Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disease that usually presents in people during old or late middle age with noticeable outward symptoms generally appearing in a person’s sixties. The phenotypes of this disorder include progressive deterioration of autonomic and motor functions, with cognitive decline in most cases. Although the underlying etiology of PD is not completely understood, the most common neuroanatomical pathology is the accumulation of misfolded alpha-synuclein (SNCA) into intracellular aggregates called Lewy Bodies (LBs), presenting throughout the enteric, peripheral and central nervous systems. Progression of the disease results in the significant loss of the dopaminergic neurons situated in the midbrain substantia nigra pars compacta [1].
Even so, several therapeutic strategies are available to treat the dopamine deficiency of PD and improve motor symptoms. Drugs that slow the progression of dopamine loss are rare, and pramipexole (PPX) is one of them [2]. PPX is a dopamine D2/D3 receptor agonist with proven efficacy in the treatment of PD motor symptoms in early and advanced PD. In studies of cells, rodents and primates, neuroprotective properties that seemed to arise partly via a mitochondria-mediated anti-apoptotic mechanism were shown [2]. Additionally, PDX is a non-ergot dopamine agonist with relatively high in vitro specificity and full intrinsic activity at the D2 subfamily of dopamine receptors, with a higher binding affinity to D3 than to D4 or D2 receptor subtypes. PDX can be advantageously administered as a monotherapy or an adjunctive therapy to levodopa to decrease side effects and increase effectiveness in both early and advanced PD treatments [3]. These results were the basis for considering whether there were other mechanisms involved in PPX treatment of PD by regulating gene expression.
Circular RNA (circRNA), consisting of a circular configuration through a typical 5′ to 3′-phosphodiester bond, was recently recognized as a new class of functional molecules. CircRNA consists of no 5′ or 3′ free terminus and is much more stable in cells. The discovery of RNA molecules with circular configurations tracks back to four decades ago [4]. Early studies found some transcripts with non-colinear or shuffled order and implied that these transcripts might be a byproduct of mis-splicing [5]. Later, accumulative evidence consolidated the existence of circular configured RNA molecules such as transcripts of mouse Sry, human ETS1, and DCC [6,7]. Although these pioneer studies have drafted a blueprint for the current circRNA research, the lack of biological functions and comprehensive analysis halted the progression of circRNA research. In the past few years, the advancement of next-generation sequencing technology enabled scientists to perform genome-wide analysis of the expression of circRNAs and to characterize the diverse origins and compositions of circRNAs. In addition, the well-established roles of miRNAs and the theory of competitive endogenous RNA (ceRNA) facilitated the large leap of circRNA research [8]. CircRNAs are abundant in the brain and exosomes, with the capability of traversing the blood–brain barrier [9]. Therefore, they are perfect candidates as potential diagnostic tools for PD.
In our research, we investigated the interactions between PPX and circSNCA to reveal the mechanism of PPX treatment in PD. Additionally, circSNCA was identified as a ceRNA of miR-7 in PD, and its expression was strongly associated with cell apoptosis and autophagy. Our findings provide novel insights into PPX effects and suggest that circSNCA might be a potential target of PD.
Results
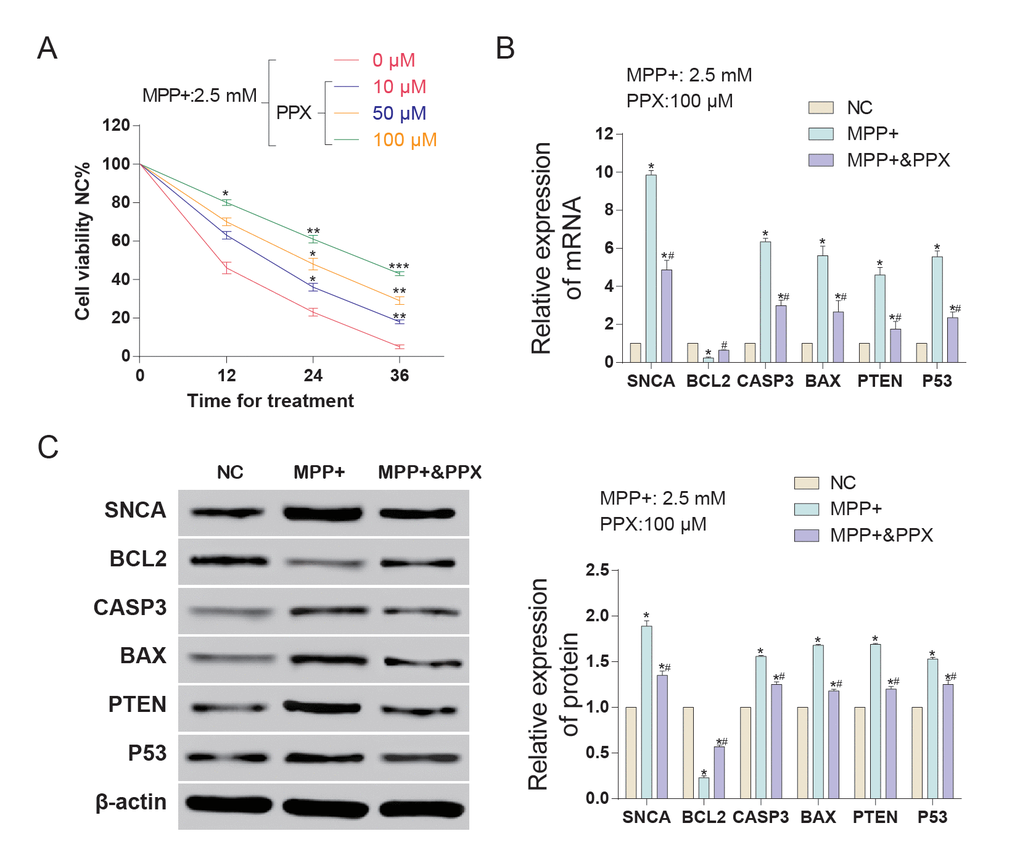
CircSNCA expression was inhibited by PPX
SNCA mRNA and circSNCA are homology-dependent genes. Hsa_circ_0070441 (143 bp) matures from the CDS region of SNCA mRNA, and hsa_circ_0127305 (114 bp) matures from the 3’UTR of SNCA (Figure 3A). According to qRT-PCR analysis, both had higher expression levels after being treated with MPP+, while the former responded more drastically. When being treated with both MPP+ and PPX, the hsa_circ_0127305 level decreased but was still higher than that of NC. However, there was no significant difference between the MPP+ and MPP+ & PPX groups for hsa_circ_0070441 (Figure 3B). Targeted miRNAs of hsa_circ_0127305, miR-580 and miR-7 were predicted using Circular RNA Interactome algorithm [14]. The relative expression of miR-580 was not detected, and miR-7 was significantly reduced in the MPP+ group compared with NC group. The level of miR-7 was the highest in the MPP+ & PPX group compared with the MPP+ group (Figure 3C). We also tested the circSNCA RNA level under different concentrations of PPX and treatment times (Figure 3D and 3E). The circSNCA level decreased with the increase in the PPX concentration (Figure 3D). For the treatment time, after the first 4 h after PPX treatment, no significant change was evident; however, 8 h after PPX treatment, the circSNCA level decreased, and 12 h after the treatment, the level of circSNCA decreased to the lowest level and remained stable thereafter (Figure 3E).
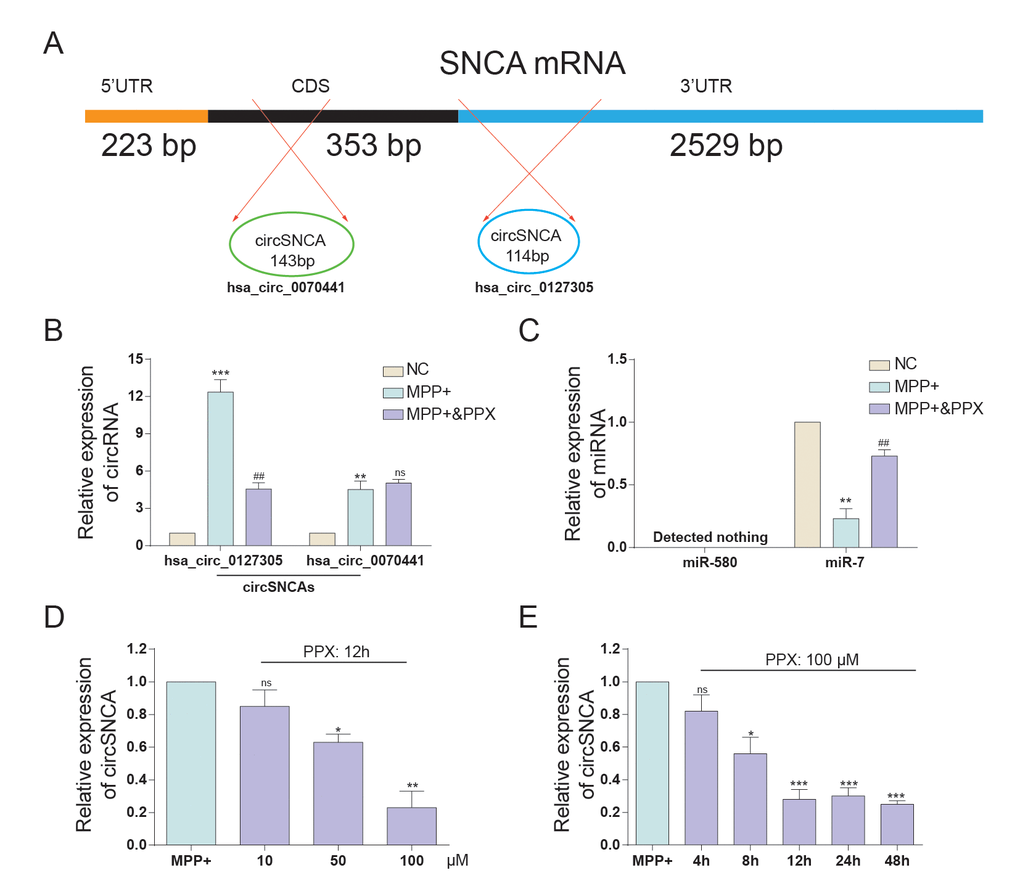
Figure 3. The expression of circSNCA and miRNA after PPC treatment. (A) SNCA mRNA has two corresponding circRNAs, respectively matured from CDS and 3’-UTR. (B) The relative expression of hsa_circ_0127305 and hsa_circ_0070441 detected by qRT-PCR increased after PPX treatment. *P < 0.05, compared with NC, #P < 0.05 compared with MPP+, and ns meant no significant difference. (C) The relative expression of miR-580 and miR-7 detected by qRT-PCR decreased with PPX treatment. *P < 0.05 meant MPP+ compared with NC, #P < 0.05 meant MPP+ & PPX compared with MPP+. (D) The relative expression of circSNCA detected by qRT-PCR decreased as the concentration of PPX increased. *P < 0.05 compared with MPP+, and ns meant no significant difference. (E) The relative expression of circSNCA detected by qRT-PCR decreased as the time for PPX (100 μM) treatment increased. *P < 0.05 compared with MPP+, and ns meant no significant difference.
Endogenous competition mechanism exists in the circSNCA/miR-7/SNCA network
CircSNCA and SNCA 3’UTR had the same target sites of the miR-7 seed region (Figure 4A-a). Double luciferase reporter assays were performed to detect the relationship between circSNCA and miR-7 or miR-7 and SNCA mRNA (Figure 4A-b and c). The dual luciferase reporter gene assay results demonstrated that only when circSNCA-WT and SNCA 3’UTR–WT were co-transfected with miR-7 was there a sharp reduction in luciferase activity (Figure 4A-b and c). Specific probes for detecting circSNCA (green dots) and miR-7 (red-dots) were transfected into SH-SY5Y cells to evaluate the space sites of circSNCA and miR-7 (Figure 4B). Both green dots and red dots were located in the cytoplasm of SH-SY5Y, with strong space overlap. These two experiments implied that there was endogenous competition between circSNCA and SNCA mRNA for miR-7 binding (Figure 5A). To test this hypothesis, we either overexpressed or knocked down circSNCA to investigate how circSNCA regulated miR-7 expression (Figure 5B and 5C). With circSNCA overexpression, the miR-7 level decreased, while with circSNCA knockdown, the miR-7 level increased (Figure 5D and 5E). Compared to the siRNAs (Si-Circ-1, Si-Circ-2, Si-Circ-3) for circSNCA, Si-Circ-1 showed the best effects on circSNCA knockdown. Western blot showed that in the MPP+ group, SNCA expression was enhanced with circSNCA overexpression, declined with circSNCA knockdown, and slightly increased with both circSNCA overexpression and PPX treatment, compared to the NC group. Similar results were observed in terms of CASP3, BAX, PTEN and P53 expression, while opposite results were observed for BCL2 (Figure 5F). It could be speculated and concluded that PPX had a negative regulation effect on the expression of SNCA and pro-apoptotic proteins and a positive regulatory effect on anti-apoptotic proteins. CircSNCA could attenuate the therapeutic effects of PPX in an in vitro PD model.
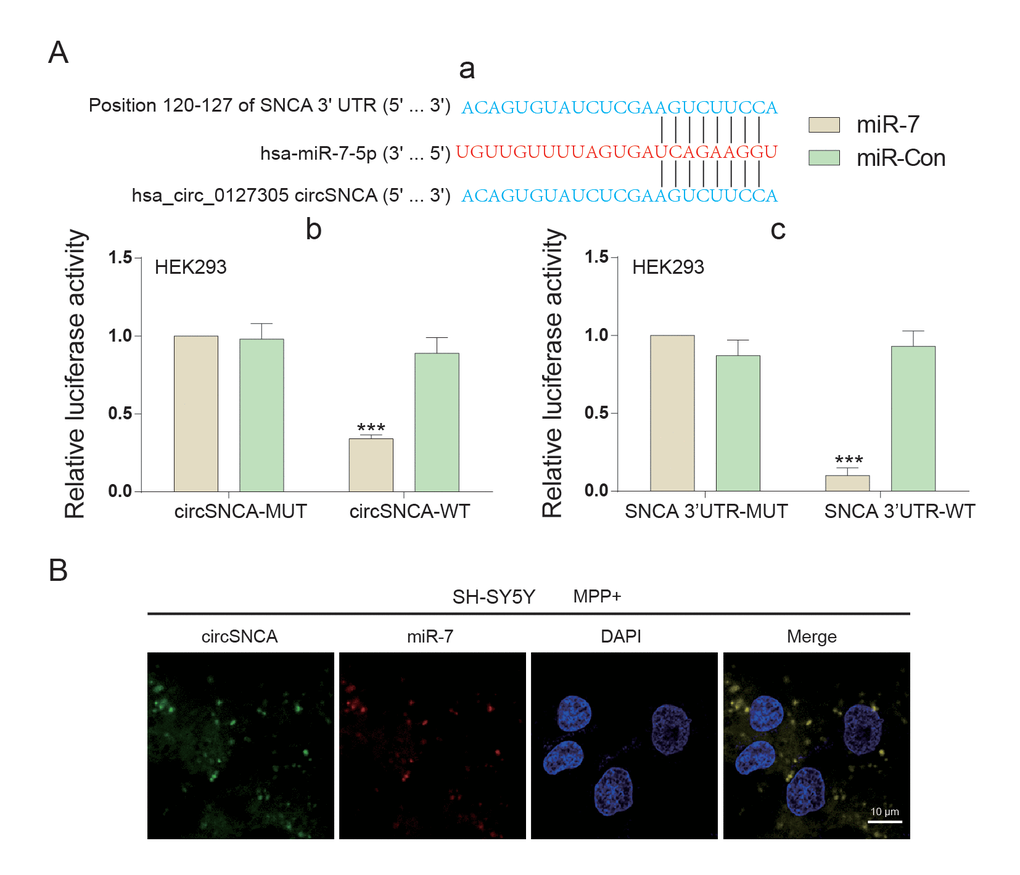
Figure 4. The target relationship among circSNCA, miR-7 and SNCA mRNA of PD. (A) (a) 3’-UTR region of SNCA mRNA and hsa_circ_0127305 were both found to harbor a binding site for miR-7. (b) Luciferase reporter assay results showed that miR-7 exclusively reduced the luciferase activity of the wild-type reporter plasmids of circSNCA. (c) Luciferase reporter assay results showed that miR-7 exclusively reduced luciferase activity of the wild-type reporter plasmids of circSNCA. (B) RNA FISH for co-localization of circSNCA and miR-7 in cytoplasm of SH-SY5Ys. CircSNCA and miR-7 probes were labeled with Alexa 488 and Cy-5, respectively. Nuclei were stained with DAPI. Scale bar = 10 μm.
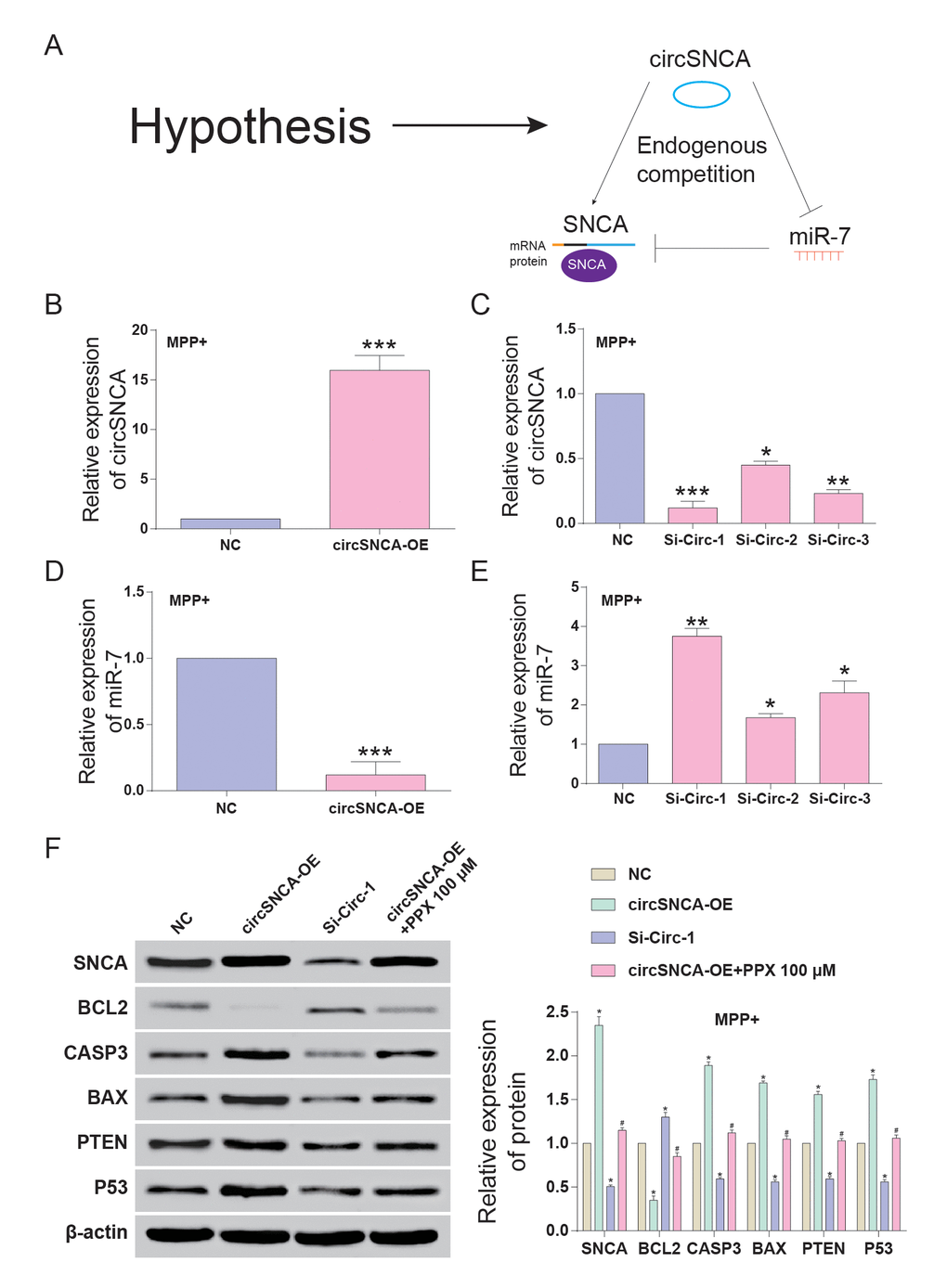
Figure 5. CircSNCA increased SNCA expression by downregulating miR-7 and influenced the expression of apoptosis-related genes. (A) Endogenous competition between circSNCA and SNCA mRNA for miR-7 binding. (B) The relative expression of circSNCA detected by qRT-PCR increased after overexpression (circSNCA-OE) in the MPP+ group. *P < 0.05 compared with NC. (C) The relative expression of circSNCA detected by qRT-PCR decreased after circSNCA knockdown in the MPP+ group. *P < 0.05 compared with NC. (D) The relative expression of miR-7 detected by qRT-PCR decreased after circSNCA overexpression in the MPP+ group. *P < 0.05 compared with NC. (E) The relative expression of miR-7 detected by qRT-PCR increased after circSNCA knockdown in the MPP+ group. *P < 0.05 compared with NC. (F) The protein expression of SNCA and pro-apoptotic genes (CASP3, BAX, PTEN and P53) detected by western blot increased in the circSNCA overexpression group, decreased in the circSNCA knockdown group, while that of the anti-apoptotic gene (BCL2) showed the opposite tendency. *P < 0.05 compared with NC group, #P < 0.05 compared with circSNCA-OE.
Furthermore, autophagy-associated protein, LC3B, was also detected by western blot. The LC3B-I level showed no significant change to circSNCA overexpression/ knockdown or PPX treatment. However, the LC3B-II level was low with circSNCA overexpression, high with circSNCA knockdown and slightly low with both circSNCA overexpression and PPX treatment (Figure 6A). To conclude, MPP+ induced an increase in circSNCA in a PD cell model, while PPX reversed it (Figure 6B). The upregulation of circSNCA could sponge and degrade miR-7 through the target sequences and Ago2, which may lead to attenuated inhibition of miR-7 on SNCA mRNA and the increased expression of SNCA. CircSNCA upregulation also positively correlated with the increasing expression levels of pro-apoptotic proteins (CASP3, BAX, PTEN and P53) and the decreasing levels of anti-apoptotic protein BCL2 and autophagy-associated protein LC3B-II.
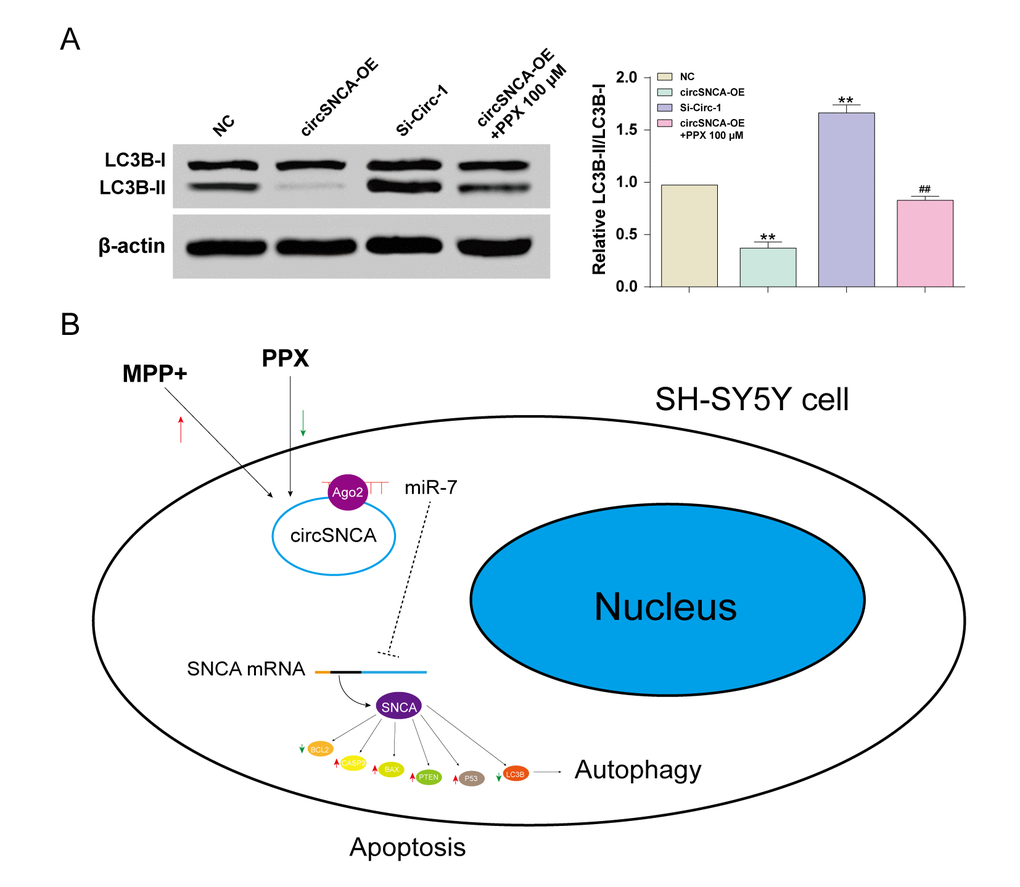
Figure 6. CircSNCA influenced the expression of autophagy-related proteins. (A) The expression of autophagy-related protein, LC3B-II, detected by western blot decreased in the circSNCA overexpression group, and increased in the circSNCA knockdown group, while that of LC3B-I showed little difference. *P < 0.05 compared with NC group, #P < 0.05 compared with circSNCA-OE. (B) PPX mediated apoptosis and autophagy of MPP+ treated SH-SY5Y cells by regulating circSNCA, which targeted miR-7.
Discussion
In previous studies, the mechanism of the suppressive effect of PPX on PD has not been well understood. In this study, we first identified the significantly reduced expression of SNCA and circSNCA after PPX treatment. Furthermore, we investigated the endogenous competition between circSNCA and SNCA mRNA and found that circSNCA was a ceRNA of miR-7 in PD, binding with miR-7 and upregulating its target gene, SNCA. Additionally, the expression of pro-apoptotic genes (CASP3, BAX, PTEN and P53) was reduced, while that of anti-apoptotic protein BCL2 and autophagy-related protein LC3B-II was increased with the downregulation of circSNCA, revealing the inhibition of apoptosis and the promotion of autophagy in PD.
Since circRNAs were newly identified as players in the regulation of post-transcriptional gene expression, studies on their effects on PD have been limited. Interacting with disease-associated miRNAs is one of the important mechanisms of circRNA involvement in disease progression [15]. Multiple previous studies have discussed the function of circRNAs as a sponge of miRNAs to influence pathological processes [16]. In PD, circRNA zip-2 knockdown can lead to the reduced aggregation of SNCA protein by sponging miR-60, thus leading to better survival outcomes of PD patients [17]. By sponging miR-7, circRNA s-7 can promote vital genes associated with PD and AD [18]. In our study, we also identified a similar mechanism of circSNCA, which acted as a ceRNA of miR-7 and upregulated SNCA in PD. In addition, we found that circSNCA expression was closely related to PPX treatment. It could be speculated that PPX treatment attenuated the progression of PD partly due to its suppressive effects on circSNCA expression.
CircSNCA’s function in PD was revealed for the first time in this study. However, the effect of circSNCA strongly relied on its direct and indirect regulation on miR-7 and SNCA, respectively. MiR-7 was believed to be closely coupled to ciRS-7, and the fine-tuning of the miR-7/miR-671/ciRS-7 axis likely plays profound roles in human cancer development [19]. MiR-7 was reported to bind the 3’ UTR of SNCA and inhibited its translation, which was confirmed in our study [20]. Tarale et al. proved that the low level of miR-7 implied a higher risk of idiopathic PD [21]. Zhou et al. suggested that miR-7 inhibited neuroinflammation in the pathogenesis of PD through targeting Nod-like receptors [22]. Li et al. demonstrated that miR-7 exerted inhibitory effects on neuronal apoptosis of PD by targeting BAX and Sirt2 [23]. In this specific case of PD, circSNCA facilitated the pathological processes as a miR-7 inhibitor, further verifying that miR-7 was a suppressive player for PD.
SNCA is of great importance in the occurrence and development of PD as accumulated evidence has proved its association with this disease. With some correlation experiments, SNCA was found related to neurotoxicity and the anti-apoptosis pathway [24]. When the extracellular environment is broken, the unbalance of gene expression appeared in neurocytes, such as an abnormal level of SNCA, followed by the changes of cell autophagy and cell apoptosis [25]. Maybe in the following process, the nerve cell damage and apoptosis in turn accelerated SNCA expression (anti-apoptosis) [26]. The rapid expression causes misfolding and aggregation of alpha-synuclein, one of the typical features of Parkinson’s [27]. Abnormal SNCA aggregation in LBs has been suggested as one of the main causes for PD, which is related to a deficiency in the ubiquitin-proteasome system and the autophagy-lysosomal pathway [28]. It was reported to be closely connected with cell apoptosis and autophagy [29]. During neuronal apoptosis, the aggregation of SNCA was realized by histones [26]. Its toxicity was partly due to the defects of autophagy-mediated clearance, and autophagy mediated by transcription factor EB could rescue the midbrain dopamine neurons from SNCA toxicity [30]. In this study, we also studied some apoptosis- and autophagy-related genes and found that apoptosis was reduced while autophagy was promoted with the downregulation of SNCA, which could help slow down the deterioration of PD. Since SNCA downregulation resulted from circSNCA knockdown, it could prove that circSNCA inhibition was effective in PD treatment.
Some limitations existed in this study. For instance, only cell experiments were conducted, and animal experiments must be carried out to prove this mechanism. Additionally, the mechanism itself should be explored more deeply and thoroughly, and some details are still not clear.
In summary, we verified that PPX treatment for PD could downregulate circSNCA. Since circSNCA served as a ceRNA that sponged miR-7 and upregulated SNCA, its downregulation by PPX treatment could reduce the expression of SNCA. The inhibition of circSNCA and SNCA reduced apoptosis and promoted the autophagy of SH-SY5Y cells, attenuating the progression of PD.
Materials and Methods
Bioinformatics retrieval
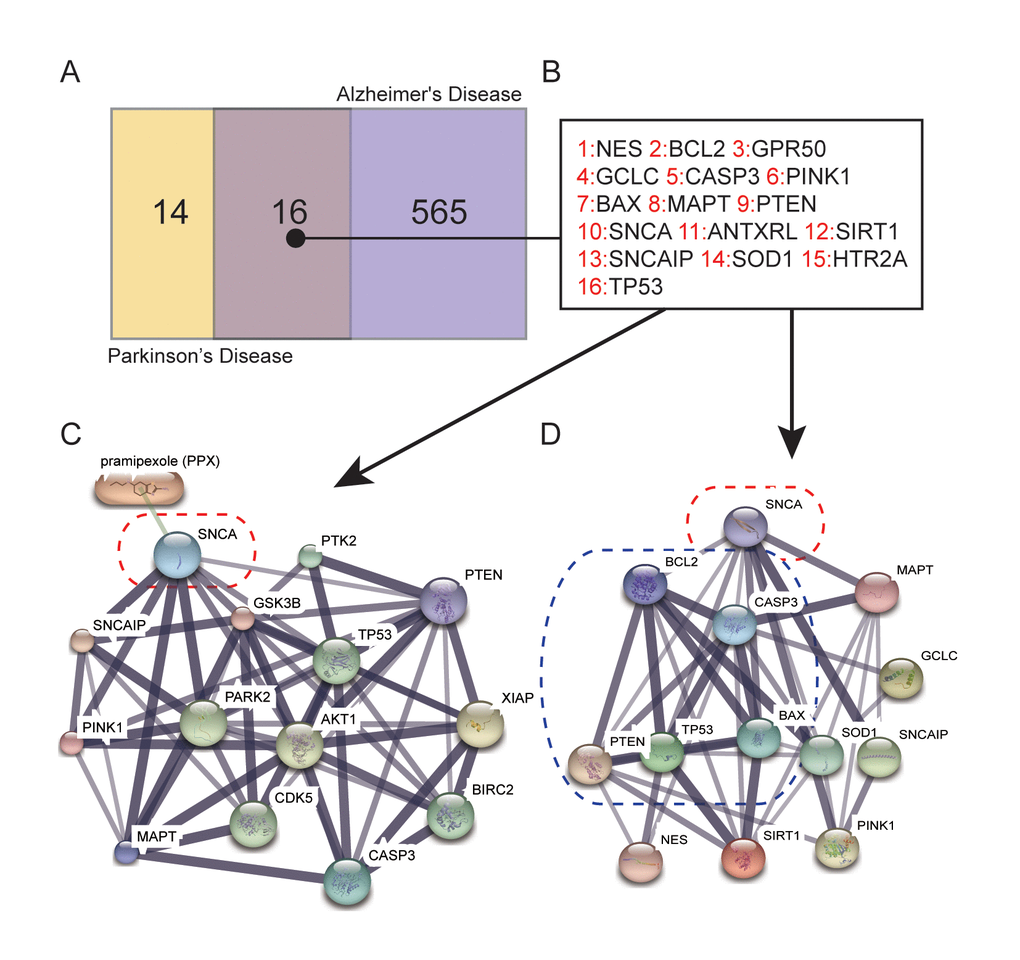
DiGSeE (http://210.107.182.61/geneSearch/) is a search platform for genetic bases of human diseases. “Parkinson” and “Alzheimer’s Disease” were used as keywords during co-existing gene selection. The interactions between these genes and PPX were determined and plotted via STITCH (http://stitch.embl.de/) and protein-protein interactions were analyzed on STRING (https://string-db.org/), with the calculation performed by Dijkstra algorithm.
Reagents and antibodies
PPX was purchased from Tocris Bioscience. MPP+ was purchased from Sigma (St. Louis, MO, USA). The primary antibodies for immunoblot analysis are listed as follows: SNCA (CST, Danvers, MA, USA, 2642), LC3BI/II (Abcam, ab51520), CASP3 (Abcam, ab2302), BAX (Abcam, ab32503), PTEN (Abcam, ab32199), P53 (Abcam, ab1431), BCL2 (Abcam, ab32124) and β-actin (Abcam, ab8227). SiRNAs for knockdown of circSNCA were synthesized by GenePharma company (Shanghai, China). pLCDH-ciR (GeneSeed, China) was used to overexpress circSNCA. FITC-labeled circSNCA probe and Cy3-labeled miR-7 probe were synthesized by Sangon Biotech. The transfection reagent utilized was Lipofectamine 2000 (Invitrogen, Shanghai, China).
Cell lines and cell culture
SH-SY5Y cells (BNCC338056, BeNa Culture Collection, Beijing, China) were grown in high-glucose Dulbecco’s-modified eagle medium (DMEM-H) with 10% fetal bovine serum (FBS) containing glutamine and sodium pyruvate, in a 5% CO2 humidified incubator at 37°C.
MTT assay
After 12 h treatment of 2.5 mM MPP+ with 10, 50, or 100 μM PPX, the viability of SH-SY5Y cells was identified via MTT assay. 1 mg MTT was added to each milliliter of medium and incubated at 37°C for 4 h. After 4 h, the medium in the plate was discarded, and in each well, 200 μL dimethyl sulfoxide (DMSO) was added before 1-min shaking for dissolution in a microplate reader (Bio-Rad Model 680; Bio-Rad, Hercules, CA, USA). The absorbance of cells in each well was measured at 570 nm, and the cell growth curve was drawn based on an average of five wells. The experiment was repeated in triplicate.
Western blot
SH-SY5Y cells were lysed in radio-immunoprecipitation assay (RIPA) buffer (BioVision, Milpitas, CA, USA). Total protein in supernatants was quantified using the BCA-200 protein assay kit (Pierce Biotechnology, Rockford, IL, USA). The protein was separated with 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto a polyvinylidene fluoride (PVDF) membrane (500 mA). The membrane was sealed in Tris Buffered Saline Tween (TBST) with 5% skim milk at room temperature for 1 h, and subsequently incubated with primary antibodies at 4°C overnight. After TBST-washing three times, the membrane was incubated for 1 h at room temperature with secondary antibody. Protein bands were visualized using enhanced chemiluminescence (Santa Cruz Biotechnology; Santa Cruz, CA, USA). Quantity One software (Bio-Rad) was used for image analysis. The results were analyzed by Image-Pro Plus 5.0 (Media, Cybernetics, USA). β-actin was included as the internal control.
Reverse transcription and quantitative PCR (qRT-PCR)
The total RNA was extracted using Trizol (Invitrogen, Carlsbad, CA, USA). The obtained RNA was reverse transcribed to cDNA using RevertAid First Strand cDNA Synthesis Kit (Thermo, Shanghai, China), and Power SYBR Green PCR Master Mix (Thermo, Shanghai, China) was used for determination. PCR program: predenaturation at 95oC for 10 min, followed by 40 cycles of denaturation at 95oC for 15 s, annealing at 60oC for 30 s, and extension at 72oC for 30 s. Subsequently, the dissolution curve of PCR products was generated. With GAPDH expression as the standard for mRNA and U6 expression as the standard for miRNA, relative mRNA and miRNA expression was calculated by the 2-ΔΔCt method. The primer sequences are supplied in Table 2.
Table 2. QRT-PCR primers and circSNCA siRNAs.
| Gene | Farword(5'→3') | Reverse(5'→3') |
| SNCA | CCTCAGCCCAGAGCCTTTC | CCTCTGCCACACCCTGCTT |
| BCL2 | GAGGATTGTGGCCTTCTTTG | CGTTATCCTGGATCCAGGTG |
| CASP3 | TCTGGTTTTCGGTGGGTGTG | CGCTTCCATGTATGATCTTTGGTTC |
| BAX | GTGAGCGGCTGCTTGTCTGG | CTTCCAGATGGTGAGCGAGG |
| PTEN | GGAAAGGGACGGACTGGTGT | GACTGGGAATTGTGACTCCC |
| P53 | GTCGGACAAGCGGCAGATTG | CCTTCGTCTTAGGGTGAGGC |
| circ-0127305 | CCATCAGCAGTGATTGAAATCTG | ACTGGGCACATTGGAACTGA |
| circ-0070441 | AGAAGACAGTGGAGGGAGCA | GGCTACTGCTGTCACACCC |
| GAPDH | TCGGAGTCAACGGATTTGGT | TTCCCGTTCTCAGCCTTGAC |
| miR-580 | GCGCTTGAGAATGATGAATC | GAATACCTCGGACCCTGC |
| miR-7 | GCCTGGAAGACTAGTGATTT | GAATACCTCGGACCCTGC |
| U6 | CTCGCTTCGGCAGCACA | AACGCTTCACGAATTTGCGT |
| has_circ_0127305 siRNAs (5'→3') | ||
| Si-circ-1 | GATTGAAATCTGCTGACAGAT | |
| Si-circ-2 | AGTGATTGAAATCTGCTGACA | |
| Si-circ-3 | GCAGTGATTGAAATCTGCTGA | |
Dual luciferase reporter gene assay
Luciferase reporter gene recombinant plasmids were inserted with the sequences of wild-type (WT) and mutated type (MUT) circSCNA, WT and MUTSCNA 3’-untranslated region (3’-UTR). MiR-7 mimics or control were co-transfected with WT and MUT circSCNA into the 293 cell line (BeNa Culture Collection, Beijing, China) using Lipofectamine 2000 (Invitrogen). Luciferase Dual Assay Kit (Thermo Fisher Scientific) was used for dual-luciferase reporter gene assay 48 h after cells were transfected.
Immunofluorescent localization
3×104 SH-SY5Y cells were plated onto slides for 24 h of growth in advance of the probe transfection experiments. 20 nM probes of FITC-labeled circSNCA or Cy3-labeled miR-7 (Sangon Biotech) were co-transfected into SH-SY5Y cells for 36 h. After culture incubation, the cells were digested by trypsin and fixed onto slides. The nuclei were stained with DAPI, and the images were collected using fluorescence microscopy (Carl Zeiss, Jena, Germany).
Author Contributions
Research conception and design: Qiuling Sang and Yajuan Sun. Data analysis and interpretation: Libo Wang, Ling Qi and Wenping Sun. Statistical analysis: Xiaoyang Liu and Weiyao Wang. Drafting of the manuscript: Qiuling Sang. Critical revision of the manuscript: Haina Zhang. Approval of final manuscript: all authors.
Conflicts of Interest
The authors confirm that there are no conflicts of interest.
Funding
This study was supported by National Natural Science Foundation of China (81201671); Foundation of Science and Technology Department of Jilin Province (20180101306JC, 20180101144JC).
References
- 1. Smeyne M, Smeyne RJ. Glutathione metabolism and Parkinson’s disease. Free Radic Biol Med. 2013; 62:13–25. https://doi.org/10.1016/j.freeradbiomed.2013.05.001 [PubMed]
- 2. Schapira AH, McDermott MP, Barone P, Comella CL, Albrecht S, Hsu HH, Massey DH, Mizuno Y, Poewe W, Rascol O, Marek K. Pramipexole in patients with early Parkinson’s disease (PROUD): a randomised delayed-start trial. Lancet Neurol. 2013; 12:747–55. https://doi.org/10.1016/S1474-4422(13)70117-0 [PubMed]
- 3. Silindir M, Ozer AY. The benefits of pramipexole selection in the treatment of Parkinson’s disease. Neurol Sci. 2014; 35:1505–11. https://doi.org/10.1007/s10072-014-1891-5 [PubMed]
- 4. Hsu MT, Coca-Prados M. Electron microscopic evidence for the circular form of RNA in the cytoplasm of eukaryotic cells. Nature. 1979; 280:339–40. https://doi.org/10.1038/280339a0 [PubMed]
- 5. Cocquerelle C, Mascrez B, Hétuin D, Bailleul B. Mis-splicing yields circular RNA molecules. FASEB J. 1993; 7:155–60. https://doi.org/10.1096/fasebj.7.1.7678559 [PubMed]
- 6. Capel B, Swain A, Nicolis S, Hacker A, Walter M, Koopman P, Goodfellow P, Lovell-Badge R. Circular transcripts of the testis-determining gene Sry in adult mouse testis. Cell. 1993; 73:1019–30. https://doi.org/10.1016/0092-8674(93)90279-Y [PubMed]
- 7. Cocquerelle C, Daubersies P, Majérus MA, Kerckaert JP, Bailleul B. Splicing with inverted order of exons occurs proximal to large introns. EMBO J. 1992; 11:1095–98. [PubMed]
- 8. Hsiao KY, Sun HS, Tsai SJ. Circular RNA - New member of noncoding RNA with novel functions. Exp Biol Med (Maywood). 2017; 242:1136–41. https://doi.org/10.1177/1535370217708978 [PubMed]
- 9. Li Y, Zheng Q, Bao C, Li S, Guo W, Zhao J, Chen D, Gu J, He X, Huang S. Circular RNA is enriched and stable in exosomes: a promising biomarker for cancer diagnosis. Cell Res. 2015; 25:981–84. https://doi.org/10.1038/cr.2015.82 [PubMed]
- 10. Kim J, So S, Lee HJ, Park JC, Kim JJ, Lee H. DigSee: disease gene search engine with evidence sentences (version cancer). Nucleic Acids Res. 2013; 41:W510-7. https://doi.org/10.1093/nar/gkt531 [PubMed]
- 11. Szklarczyk D, Santos A, von Mering C, Jensen LJ, Bork P, Kuhn M. STITCH 5: augmenting protein-chemical interaction networks with tissue and affinity data. Nucleic Acids Res. 2016; 44:D380–84. https://doi.org/10.1093/nar/gkv1277 [PubMed]
- 12. Wang JD, Cao YL, Li Q, Yang YP, Jin M, Chen D, Wang F, Wang GH, Qin ZH, Hu LF, Liu CF. A pivotal role of FOS-mediated BECN1/Beclin 1 upregulation in dopamine D2 and D3 receptor agonist-induced autophagy activation. Autophagy. 2015; 11:2057–73. https://doi.org/10.1080/15548627.2015.1100930 [PubMed]
- 13. Kim MJ, Park M, Kim DW, Shin MJ, Son O, Jo HS, Yeo HJ, Cho SB, Park JH, Lee CH, Kim DS, Kwon OS, Kim J, et al. Transduced PEP-1-PON1 proteins regulate microglial activation and dopaminergic neuronal death in a Parkinson’s disease model. Biomaterials. 2015; 64:45–56. https://doi.org/10.1016/j.biomaterials.2015.06.015 [PubMed]
- 14. Dudekula DB, Panda AC, Grammatikakis I, De S, Abdelmohsen K, Gorospe M. CircInteractome: A web tool for exploring circular RNAs and their interacting proteins and microRNAs. RNA Biol. 2016; 13:34–42. https://doi.org/10.1080/15476286.2015.1128065 [PubMed]
- 15. Ghosal S, Das S, Sen R, Basak P, Chakrabarti J. Circ2Traits: a comprehensive database for circular RNA potentially associated with disease and traits. Front Genet. 2013; 4:283. https://doi.org/10.3389/fgene.2013.00283 [PubMed]
- 16. Guo JU, Agarwal V, Guo H, Bartel DP. Expanded identification and characterization of mammalian circular RNAs. Genome Biol. 2014; 15:409. https://doi.org/10.1186/s13059-014-0409-z [PubMed]
- 17. Kumar L, Shamsuzzama, Jadiya P, Haque R, Shukla S, Nazir A. Functional characterization of novel circular RNA molecule, circzip-2 and its synthesizing gene zip-2 in C. elegans model of Parkinson’s Disease. Mol Neurobiol. 2018. https://doi.org/10.1007/s12035-018-0903-5 [PubMed]
- 18. Lukiw WJ. Circular RNA (circRNA) in Alzheimer’s disease (AD). Front Genet. 2013; 4:307. https://doi.org/10.3389/fgene.2013.00307 [PubMed]
- 19. Hansen TB, Kjems J, Damgaard CK. Circular RNA and miR-7 in cancer. Cancer Res. 2013; 73:5609–12. https://doi.org/10.1158/0008-5472.CAN-13-1568 [PubMed]
- 20. McMillan KJ, Murray TK, Bengoa-Vergniory N, Cordero-Llana O, Cooper J, Buckley A, Wade-Martins R, Uney JB, O’Neill MJ, Wong LF, Caldwell MA. Loss of MicroRNA-7 regulation leads to alpha-synuclein accumulation and dopaminergic neuronal loss in vivo. Mol Ther. 2017; 25:2404–14. https://doi.org/10.1016/j.ymthe.2017.08.017 [PubMed]
- 21. Tarale P, Daiwile AP, Sivanesan S, Stöger R, Bafana A, Naoghare PK, Parmar D, Chakrabarti T, Krishnamurthi K. Manganese exposure: linking down-regulation of miRNA-7 and miRNA-433 with α-synuclein overexpression and risk of idiopathic Parkinson’s disease. Toxicol In Vitro. 2018; 46:94–101. https://doi.org/10.1016/j.tiv.2017.10.003 [PubMed]
- 22. Zhou Y, Lu M, Du RH, Qiao C, Jiang CY, Zhang KZ, Ding JH, Hu G. MicroRNA-7 targets Nod-like receptor protein 3 inflammasome to modulate neuroinflammation in the pathogenesis of Parkinson’s disease. Mol Neurodegener. 2016; 11:28. https://doi.org/10.1186/s13024-016-0094-3 [PubMed]
- 23. Li S, Lv X, Zhai K, Xu R, Zhang Y, Zhao S, Qin X, Yin L, Lou J. MicroRNA-7 inhibits neuronal apoptosis in a cellular Parkinson’s disease model by targeting Bax and Sirt2. Am J Transl Res. 2016; 8:993–1004. [PubMed]
- 24. Xu J, Kao SY, Lee FJ, Song W, Jin LW, Yankner BA. Dopamine-dependent neurotoxicity of alpha-synuclein: a mechanism for selective neurodegeneration in Parkinson disease. Nat Med. 2002; 8:600–06. https://doi.org/10.1038/nm0602-600 [PubMed]
- 25. Musgrove RE, King AE, Dickson TC. α-Synuclein protects neurons from apoptosis downstream of free-radical production through modulation of the MAPK signalling pathway. Neurotox Res. 2013; 23:358–69. https://doi.org/10.1007/s12640-012-9352-5 [PubMed]
- 26. Jiang P, Gan M, Yen SH, McLean PJ, Dickson DW. Histones facilitate α-synuclein aggregation during neuronal apoptosis. Acta Neuropathol. 2017; 133:547–58. https://doi.org/10.1007/s00401-016-1660-z [PubMed]
- 27. Ibáñez P, Bonnet AM, Débarges B, Lohmann E, Tison F, Pollak P, Agid Y, Dürr A, Brice A. Causal relation between alpha-synuclein gene duplication and familial Parkinson’s disease. Lancet. 2004; 364:1169–71. https://doi.org/10.1016/S0140-6736(04)17104-3 [PubMed]
- 28. Lehri-Boufala S, Ouidja MO, Barbier-Chassefière V, Hénault E, Raisman-Vozari R, Garrigue-Antar L, Papy-Garcia D, Morin C. New roles of glycosaminoglycans in α-synuclein aggregation in a cellular model of Parkinson disease. PLoS One. 2015; 10:e0116641. https://doi.org/10.1371/journal.pone.0116641 [PubMed]
- 29. Lenart J, Zieminska E, Diamandakis D, Lazarewicz JW. Altered expression of genes involved in programmed cell death in primary cultured rat cerebellar granule cells acutely challenged with tetrabromobisphenol A. Neurotoxicology. 2017; 63:126–36. https://doi.org/10.1016/j.neuro.2017.09.014 [PubMed]
- 30. Decressac M, Mattsson B, Weikop P, Lundblad M, Jakobsson J, Björklund A. TFEB-mediated autophagy rescues midbrain dopamine neurons from α-synuclein toxicity. Proc Natl Acad Sci USA. 2013; 110:E1817–26. https://doi.org/10.1073/pnas.1305623110 [PubMed]