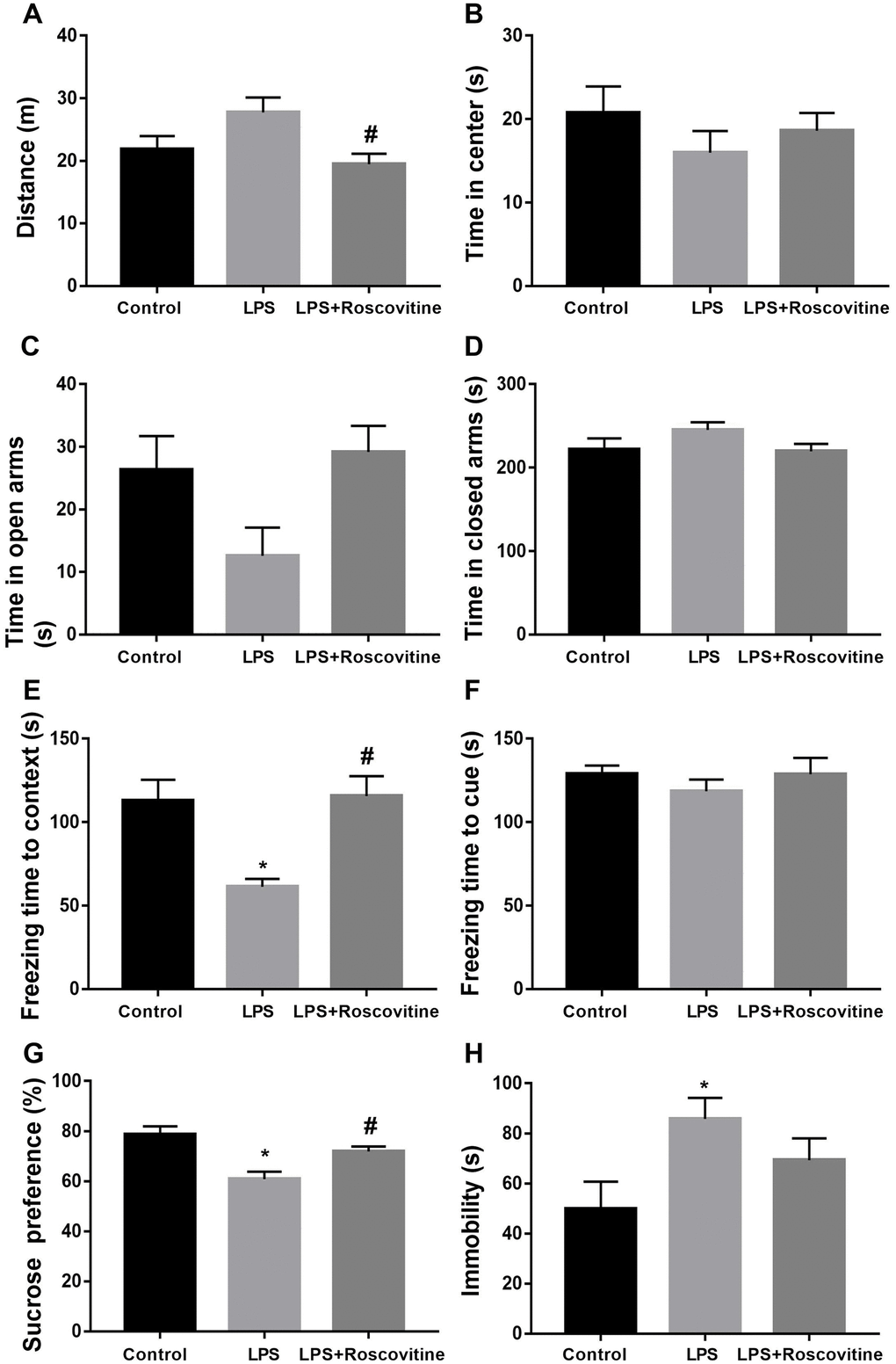
Figure 9. LPS-induced neurobehavioral abnormities were attenuated by roscovitine. (A) LPS had no effect on the total distance traveled, while roscovitine treatment significantly increased total distance traveled in LPS + roscovitine group compared with LPS group. (B) No difference in time spent in the center of the open arena was observed among groups. (C, D) There was no difference in time in the open arms and closed arms between groups. (E) LPS-induced significantly decreased the freezing time to context was reversed by roscovitine treatment. (F) There was no difference in freezing time to tone in the auditory-cued fear test among groups. (G) Decreased preference for sucrose in LPS-exposed mice was reversed by roscovitine treatment. (H) LPS significantly increased immobility compared with control group, which was not prevented by roscovitine treatment. Data are presented as the mean ± SEM, n = 10-12, *P < 0.05 vs control group; #P < 0.05 vs LPS group. LPS, lipopolysaccharide.
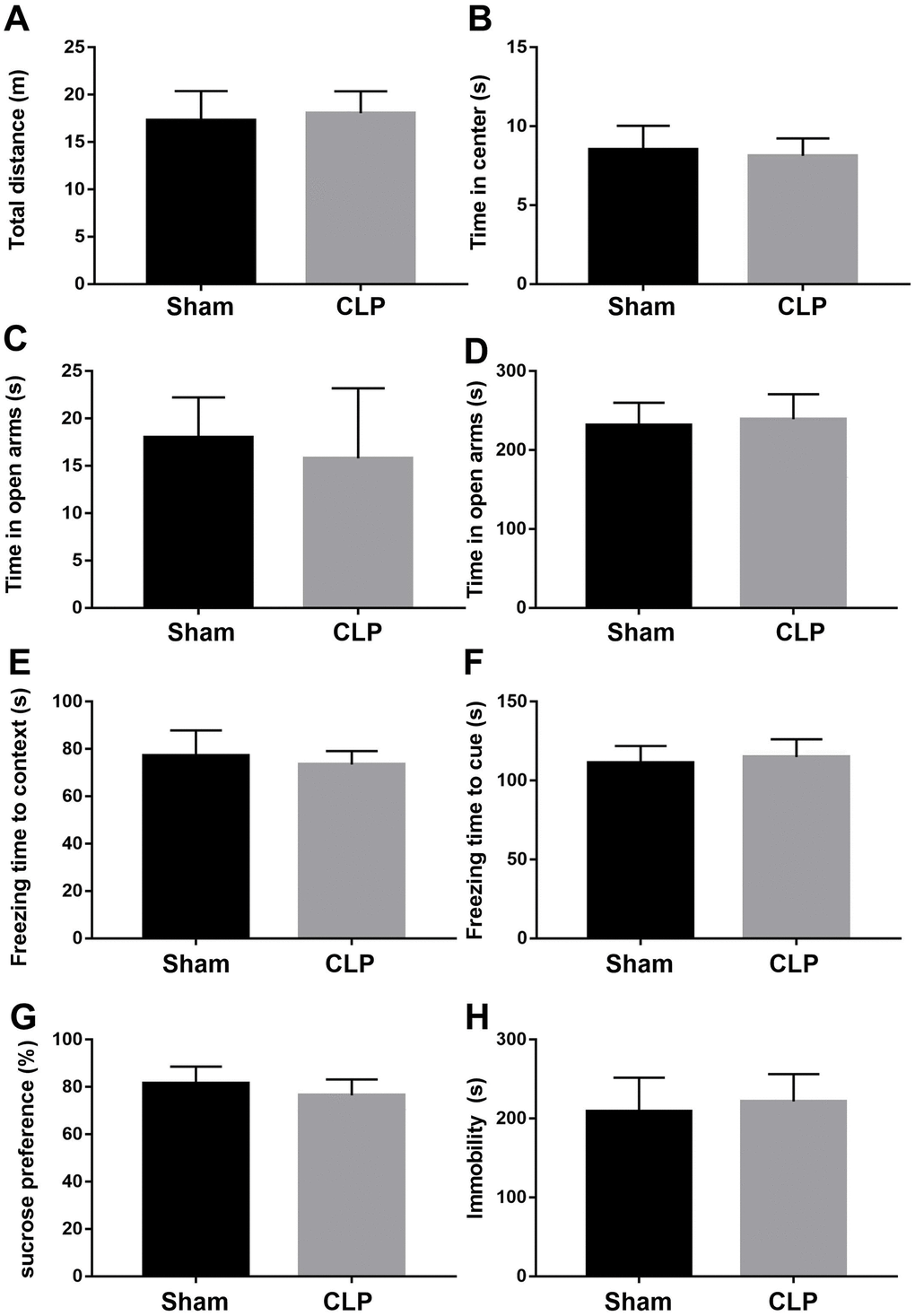
Figure 10. CLP did not induce neurobehavioral abnormities. (A, B) CLP had no effect on the total distance traveled and time spent in the center of the open arena compared with sham group. (C, D) There was no difference in time in the open arms and closed arms between groups. (E, F) There was no difference in freezing time to context or tone in fear conditioning tests between groups. (G, H) CLP had no effect on preference for sucrose or immobility compared with sham group. Data are presented as the mean ± SEM, n = 10-12, *P < 0.05 vs sham group. CLP, cecal ligation and puncture.
We performed the fear conditioning tests to evaluate whether LPS challenge impaired contextual fear memory. As revealed in Figure 9E, 9F, LPS significantly decreased the freezing time to context relative to that of control group (F2,31 = 7.384, P = 0.0024), which was reversed by roscovitine treatment. However, there was no difference in freezing time to tone in the auditory-cued fear test among groups (F2,31 = 0.561, P = 0.5763). Also, there was no difference in freezing time to context (t = 0.3019, P = 0.7655, Figure 10E) or tone (t = 0.2329, P = 0.818, Figure 10F) between the sham and CLP groups in the fear conditioning tests.
In the sucrose preference test, LPS-exposed mice displayed significantly decreased preference for sucrose relative to that of control group (F2,31 = 10.08, P = 0.0004, Figure 9G). In the forced swim test, LPS significantly increased immobility compared with control group (F2,31 = 3.46, P = 0.044, Figure 9H), suggesting LPS induced depression like behavior. However, roscovitine treatment only reversed the sucrose preference but not immobility. Again, CLP had no effect on the preference for sucrose (t = 1.778, P = 0.0893, Figure 10G) or immobility (t = 1.195, P = 0.2477, Figure 10H) in the CLP group compared with the sham group.
Discussion
The long-term consequences of sepsis and its pathophysiological mechanisms are complex, and have not been fully elucidated. The large-scale proteomic analysis provides comprehensive information about the regulation of inflammation-associated proteins after sepsis. To the best of our knowledge, this is the first study investigating the long-term neurobehavioral abnormities following LPS exposure. More importantly, we showed that pSynGAP1 disturbance plays a key role in hippocampal oscillation network impairment, which might contribute to long-term neurobehavioral abnormities in sepsis survivors.
Systemic inflammation can impair cognition with relevance to dementia, implying that these acute events induce or exacerbate central nervous system (CNS) pathology, even in the absence of overt invasion of bacteria into the CNS [15]. In animal models of sepsis, induction of neuroinflammation by LPS or CLP increased intracellular accumulation of amyloid precursor protein and amyloid β peptide and consequent cognitive impairments [16, 17]. In human studies, it has been demonstrated that patients with delirium due to sepsis in the Intensive Care Unit showed significant cognitive impairments at 12-18 months after hospital discharge when compared with controls [1]. Recent epidemiological study reports that human survivors of sepsis have an increased risk of long-term cognitive decline such as AD [18]. These classical pathology hallmarks are accompanied by neuroinflammation, synaptic loss, and brain atrophy [4, 5, 19]. Although increasing evidence has suggested that CLP is more clinically relevant, our study showed that LPS challenge but not CLP led to long-term neurobehavioral abnormities. This can be explained by the reason that our model of CLP is mild and thus does not significantly affect brain function. Yet, the mechanism underlying LPS-induced long-term neurobehavioral abnormities remains to be elucidated.
Transcriptomic analysis provided information about the regulation of mRNAs in animal models with sepsis [20]. However, the findings from transcriptomic studies do not always translate into proteome alterations due to post-transcriptional and post-translational regulation mechanisms. The development of quantitative proteomics approaches has greatly accelerated the understanding of various cellular and physiological processes and how these are affected by disease allowing the identification of novel biomarkers. Developments in LC-MS–based proteomics and phosphoproteomics in particular, enable the comprehensive characterization of proteomes and tens of thousands of phosphorylation events [11, 21]. It is a powerful tool for identifying novel molecule biomarkers and also provides insights into the pathophysiology of neurodegenerative and other brain-related diseases [22]. Protein functions can be switched on or off by site-specific phosphorylation, or modulated by cumulative phosphorylation of multiple sites, which is an important posttranslational modification that regulates protein function and plays a prominent role in diverse biological phenomena [23]. Thus, this approach becomes an efficient tool to investigate global signaling-level changes in biological systems [24]. It is estimated that one-third of all proteins are likely to be phosphorylated, thus phosphoproteomic analysis offers an excellent potential for the identification of candidate regulatory proteins in various cellular states. To our knowledge, however, no previous study has utilized this method to investigate long-term hippocampal phosphoproteomic alterations following sepsis development. In the present study, we showed that several cellular signaling cascades related to apoptosis, mitochondria dysfunction, and microtubule-associated protein were significantly up-regulated following LPS, including increased phosphorylation of Cdk11b, Atp8a1, Iscu, Map1b, Dctn1, Map2, and Ank2. Thus, we further performed some of these functional changes and showed that LPS exposure induced apoptosis and mitochondria dysfunction, as reflected by significantly increased cleaved caspase-3 and cytochrome C expressions in the hippocampus. These results are consistent with previous findings in the literature indicating that apoptosis, mitochondria dysfunction, and disturbance in microtubule-associated protein are required for hippocampus-dependent learning [25–27]. However, LPS did not significantly affect microtubule-associated protein such as MAP-2, suggesting microtubule-associated protein dysfunction may be not an important factor contributing to long-term neurobehavioral abnormities induced by LPS. In addition to neuronal cell signaling pathway, we have also identified several biological functions altered in LPS-exposed mice, such as Syngap1, Bsn, Shisa6, Synpo, Pclo, Ppp1r9b, and Dlgap2 for synaptic proteins.
Indeed, synaptic dysfunction is widely proposed as an initial insult leading to the neurodegeneration observed in AD [19, 21]. Although some of the protein phosphorylation changes observed were not related to cognition, use of this discovery-based approach represents a novel method for determining signaling events involved in specific memory processes. However, it should be noted that using stricter threshold to define differences in protein expressions will provide more solid evidence. In addition, morphology or functional analysis of these protein phosphorylation changes caused by LPS are also needed in our future studies.
Based on the results of protein-protein interaction analysis, Syngap1 is identified among the hub of synaptic plasticity. SynGAP is a protein abundant at the postsynaptic density of glutamatergic neurons and modulates synaptic strength by regulating the incorporation of AMPA receptors at the synapse [28]. Structurally, SynGAP is linked to postsynaptic scaffold proteins, which is critically involved in synapse density, synaptic physiology, and long-term potentiation [29]. In contrast, SynGAP disturbance has been linked to many neuropsychical diseases such as intellectual disability and autism spectrum disorders [30]. 1t has been shown that pSynGAP1 is required for AMPA receptor insertion and spine enlargement, whereas inhibition of SynGAP dispersion by CaMKII inhibitor prevents long- term potentiation [31]. This suggested that pSynGAP1 is regulated by pCaMKII and critically involved in synaptic plasticity. To test this hypothesis, we used a Cdk5 inhibitor roscovitine, which has been shown to upregulate pSynGAP1 level and improve cognitive impairment [13, 32, 33]. Consistently, we also showed that roscovitine reversed LPS-induced synaptic loss in the hippocampus. On the other hand, roscovitine is a Cdk5 inhibitor and is reported to have other effects such as anti-inflammatory property [34]. Thus, in addition to unregulated pSynGAP1, other mechanisms might also be involved in the beneficial effects of roscovitine for sepsis survivors.
Brain functions such as perception and cognition are based on particular functional network. It has been shown that neural assemblies involved in these cognitive functions will oscillate in a synchronized manner at specific frequencies while processing information [35]. In particular, theta oscillations are linked to various cognitive processes, especially for hippocampal-dependent memory function [36], whereas gamma oscillations are implicated in perception, learning, and memory [37]. To test whether altered oscillations in the brain are involved in LPS-induced long-term neurobehavioral abnormities, we used multichannel microwire array to record in vivo LFP, we found significantly decreased theta and gamma oscillations during locomotion in LPS-exposed animals, but did not significantly affect alpha and beta oscillation power. Although alpha and beta oscillations have also been involved in cognition [38], we found LPS selectively induced impairments in theta and gamma oscillations. Notably, we showed roscovitine rescued LPS-induced hippocampal oscillation disturbance and consequent neurobehavioral abnormities following LPS exposure, suggesting disturbance of pSynGAP1 is critical for hippocampal oscillation network impairment and may serve as a therapeutic target for sepsis related cognitive disorder.
In conclusion, our study provides new evidence that pSynGAP1 disturbance-mediated hippocampal oscillation network impairment might contribute to long-term neurobehavioral abnormities in sepsis survivors. However, further studies using more specific approach are needed to confirm our results. Moreover, other animal models of sepsis should also be used to confirm our current results.
Materials and Methods
Animal model
The animal care and the experiment were approved by the Ethics Committee of First Affiliated Hospital of Zhengzhou University, Zhengzhou, China and were performed according to the Guide for the Care and Use of Laboratory Animals approved by the National Institutes of Health of the United States. One hundred seventy-two male C57BL/6 mice (3-4 months) were purchased from the Animal Center of Jinling Hospital, Nanjing, China. Due to animal death and different experimental purposes, we intentionally allocated more animals in the LPS or CLP group. Animals were randomly allocated to the following groups: control group (n =25), LPS group (n = 40), LPS + roscovitine group (n = 33), sham (n = 22) or CLP (n = 52) group. The mice were housed 4-5 per cage on a 12-h light–dark cycle in a room of 22-25 °C with food and water available ad libitum. Before the experimental study, animals were allowed to acclimatize for at least one week.
Animal models of sepsis
We established the animal models of sepsis by utilizing LPS in a rodent model of sepsis as previously described [5, 6]. For LPS injection, mice received LPS (Escherichia coli endotoxin 0111: B4, Lot # 064M4125V, Sigma, Shanghai, China, 5 mg/kg). All the procedures were performed by an experienced investigator to keep the model stable.
CLP model was induced as we previously described [4]. Briefly, animals were anesthetized with 2% sodium pentobarbital (50 mg/kg; Sigma Chemical Co, St. Louis, MO, USA) by intraperitoneal (i.p.) injection and a 1-cm ventral midline laparotomy was performed. After then, the cecum was carefully exposed and ligated with a 4.0 silk suture, about 0.5 cm below the ileocecal valve. Subsequently, the cecum was perforated with 22–gauge needle and gently compressed to extrude a small amount of feces. Finally, the cecum was returned to the peritoneal cavity and the laparotomy was closed with 4.0 silk sutures. Immediately after the operation, animals received fluid resuscitation with normal saline solution (subcutaneously, 20 ml/kg of body weight) and antibiotic therapy (ertapenem, 20 mg kg-1; Merck Research Laboratory, USA). All mice were returned to their cages with free access to food and water. For sham group, animals were treated identically without ligation or puncture of the cecum.
Drugs
Roscovitine (20 mg/kg, R-1234; LC Laboratories) or equal volume vehicle (0.2% dimethylsulfoxide) was injected i.p. daily for 3 days before and until the end of behavioral testing. The dosage of roscovitine was used based on previous study that 20 mg/kg/day roscovitine attenuated diabetes-related cognitive deficits [13].
Behavioral experiments
Twelve months following LPS injection, a battery of well-established behavioral tests was used to assess behavioral alterations as we previously described [4, 39]. All behavioral studies were performed between 11:00 AM and 17:00 PM under dim lighting conditions. All the behavior of mice was recorded by a video camera (XR-XZ301, Shanghai Softmaze Information Technology Co. Ltd., Shanghai, China).
Open field test
The exploratory activities of the mice were evaluated by the open field test. Each mouse was released in the center of the white plastic chamber (50 cm × 50 cm × 40 cm), and allowed to explore for 5 min. Total distance traveled and time spent in the center of the arena were automatically recorded by a video tracking system. At the end of testing, the arena was cleaned with 75% alcohol to avoid the presence of olfactory cues.
Sucrose preference test
Anhedonia was measured by preference for a sucrose solution over water, using a two-bottle free choice method. Before test, mice were trained to consume two bottles of 1% sucrose solution for 24 h. On the testing day, each mouse was given two bottles of drinking containing either 1% sucrose solution or water for 24 h, where the position of the two bottles was switched to control for a side preference in drinking behavior at 12 h. Sucrose preference was calculated as sucrose consumption/(sucrose consumption + water consumption) × 100%).
Fear conditioning test
The mouse was placed in the conditioning chamber (32 cm × 25 cm × 25 cm) for 3 min as an accommodation period and then one tone-foot-shock pairing (tone, 30 s, 65 dB, 1 kHz; foot-shock, 2 s, 0.8 mA) was delivered. Twenty-four hours later, mouse was placed back into the same chamber for 5 min without the tone and shock. The tone fear conditioning test was assessed 2 h after the contextual fear conditioning test in a novel chamber changed in the shape, color, and smell and the training tone was delivered for 3 min. The freezing behavior in these two chambers was video recorded, which was defined as the absence of all visible movement of the body except for respiration.
Elevated plus maze test
Anxiety-like behavior was assessed by elevated plus maze test, where a central platform is connected to four arms (50 cm long, 10 cm wide, 70 cm above ground). Two opposite arms were enclosed by 20 cm high walls. Animals were placed onto the center platform with the head toward an open arm and allowed to move freely for 5 min. The sessions were videotaped by a camera over the center of the maze and the time spent in the open and closed arms. The maze was thoroughly cleaned with 70% ethanol between each test session.
Forced swim test
This test measures depressive-like behavior with immobility taken as the dependent measure of behavioral despair. Mice were placed singly in a 4 litre clear plexiglass beaker (15 cm diameter 30 cm height) filled with water (20-24 °C) for 6 min, with the immobility scored in the final 4 min only. Time spent immobile (absence of movement except leg kicks to stay afloat) is then used as a measure of behavioral despair and helplessness.
Electrophysiological recordings and analysis
Electrophysiological recordings and analysis were performed as we previously described [40]. Mice were anesthetized with 2% sodium pentobarbital in saline (40 mg/kg, i.p.; Sigma, St Louise, MO, USA) and fixed in a stereotaxic apparatus on a temperature-regulated heating pad set to maintain body temperature at 36–37 °C. After surgical preparation and craniotomies, local field potentials (LFP) were recorded from hippocampal CA1 region (2.1 mm posterior, 1.5 mm lateral, and 1.5mm depth) using a 8-channel microwire array. All electrodes were joined to a miniature connector and were then fixed to the skull using dental acrylic. After 7-day recovery period, LFP were recorded continuously (sampling rate = 1000 Hz; bandpass filter = 1–400 Hz) when the animals explored in the open arena. The recorded LFP were filtered by a 50 Hz notching filter to remove the powerline artifact. At the end of recordings, animals were deeply anesthetized and brains were removed and fixed for verification of electrode placement. All data analyses were performed by Neuroexplorer (Plexon Inc., Dallas, TX) software.
Protein extraction, protein digestion, iTRAQ labeling, and MS/MS analysis
Protein extraction, protein digestion, iTRAQ labeling, and MS/MS analysis were described as previously [11, 12]. Briefly, the hippocampal tissues of 6 mice from each group were sacrificed by pentobarbital injection (50 mg/kg i.p.). Mouse tissues were quickly collected, rinsed with phosphate buffered saline (PBS) and flash frozen in liquid nitrogen. Samples were extracted and digested, and the tryptic peptides were labeled using the iTRAQ Reagent-8plex Multiplex Kit. The samples from the control group were labeled with iTRAQ tags 113 and 114, while tags 115 and 116 were used for the LPS group. iTRAQ labeling and tandem mass spectrometry analysis were carried out by Proteome Discoverer 2.1 (Thermo Fisher Scientific). The following parameters thresholds were set as : FDR ≤ 0.01, P value <0.05, and 1.2-fold change (expression difference >1.2-fold or <0.83-fold).
Western blot analysis
Proteins extracted from the hippocampus were processed for western blot as we described previously [4]. In brief, an equal amount of protein (20 μg) was separated on 10% SDS-PAGE and transferred to PVDF membrane. Membranes were blocked with blocking buffer (5% BSA, 10 mM Tris pH, 7.5, 100 mM NaCl, and 0.1% tween-20) for 2 h. Membranes were then incubated overnight at 4 °C with primary antibodies rabbit anti-cleaved caspase-3 (1:1000; Cell Signaling Technology, USA), anti-cytochrome C (1:1000; Servicebio, Wuhan, China), anti-MAP2 (1:1000; Servicebio, Wuhan, China), anti-SynGAP1 (1:500; APExBIO, USA), anti-CamKII (1:1000; Abcam, Cambridge, UK), anti-pCamKII (1:1000; APExBIO, USA), anti-pSynGAP (Ser 1123, 1:500; bs-10392R, Beijing, China), and GAPDH (1:5000; Sigma St. Louis, MO, USA). After washing, membranes were incubated with respective HRP-conjugated secondary antibodies (1:5000; Santa Cruz Biotechnology, USA) for 2 h at room temperature. Protein bands were visualized and quantitated using ImageJ software (NIH Image, Bethesda, USA).
Golgi staining
The brains of mice were assessed by a Golgi Stain Kit (#PK401, FD NeuroTechnologies, Columbia, MD, USA) at the ending of the behavioral tests as we previously described [41]. Briefly, mice were deeply anesthetized by sodium pentobarbital (60 mg/kg, i.p.; Sigma, St Louise, MO, USA) and rapidly sacrificed. The brains were immersed in impregnation solution (a mixture of solution A and B) and stored in the dark at room temperature for 3 weeks. Then, the brains were transferred into Solution C and stored for 7 days. Finally, the brains were sliced at a thickness of 100 μm, stained and then mounted on gelatin-coated slides. The dendrites from hippocampal neurons in CA1 region were captured with a confocal microscope (× 100 oil objective). Dendritic spine density were detected along CA1 secondary dendrites starting from their point of origin on the primary dendrite and the counting was performed by an experimenter blinded to the group of each sample.
Statistical analysis
Statistical analysis was performed using GraphPad Prism 7.0 (GraphPad Software, La Jolla, CA, USA). Data are displayed as mean ± SEM. Comparisons between two groups were performed by independent-t test or Mann-Whitney U test where appropriate. Differences among multiple groups were assessed with one-way ANOVA followed by post-hoc Tukey multiple comparisons. The survival rate was estimated by Kaplan–Meier method and compared by the log–rank test. A P < 0.05 was considered statistically significant.
Author Contributions
Yong Wang performed the majority of the experiments and prepared the manuscript. Hua Wei performed the experiments and prepared the manuscript. Jianhua Tong performed the experiments and literature searches. Muhuo Ji and Jianjun Yang designed the entire manuscript and guided the project.
Conflicts of Interest
All authors listed declare that there are no conflicts of interest related to this work.
Funding
This study was supported by the grants from the National Natural Science Foundation of China (Nos., 81771156, 81772126, 81971892).
References
- 1. Iwashyna TJ, Ely EW, Smith DM, Langa KM. Long-term cognitive impairment and functional disability among survivors of severe sepsis. JAMA. 2010; 304:1787–94. https://doi.org/10.1001/jama.2010.1553 [PubMed]
- 2. Pandharipande PP, Girard TD, Jackson JC, Morandi A, Thompson JL, Pun BT, Brummel NE, Hughes CG, Vasilevskis EE, Shintani AK, Moons KG, Geevarghese SK, Canonico A, et al, and BRAIN-ICU Study Investigators. Long-term cognitive impairment after critical illness. N Engl J Med. 2013; 369:1306–16. https://doi.org/10.1056/NEJMoa1301372 [PubMed]
- 3. Rengel KF, Hayhurst CJ, Pandharipande PP, Hughes CG. Long-term cognitive and functional impairments after critical illness. Anesth Analg. 2019; 128:772–80. https://doi.org/10.1213/ANE.0000000000004066 [PubMed]
- 4. Ji MH, Qiu LL, Tang H, Ju LS, Sun XR, Zhang H, Jia M, Zuo ZY, Shen JC, Yang JJ. Sepsis-induced selective parvalbumin interneuron phenotype loss and cognitive impairments may be mediated by NADPH oxidase 2 activation in mice. J Neuroinflammation. 2015; 12:182. https://doi.org/10.1186/s12974-015-0401-x [PubMed]
- 5. Zhang S, Wang X, Ai S, Ouyang W, Le Y, Tong J. Sepsis-induced selective loss of NMDA receptors modulates hippocampal neuropathology in surviving septic mice. PLoS One. 2017; 12:e0188273. https://doi.org/10.1371/journal.pone.0188273 [PubMed]
- 6. Anderson ST, Commins S, Moynagh PN, Coogan AN. Lipopolysaccharide-induced sepsis induces long-lasting affective changes in the mouse. Brain Behav Immun. 2015; 43:98–109. https://doi.org/10.1016/j.bbi.2014.07.007 [PubMed]
- 7. Maheshwari P, Eslick GD. Bacterial infection and Alzheimer’s disease: a meta-analysis. J Alzheimers Dis. 2015; 43:957–66. https://doi.org/10.3233/JAD-140621 [PubMed]
- 8. Keshishian H, Burgess MW, Specht H, Wallace L, Clauser KR, Gillette MA, Carr SA. Quantitative, multiplexed workflow for deep analysis of human blood plasma and biomarker discovery by mass spectrometry. Nat Protoc. 2017; 12:1683–701. https://doi.org/10.1038/nprot.2017.054 [PubMed]
- 9. Xie H, Huang H, Tang M, Wu Y, Huang R, Liu Z, Zhou M, Liao W, Zhou J. iTRAQ-based quantitative proteomics suggests synaptic mitochondrial dysfunction in the hippocampus of rats susceptible to chronic mild stress. Neurochem Res. 2018; 43:2372–83. https://doi.org/10.1007/s11064-018-2664-y [PubMed]
- 10. Liu X, Zheng W, Wang W, Shen H, Liu L, Lou W, Wang X, Yang P. A new panel of pancreatic cancer biomarkers discovered using a mass spectrometry-based pipeline. Br J Cancer. 2017; 117:1846–54. https://doi.org/10.1038/bjc.2017.365 [PubMed]
- 11. Keshishian H, Burgess MW, Gillette MA, Mertins P, Clauser KR, Mani DR, Kuhn EW, Farrell LA, Gerszten RE, Carr SA. Multiplexed, quantitative workflow for sensitive biomarker discovery in plasma yields novel candidates for early myocardial injury. Mol Cell Proteomics. 2015; 14:2375–93. https://doi.org/10.1074/mcp.M114.046813 [PubMed]
- 12. Henningsen K, Palmfeldt J, Christiansen S, Baiges I, Bak S, Jensen ON, Gregersen N, Wiborg O. Candidate hippocampal biomarkers of susceptibility and resilience to stress in a rat model of depression. Mol Cell Proteomics. 2012; 11:M111.016428. https://doi.org/10.1074/mcp.M111.016428 [PubMed]
- 13. Posada-Duque RA, Ramirez O, Härtel S, Inestrosa NC, Bodaleo F, González-Billault C, Kirkwood A, Cardona-Gómez GP. CDK5 downregulation enhances synaptic plasticity. Cell Mol Life Sci. 2017; 74:153–72. https://doi.org/10.1007/s00018-016-2333-8 [PubMed]
- 14. Berryer MH, Chattopadhyaya B, Xing P, Riebe I, Bosoi C, Sanon N, Antoine-Bertrand J, Lévesque M, Avoli M, Hamdan FF, Carmant L, Lamarche-Vane N, Lacaille JC, et al. Decrease of SYNGAP1 in GABAergic cells impairs inhibitory synapse connectivity, synaptic inhibition and cognitive function. Nat Commun. 2016; 7:13340. https://doi.org/10.1038/ncomms13340 [PubMed]
- 15. Gofton TE, Young GB. Sepsis-associated encephalopathy. Nat Rev Neurol. 2012; 8:557–66. https://doi.org/10.1038/nrneurol.2012.183 [PubMed]
- 16. Eimerbrink MJ, Pendry RJ, Hodges SL, Wiles JD, Peterman JL, White JD, Hayes HB, Chumley MJ, Boehm GW. The α5-GABAAR inverse agonist MRK-016 upregulates hippocampal BDNF expression and prevents cognitive deficits in LPS-treated mice, despite elevations in hippocampal Aβ. Behav Brain Res. 2019; 359:871–77. https://doi.org/10.1016/j.bbr.2018.07.013 [PubMed]
- 17. Schwalm MT, Pasquali M, Miguel SP, Dos Santos JP, Vuolo F, Comim CM, Petronilho F, Quevedo J, Gelain DP, Moreira JC, Ritter C, Dal-Pizzol F. Acute brain inflammation and oxidative damage are related to long-term cognitive deficits and markers of neurodegeneration in sepsis-survivor rats. Mol Neurobiol. 2014; 49:380–85. https://doi.org/10.1007/s12035-013-8526-3 [PubMed]
- 18. Lövheim H, Gilthorpe J, Johansson A, Eriksson S, Hallmans G, Elgh F. Herpes simplex infection and the risk of Alzheimer’s disease: a nested case-control study. Alzheimers Dement. 2015; 11:587–92. https://doi.org/10.1016/j.jalz.2014.07.157 [PubMed]
- 19. Bos I, Vos S, Verhey F, Scheltens P, Teunissen C, Engelborghs S, Sleegers K, Frisoni G, Blin O, Richardson JC, Bordet R, Tsolaki M, Popp J, et al. Cerebrospinal fluid biomarkers of neurodegeneration, synaptic integrity, and astroglial activation across the clinical Alzheimer’s disease spectrum. Alzheimers Dement. 2019; 15:644–54. https://doi.org/10.1016/j.jalz.2019.01.004 [PubMed]
- 20. Srinivasan K, Friedman BA, Larson JL, Lauffer BE, Goldstein LD, Appling LL, Borneo J, Poon C, Ho T, Cai F, Steiner P, van der Brug MP, Modrusan Z, et al. Untangling the brain’s neuroinflammatory and neurodegenerative transcriptional responses. Nat Commun. 2016; 7:11295. https://doi.org/10.1038/ncomms11295 [PubMed]
- 21. Chen C, Jiang X, Li Y, Yu H, Li S, Zhang Z, Xu H, Yang Y, Liu G, Zhu F, Ren X, Zou L, Xu B, et al. Low-dose oral copper treatment changes the hippocampal phosphoproteomic profile and perturbs mitochondrial function in a mouse model of Alzheimer’s disease. Free Radic Biol Med. 2019; 135:144–56. https://doi.org/10.1016/j.freeradbiomed.2019.03.002 [PubMed]
- 22. Hosp F, Mann M. A primer on concepts and applications of proteomics in neuroscience. Neuron. 2017; 96:558–71. https://doi.org/10.1016/j.neuron.2017.09.025 [PubMed]
- 23. Wang Z, Ma J, Miyoshi C, Li Y, Sato M, Ogawa Y, Lou T, Ma C, Gao X, Lee C, Fujiyama T, Yang X, Zhou S, et al. Quantitative phosphoproteomic analysis of the molecular substrates of sleep need. Nature. 2018; 558:435–39. https://doi.org/10.1038/s41586-018-0218-8 [PubMed]
- 24. Derouiche A, Cousin C, Mijakovic I. Protein phosphorylation from the perspective of systems biology. Curr Opin Biotechnol. 2012; 23:585–90. https://doi.org/10.1016/j.copbio.2011.11.008 [PubMed]
- 25. Cao Y, Li Q, Liu L, Wu H, Huang F, Wang C, Lan Y, Zheng F, Xing F, Zhou Q, Li Q, Shi H, Zhang B, et al. Modafinil protects hippocampal neurons by suppressing excessive autophagy and apoptosis in mice with sleep deprivation. Br J Pharmacol. 2019; 176:1282–97. https://doi.org/10.1111/bph.14626 [PubMed]
- 26. Liu Y, Yan J, Sun C, Li G, Li S, Zhang L, Di C, Gan L, Wang Y, Zhou R, Si J, Zhang H. Ameliorating mitochondrial dysfunction restores carbon ion-induced cognitive deficits via co-activation of NRF2 and PINK1 signaling pathway. Redox Biol. 2018; 17:143–57. https://doi.org/10.1016/j.redox.2018.04.012 [PubMed]
- 27. Palenzuela R, Gutiérrez Y, Draffin JE, Lario A, Benoist M, Esteban JA. MAP1B light chain modulates synaptic transmission via AMPA receptor intracellular trapping. J Neurosci. 2017; 37:9945–63. https://doi.org/10.1523/JNEUROSCI.0505-17.2017 [PubMed]
- 28. Creson TK, Rojas C, Hwaun E, Vaissiere T, Kilinc M, Jimenez-Gomez A, Holder JL
Jr , Tang J, Colgin LL, Miller CA, Rumbaugh G. Re-expression of SynGAP protein in adulthood improves translatable measures of brain function and behavior. Elife. 2019; 8:e46752. https://doi.org/10.7554/eLife.46752 [PubMed] - 29. Jeyabalan N, Clement JP. SYNGAP1: mind the gap. Front Cell Neurosci. 2016; 10:32. https://doi.org/10.3389/fncel.2016.00032 [PubMed]
- 30. Verma V, Paul A, Amrapali Vishwanath A, Vaidya B, Clement JP. Understanding intellectual disability and autism spectrum disorders from common mouse models: synapses to behaviour. Open Biol. 2019; 9:180265. https://doi.org/10.1098/rsob.180265 [PubMed]
- 31. Araki Y, Zeng M, Zhang M, Huganir RL. Rapid dispersion of SynGAP from synaptic spines triggers AMPA receptor insertion and spine enlargement during LTP. Neuron. 2015; 85:173–89. https://doi.org/10.1016/j.neuron.2014.12.023 [PubMed]
- 32. Walkup WG
4th , Washburn L, Sweredoski MJ, Carlisle HJ, Graham RL, Hess S, Kennedy MB. Phosphorylation of synaptic GTPase-activating protein (synGAP) by Ca2+/calmodulin-dependent protein kinase II (CaMKII) and cyclin-dependent kinase 5 (CDK5) alters the ratio of its GAP activity toward ras and rap GTPases. J Biol Chem. 2015; 290:4908–27. https://doi.org/10.1074/jbc.M114.614420 [PubMed] - 33. Liu W, Zhou Y, Liang R, Zhang Y. Inhibition of cyclin-dependent kinase 5 activity alleviates diabetes-related cognitive deficits. FASEB J. 2019; 33:14506–15. https://doi.org/10.1096/fj.201901292R [PubMed]
- 34. Pfänder P, Fidan M, Burret U, Lipinski L, Vettorazzi S. Cdk5 deletion enhances the anti-inflammatory potential of GC-mediated GR activation during inflammation. Front Immunol. 2019; 10:1554. https://doi.org/10.3389/fimmu.2019.01554 [PubMed]
- 35. Cole SR, Voytek B. Brain oscillations and the importance of waveform shape. Trends Cogn Sci. 2017; 21:137–49. https://doi.org/10.1016/j.tics.2016.12.008 [PubMed]
- 36. Makino Y, Polygalov D, Bolaños F, Benucci A, McHugh TJ. Physiological signature of memory age in the prefrontal-hippocampal circuit. Cell Rep. 2019; 29:3835–46.e5. https://doi.org/10.1016/j.celrep.2019.11.075 [PubMed]
- 37. Lega B, Burke J, Jacobs J, Kahana MJ. Slow-theta-to-gamma phase-amplitude coupling in human hippocampus supports the formation of new episodic memories. Cereb Cortex. 2016; 26:268–78. https://doi.org/10.1093/cercor/bhu232 [PubMed]
- 38. Griffiths BJ, Parish G, Roux F, Michelmann S, van der Plas M, Kolibius LD, Chelvarajah R, Rollings DT, Sawlani V, Hamer H, Gollwitzer S, Kreiselmeyer G, Staresina B, et al. Directional coupling of slow and fast hippocampal gamma with neocortical alpha/beta oscillations in human episodic memory. Proc Natl Acad Sci USA. 2019; 116:21834–42. https://doi.org/10.1073/pnas.1914180116 [PubMed]
- 39. Ji MH, Jia M, Zhang MQ, Liu WX, Xie ZC, Wang ZY, Yang JJ. Dexmedetomidine alleviates anxiety-like behaviors and cognitive impairments in a rat model of post-traumatic stress disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2014; 54:284–88. https://doi.org/10.1016/j.pnpbp.2014.06.013 [PubMed]
- 40. Ji M, Li S, Zhang L, Gao Y, Zeng Q, Mao M, Yang J. Sepsis induced cognitive impairments by disrupting hippocampal parvalbumin interneuron-mediated inhibitory network via a D4-receptor mechanism. Aging (Albany NY). 2020; 12:2471–84. https://doi.org/10.18632/aging.102755 [PubMed]
- 41. Ji MH, Wang XM, Sun XR, Zhang H, Ju LS, Qiu LL, Yang JJ, Jia M, Wu J, Yang J. Environmental enrichment ameliorates neonatal sevoflurane exposure-induced cognitive and synaptic plasticity impairments. J Mol Neurosci. 2015; 57:358–65. https://doi.org/10.1007/s12031-015-0627-1 [PubMed]



