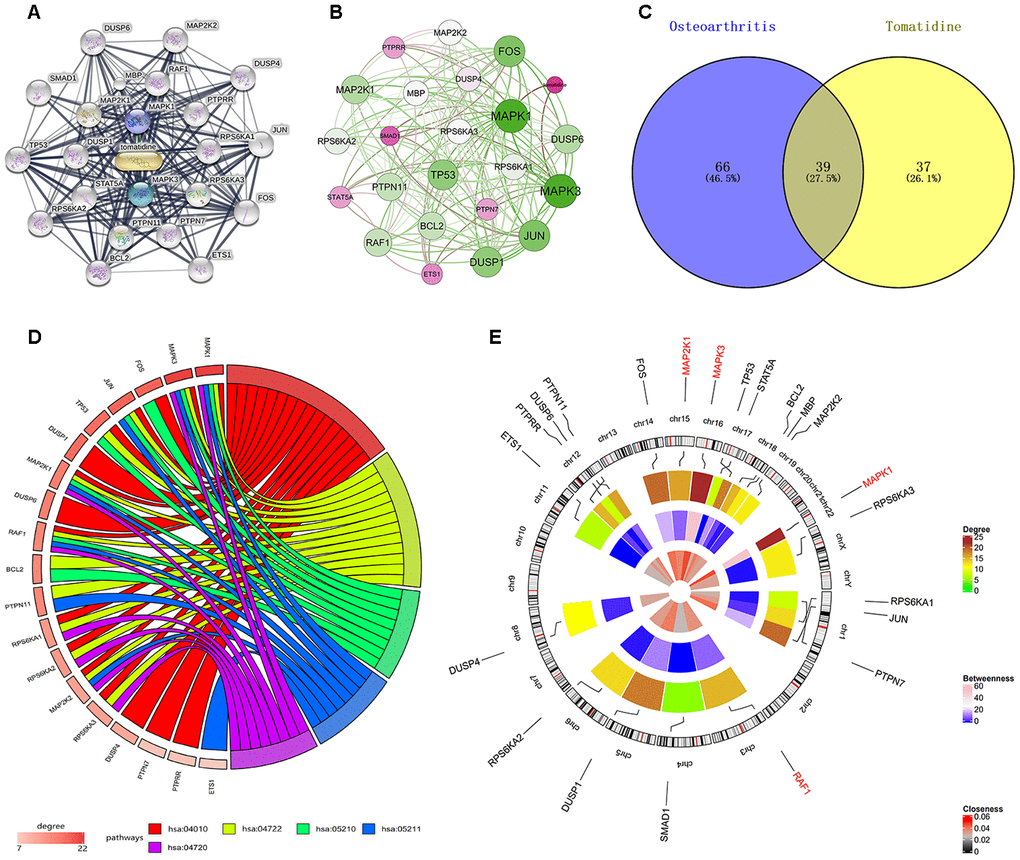
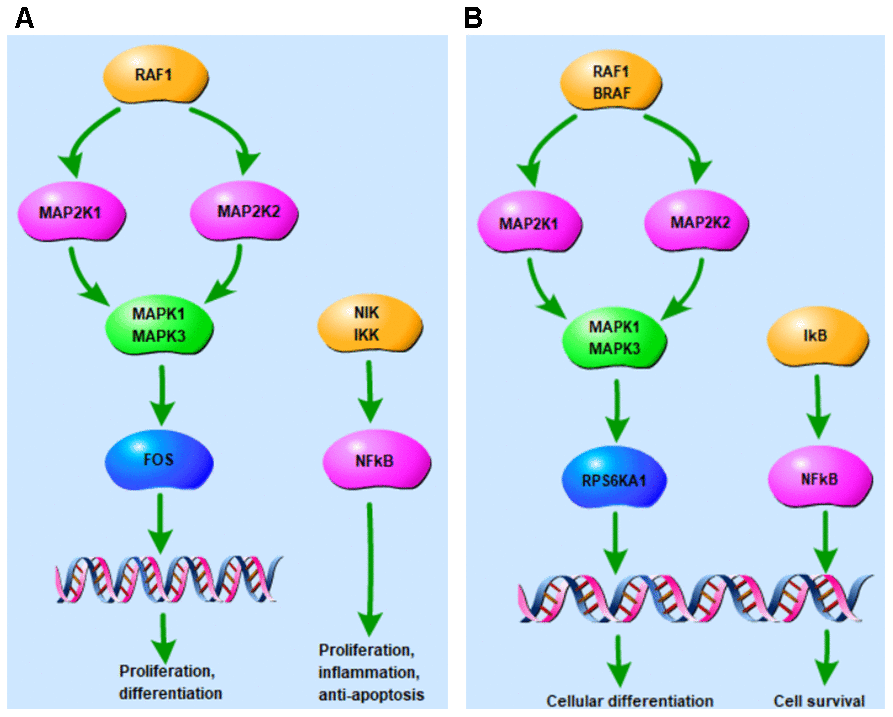
Introduction
Osteoarthritis (OA) is a degenerative joint disease associated with chronic pain and disability; it is characterized by articular cartilage breakdown, synovial inflammation, and bone hypertrophy [1]. The constantly increasing numbers of OA patients worldwide incurs an expenditure of 1.0% to 2.5% Gross Domestic Product (GDP) in developed countries [2, 3]. OA is a progressive joint disease without any effective curative treatment [4–6]. Therefore, it is of paramount importance to identify the underlying pathways involved in OA pathology and discover new therapies that can improve the quality of life for patients with OA.
The hallmark of OA is the imbalance between pro-inflammatory and anti-inflammatory cytokines [7]. The most critical pro-inflammatory cytokine involved in OA pathogenesis is interleukin-1β (IL-1β), which downregulates key extracellular matrix (ECM) proteins, type-II collagen and aggrecan, and upregulates matrix metalloproteinases (MMPs) and ADAMTS-5, which breakdown the cartilage tissue [8, 9]. Therefore, anti-inflammatory drugs are potential therapeutic candidates against OA.
Tomatidine, the major glycoalkaloid produced by the tomato plant, is associated with anti-apoptotic, anti-inflammatory, anti-bacterial, and anti-cancer properties [10–14]. Tomatidine suppresses inflammation in LPS-stimulated murine macrophages by inhibiting the NF-κB and JNK signaling pathways [15]. However, the therapeutic effects of tomatidine in OA are unknown. Therefore, in this study, we investigated the effects of tomatidine on IL-1β-induced inflammation in primary articular chondrocytes and cartilage degeneration in the rat OA model.
Results
Low-dose tomatidine does not affect viability of IL-1β-treated primary chondrocytes
CCK-8 assay showed that pretreatment of primary chondrocytes with 2.5, 5, or 10μM tomatidine followed by 10ng/ml IL-1β did not affect cell viability compared to the controls (P > 0.05; Figure 3A–3C). However, pretreatment with 20μM tomatidine significantly decreased the viability of primary chondrocytes (P < 0.05; Figure 3B, 3C). We chose 2.5, 5, and 10μM tomatidine doses for subsequent experiments.
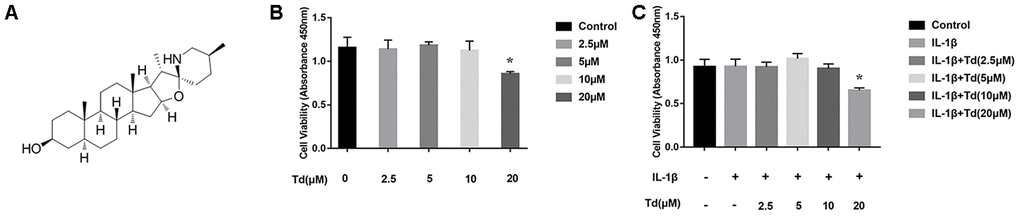
Figure 3. Effects of tomatidine on the viability of primary chondrocytes. (A) Chemical structure of tomatidine. (B, C) CCK-8 assay shows the viability of primary chondrocytes treated for 24 h with (B) 2.5, 5, 10 or 20 μM tomatidine alone or in combination with 10 ng/mlIL-1β (C). DMSO was used as control. As shown, treatments with 2.5, 5, or 10μM tomatidine or 10ng/mlIL-1β do not affect cell viability. Viability of primary chondrocytes is significantly affected by treatment with 20μM tomatidine in presence or absence of 10ng/mlIL-1β. The values are shown as means ± SD. *p < 0.05compared with the control group.
Tomatidine inhibits IL-1β-induced iNOS and COX-2 expression in primary chondrocytes
We analyzed the effects of tomatidine pre-treatment on the IL-1β-induced expression of iNOS and COX-2 in primary chondrocytes. IL-1β-treated primary chondrocytes showed significantly higher levels of iNOS and COX-2 proteins compared to the blank control, but, pre-treatment with 2.5, 5.0 and 10μM tomatidine significantly reduced iNOS and COX-2 expression in a concentration-dependent manner (Figure 4A, 4B). These data demonstrate that tomatidine inhibits IL-1β-induced iNOS and COX-2 expression in primary chondrocytes.
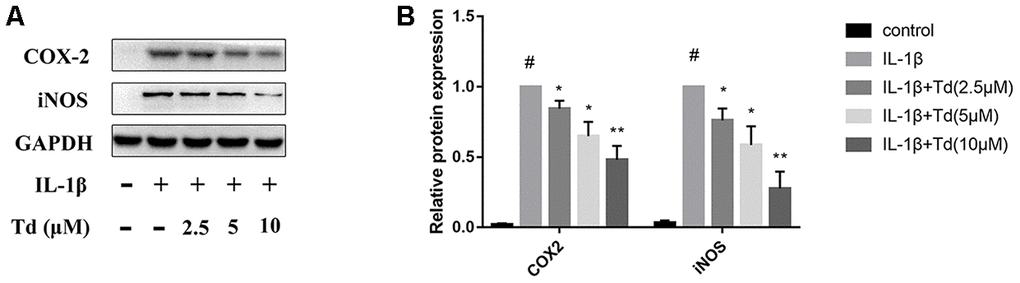
Figure 4. Tomatidine inhibits IL-1β-induced iNOS and COX-2 expression in primary chondrocytes. (A) Representative western blot images and (B) Histogram plots show the levels of iNOS and COX-2 proteins relative to GAPDH (internal control) levels in primary chondrocytes treated for 24 h with 2.5, 5, or 10μM tomatidine alone or in combination with 10ng/mlIL-1β. DMSO was used as control. The values are shown as means ± SD of triplicate experiments. #p < 0.05 compared with the control group; *p < 0.05 and **p < 0.01 compared with the IL-1β group.
Tomatidine inhibits IL-1β induced MMPs and ADAMTS-5 in chondrocytes
Next, we analyzed the effects of tomatidine on IL-1β-induced expression of MMPs (MMP1, MMP3, and MMP13) and ADAMTS-5 in primary chondrocytes. IL-1β-treated primary chondrocytes showed significantly higher expression of MMPs and ADAMTS-5 compared to the blank controls, but, tomatidine significantly reduced the expression of MMPs and ADAMTS-5 in a concentration-dependent manner (Figure 5A, 5B). These findings show that tomatidine protects against OA progression by suppressing the expression of MMPs and ADAMTS-5 in primary chondrocytes.
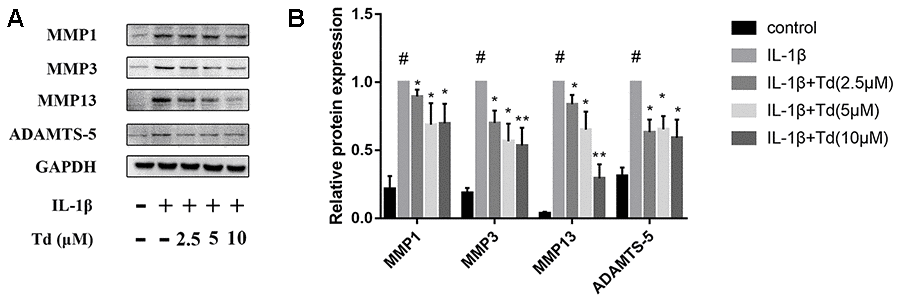
Figure 5. Tomatidine inhibits IL-1β induced MMPs and ADAMTS-5 in chondrocytes. (A) Representative western blot images and (B) Histogram plots show the levels of MMP1, MMP3, MMP13 and ADAMTS-5proteins relative to GAPDH (internal control) levels in primary chondrocytes treated for 24 h with 2.5, 5, or 10μM tomatidine alone or in combination with 10ng/mlIL-1β. DMSO was used as control. The values are shown as means ± SD of triplicate experiments. #p < 0.05 compared with the control group. *p < 0.05 and **p < 0.01 compared with the IL-1β group.
Tomatidine suppresses IL-1β induced degradation of aggrecan and collagen-II in primary chondrocytes
IL-1β-treated primary chondrocytes showed significantly higher degradation of ECM proteins, aggrecan and collagen-II, compared with the blank controls based on western blotting and immunofluorescence staining assays (Figure 6A–6C). However, tomatidine significantly reduced the degradation of aggrecan and collagen-II in the IL-1β-treated primary chondrocytes in a concentration dependent manner (Figure 6A–6C).
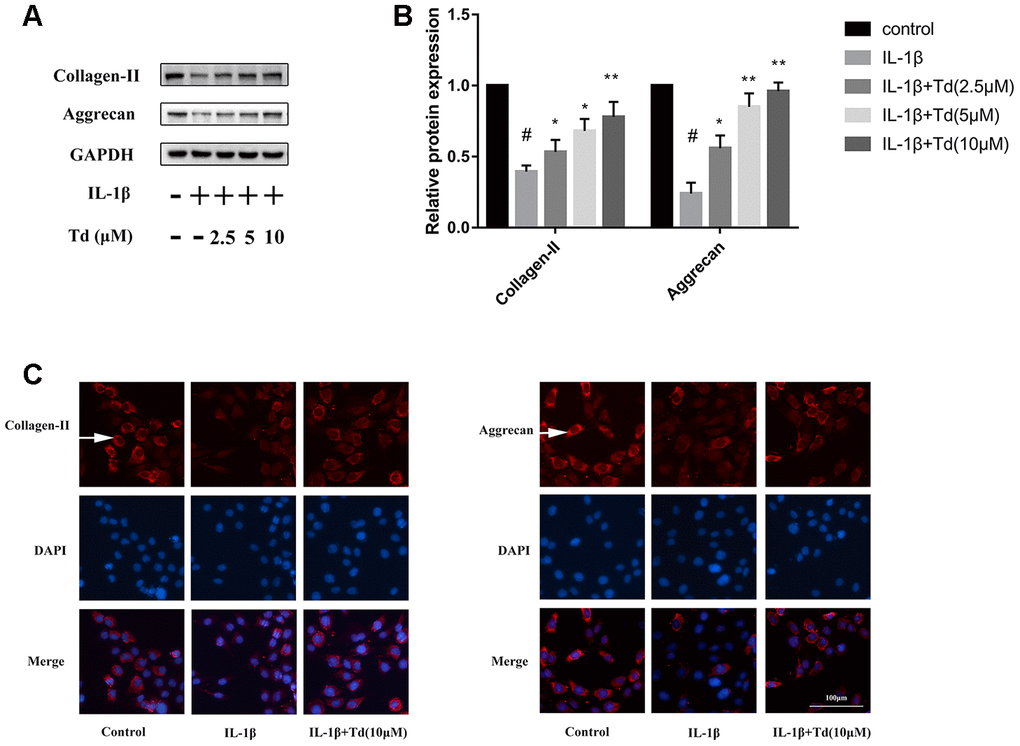
Figure 6. Tomatidine suppresses IL-1β induced degradation of aggrecan and collagen-II in chondrocytes. (A) Representative western blot images and (B) Histogram plot shows the levels of aggrecan and collagen-II proteins relative to GAPDH (internal control) levels in the primary chondrocytes treated for 24 h with 2.5, 5, or 10μM tomatidine alone or in combination with 10ng/mlIL-1β. DMSO was used as control. (C) Immunofluorescence staining images show aggrecan and collagen-II expression in the primary chondrocytes treated for 24 h with 2.5, 5, or 10μM tomatidine alone or in combination with 10ng/mlIL-1β. The nuclei are stained with DAPI. The white arrows indicate the expression of aggrecan and collagen-II. The values are shown as means ± SD of triplicate experiments. #p < 0.05 compared with the control group; *p < 0.05 and **p < 0.01 compared with the IL-1β group.
Tomatidine suppresses IL-1β induced activation of NF-κB in primary chondrocytes
We analyzed the effects of tomatidine on IL-1β-induced activation of NF-κB in primary chondrocytes. IL-1β-treated primary chondrocytes showed significantly higher levels of phospho-P65 compared to the blank controls, but, pre-treatment with tomatidine significantly reduced phospho-P65 levels in a concentration-dependent manner (Figure 7A, 7B; P < 0.05). Immunofluorescence assay showed that tomatidine inhibits nuclear translocation of P65 inIL-1β-treated primary chondrocytes in a concentration dependent manner (Figure 7C). These results demonstrate that tomatidine suppresses activation of the NF-κB in IL-1β-treated primary chondrocytes.
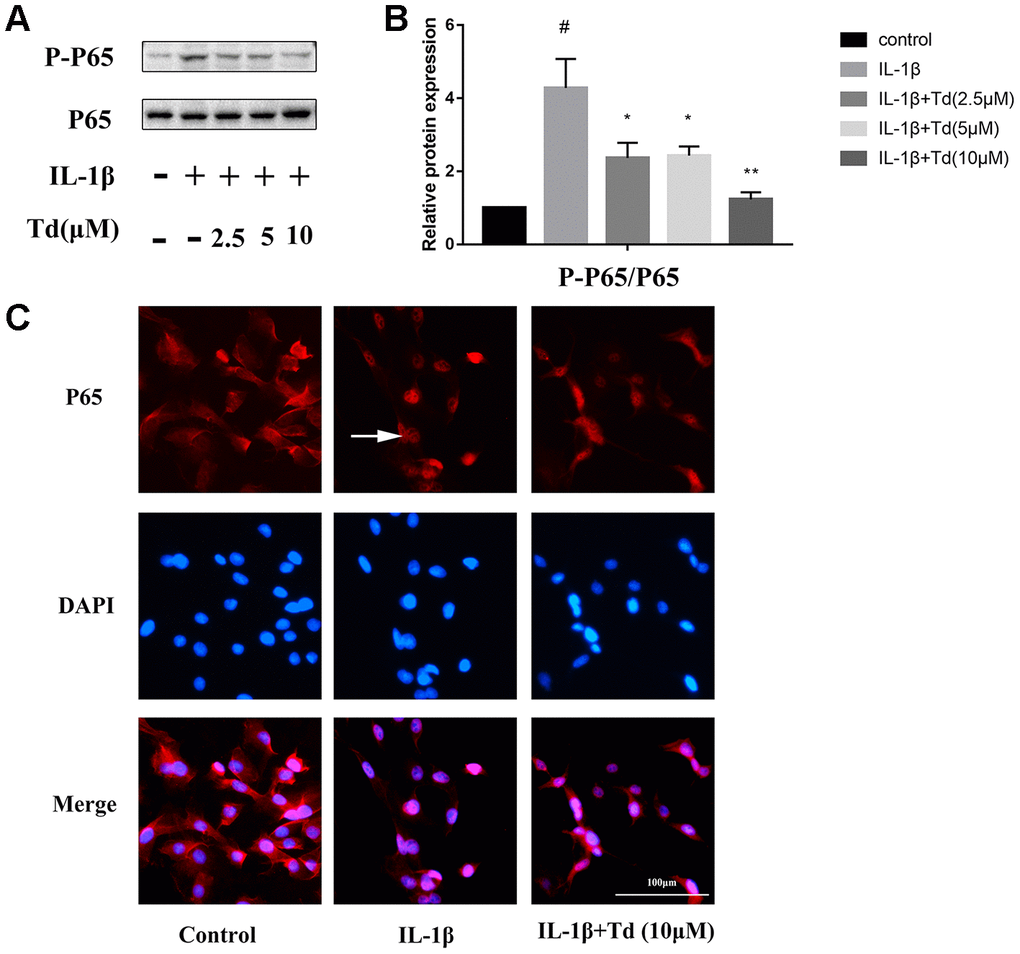
Figure 7. Tomatidine suppresses IL-1β-induced NF-κB activation in primary chondrocytes. (A) Representative western blot images and (B) Histogram plots show the levels of phospho-P65 and P65 proteins relative to GAPDH (internal control) levels in the primary chondrocytes treated for 30 mins with 2.5, 5, or 10μM tomatidine alone or in combination with 10 ng/mlIL-1β. DMSO was used as control. (C) Immunofluorescence staining images show P65 localization in primary chondrocytes treated for 30 mins with 2.5, 5, or 10μM tomatidine alone or in combination with 10ng/mlIL-1β. The white arrow indicates nuclear translocation of P65. The values are shown as means ± SD of triplicate experiments. #p < 0.05 compared with the control group; *p < 0.05 and **p < 0.01 compared with the IL-1β group.
Tomatidine suppresses IL-1β-induced activation of MAPK signaling pathway in primary chondrocytes
Next, we analyzed the effects of tomatidine on the activation status of the MAPK signaling pathway in IL-1β-treated primary chondrocytes. As shown in Figure 8A, 8B, IL-1β-treated primary chondrocytes showed significantly higher levels of phospho-P38, phospho-ERK, and phospho-JNK compared to the blank controls, but, tomatidine suppressed P38 activation in a concentration-dependent manner (Figure 8A, 8B; P < 0.05). These data demonstrate that tomatidine reduces inflammation in primary chondrocytes by inhibiting the activation of p38 MAPK signaling pathway.
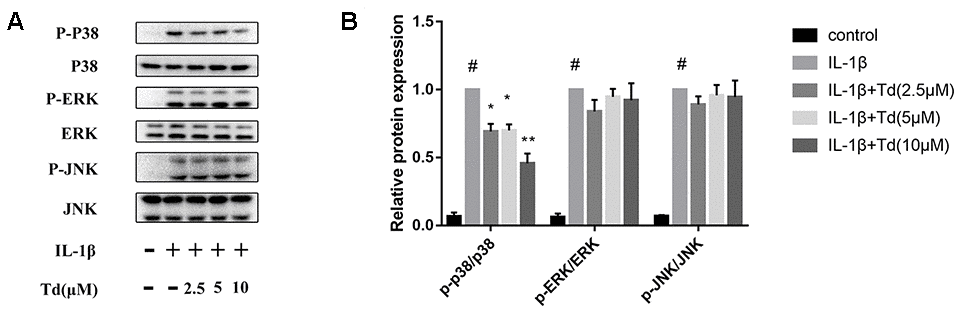
Figure 8. Tomatidine inhibits IL-1β-induced MAPK activation in primary chondrocytes. (A) Representative western blot images and (B) Histogram plot shows the levels of phospho-P38, P38, phospho-ERK, ERK, phospho-JNK, and JNK proteins relative to GAPDH (internal control) levels in the primary chondrocytes treated for 30 mins with 2.5, 5, or 10μM tomatidine alone or in combination with 10ng/mlIL-1β. DMSO was used as control. The values are means ± SD of triplicate experiments. #p < 0.05 compared with the control group. *p < 0.05 and **p < 0.01 compared with the IL-1β group.
Tomatidine ameliorates OA progression in the rat OA model
Next, we analyzed the effects of tomatidine on the progression of OA in the rat model. Histological analysis showed that damage to the articular cartilage was significantly reduced in the experimental group rats (OA model rats treated with tomatidine) compared to the OA group (Figure 9A). Furthermore, immunohistochemical staining showed that tomatidine significantly reduced INOS and MMP3levels in the articular cartilage of experimental group rats compared to the OA group (Figure 9B, 9C). This demonstrates that tomatidine ameliorates OA progression in the rat OA model.
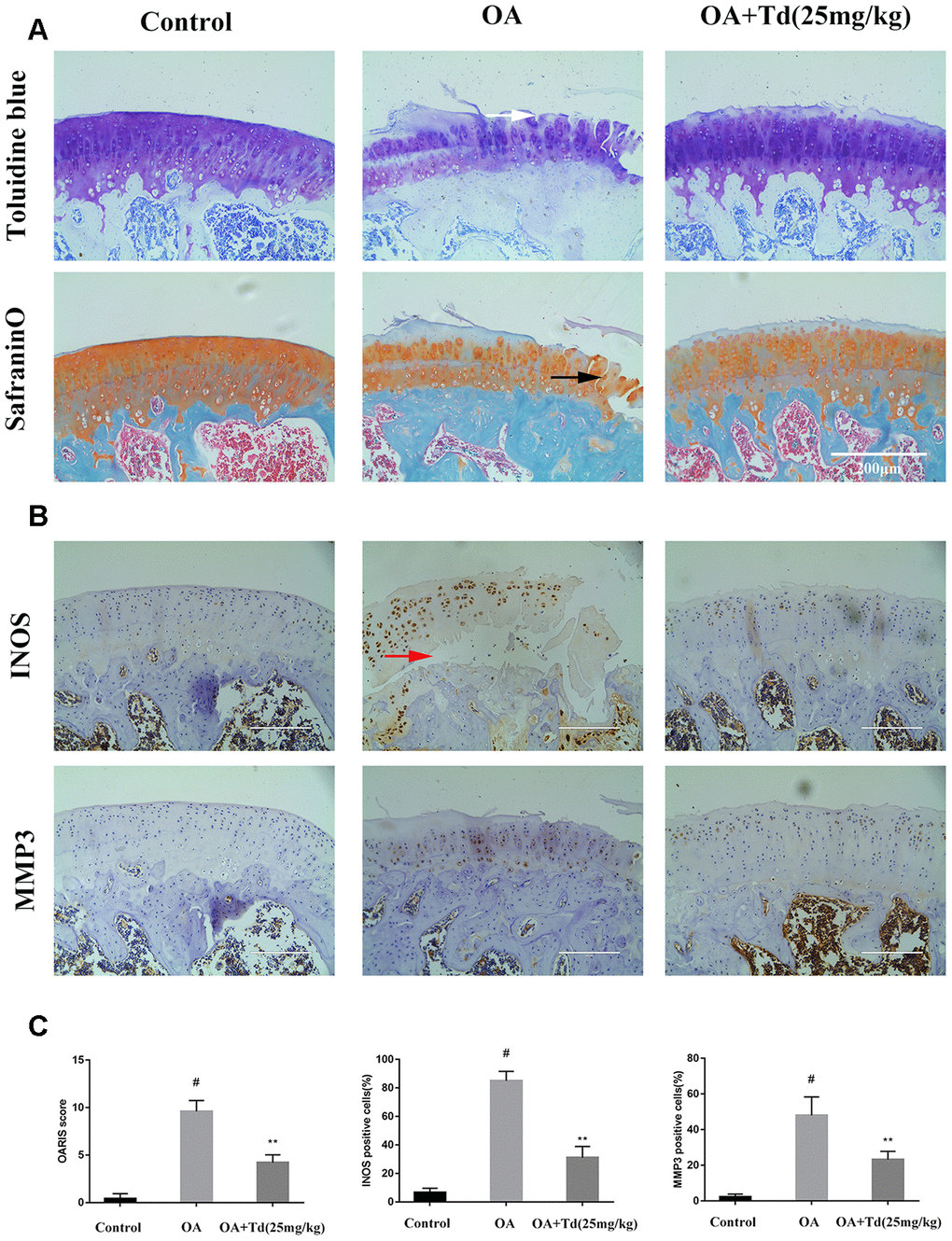
Figure 9. Tomatidine ameliorates OA progression in the rat OA model. Sprague Dawley rats (n=5/group) were randomly divided into blank control, OA, and experimental groups. The experimental group rats were fed a diet that included 25mg/kg/day tomatidine. The rats were maintained in these groups for 12 weeks and then euthanized and their tibiofemoral joints were obtained and processed (A) Representative images show safranin O-fast green(S-O) and toluidine blue stained sections of tibiofemoral joints from blank control, OA and experimental group rats. The vertical fissures (black arrow), surface discontinuity (white arrow) and delamination (red arrow) are indicated in the relevant samples as shown. (B) Representative immunohistochemical stained images show the expression of iNOS and MMP3 proteins in tibiofemoral joint sections from blank control, OA and experimental group rats. (C) The OA grades of blank control, OA and experimental group rats at 12 weeks according to the Osteoarthritis Research Society International (OARSI) scores are shown. The scoring was performed in a blinded manner. The iNOS and MMP3 positive cells were counted in each tibiofemoral joint section from blank control, OA and experimental group rats and quantified using the Image-J software. #p < 0.05 compared with the control group. *p < 0.05 and **p < 0.01 compared with the OA group.
μ-CT evaluation of the effects of tomatidine treatment on remodeling of the subchondral bone in OA model rats
We performed μ-CT to determine the effects of tomatidine on the subchondral bone remodeling in the OA model rats. The OA group rats showed decreased BV/TV and trabecular numbers and increased trabecular separation in the subchondral bones compared to the blank control group (Figure 10A, 10B; P < 0.05). However, the BV/TV, trabecular numbers and trabecular separation parameters were similar for both OA and tomatidine-treated experimental group rats (Figure 10A, 10B; P > 0.05). These results show that tomatidine does not affect subchondral bone remodeling in OA model rats.
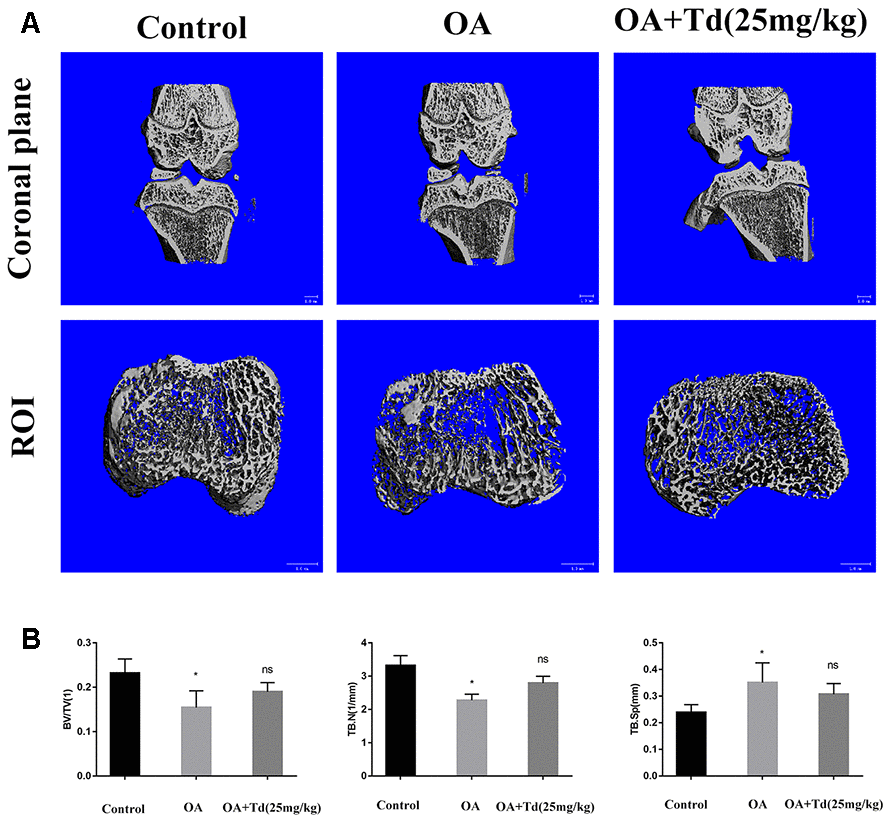
Figure 10. μ-CT evaluation of the subchondral bone at the tibial plateau in the OA model rats. (A) Representative images show the 3D μ-CT coronal views of the right knee and axial view of ROI from blank control, OA and experimental group rats. (B) Histogram plots show the analysis of trabecular structure analysis, including bone volume/tissue volume (BV/TV), trabecular number (Tb. N) and trabecular separation (Tb. Sp)in the blank control, OA and experimental group rats. *p < 0.05 compared with the control group; ns: not significant compared with the OA group.
Discussion
Our study demonstrates that tomatidine inhibits IL-1β-induced expression of iNOS, COX-2, and MMPs, and degradation of aggrecan and collagen-II in primary chondrocytes by suppressing the activation of NF- κB and MAPK signaling pathways. These results show that tomatidine sustains the levels of ECM proteins by suppressing the expression of proteins involved in inflammation and degradation of ECM proteins. Moreover, tomatidine reduces cartilage degeneration in the rat OA model. These findings reveal that tomatidine is a promising drug for OA treatment.
IL-1β is a major pro-inflammatory cytokine that is involved in OA pathology by inducing inflammatory responses and catabolic effects [9, 17–19]. IL-1β promotes the expression of MMPs, which are involved in the degradation of cartilage components [20]. IL-1β also induces ADAMTS-5, which promotes breakdown of aggrecan [21, 22]. IL-1β stimulates the expression of iNOS and COX-2, which promote the release of well known inflammatory mediators, NO and PGE2 [23, 24]. Several studies suggest that inhibition of these pro-inflammatory mediators hinders OA progression [19, 20].
In this study, we hypothesized that the anti-inflammatory properties of tomatidine will delay or abrogate OA progression in the primary chondrocytes and cartilage tissue. Chiu et al. reported that tomatidine inhibits the inflammatory response in LPS-stimulated mouse macrophages by suppressing the NF-κB and JNK signaling pathways [15]. The activation of NF-κB signaling pathway is implicated in the onset and progression of OA [25]. NF-κB pathway also regulates the expression of iNOS, COX-2, and MMPs [26, 27]. Therefore, targeted inhibition of the NF- κB signaling pathway is a potential therapy for patients with OA. We demonstrate that tomatidine significantly inhibits IL-1β-induced expression of inflammatory factors by suppressing NF-κB activation. MAPK signaling is another key pathway involved in OA pathogenesis [28]. The OA chondrocytes demonstrate significantly higher levels of phospho-p38, phospho-JNK, and phospho-ERK compared to normal chondrocytes [29]. Our study demonstrates that tomatidine blocks IL-1β-stimulated MAPK pathway activation in primary articular chondrocytes. Overall, our study demonstrates that tomatidine exerts anti-inflammatory effects on IL-1β-stimulated chondrocytes by suppressing the NF- κB and MAPK signaling pathways.
Our study has several limitations. Firstly, it is not clear if tomatidine inhibits NF- κB and MAPK pathways directly or indirectly through mediators. Secondly, a higher concentration of tomatidine does not demonstrate anti-inflammatory and cell survival effects, probably because it exceeds the optimal dosage.
Conclusions
In summary, our study shows that tomatidine inhibits IL-1β-stimulated inflammation in primary chondrocytes by suppressing the NF-κB and MAPK signaling pathways (Figure 11). Moreover, tomatidine decreases the articular cartilage pathology in the rat OA model. These findings suggest that tomatidine is a potential treatment for OA, which needs to be ascertained by further investigations.
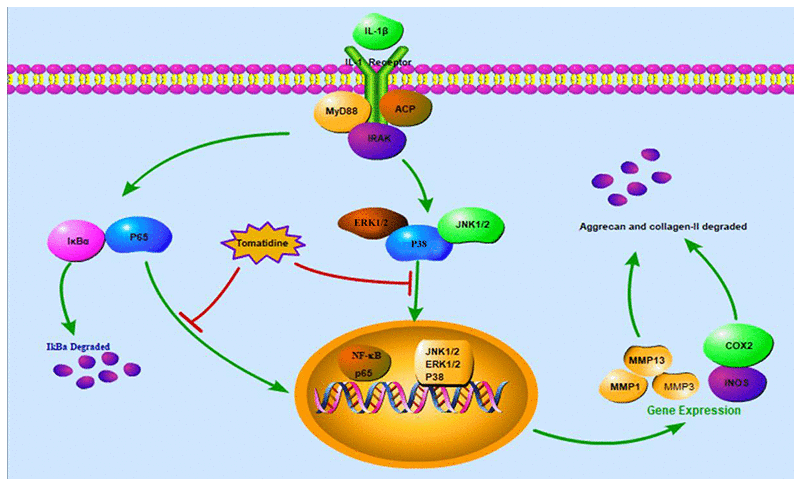
Figure 11. The anti-inflammatory mechanism of action of tomatidine in primary articular chondrocytes and the OA model rats. The schematic diagram shows that tomatidine suppresses IL-1β-induced expression of iNOS, COX-2, MMPs and ADAMTS-5, and degradation of aggrecan and collagen-II by inhibiting the NF-κB and MAPK pathways.
Materials and Methods
Chemicals and reagents
Tomatidine (purity>98%) was purchased from MedChem Express (Princeton, NJ). Collagenase type II, recombinant rat IL-1β, and dimethylsulfoxide (DMSO) were obtained from Sigma Chemical Co. (St. Louis, MO, USA). Cell Counting Kit-8 (CCK-8) was obtained from Boster (Wuhan, China). The primary antibody against aggrecan was purchased from Abcam (Cambridge, UK). The antibodies against type-II collagen, iNOS, and MMP13 were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA), whereas, antibodies against COX2, MMP3, P38, P-P38, ERK, P-ERK, JNK, P-JNK, P65, P-P65 were obtained from the Cell Signaling Technology Incorporated (Beverly, MA, USA). The antibodies against GAPDH, ADAMTS-5 and MMP1 were obtained from Boster (Wuhan, China). Fetal bovine serum (FBS), bovine serum albumin (BSA), and Dulbecco’s modified Eagle’ ’s medium F12 (DMEM/F12) were purchased from Gibco (NY, USA).
Primary chondrocyte isolation and culture
The primary chondrocytes were isolated as described previously [36]. Briefly, the articular cartilage was obtained from the knees of male Sprague-Dawley rats, cut into 1 mm3pieces, and digested with trypsin for 30 min at 37°C. Then, the primary chondrocytes were obtained by digestion with 0.2% collagenase II for 4-6 h at 37°C. The primary chondrocytes were cultured in DMEM/F12 medium supplemented with 10% FBS and 1% penicillin/streptomycin at 37°C and 5% CO2. The medium was changed every two days. The primary chondrocytes from 2nd and 3rdpassages were used for further experiments.
Cell viability assay
Tomatidine cytotoxicity was analyzed using the CCK-8 assay according to manufacturer’s instructions. Briefly, the primary chondrocytes were seeded in a 96-well plate (5 × 103/well) for 24h, pre-treated with different concentrations of tomatidine (2.5, 5, 10 or 20μM) for 2 h then treated with10ng/mlIL-1β for 24h. Primary chondrocytes treated with DMSO, tomatidine or IL-1β alone were used as controls. Subsequently, the cells were incubated with 10μl CCK-8 solution for 1h at 37°C. The absorbance was measured at 450nm using a plate reader (Bio-Rad, Richmond, CA, USA).
Western blotting
The primary chondrocytes were seeded in a 6-well plate at 37°C and 5% CO2 in a humidified chamber. The cells were treated with DMSO (control), 10ng/ml IL-1β alone or 2.5, 5 or 10μM tomatidine pretreatment for 2 h followed by 10ng/ml IL-1β for 24h. Then, the total cellular proteins were extracted using the RIPA buffer containing 1% protease inhibitor and 1% phosphatase inhibitor (Boster, Wuhan, China). The concentration of total protein lysates was measured using the bicinchoninic acid (BCA) protein assay kit (Boster, Wuhan, China). Equal amounts of protein samples were separated on a 12% polyacrylamide gel, transferred onto 0.45μm polyvinylidene difluoride (PVDF) membranes (Millipore, Billerica, MA), andb locked with 5% BSA in Tris-buffered Saline-Tween solution (TBST) for 1h at room temperature. Then, the membranes were incubated overnight at 4°C with the primary antibodies against iNOS, MMP13, and GAPDH at 1:500 dilution, and antibodies against COX-2, MMP1, MMP3, ADAMTS-5, phospho-P38, P38, phospho-ERK, ERK, phospho-JNK, JNK, phospho-P65 and P65 at 1:1000 dilution. Then, after washing with TBST, the membranes were incubated with the HRP-conjugated secondary antibody for 1h at room temperature. The blots were developed with the Enhanced Chemiluminescence (ECL) reagent (ECL; Boster, Wuhan, China), and the protein bands were quantified using the Bio-Rad Image Lab 5.0 system(Bio-Rad, USA).
Immunofluorescence staining
The primary chondrocytes were seeded in a 24-well plate and treated with 10ng/ml IL-1β alone or in combination with 10μM tomatidine. The cells were then fixed with 4% paraformaldehyde for 10min, permeated with 0.1% Triton X-100 for 10min, and blocked with 1% BSA for 30min. Subsequently, the cells were incubated overnight at 4 °C with antibodies against collagen-II, aggrecan, and P65 at 1:200 dilutions. Then, after washing thrice with PBS, the cells were incubated with Cy3-conjugated goat anti-rabbit or Cy3- conjugated goat anti-rat secondary antibodies at a 1:75 dilution for 1h. The cells were incubated with 4–6-Diamidino-2-phenylindole (DAPI) for 10 min to stain the nuclei, and examined using a automated EVOS fluorescence microscope (Life Technologies, USA).
Animal experiments and histological analysis
The animal protocol was approved by the Ethics and Animal Research Committee of Huazhong University of Science and Technology. We purchased male Sprague-Dawley rats weighing 250-300g from the Experimental Animal Center of Tongji Medical College (Wuhan, China) and induced OA as described previously [37]. The animals (n=5/group) were randomly divided into blank control, OA, and experimental groups. OA pathogenesis was induced by surgery involving anterior cruciate ligament transaction and partial medial meniscectomy [37]. The experimental group rats were fed with 25 mg/ Kg/ day tomatidine as previously described [38]. After 12 weeks, the animals were sacrificed, the tibiofemoral joints were collected, fixed with 4% paraformaldehyde for 24h, and decalcified by incubation in 10% EDTA for 20 days. Then, the specimens were embedded in wax blocks, cut into 3-5μm thick sections, and stained with safranin O-fast green(S-O) and toluidine blue. The stained specimens from all 3 rat groups were scored according to the Osteoarthritis Research Society International (OARSI) recommendations and graded in a blinded manner. Another set of tibiofemoral joint sections from the 3 groups were incubated with antibodies against INOS and MMP3. The numbers of INOS and MMP3 positive cells were determined using the Image-J software.
Micro-computed tomography (μ-CT)
We scanned the right knee joints of the rats using the μ-Computed Tomography system (Scanco Medical, Bassersdorf, Switzerland). We obtained 21.0μm resolution μ-CT images at 70kV and 113μA and generated 3D images using the CT system software. The same region of interest (ROI) was drawn on 100 consecutive slices of 2.1mm total thickness representing the subchondral bone at the tibial plateau and the values for bone parameters such as bone volume/tissue volume (BV/TV), trabecular numbers (Tb.N) and trabecular separation (Tb.Sp) were determined.
Statistical analysis
All experiments were performed in triplicate and the data presented as means ± standard deviation (SD). All data were analyzed using the Shapiro-Wilk test for normal distribution and Levene’s test for homogeneity of variances. The statistical differences between normally distributed groups were analyzed by one-way analysis of variance (ANOVA) and Student’s t-test. The nonparametric data (OARSI scores) were compared using the Kruskal-Wallis H test. P-value < 0.05 was considered statistically significant.
Author Contributions
HY designed the experiments; XC, TY, XH and YX performed the experiments; BN performed instrument operations; RZ analyzed the data; XC and TY prepared the manuscript. All authors read and consented to the final manuscript.
Conflicts of Interest
All authors declare that there are no conflicts of interest.
Funding
This research was supported by the Fundamental Research Funds for the Central Universities (Grant No. 2017KFYXJJ104) and the National Natural Science Foundation of China (Grant No. 81772390).
References
- 1. Glyn-Jones S, Palmer AJ, Agricola R, Price AJ, Vincent TL, Weinans H, Carr AJ. Osteoarthritis. Lancet. 2015; 386:376–87. https://doi.org/10.1016/S0140-6736(14)60802-3 [PubMed]
- 2. Hiligsmann M, Cooper C, Arden N, Boers M, Branco JC, Luisa Brandi M, Bruyère O, Guillemin F, Hochberg MC, Hunter DJ, Kanis JA, Kvien TK, Laslop A, et al. Health economics in the field of osteoarthritis: an expert's consensus paper from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Semin Arthritis Rheum. 2013; 43:303–13. https://doi.org/10.1016/j.semarthrit.2013.07.003 [PubMed]
- 3. Mandl LA. Osteoarthritis year in review 2018: clinical. Osteoarthritis Cartilage. 2019; 27:359–64. https://doi.org/10.1016/j.joca.2018.11.001 [PubMed]
- 4. Scanzello CR, Goldring SR. The role of synovitis in osteoarthritis pathogenesis. Bone. 2012; 51:249–57. https://doi.org/10.1016/j.bone.2012.02.012 [PubMed]
- 5. Bruyère O, Cooper C, Pelletier JP, Branco J, Luisa Brandi M, Guillemin F, Hochberg MC, Kanis JA, Kvien TK, Martel-Pelletier J, Rizzoli R, Silverman S, Reginster JY. An algorithm recommendation for the management of knee osteoarthritis in Europe and internationally: a report from a task force of the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Semin Arthritis Rheum. 2014; 44:253–63. https://doi.org/10.1016/j.semarthrit.2014.05.014 [PubMed]
- 6. McAlindon TE, Bannuru RR, Sullivan MC, Arden NK, Berenbaum F, Bierma-Zeinstra SM, Hawker GA, Henrotin Y, Hunter DJ, Kawaguchi H, Kwoh K, Lohmander S, Rannou F, et al. OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthritis Cartilage. 2014; 22:363–88. https://doi.org/10.1016/j.joca.2014.01.003 [PubMed]
- 7. Wojdasiewicz P, Poniatowski ŁA, Szukiewicz D. The role of inflammatory and anti-inflammatory cytokines in the pathogenesis of osteoarthritis. Mediators Inflamm. 2014; 2014:561459. https://doi.org/10.1155/2014/561459 [PubMed]
- 8. Kapoor M, Martel-Pelletier J, Lajeunesse D, Pelletier JP, Fahmi H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011; 7:33–42. https://doi.org/10.1038/nrrheum.2010.196 [PubMed]
- 9. Kobayashi M, Squires GR, Mousa A, Tanzer M, Zukor DJ, Antoniou J, Feige U, Poole AR. Role of interleukin-1 and tumor necrosis factor alpha in matrix degradation of human osteoarthritic cartilage. Arthritis Rheum. 2005; 52:128–35. https://doi.org/10.1002/art.20776 [PubMed]
- 10. Friedman M. Tomato glycoalkaloids: role in the plant and in the diet. J Agric Food Chem. 2002; 50:5751–80. https://doi.org/10.1021/jf020560c [PubMed]
- 11. Song SE, Shin SK, Cho HW, Im SS, Bae JH, Woo SM, Kwon TK, Song DK. Tomatidine inhibits tumor necrosis factor-α-induced apoptosis in C2C12 myoblasts via ameliorating endoplasmic reticulum stress. Mol Cell Biochem. 2018; 444:17–25. https://doi.org/10.1007/s11010-017-3226-3 [PubMed]
- 12. Kuo CY, Huang WC, Liou CJ, Chen LC, Shen JJ, Kuo ML. Tomatidine attenuates airway hyperresponsiveness and inflammation by suppressing Th2 cytokines in a mouse model of asthma. Mediators Inflamm. 2017; 2017:5261803. https://doi.org/10.1155/2017/5261803 [PubMed]
- 13. Guay I, Boulanger S, Isabelle C, Brouillette E, Chagnon F, Bouarab K, Marsault E, Malouin F. Tomatidine and analog FC04-100 possess bactericidal activities against listeria, bacillus and staphylococcus SPP. BMC Pharmacol Toxicol. 2018; 19:7. https://doi.org/10.1186/s40360-018-0197-2 [PubMed]
- 14. Yan KH, Lee LM, Yan SH, Huang HC, Li CC, Lin HT, Chen PS. Tomatidine inhibits invasion of human lung adenocarcinoma cell A549 by reducing matrix metalloproteinases expression. Chem Biol Interact. 2013; 203:580–87. https://doi.org/10.1016/j.cbi.2013.03.016 [PubMed]
- 15. Chiu FL, Lin JK. Tomatidine inhibits iNOS and COX-2 through suppression of NF-kappaB and JNK pathways in LPS-stimulated mouse macrophages. FEBS Lett. 2008; 582:2407–12. https://doi.org/10.1016/j.febslet.2008.05.049 [PubMed]
- 16. Dweep H, Gretz N. miRWalk2.0: a comprehensive atlas of microRNA-target interactions. Nat Methods. 2015; 12:697. https://doi.org/10.1038/nmeth.3485 [PubMed]
- 17. Blasioli DJ, Kaplan DL. The roles of catabolic factors in the development of osteoarthritis. Tissue Eng Part B Rev. 2014; 20:355–63. https://doi.org/10.1089/ten.TEB.2013.0377 [PubMed]
- 18. Tetlow LC, Adlam DJ, Woolley DE. Matrix metalloproteinase and proinflammatory cytokine production by chondrocytes of human osteoarthritic cartilage: associations with degenerative changes. Arthritis Rheum. 2001; 44:585–94. https://doi.org/10.1002/1529-0131(200103)44:3<585::AID-ANR107>3.0.CO;2-C [PubMed]
- 19. Santangelo KS, Nuovo GJ, Bertone AL. In vivo reduction or blockade of interleukin-1β in primary osteoarthritis influences expression of mediators implicated in pathogenesis. Osteoarthritis Cartilage. 2012; 20:1610–18. https://doi.org/10.1016/j.joca.2012.08.011 [PubMed]
- 20. Mao Z, Wang P, Pan Q, Huang X, Zhang R, Shang X, Ma X, You H. Pravastatin alleviates interleukin 1β-induced cartilage degradation by restoring impaired autophagy associated with MAPK pathway inhibition. Int Immunopharmacol. 2018; 64:308–318. https://doi.org/10.1016/j.intimp.2018.09.018 [PubMed]
- 21. Larkin J, Lohr TA, Elefante L, Shearin J, Matico R, Su JL, Xue Y, Liu F, Genell C, Miller RE, Tran PB, Malfait AM, Maier CC, Matheny CJ. Translational development of an ADAMTS-5 antibody for osteoarthritis disease modification. Osteoarthritis Cartilage. 2015; 23:1254–66. https://doi.org/10.1016/j.joca.2015.02.778 [PubMed]
- 22. Ji Q, Xu X, Zhang Q, Kang L, Xu Y, Zhang K, Li L, Liang Y, Hong T, Ye Q, Wang Y. The IL-1β/AP-1/miR-30a/ADAMTS-5 axis regulates cartilage matrix degradation in human osteoarthritis. J Mol Med (Berl). 2016; 94:771–85. https://doi.org/10.1007/s00109-016-1418-z [PubMed]
- 23. Zhong L, Schivo S, Huang X, Leijten J, Karperien M, Post JN. Nitric oxide mediates crosstalk between interleukin 1β and Wnt signaling in primary human chondrocytes by reducing DKK1 and FRZB expression. Int J Mol Sci. 2017; 18:2491. https://doi.org/10.3390/ijms18112491 [PubMed]
- 24. Li TF, Yukata K, Yin G, Sheu T, Maruyama T, Jonason JH, Hsu W, Zhang X, Xiao G, Konttinen YT, Chen D, O’Keefe RJ. BMP-2 induces ATF4 phosphorylation in chondrocytes through a COX-2/PGE2 dependent signaling pathway. Osteoarthritis Cartilage. 2014; 22:481–89. https://doi.org/10.1016/j.joca.2013.12.020 [PubMed]
- 25. Rigoglou S, Papavassiliou AG. The NF-κB signalling pathway in osteoarthritis. Int J Biochem Cell Biol. 2013; 45:2580–84. https://doi.org/10.1016/j.biocel.2013.08.018 [PubMed]
- 26. Ulivi V, Giannoni P, Gentili C, Cancedda R, Descalzi F. p38/NF-kB-dependent expression of COX-2 during differentiation and inflammatory response of chondrocytes. J Cell Biochem. 2008; 104:1393–406. https://doi.org/10.1002/jcb.21717 [PubMed]
- 27. Khan NM, Haseeb A, Ansari MY, Devarapalli P, Haynie S, Haqqi TM. Dataset of effect of wogonin, a natural flavonoid, on the viability and activation of NF-κB and MAPKs in IL-1β-stimulated human OA chondrocytes. Data Brief. 2017; 12:150–55. https://doi.org/10.1016/j.dib.2017.03.054 [PubMed]
- 28. Zhang Y, Pizzute T, Pei M. A review of crosstalk between MAPK and Wnt signals and its impact on cartilage regeneration. Cell Tissue Res. 2014; 358:633–49. https://doi.org/10.1007/s00441-014-2010-x [PubMed]
- 29. Boileau C, Martel-Pelletier J, Brunet J, Schrier D, Flory C, Boily M, Pelletier JP. PD-0200347, an alpha2delta ligand of the voltage gated calcium channel, inhibits in vivo activation of the Erk1/2 pathway in osteoarthritic chondrocytes: a PKCalpha dependent effect. Ann Rheum Dis. 2006; 65:573–80. https://doi.org/10.1136/ard.2005.041855 [PubMed]
- 30. Szklarczyk D, Santos A, von Mering C, Jensen LJ, Bork P, Kuhn M. STITCH 5: augmenting protein-chemical interaction networks with tissue and affinity data. Nucleic Acids Res. 2016; 44:D380–84. https://doi.org/10.1093/nar/gkv1277 [PubMed]
- 31. Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 2003; 13:2498–504. https://doi.org/10.1101/gr.1239303 [PubMed]
- 32. Huang da W, Sherman BT, Lempicki RA. Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009; 37:1–13. https://doi.org/10.1093/nar/gkn923 [PubMed]
- 33. Huang da W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009; 4:44–57. https://doi.org/10.1038/nprot.2008.211 [PubMed]
- 34. Walter W, Sánchez-Cabo F, Ricote M. GOplot: an R package for visually combining expression data with functional analysis. Bioinformatics. 2015; 31:2912–14. https://doi.org/10.1093/bioinformatics/btv300 [PubMed]
- 35. Gu Z, Gu L, Eils R, Schlesner M, Brors B. Circlize implements and enhances circular visualization in R. Bioinformatics. 2014; 30:2811–12. https://doi.org/10.1093/bioinformatics/btu393 [PubMed]
- 36. Huang X, Xi Y, Pan Q, Mao Z, Zhang R, Ma X, You H. Caffeic acid protects against IL-1β-induced inflammatory responses and cartilage degradation in articular chondrocytes. Biomed Pharmacother. 2018; 107:433–439. https://doi.org/10.1016/j.biopha.2018.07.161 [PubMed]
- 37. Chu X, You H, Yuan X, Zhao W, Li W, Guo X. Protective effect of lentivirus-mediated siRNA targeting ADAMTS-5 on cartilage degradation in a rat model of osteoarthritis. Int J Mol Med. 2013; 31:1222–28. https://doi.org/10.3892/ijmm.2013.1318 [PubMed]
- 38. Fujiwara Y, Kiyota N, Tsurushima K, Yoshitomi M, Horlad H, Ikeda T, Nohara T, Takeya M, Nagai R. Tomatidine, a tomato sapogenol, ameliorates hyperlipidemia and atherosclerosis in apoE-deficient mice by inhibiting acyl-CoA:cholesterol acyl-transferase (ACAT). J Agric Food Chem. 2012; 60:2472–79. https://doi.org/10.1021/jf204197r [PubMed]