Introduction
Dilated cardiomyopathy (DCM) is a non-ischemic heart muscle disease with structural and functional myocardial abnormalities characterized by dilation of the ventricular chamber and impaired contraction [1]. Myocardial damage triggers inflammation followed by the recruitment of immune cells to the injured site and the release of cytokines [2, 3].
Furthermore, aging is considered to be a risk factor for cardiovascular diseases and to have a negative impact on ventricular function [4–7]. On the molecular level, aging is accompanied by a disturbance in energy homeostasis, mitochondrial dysfunction, and increased inflammation [4]. Importantly, sex differences in the mitochondrial function in several pathologies, including cardiovascular diseases, have been demonstrated [8]. Mitochondrial biogenesis and respiration are regulated by PGC-1α, a transcriptional coactivator, and its downregulation has been shown to lead to hypertrophy and heart failure [9–11]. Furthermore, the reduced activity of PGC-1α itself, as well as its modulators, especially AMP-activated kinase (AMPK) among others, has been associated with aging [12]. A decline in AMPK activity, a crucial regulator of energy metabolic homeostasis, has been shown in older subjects, and increasing this AMPK activity may extend lifespans [13]. Sirtuins (Sirt), a conserved family of global metabolic regulators with NAD+-dependent deacetylase activity [14], and Sirt1 (an evolutionarily conserved AMPK partner) in particular [15, 16], have been widely reported to protect against age-associated diseases and therefore, to increase health span and life span [17–21]. Consistent reduction of NAD+ levels in older mice is accompanied by a decrease in Sirt1 activity, while its genetic or pharmacological restoration promotes longevity [18, 22, 23].
Among other factors, AMPK and Sirt1 play essential roles in mitochondrial biology. Particularly, AMPK and Sirt1 promote PGC-1α activity and, thus, mitochondrial biogenesis. Furthermore, AMPK controls mitochondrial clearance, i.e., mitophagy [24] and a decline in the AMPK activity leads to impaired mitophagy, accumulation of dysfunctional mitochondria, and ROS formation, which may trigger inflammation- and aging-related diseases [4, 25, 26]. Additionally, the release of mtDNA from damaged mitochondria into the cytosol may lead to an inflammatory response, via Toll-like receptors and STING-dependent inflammasome activation [25]. Similarly, emerging data have suggested that Sirt1 may play an anti-inflammatory role [27–29].
In addition to Sirt1, Sirt3 is another sirtuin playing an essential role in mitochondrial biology. Sirt3 is the main mitochondrial-localized deacetylase that maintains the activity of numerous mitochondrial enzymes, e.g. SOD2, and thus, supports metabolic and redox balance in mitochondria [30]. Both we and others have shown a decline in Sirt3 expression in older human myocardium [31, 32]. It is important to note that an anti-inflammatory role of mitochondria-localized Sirt3 has been reported [33, 34].
Sex is another potential risk factor associated with cardiovascular diseases. Men have an increased incidence and severity of atherosclerosis, myocardial infarction, heart failure, and DCM [35–37], whereas women with DCM have better chances of survival than men [38]. Heart failure is associated with cardiomyocyte hypertrophy, apoptosis, inflammation, and interstitial fibrosis, which all occur in a sex-specific manner [39]. Estrogen (E2) seems to play a protective role, as a decline in its levels is associated with deleterious left ventricle remodeling and cardiac dysfunction [40].
Altogether, sex and age have significant impacts on cardiomyopathy, however, there is still a large gap in the research about signaling in heart diseases dependent on age or sex. Here, we investigated age- and sex-related alterations in the expression of metabolic regulators, i.e., AMPK and Sirt1, mitochondrial biogenesis, and inflammation parameters in patients with DCM. The analyses revealed a decreased Sirt1 and Sirt3 expression in older DCM patients in both sexes, whereas the activity of AMPK was increased only in men. Sex differences were also found in mitochondrial antioxidant capacity, e.g., SOD2. DCM in the hearts of older patients was associated with a reduced expression of mitochondrial proteins and increased inflammation, which were both sex dependent and independent.
Results
Discussion
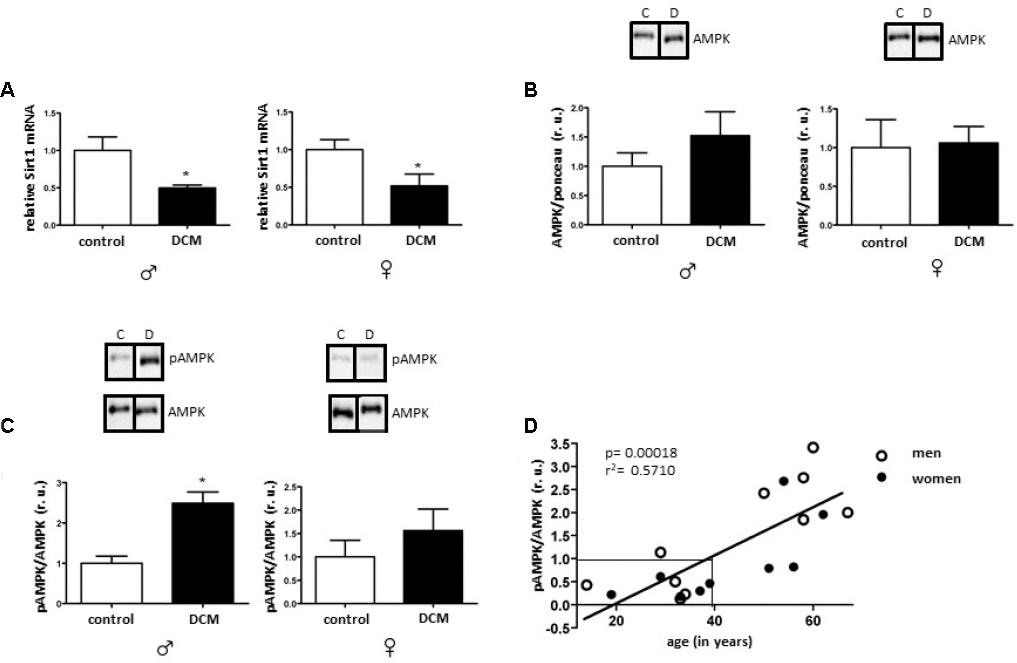
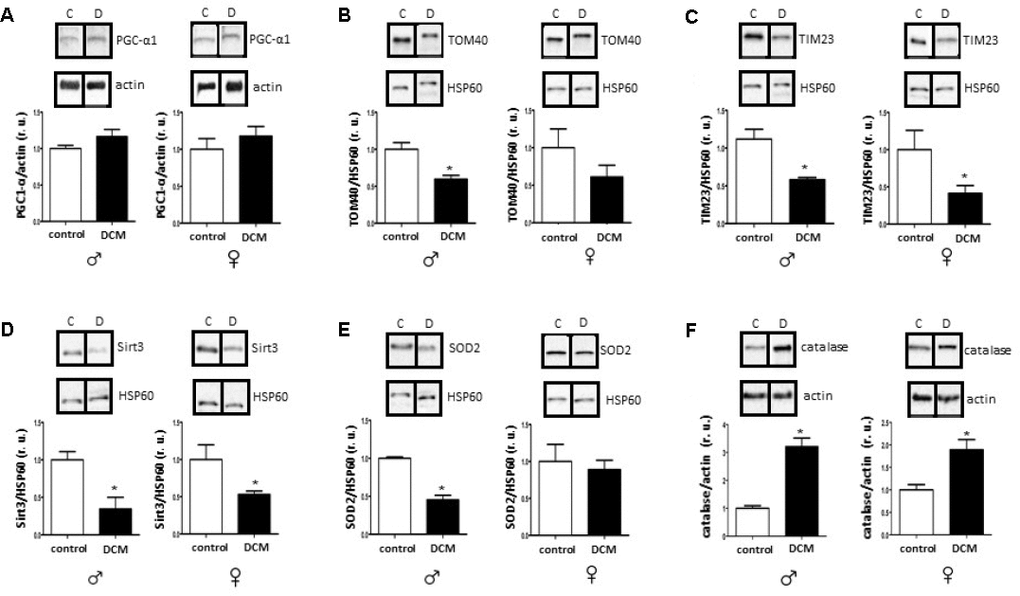
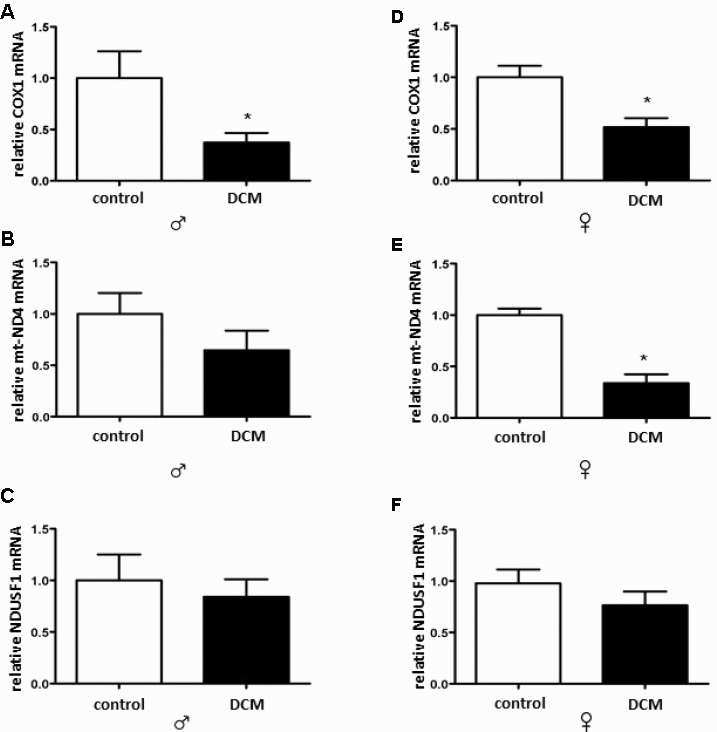
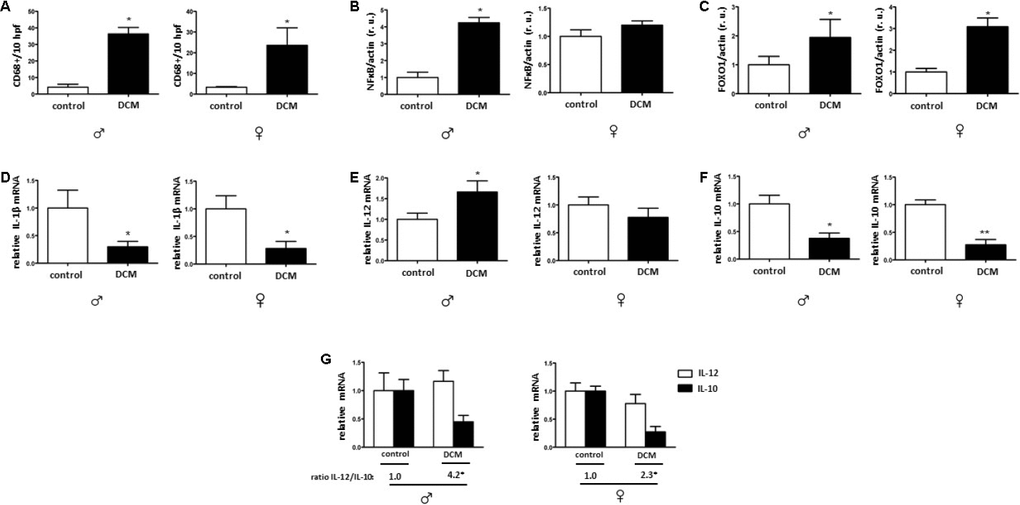
In the current study, we investigated age- and sex-related alterations in metabolic, mitochondrial, and inflammatory markers in DCM patients. The main findings are as follows: 1) In older patients, DCM is associated with significant Sirt1 downregulation in both sexes, while pAMPK/AMPK ratio upregulation only appeared in males; 2) expression of several mitochondrial proteins (TOM40, TIM23, and Sirt3), including a main mitochondrial antioxidant SOD2, and mRNA (cox1, nd4) is reduced in older but not in younger DCM patients; 3) expression of the inflammatory markers in older hearts with DCM is increased, with a stronger pro-inflammatory response in older males with DCM. Taken together, these findings indicate a disturbance of metabolic sensing, impairment of mitochondrial biogenesis, and increased inflammation in older individuals with DCM in both sex-dependent and -independent manners.
The crucial role of Sirt1 in inflammation, DNA repair, apoptosis, and aging has already been investigated [48, 49]. Its reduced expression and activity have been associated with various pathologies, e.g., diabetes, Alzheimer's disease and coronary artery disease [50–52]. Sirt1 activation by resveratrol tends to decelerate aging processes and the onset of chronic diseases [53–55]. In the present study, the Sirt1 expression was significantly diminished in DCM hearts in older individuals in a sex-independent manner, while no alterations were observed in younger DCM patients. In accordance with our data, consistent reduction of Sirt1 expression was reported in older mice [56] as well as in control hearts of older women. Likewise, Sirt1-deficient mice showed a progressive DCM strongly associated with mitochondrial dysfunction [57]. Furthermore, reduced Sirt1 expression was reported in monocytes of older patients with cardiovascular diseases [52], suggesting that aging directly aggravates cardiac dysfunction via disruption of Sirt1 signaling, promoting mitochondrial dysfunction, apoptosis, and inflammation as a result.
AMPK is a key Sirt1 partner in regulating metabolic activity and inflammation that is activated under metabolic stress [58]. AMPK and SIRT1 may regulate each other and share many common targets [15]. An increasing number of reports emphasize the supporting role of AMPK activity in cardiac metabolic homeostasis [59]. From the other side, persistent AMPK overactivation might be deleterious in some cases, with a chronic AMPK activation appearing to aggravate the pathological damage that strokes and myocardial ischemia cause [13, 60, 61], by, e.g., decreasing fatty acid oxidation [62]. Furthermore, sustained AMPK activation leads to excessive mitochondrial fragmentation and mitophagy, leading to the depletion of functional mitochondria [63]. Here, DCM in older patients was associated with increased phosphorylation of AMPK compared to the control group, whereas in young DCM patients AMPK phosphorylation was markedly reduced. Though diminished AMPK activity in hearts with DCM has been previously observed [61], no age dependence has been demonstrated. In fact, both we and others [13, 32] observed a marked reduction of the cardiac AMPK phosphorylation in older, control individuals. It is tempting to speculate that, with the reduced AMPK phosphorylation in older hearts, additional DCM-related metabolic impairment may trigger the compensatory activation of the AMPK phosphorylation. Fitting in with this view, decreased ATP levels are commonly observed in DCM patients [64].
The disturbance of metabolic homeostasis in older DCM hearts is further highlighted by the reduced mitochondrial biogenesis observed in the present study. Particularly, a reduced Sirt3 expression in older DCM hearts, a main mitochondria-localized deacetylase involved in the regulation of mitochondrial enzymes activity [65, 66], argues for a disturbed mitochondrial function. The pivotal role of Sirt3 in cardiovascular health/diseases is largely accepted and Sirt3 dysfunction is associated with numerous pathologies [67, 68].
Aside from impaired mitochondrial biogenesis and acetylation capacity, numerous mitochondrial abnormalities have been reported in patients with DCM elsewhere [69–71], which may additionally contribute to the mitochondrial dysfunction in older DCM hearts. Mitochondrial dysfunction is typically accompanied by impaired ATP synthesis with enhanced ROS formation [72]. Though we did not measure adenine nucleotide homeostasis in the present study, the enhanced activation of key metabolic sensors, i.e., AMPK, solely in older male DCM hearts suggests the elevation of the AMP/ATP or ADP/ATP ratio. Interestingly, a male-specific downregulation of the main mitochondrial antioxidant enzyme SOD2 was found in older DCM hearts. Altogether, the data suggest that DCM aggravates metabolic and oxidative stress in older hearts in both a sex-dependent and -independent manner.
The results of research conducted thus far suggests that the higher incidence of cardiac diseases, particularly of DCM, in males may be due to the fact that females are protected by higher levels of steroid hormones, 17β-estradiol among others. However, in older, post-menopausal females, the protective effect of 17β-estradiol is lost. In fact, older men have higher 17β-estradiol concentrations in their blood compared to older women [73]. Our recent report [32] showed that cardiac aging is defined by the decline in mitochondrial anti-oxidative defense and a pro-inflammatory shift in females but not in males.
An increased inflammatory response accompanied by decreased mitochondrial function [74, 75] is typical for older hearts.
NFκB promotes inflammation in myocardial tissue in age-related diseases [76]. In accordance with this function, our results showed enhanced NFκB expression in DCM hearts in older males. [27, 77]. Pro-inflammatory mediators are increased in aging under certain physiological conditions [78]. In the present study, the expression of pro-inflammatory factor IL-12 was elevated in older DCM hearts only in males, while a substantial decrease in IL-10 expression was observed in older DCM hearts in a sex-independent manner. A reduced IL-10 expression is a typical fingerprint observed in cardiac aging [32] as well as in aging macrophages [79]; furthermore, IL-10 deficiency promotes a detrimental course of inflammation [80]. The total amount of cardiac macrophages was elevated in DCM patients in this study and accompanied by a shift in the IL-12/IL-10 ratio to IL-12 side, which is a marker of pro-inflammatory macrophages [81].
Our results, in combination with those from other previous studies, suggest an increased inflammatory phenotype in older DCM hearts, which is even more pronounced in males.
In conclusion, the present study revealed that DCM is associated with the downregulation of key metabolic regulator Sirt1, diminished mitochondrial biogenesis and anti-oxidative defense, and an increased inflammatory response in older DCM hearts in both sex-dependent and -independent manners.
Materials and Methods
Human samples
Human lateral left ventricular (LV) wall tissue from patients with idiopathic end-stage DCM was collected during organ transplantation (men=10 and women=10) and from healthy organ donors (men=16 and women=15). The informed consent from all donors or their legal guardians was obtained. The patients with DCM had ejection fractions (EF) <30%. The tissue was frozen in liquid nitrogen immediately after collection and stored at -80° C. The patients were between 19 and 70 years old, while healthy donors were between 17 and 68 years of age. The control (non-diseased) LV samples were divided into 4 groups of young (17-40 years; male: n=7 and female: n=7) and old (50-68 years; male: n=9 and female: n=8) individuals; DCM samples were divided into 4 groups of young (19-40 years; male: n=5 and female: n=5) and old (50-70 years; male: n=5 and female: n=5) individuals.
For DCM: Sample collection and the experimental protocols were approved by the scientific boards at the Heart and Diabetes Centre (HDZ) NRW (21/2013) and at the Charité – Universitätsmedizin Berlin (EA2/158/16). All research was performed in accordance with the guidelines from the relevant regulatory German authorities.
For healthy donors: The scientific board at the Hungarian Ministry of Health (ETT-TUKEB: 4991-0/2010-1018EKU) approved the sample collection and the experimental protocols. All research was performed in accordance with the German and Hungarian regulatory guidelines.
RNA extraction and quantitative real-time PCR
Total RNA from cardiac human tissue was homogenized in RNA-Bee (Amsbio, Abbingdon, UK) and the Phenol/Chloroform (Roth, Karlsruhe, Germany) extraction protocol was used for the RNA isolation. The Caliper LabChip bioanalyzer (Agilent Technologies, Ratingen Germany) was used to analyze the purity of the isolated RNA. Quantitative real-time PCR were performed using the Brilliant SYBR Green qPCR master mix (Applied Biosystems, Foster City, CA, USA). The relative amount of target mRNA was determined using the comparative threshold (Ct) method as previously described [34]. The mRNA contents of target genes were normalized to the expression of hypoxanthine phosphoribosyl transferase (HPRT).
Protein extraction and immunoblotting
LV samples from DCM and control hearts were homogenized in a Laemmli buffer (253mM Tris/HCL pH 6.8, 8% SDS, 40% glycerin, 200mM DTT, 0.4% bromophenol blue). Proteins were quantified using the BCA Assay (Thermo Scientific Pierce Protein Biology, Schwerte, Germany). Equal amounts of total proteins were separated on SDS-polyacrylamide gels and transferred to a nitrocellulose membrane. The membranes were immunoblotted overnight with the following primary antibodies: AMPK (1:2000, Cell Signaling, USA), p-AMPK (1:2000, Thr172, Cell Signaling, USA), PGC-1α (1:1000 Abcam, UK), TOM40 (1:1000, Abcam, UK), TIM 23 (1:5000, BD, USA), Sirt3 (1:1000, Cell Signaling, USA), SOD2 (1:1000, Santa Cruz, USA), catalase (1:1000, Cell Signaling, USA), NFκBp65 (1:200, Santa Cruz, USA) and FOXO1 (1:1000, Cell Signaling, USA). Equal sample loading was confirmed by analysis of actin (1:1000, Santa Cruz, USA), HSP60 (1:1000, Cell Signaling, USA) or Ponceau S staining. Immunoreactive proteins were detected using ECL Plus (GE Healthcare, Buckinghamshire, UK) and quantified with ImageLab (version 5.2.1 build 11, Bio-Rad Laboratories (USA)).
Immunohistochemistry
For immunohistochemistry, 5 μm cryo-sections of human LV were fixed in formalin for 1 hour at room temperature and subjected to a heat-induced epitope retrieval step prior to incubation with anti-CD68 antibody (clone PGM-1, Agilent Technologies, Santa Clara, CA, USA). The detection was performed by the LSAB method applying the Dako REAL™ Detection System (Agilent Technologies, Santa Clara, CA, USA). Nuclei were counterstained with hematoxylin and mounted on slides with glycerol gelatin (both Merck KGaA, Darmstadt, Germany). Negative controls were performed by omitting the primary antibody. Images were acquired using an AxioImager Z1 microscope (Carl Zeiss MicroImaging, Inc.). Positive cells were quantified in 10 high power fields (hpf) (field of vision in x400 original magnification). All evaluations were performed in a blinded manner.
Statistical analysis
The data are given as the mean ± SEM. The GraphPad Prism 5 (GraphPad Software, 2003, San Diego, USA) was used for statistical analysis. The data were evaluated using the non-parametric test (Mann-Whitney test for two independent groups). A simple linear regression analysis was performed with function lm() in R. Statistical significance was accepted when p < 0.05.
Supplementary Materials
Author Contributions
M.B. conceived the project, analyzed the data, prepared the figures, and wrote the main manuscript text. S.P. analyzed the data and wrote the main manuscript text. N.H. performed western blot experiments and analyzed the data. P.B. analyzed data and revised the manuscript. A.A.K. performed the immunohistochemical experiments, analyzed the data, and revised the manuscript. H.M. acquired the human DCM tissue and revised the manuscript. I.B. acquired the human tissue and revised the manuscript. Y.L. analyzed the data, and wrote and revised the manuscript. V.R.Z. generated research funds, initiated, and coordinated the project. All authors commented on the manuscript.
Acknowledgments
We would like to thank Michael Nnaji for revising the manuscript. We would also like to express our gratitude to Jenny Jansen and Arne Kühne for their technical assistance.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
This work was funded by the DZHK (German Centre for Cardiovascular Research; 81Z2100201) (including salaries and consumables). Control cardiac tissue collection and management was supported by the Hungarian National Research, Development and Innovation Office (GINOP-2.3.2-15-2016-00040) and the Ministry of Human Capacities Hungary (20391-3/2018/FEKUSTRAT) to IB. We acknowledge support from the German Research Foundation (DFG) and the Open Access Publication Fund of Charité – Universitätsmedizin Berlin.
References
- 1. Schultheiss HP, Fairweather D, Caforio AL, Escher F, Hershberger RE, Lipshultz SE, Liu PP, Matsumori A, Mazzanti A, McMurray J, Priori SG. Dilated cardiomyopathy. Nat Rev Dis Primers. 2019; 5:32. https://doi.org/10.1038/s41572-019-0084-1 [PubMed]
- 2. Epelman S, Liu PP, Mann DL. Role of innate and adaptive immune mechanisms in cardiac injury and repair. Nat Rev Immunol. 2015; 15:117–29. https://doi.org/10.1038/nri3800 [PubMed]
- 3. Li AH, Liu PP, Villarreal FJ, Garcia RA. Dynamic changes in myocardial matrix and relevance to disease: translational perspectives. Circ Res. 2014; 114:916–27. https://doi.org/10.1161/CIRCRESAHA.114.302819 [PubMed]
- 4. Gude NA, Broughton KM, Firouzi F, Sussman MA. Cardiac ageing: extrinsic and intrinsic factors in cellular renewal and senescence. Nat Rev Cardiol. 2018; 15:523–42. https://doi.org/10.1038/s41569-018-0061-5 [PubMed]
- 5. Strait JB, Lakatta EG. Aging-associated cardiovascular changes and their relationship to heart failure. Heart Fail Clin. 2012; 8:143–64. https://doi.org/10.1016/j.hfc.2011.08.011 [PubMed]
- 6. Cianflone E, Torella M, Chimenti C, De Angelis A, Beltrami AP, Urbanek K, Rota M, Torella D. Adult cardiac stem cell aging: a reversible stochastic phenomenon? Oxid Med Cell Longev. 2019; 2019:5813147. https://doi.org/10.1155/2019/5813147 [PubMed]
- 7. Olivetti G, Melissari M, Capasso JM, Anversa P. Cardiomyopathy of the aging human heart. Myocyte loss and reactive cellular hypertrophy. Circ Res. 1991; 68:1560–68. https://doi.org/10.1161/01.res.68.6.1560 [PubMed]
- 8. Ventura-Clapier R, Moulin M, Piquereau J, Lemaire C, Mericskay M, Veksler V, Garnier A. Mitochondria: a central target for sex differences in pathologies. Clin Sci (Lond). 2017; 131:803–22. https://doi.org/10.1042/CS20160485 [PubMed]
- 9. Arany Z, He H, Lin J, Hoyer K, Handschin C, Toka O, Ahmad F, Matsui T, Chin S, Wu PH, Rybkin II, Shelton JM, Manieri M, et al. Transcriptional coactivator PGC-1 alpha controls the energy state and contractile function of cardiac muscle. Cell Metab. 2005; 1:259–71. https://doi.org/10.1016/j.cmet.2005.03.002 [PubMed]
- 10. Riehle C, Abel ED. PGC-1 proteins and heart failure. Trends Cardiovasc Med. 2012; 22:98–105. https://doi.org/10.1016/j.tcm.2012.07.003 [PubMed]
- 11. Rowe GC, Jiang A, Arany Z. PGC-1 coactivators in cardiac development and disease. Circ Res. 2010; 107:825–38. https://doi.org/10.1161/CIRCRESAHA.110.223818 [PubMed]
- 12. Wenz T. Mitochondria and PGC-1α in aging and age-associated diseases. J Aging Res. 2011; 2011:810619. https://doi.org/10.4061/2011/810619 [PubMed]
- 13. Salminen A, Kaarniranta K. AMP-activated protein kinase (AMPK) controls the aging process via an integrated signaling network. Ageing Res Rev. 2012; 11:230–41. https://doi.org/10.1016/j.arr.2011.12.005 [PubMed]
- 14. Guarente L. The many faces of sirtuins: sirtuins and the warburg effect. Nat Med. 2014; 20:24–25. https://doi.org/10.1038/nm.3438 [PubMed]
- 15. Ruderman NB, Xu XJ, Nelson L, Cacicedo JM, Saha AK, Lan F, Ido Y. AMPK and SIRT1: a long-standing partnership? Am J Physiol Endocrinol Metab. 2010; 298:E751–60. https://doi.org/10.1152/ajpendo.00745.2009 [PubMed]
- 16. Cantó C, Gerhart-Hines Z, Feige JN, Lagouge M, Noriega L, Milne JC, Elliott PJ, Puigserver P, Auwerx J. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature. 2009; 458:1056–60. https://doi.org/10.1038/nature07813 [PubMed]
- 17. Cohen HY, Miller C, Bitterman KJ, Wall NR, Hekking B, Kessler B, Howitz KT, Gorospe M, de Cabo R, Sinclair DA. Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science. 2004; 305:390–92. https://doi.org/10.1126/science.1099196 [PubMed]
- 18. Satoh A, Brace CS, Rensing N, Cliften P, Wozniak DF, Herzog ED, Yamada KA, Imai S. Sirt1 extends life span and delays aging in mice through the regulation of Nk2 homeobox 1 in the DMH and LH. Cell Metab. 2013; 18:416–30. https://doi.org/10.1016/j.cmet.2013.07.013 [PubMed]
- 19. Kayashima Y, Katayanagi Y, Tanaka K, Fukutomi R, Hiramoto S, Imai S. Alkylresorcinols activate SIRT1 and delay ageing in drosophila melanogaster. Sci Rep. 2017; 7:43679. https://doi.org/10.1038/srep43679 [PubMed]
- 20. Satoh A, Imai S. Systemic regulation of mammalian ageing and longevity by brain sirtuins. Nat Commun. 2014; 5:4211. https://doi.org/10.1038/ncomms5211 [PubMed]
- 21. Lee SH, Lee JH, Lee HY, Min KJ. Sirtuin signaling in cellular senescence and aging. BMB Rep. 2019; 52:24–34. https://doi.org/10.5483/BMBRep.2019.52.1.290 [PubMed]
- 22. Mouchiroud L, Houtkooper RH, Moullan N, Katsyuba E, Ryu D, Cantó C, Mottis A, Jo YS, Viswanathan M, Schoonjans K, Guarente L, Auwerx J. The NAD(+)/Sirtuin pathway modulates longevity through activation of mitochondrial UPR and FOXO signaling. Cell. 2013; 154:430–41. https://doi.org/10.1016/j.cell.2013.06.016 [PubMed]
- 23. Chang HC, Guarente L. SIRT1 mediates central circadian control in the SCN by a mechanism that decays with aging. Cell. 2013; 153:1448–60. https://doi.org/10.1016/j.cell.2013.05.027 [PubMed]
- 24. Novak I. Mitophagy: a complex mechanism of mitochondrial removal. Antioxid Redox Signal. 2012; 17:794–802. https://doi.org/10.1089/ars.2011.4407 [PubMed]
- 25. Sun N, Youle RJ, Finkel T. The Mitochondrial Basis of Aging. Mol Cell. 2016; 61:654–666. https://doi.org/10.1016/j.molcel.2016.01.028 [PubMed]
- 26. Picca A, Mankowski RT, Burman JL, Donisi L, Kim JS, Marzetti E, Leeuwenburgh C. Mitochondrial quality control mechanisms as molecular targets in cardiac ageing. Nat Rev Cardiol. 2018; 15:543–54. https://doi.org/10.1038/s41569-018-0059-z [PubMed]
- 27. Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, Frye RA, Mayo MW. Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J. 2004; 23:2369–80. https://doi.org/10.1038/sj.emboj.7600244 [PubMed]
- 28. Gallí M, Van Gool F, Leo O. Sirtuins and inflammation: friends or foes? Biochem Pharmacol. 2011; 81:569–76. https://doi.org/10.1016/j.bcp.2010.12.010 [PubMed]
- 29. Mendes KL, Lelis DF, Santos SH. Nuclear sirtuins and inflammatory signaling pathways. Cytokine Growth Factor Rev. 2017; 38:98–105. https://doi.org/10.1016/j.cytogfr.2017.11.001 [PubMed]
- 30. Parodi-Rullán RM, Chapa-Dubocq XR, Javadov S. Acetylation of mitochondrial proteins in the heart: the role of SIRT3. Front Physiol. 2018; 9:1094. https://doi.org/10.3389/fphys.2018.01094 [PubMed]
- 31. Brown K, Xie S, Qiu X, Mohrin M, Shin J, Liu Y, Zhang D, Scadden DT, Chen D. SIRT3 reverses aging-associated degeneration. Cell Rep. 2013; 3:319–27. https://doi.org/10.1016/j.celrep.2013.01.005 [PubMed]
- 32. Barcena de Arellano ML, Pozdniakova S, Kühl AA, Baczko I, Ladilov Y, Regitz-Zagrosek V. Sex differences in the aging human heart: decreased sirtuins, pro-inflammatory shift and reduced anti-oxidative defense. Aging (Albany NY). 2019; 11:1918–33. https://doi.org/10.18632/aging.101881 [PubMed]
- 33. Koyama T, Kume S, Koya D, Araki S, Isshiki K, Chin-Kanasaki M, Sugimoto T, Haneda M, Sugaya T, Kashiwagi A, Maegawa H, Uzu T. SIRT3 attenuates palmitate-induced ROS production and inflammation in proximal tubular cells. Free Radic Biol Med. 2011; 51:1258–67. https://doi.org/10.1016/j.freeradbiomed.2011.05.028 [PubMed]
- 34. Barger JL, Anderson RM, Newton MA, da Silva C, Vann JA, Pugh TD, Someya S, Prolla TA, Weindruch R. A conserved transcriptional signature of delayed aging and reduced disease vulnerability is partially mediated by SIRT3. PLoS One. 2015; 10:e0120738. https://doi.org/10.1371/journal.pone.0120738 [PubMed]
- 35. Fairweather D, Cooper LT
Jr , Blauwet LA. Sex and gender differences in myocarditis and dilated cardiomyopathy. Curr Probl Cardiol. 2013; 38:7–46. https://doi.org/10.1016/j.cpcardiol.2012.07.003 [PubMed] - 36. Coronado MJ, Brandt JE, Kim E, Bucek A, Bedja D, Abston ED, Shin J, Gabrielson KL, Mitzner W, Fairweather D. Testosterone and interleukin-1β increase cardiac remodeling during coxsackievirus B3 myocarditis via serpin A 3n. Am J Physiol Heart Circ Physiol. 2012; 302:H1726–36. https://doi.org/10.1152/ajpheart.00783.2011 [PubMed]
- 37. Cocker MS, Abdel-Aty H, Strohm O, Friedrich MG. Age and gender effects on the extent of myocardial involvement in acute myocarditis: a cardiovascular magnetic resonance study. Heart. 2009; 95:1925–30. https://doi.org/10.1136/hrt.2008.164061 [PubMed]
- 38. Halliday BP, Gulati A, Ali A, Newsome S, Lota A, Tayal U, Vassiliou VS, Arzanauskaite M, Izgi C, Krishnathasan K, Singhal A, Chiew K, Gregson J, et al. Sex- and age-based differences in the natural history and outcome of dilated cardiomyopathy. Eur J Heart Fail. 2018; 20:1392–400. https://doi.org/10.1002/ejhf.1216 [PubMed]
- 39. Kessler EL, Rivaud MR, Vos MA, van Veen TA. Sex-specific influence on cardiac structural remodeling and therapy in cardiovascular disease. Biol Sex Differ. 2019; 10:7. https://doi.org/10.1186/s13293-019-0223-0 [PubMed]
- 40. Li S, Gupte AA. The role of estrogen in cardiac metabolism and diastolic function. Methodist Debakey Cardiovasc J. 2017; 13:4–8. https://doi.org/10.14797/mdcj-13-1-4 [PubMed]
- 41. Rosca MG, Hoppel CL. Mitochondrial dysfunction in heart failure. Heart Fail Rev. 2013; 18:607–22. https://doi.org/10.1007/s10741-012-9340-0 [PubMed]
- 42. Higashida K, Kim SH, Jung SR, Asaka M, Holloszy JO, Han DH. Effects of resveratrol and SIRT1 on PGC-1α activity and mitochondrial biogenesis: a reevaluation. PLoS Biol. 2013; 11:e1001603. https://doi.org/10.1371/journal.pbio.1001603 [PubMed]
- 43. Amat R, Planavila A, Chen SL, Iglesias R, Giralt M, Villarroya F. SIRT1 controls the transcription of the peroxisome proliferator-activated receptor-gamma co-activator-1alpha (PGC-1alpha) gene in skeletal muscle through the PGC-1alpha autoregulatory loop and interaction with MyoD. J Biol Chem. 2009; 284:21872–80. https://doi.org/10.1074/jbc.M109.022749 [PubMed]
- 44. Toba H, de Castro Brás LE, Baicu CF, Zile MR, Lindsey ML, Bradshaw AD. Secreted protein acidic and rich in cysteine facilitates age-related cardiac inflammation and macrophage M1 polarization. Am J Physiol Cell Physiol. 2015; 308:C972–82. https://doi.org/10.1152/ajpcell.00402.2014 [PubMed]
- 45. Akkafa F, Halil Altiparmak I, Erkus ME, Aksoy N, Kaya C, Ozer A, Sezen H, Oztuzcu S, Koyuncu I, Umurhan B. Reduced SIRT1 expression correlates with enhanced oxidative stress in compensated and decompensated heart failure. Redox Biol. 2015; 6:169–73. https://doi.org/10.1016/j.redox.2015.07.011 [PubMed]
- 46. Lamkanfi M, Dixit VM. Mechanisms and functions of inflammasomes. Cell. 2014; 157:1013–22. https://doi.org/10.1016/j.cell.2014.04.007 [PubMed]
- 47. Chung S, Lee TJ, Reader BF, Kim JY, Lee YG, Park GY, Karpurapu M, Ballinger MN, Qian F, Rusu L, Chung HY, Unterman TG, Croce CM, Christman JW. FoxO1 regulates allergic asthmatic inflammation through regulating polarization of the macrophage inflammatory phenotype. Oncotarget. 2016; 7:17532–46. https://doi.org/10.18632/oncotarget.8162 [PubMed]
- 48. Anastasiou D, Krek W. SIRT1: linking adaptive cellular responses to aging-associated changes in organismal physiology. Physiology (Bethesda). 2006; 21:404–10. https://doi.org/10.1152/physiol.00031.2006 [PubMed]
- 49. Qin W, Yang T, Ho L, Zhao Z, Wang J, Chen L, Zhao W, Thiyagarajan M, MacGrogan D, Rodgers JT, Puigserver P, Sadoshima J, Deng H, et al. Neuronal SIRT1 activation as a novel mechanism underlying the prevention of Alzheimer disease amyloid neuropathology by calorie restriction. J Biol Chem. 2006; 281:21745–54. https://doi.org/10.1074/jbc.M602909200 [PubMed]
- 50. Aditya R, Kiran AR, Varma DS, Vemuri R, Gundamaraju R. A review on SIRtuins in diabetes. Curr Pharm Des. 2017; 23:2299–307. https://doi.org/10.2174/1381612823666170125153334 [PubMed]
- 51. Lutz MI, Milenkovic I, Regelsberger G, Kovacs GG. Distinct patterns of sirtuin expression during progression of Alzheimer’s disease. Neuromolecular Med. 2014; 16:405–14. https://doi.org/10.1007/s12017-014-8288-8 [PubMed]
- 52. Chan SH, Hung CH, Shih JY, Chu PM, Cheng YH, Lin HC, Tsai KL. SIRT1 inhibition causes oxidative stress and inflammation in patients with coronary artery disease. Redox Biol. 2017; 13:301–09. https://doi.org/10.1016/j.redox.2017.05.027 [PubMed]
- 53. Dasgupta B, Milbrandt J. Resveratrol stimulates AMP kinase activity in neurons. Proc Natl Acad Sci USA. 2007; 104:7217–22. https://doi.org/10.1073/pnas.0610068104 [PubMed]
- 54. Wood JG, Rogina B, Lavu S, Howitz K, Helfand SL, Tatar M, Sinclair D. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature. 2004; 430:686–89. https://doi.org/10.1038/nature02789 [PubMed]
- 55. Zhang J. Resveratrol inhibits insulin responses in a SirT1-independent pathway. Biochem J. 2006; 397:519–27. https://doi.org/10.1042/BJ20050977 [PubMed]
- 56. Lu TM, Tsai JY, Chen YC, Huang CY, Hsu HL, Weng CF, Shih CC, Hsu CP. Downregulation of Sirt1 as aging change in advanced heart failure. J Biomed Sci. 2014; 21:57. https://doi.org/10.1186/1423-0127-21-57 [PubMed]
- 57. Planavila A, Dominguez E, Navarro M, Vinciguerra M, Iglesias R, Giralt M, Lope-Piedrafita S, Ruberte J, Villarroya F. Dilated cardiomyopathy and mitochondrial dysfunction in Sirt1-deficient mice: a role for Sirt1-Mef2 in adult heart. J Mol Cell Cardiol. 2012; 53:521–31. https://doi.org/10.1016/j.yjmcc.2012.07.019 [PubMed]
- 58. Steinberg GR, Kemp BE. AMPK in health and disease. Physiol Rev. 2009; 89:1025–78. https://doi.org/10.1152/physrev.00011.2008 [PubMed]
- 59. Costantino S, Paneni F, Cosentino F. Ageing, metabolism and cardiovascular disease. J Physiol. 2016; 594:2061–73. https://doi.org/10.1113/JP270538 [PubMed]
- 60. McCullough LD, Zeng Z, Li H, Landree LE, McFadden J, Ronnett GV. Pharmacological inhibition of AMP-activated protein kinase provides neuroprotection in stroke. J Biol Chem. 2005; 280:20493–502. https://doi.org/10.1074/jbc.M409985200 [PubMed]
- 61. Dyck JR, Lopaschuk GD. AMPK alterations in cardiac physiology and pathology: enemy or ally? J Physiol. 2006; 574:95–112. https://doi.org/10.1113/jphysiol.2006.109389 [PubMed]
- 62. Tian R, Musi N, D'Agostino J, Hirshman MF, Goodyear LJ. Increased adenosine monophosphate-activated protein kinase activity in rat hearts with pressure-overload hypertrophy. Circulation. 2001; 104:1664–9. https://doi.org/10.1161/hc4001.097183 [PubMed]
- 63. Toyama EQ, Herzig S, Courchet J, Lewis TL
Jr , Losón OC, Hellberg K, Young NP, Chen H, Polleux F, Chan DC, Shaw RJ. Metabolism. Amp-activated protein kinase mediates mitochondrial fission in response to energy stress. Science. 2016; 351:275–81. https://doi.org/10.1126/science.aab4138 [PubMed] - 64. Beer M, Seyfarth T, Sandstede J, Landschütz W, Lipke C, Köstler H, von Kienlin M, Harre K, Hahn D, Neubauer S. Absolute concentrations of high-energy phosphate metabolites in normal, hypertrophied, and failing human myocardium measured noninvasively with (31)P-SLOOP magnetic resonance spectroscopy. J Am Coll Cardiol. 2002; 40:1267–74. https://doi.org/10.1016/s0735-1097(02)02160-5 [PubMed]
- 65. Kanwal A. Functional and therapeutic potential of mitochondrial SIRT3 deacetylase in disease conditions. Expert Rev Clin Pharmacol. 2018; 11:1151–55. https://doi.org/10.1080/17512433.2018.1546119 [PubMed]
- 66. Sun W, Liu C, Chen Q, Liu N, Yan Y, Liu B. SIRT3: a new regulator of cardiovascular diseases. Oxid Med Cell Longev. 2018; 2018:7293861. https://doi.org/10.1155/2018/7293861 [PubMed]
- 67. Winnik S, Auwerx J, Sinclair DA, Matter CM. Protective effects of sirtuins in cardiovascular diseases: from bench to bedside. Eur Heart J. 2015; 36:3404–12. https://doi.org/10.1093/eurheartj/ehv290 [PubMed]
- 68. Hirschey MD, Shimazu T, Jing E, Grueter CA, Collins AM, Aouizerat B, Stančáková A, Goetzman E, Lam MM, Schwer B, Stevens RD, Muehlbauer MJ, Kakar S, et al. SIRT3 deficiency and mitochondrial protein hyperacetylation accelerate the development of the metabolic syndrome. Mol Cell. 2011; 44:177–90. https://doi.org/10.1016/j.molcel.2011.07.019 [PubMed]
- 69. El-Hattab AW, Scaglia F. Mitochondrial cardiomyopathies. Front Cardiovasc Med. 2016; 3:25. https://doi.org/10.3389/fcvm.2016.00025 [PubMed]
- 70. Marín-García J, Goldenthal MJ. Mitochondrial centrality in heart failure. Heart Fail Rev. 2008; 13:137–50. https://doi.org/10.1007/s10741-007-9079-1 [PubMed]
- 71. Bonora M, Wieckowski MR, Sinclair DA, Kroemer G, Pinton P, Galluzzi L. Targeting mitochondria for cardiovascular disorders: therapeutic potential and obstacles. Nat Rev Cardiol. 2019; 16:33–55. https://doi.org/10.1038/s41569-018-0074-0 [PubMed]
- 72. Braidy N, Guillemin GJ, Mansour H, Chan-Ling T, Poljak A, Grant R. Age related changes in NAD+ metabolism oxidative stress and Sirt1 activity in wistar rats. PLoS One. 2011; 6:e19194. https://doi.org/10.1371/journal.pone.0019194 [PubMed]
- 73. Sharma AN, Aoun P, Wigham JR, Weist SM, Veldhuis JD. Estradiol, but not testosterone, heightens cortisol-mediated negative feedback on pulsatile ACTH secretion and ACTH approximate entropy in unstressed older men and women. Am J Physiol Regul Integr Comp Physiol. 2014; 306:R627–35. https://doi.org/10.1152/ajpregu.00551.2013 [PubMed]
- 74. Zahn JM, Sonu R, Vogel H, Crane E, Mazan-Mamczarz K, Rabkin R, Davis RW, Becker KG, Owen AB, Kim SK. Transcriptional profiling of aging in human muscle reveals a common aging signature. PLoS Genet. 2006; 2:e115. https://doi.org/10.1371/journal.pgen.0020115.eor [PubMed]
- 75. Zahn JM, Poosala S, Owen AB, Ingram DK, Lustig A, Carter A, Weeraratna AT, Taub DD, Gorospe M, Mazan-Mamczarz K, Lakatta EG, Boheler KR, Xu X, et al. AGEMAP: a gene expression database for aging in mice. PLoS Genet. 2007; 3:e201. https://doi.org/10.1371/journal.pgen.0030201 [PubMed]
- 76. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013; 153:1194–217. https://doi.org/10.1016/j.cell.2013.05.039 [PubMed]
- 77. Ghisays F, Brace CS, Yackly SM, Kwon HJ, Mills KF, Kashentseva E, Dmitriev IP, Curiel DT, Imai SI, Ellenberger T. The N-terminal domain of SIRT1 is a positive regulator of endogenous SIRT1-dependent deacetylation and transcriptional outputs. Cell Rep. 2015; 10:1665–73. https://doi.org/10.1016/j.celrep.2015.02.036 [PubMed]
- 78. Singh T, Newman AB. Inflammatory markers in population studies of aging. Ageing Res Rev. 2011; 10:319–29. https://doi.org/10.1016/j.arr.2010.11.002 [PubMed]
- 79. Zhang B, Bailey WM, Braun KJ, Gensel JC. Age decreases macrophage IL-10 expression: implications for functional recovery and tissue repair in spinal cord injury. Exp Neurol. 2015; 273:83–91. https://doi.org/10.1016/j.expneurol.2015.08.001 [PubMed]
- 80. Genovese T, Esposito E, Mazzon E, Di Paola R, Caminiti R, Bramanti P, Cappelani A, Cuzzocrea S. Absence of endogenous interleukin-10 enhances secondary inflammatory process after spinal cord compression injury in mice. J Neurochem. 2009; 108:1360–72. https://doi.org/10.1111/j.1471-4159.2009.05899.x [PubMed]
- 81. Edwards JP, Zhang X, Frauwirth KA, Mosser DM. Biochemical and functional characterization of three activated macrophage populations. J Leukoc Biol. 2006; 80:1298–307. https://doi.org/10.1189/jlb.0406249 [PubMed]