Introduction
PIAS1, named as protein inhibitor of the activated STAT1, which is one of E3-type small ubiquitin-like modifier (SUMO) ligases and plays important roles in diverse cellular pathways, such as the STAT, p53 and steroid hormone signaling pathways. Based on the results of in vivo and in vitro experiments, PIAS1 was reported to regulate innate immune responses mediated by the interferon (IFN)-gamma- or -beta-inducible genes by inhibiting the STAT pathway [1]. The STAT1 protein is an essential transcription factor functioning in inflammatory activation in Alzheimer’s disease (AD) brains [2–5]. PIAS1 may function as an E3 ligase for ligand-activated nuclear receptor peroxisome proliferator-activated receptor (PPAR) gamma, which is modified by SUMO-1 and is a potential target for apoptosis-induction therapy in cancer cells [6]. Based on a previous study of Pias1-/- cells that employed microarray analysis, PIAS1 was also identified as a novel negative regulator of NF-κB that is associated with immunity and homeostasis in a number of human illnesses, such as chronic inflammatory diseases and cancer [7–9]. PIAS1 acts as one of the positive transcriptional coregulators of GATA-3, and its molecular interaction is reported to be important for Th2 immune responses by regulating IL-13 [10]. Two members of the PIAS family proteins, PIAS1 and PIAS3, were demonstrated to interact with TATA-binding protein (TBP) [11]. It has been showed that PIAS1 associates with protein-tyrosine phosphatase 1B (PTP1B) in mammalian fibroblasts and catalyzes sumoylation of PTP1B, thereby regulating metabolism and cell proliferation [12]. PIAS1 can promote the proliferation of fibroblasts, smooth muscle cells, and intestinal epithelial cells by interacting with the Kruppel-like factor 5 (KLF5) and enhancing its ability [13]. PIAS1 was reported to confer DNA-binding specificity to the homeoprotein Msx1 in regulating myoblast differentiation through repression of the myogenic regulatory gene MyoD [14]. An epigenetic mechanism has been elucidated that PIAS1 might recruit DNA methyltransferases and heterochromatin protein 1 by binding to the Foxp3 promoter during the natural regulatory T cell differentiation [15]. It is reported that the suppression of PIAS1 abolished the ability of arsenic trioxide, an effective treatment of acute promyelocytic leukemia (APL), to trigger apoptosis in APL cells [16]. During the adipogenesis, PIAS1 plays a dynamic role by promoting the SUMOylation of CCAAT/enhancer-binding protein beta (C/EBPbeta) [17]. The epigenetic pathway associated with PIAS1 may play important roles in regulating the hematopoietic stem cells self-renewal and differentiation [18]. A critical function of PIAS1 in controlling insulin sensitivity by inhibiting the inflammatory cascade has also been observed in adipose tissue [19]. The phosphorylation of PIAS1 was reported to mediate SUMOylation of Elk-1, which may function in promoting neuronal survival in APP/PS1 mice [20].
PIAS1 may serve to regulate the accumulation of Huntingtin (HTT) proteins and its modulation in neurons, which can alter Huntington's disease (HD)-associated phenotypes in vivo [21, 22]. However, the pathological and molecular functions of PIAS1 in other neurodegenerative disorders has not been studied, especially for AD. Therefore, it is particularly important to explore the function of PIAS1 in AD and analyze its molecular regulation mechanism.
Results and Discussion
Expression and regulation of PIAS1 in AD and HT-22 cells
Based on analyzing the differences of the PIAS1 gene expression levels in four human brain regions between the normal controls and AD patients, the expression of PIAS1 was found to be lower in both the inferior frontal gyrus (p = 0.0342) and parahippocampal gyrus (p = 0.0882) in AD samples. In the frontal cortex, the expression of PIAS1 was detected to be significantly higher (p = 0.0315) in AD samples. No significant difference was identified between the control and AD samples in the superior temporal gyrus. These results are presented in Figure 1.
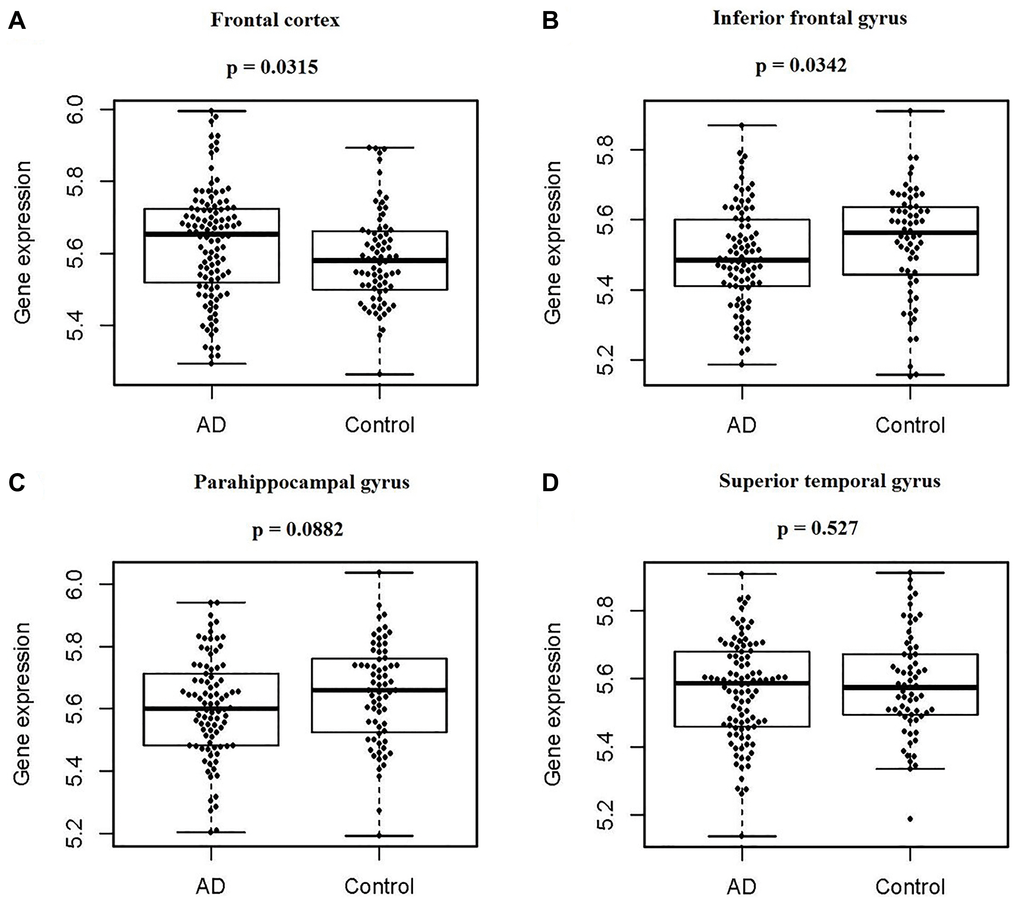
Figure 1. PIAS1 gene expression in four regions of human brains between normal controls and AD patients. Based on the Synapse database, the difference in PIAS1 gene expression in four regions of human brains between normal controls and AD patients was determined by using the Wilcoxon signed-rank test. (A) For the frontal cortex, there were 111 cases and 76 controls, and PIAS expression was significantly higher in AD patients, with a p value of 0.0315. (B) For the inferior frontal gyrus, there were 90 cases and 64 controls, and PIAS expression was significantly lower in AD patients, with a p value of 0.0342. (C) For the parahippocampal gyrus, there were 90 cases and 68 controls, and PIAS expression was not significantly changed, with a p value of 0.0882. (D) For the superior temporal gyrus, there were 102 cases and 65 controls, and PIAS expression was not significantly changed, with a p value of 0.527.
By transfecting the recombinant plasmid, the stable HT-22 cells under the overexpression of Pias1 (Pias1/+) was established. The qRT-PCR results showed that through the over expression experiment, we have significantly increased the expression of Pias1 (Figure 2A). According to the results of Western blotting (WB), the protein expression of PIAS1 was also found to be higher in Pias1/+ cells that is labeled as ~ 72 kDa protein (Figure 2B). The CCK-8 assay results showed that the proliferation of HT-22 cells was enhanced in Pias1/+ cells (Figure 2C). The neurite outgrowth of HT-22 cells could also be enhanced by the overexpression of Pias1. In a functional study of PIAS1 in the regulation of cell proliferation in human prostate cancer (HPC), PIAS1 was reported to be increased in HPC and to enhance cell proliferation through inhibition of cyclin-dependent kinase inhibitor 1A (P21) [23]. The activation of the PIAS1-modulated Smad2/4 complex was demonstrated to mediate zinc-induced apoptosis of prostate cancer cells [24]. PIAS1 has promotive effects on cell proliferation by upregulating the activity of KLF5 in various cell types [13].
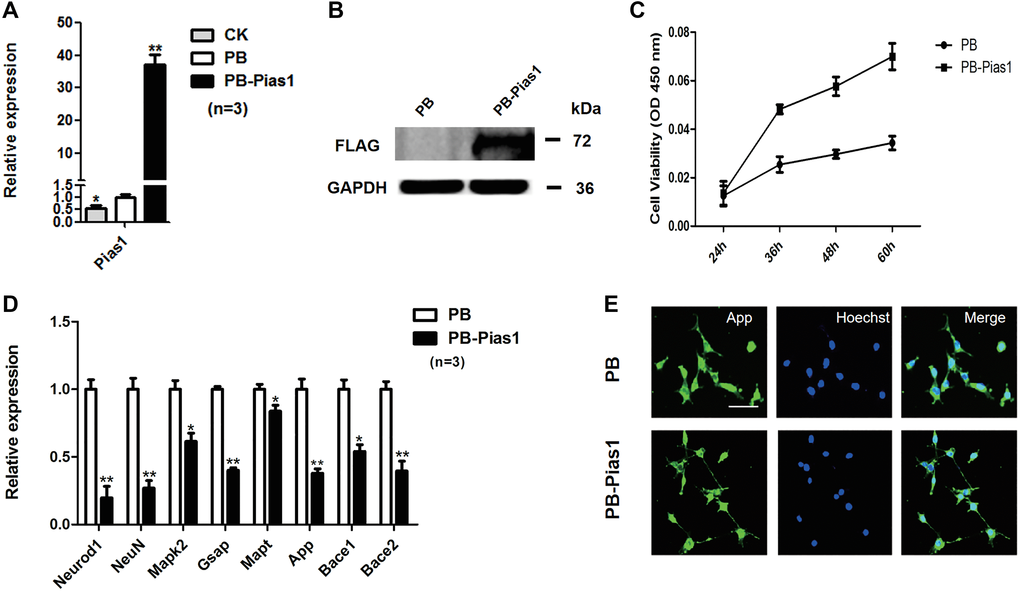
Figure 2. Overexpression of Pias1 regulates AD marker gene expression and cell proliferation in HT-22 cells. (A) qRT-PCR analysis of Pias1 mRNA expression in the hippocampal neuronal HT22 cells transfected with the indicated transgenes. Data represent the mean ± SD of three biological replicates. *p < 0.05, **p < 0.01 versus PB vector. The control check (CK) group represents the baseline expression of Pias1 in HT-22 cells. (B) Western blot analysis of PIAS1 in the hippocampal neuronal HT22 cells with stable Pias1 transgenic expression. (C) Cell proliferation was determined by CCK-8 assays in HT-22 cells transfected with PiggyBac vector (PB) or PB-Pias1. Data represent the mean ± SD. *p < 0.05, **p < 0.01. (D) qRT-PCR analysis of Neurod1, NeuN, Mapk2, Gsap, Mapt, App, Bace1 and Bace2 expression levels in PB and PB-Pias1 hippocampal neuronal HT22 cells. Data are presented as the mean ± SD of three independent experiments. *p < 0.05, **p < 0.01. (E) Immunofluorescence staining of APP in the hippocampal neuronal HT22 cells overexpressing PB-Pias1. Bar: 100 μm.
To explore the functional roles of PIAS1 in AD, some marker genes such as neuron-specific nuclear proteins including neuronal differentiation 1 (Neurod1) and NeuN encoded by FOX3, mitogen-activated protein kinase 2 (Mapk2), gamma-secretase activating protein (Gsap), microtubule associated protein tau (Mapt), amyloid beta precursor protein (App), beta-site APP-cleaving enzyme 1 (Bace1) and beta-site APP-cleaving enzyme 2 (Bace2) were investigated to determine their expression changes in response to overexpression of Pias1. All of these genes expressions were shown to be significantly decreased in the Pias1/+ group (Figure 2D). Moreover, according to the immunofluorescence analysis, it was showed that the axonal outgrowth of HT-22 cells could be increased by the overexpression of Pias1 (Figure 2E). These results suggested that Pias1 might have potential functions in the regulation of AD and neuronal development.
Differentially expressed genes analysis in response to Pias1 overexpression in HT-22 cells
To screen the downstream target genes in HT-22 cells by overexpressing Pias1, we performed RNA-seq to obtain the transcriptome data of two cell types, WT cells and Pias1/+ cells. The sequencing data quality is shown in Table 1. Our data were of high quality, exhibiting a Q20 value greater than 95% and a Q30 value greater than 90%. Based on the data preprocessing and transcriptome assembly, a total of 21,862 annotated transcripts were identified in this study. According to the differentially expressed genes (DEGs) analysis, there were 285 significantly dysregulated genes, including 79 upregulated genes (FC > 2, p < 0.05) and 206 downregulated genes (FC < 1/2, p < 0.05), as determined by the comparison of the Pias1/+ group to the WT group. The details of the up- and downregulated DEGs are shown in Supplementary Table 1. The PCA of sequencing samples, as well as the distribution and clustering of DEGs, are shown in Figure 3.
Table 1. The quality control of RNA-seq data.
| Sample | Raw reads | Clean reads | Clean reads % | Q20 % | Q30 % |
| PB-1 | 48213112 | 45042086 | 93.42 | 97.38 | 92.97 |
| Pias1-1 | 45822220 | 42887740 | 93.59 | 97.36 | 92.89 |
| PB-2 | 50567794 | 47369520 | 93.67 | 97.27 | 92.71 |
| Pias1-2 | 48265814 | 45448864 | 94.16 | 97.40 | 92.97 |
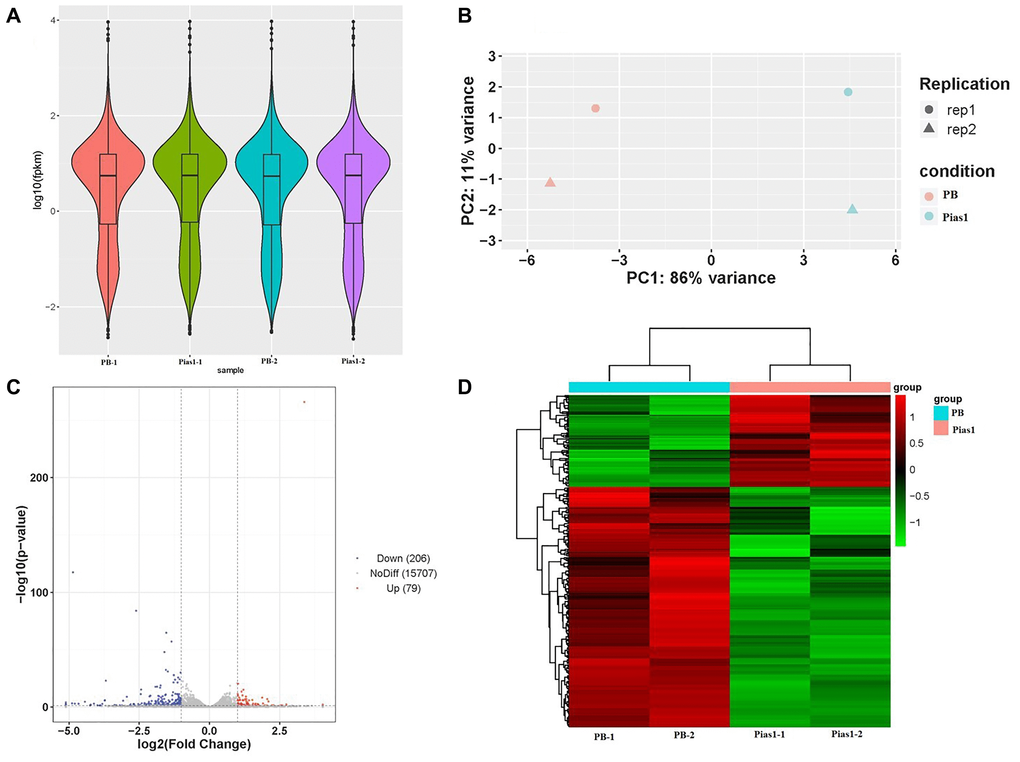
Figure 3. RNA-seq data quality and DEG analysis. (A) The violin plot based on values of log10 (fpkm) showed the whole gene expression distribution of four groups, including PB-1, PB-2, Pias1-1 and Pias1-2. (B) The PCA plot showed how well the four sequencing samples (PB-1, PB-2, Pias1-1 and Pias1-2) are separated from each other. (C) The scatter plot showed the results of DEGs based on the values of log2 (fold change) and -log10 (p value), including 206 downregulated genes, 79 upregulated genes and 15707 genes with no significant differences. (D) The heatmap showed the expression patterns of 285 significantly dysregulated genes with FC greater than 2 and p value less than 0.05, including 79 upregulated genes and 206 downregulated genes, by comparison of the Pias1/+ group to the WT group.
To test the accuracy of screening DEGs by high-throughput sequencing, we selected 11 upregulated genes and 18 downregulated genes for the following qRT-PCR validation. The results showed that 7 out of 11 upregulated genes were significantly identified in the Pias1/+ group, they were A4gnt, Cldn6, Gal3st2, Cnnm1, Rnf151, Ccr7 and Chad (Figure 4A). Human alpha1,4-N-acetylglucosaminyltransferase (A4gnt) was reported to function in O-glycan biosynthesis [25]. The expression of Claudin-6 (Cldn6) in early development was reported to be relevant to definitive endoderm derivatives [26]. In addition, 9 out of 18 downregulated genes were significantly identified in the Pias1/+ group, they were Rflna, Rbm20, Mapk15, Egr1, Fam209, Mettl24, Shcbp1l, Fos and S100b (Figure 4B). We further examined the PIAS1 regulation of cell cycle inhibitors, including p15, p18, p21, p27, p53 and p57. Among these genes, three inhibitors, p53, p18 and p21, were significantly downregulated with the overexpression of Pias1 (Figure 4C).
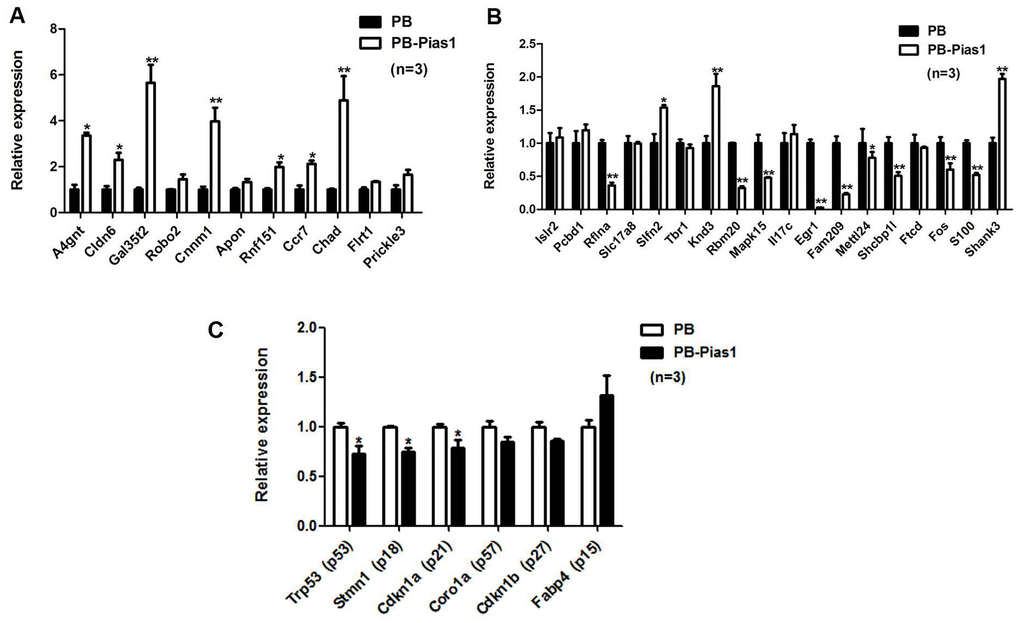
Figure 4. Validation of target gene expression by qRT-PCR. (A) For upregulated DEGs, 11 genes were selected for the following qRT-PCR validation. (B) For downregulated DEGs, 18 genes were selected for the following qRT-PCR validation. (C) The PIAS1 regulation of cell cycle inhibitors, including p15, p18, p21, p27, p53 and p57, was tested by qRT-PCR. Among them, three inhibitors, p53, p18 and p21, were significantly downregulated under the overexpression of Pias1. *p < 0.05, **p < 0.01.
Signaling pathway and gene ontology enrichment under the overexpression of Pias1 in HT-22 cells
Based on GSEA, 30 significantly enriched KEGG pathways were identified to be involved in the regulation of Pias1 in HT-22 cells, which are shown in Figure 5. Among these pathways, the top two significantly enriched pathways were the TNF signaling pathway (FDR=4.25E–08) in terms of environmental information processing and the IL-17 signaling pathway (FDR=4.88E–06) in terms of organismal systems. In addition, other signaling pathways, including the MAPK signaling pathway (FDR=2.34E–02), the ErbB signaling pathway (FDR=3.09E–02) and the NF-kappa B signaling pathway (FDR=4.11E–02), were also identified as being significantly associated with Pias1 regulation. The genes involved in each pathway are shown in Table 2.
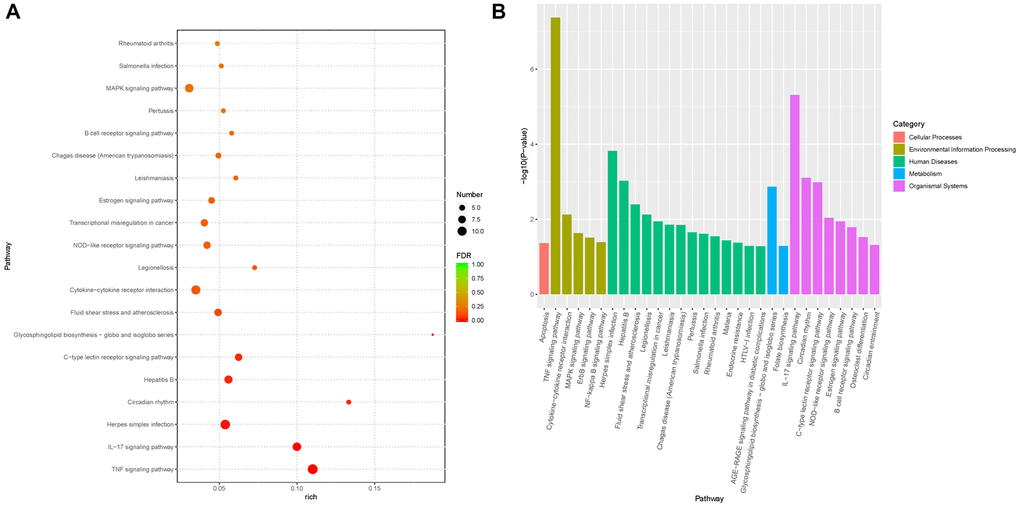
Figure 5. Functional enrichment analysis of DEGs from RNA-seq data. (A) The plot showed the top 20 significantly enriched KEGG pathways of differentially expressed genes (DEGs) associated with Pias1 regulation with an FDR value cutoff of 0.05. Round size represents the gene count of each pathway, and the color represents the significance level. (B) The bar chart showed the categories of significantly enriched pathways. There were five pathway categories including cellular processes (red), environmental information processing (green yellow), human diseases (green), metabolism (blue) and organismal systems (purple).
Table 2. Significantly enriched signaling pathways and dysregulated genes.
| Pathway | FDR | Up_genes | Down_genes |
| TNF signaling pathway | 4.25E–08 | – | Fas, Creb5, Gm5431, Jun, Lif, Junb, Fos, Ptgs2, Cxcl1, Vcam1, Ccl2, Mmp9 |
| IL-17 signaling pathway | 4.88E–06 | – | Mapk15, Fosl1, Il17c, Jun, Fos, Ptgs2, Cxcl1, Ccl2, Mmp9 |
| MAPK signaling pathway | 2.34E–02 | Hspa1l | Fas, Jun, Dusp6, Dusp1, Nr4a1, Fos, Areg, Epha2 |
| ErbB signaling pathway | 3.09E–02 | – | Jun, Nrg1, Areg, Hbegf |
| NF-kappa B signaling pathway | 4.11E–02 | Blnk | Ptgs2, Vcam1, Plau |
Moreover, we have also identified several significantly enriched GO terms, which are listed in Supplementary Table 2. The top three biological process (BP) terms were response to lipid (GO:0033993), regulation of cell population proliferation (GO:0042127) and response to steroid hormone (GO:0048545). The molecular function (MF) terms were primarily related to DNA-binding transcription factor activity, steroid hormone receptor activity and transcription regulator activity. The cellular component (CC) term of the transcription factor AP-1 complex (GO:0035976) was significantly enriched with the target genes Fos, Jun and Junb.
Protein-protein interaction network construction in response to Pias1 overexpression
In addition, a regulatory network of protein-protein interaction in response to the overexpression of Pias1 in HT-22 cells was constructed (Figure 8). A total of 43 encoding DEGs were involved in this network. Our findings provide evidence for a direct link between PIAS1 and NR3C2. The Nr3c2 gene encodes the mineralocorticoid receptor (MLR), which plays important roles in blood pressure control and fluid balance by regulating the amount of sodium in the body [36]. MLR has been determined to be involved in guiding spatial and stimulus-response learning in mice [37]. In humans, PIAS1 was reported to repress MLR ligand-dependent transcriptional activity through the interaction with the N-terminal domain of MLR through SUMO modifications [38]. The upstream gene was Agt (angiotensinogen), acting as a serpin peptidase inhibitor, which is important for the renin-angiotensin system (RAS) [39]. The pathophysiology and morphology of RAS in mice brains may be influenced by the knockout of neurolysin [40]. The cell death and production of reactive oxygen species (ROS) in the mouse brain may be restrained by the ACE2/Ang-(1-7)/Mas pathway activation with angiotensin II overproduction [41].
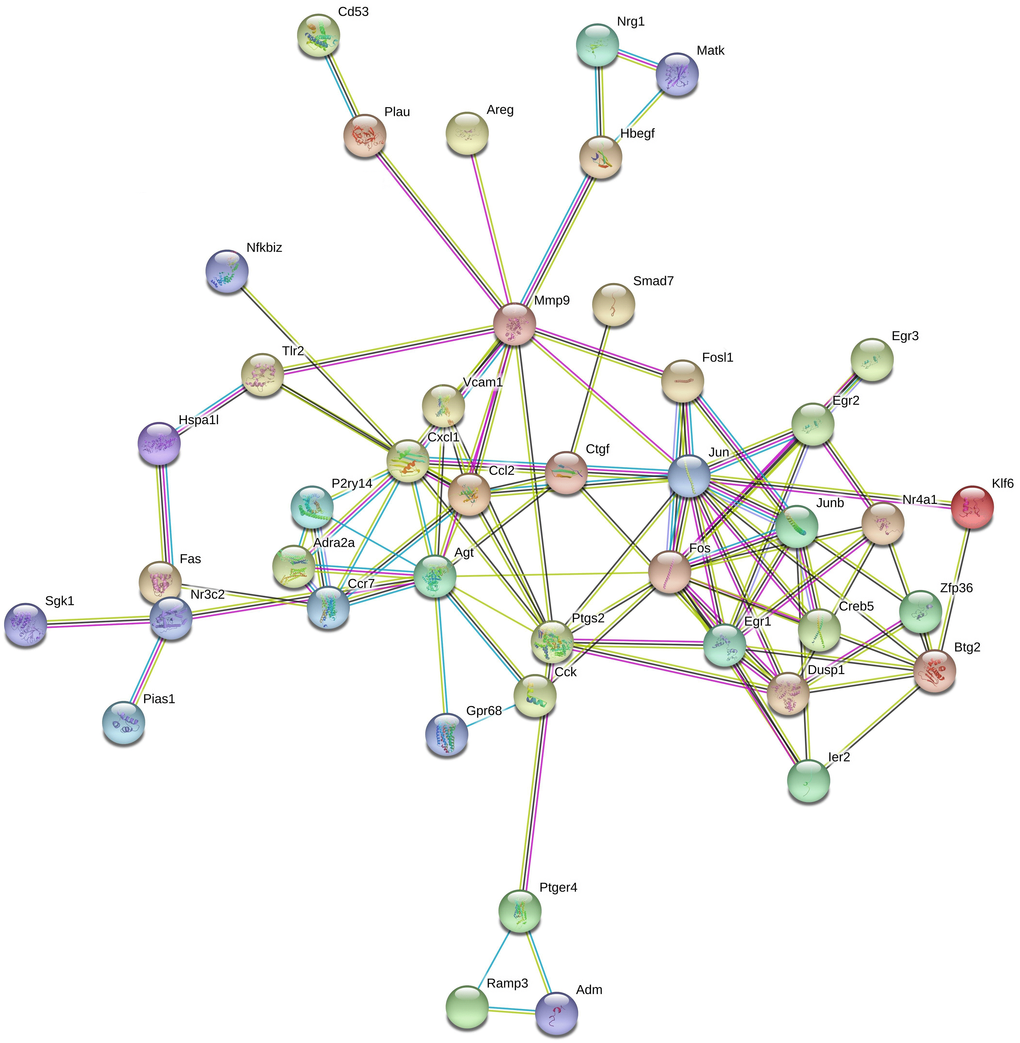
Figure 8. Protein-protein interaction network associated with Pias1 overexpression in HT-22 cells. The protein-protein interaction network was reconstructed by the top 43 DEG proteins in response to Pias1 overexpression in HT-22 cells, such as Nr3c2.
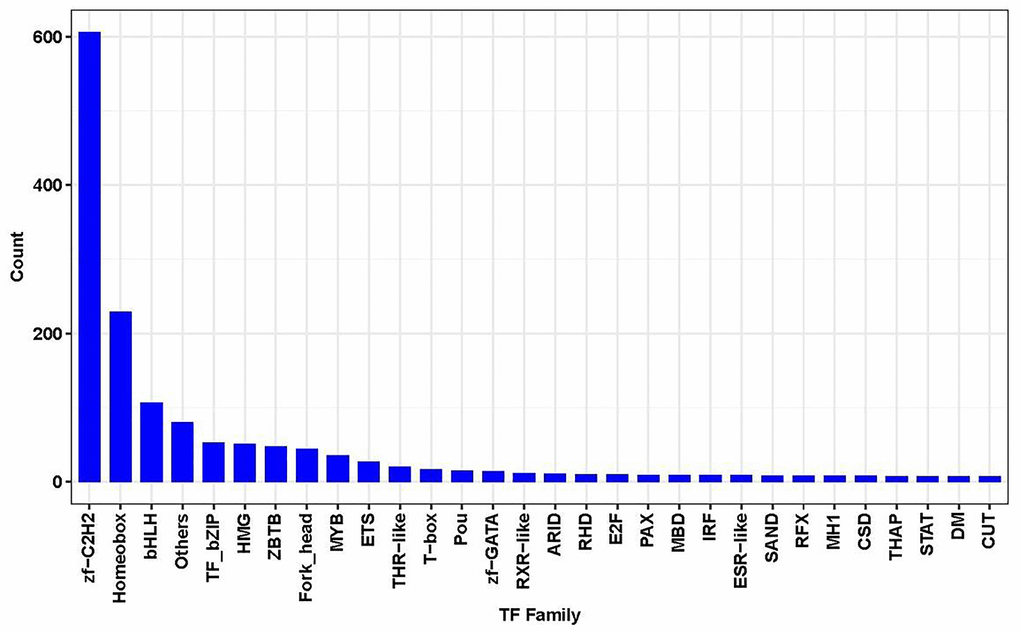
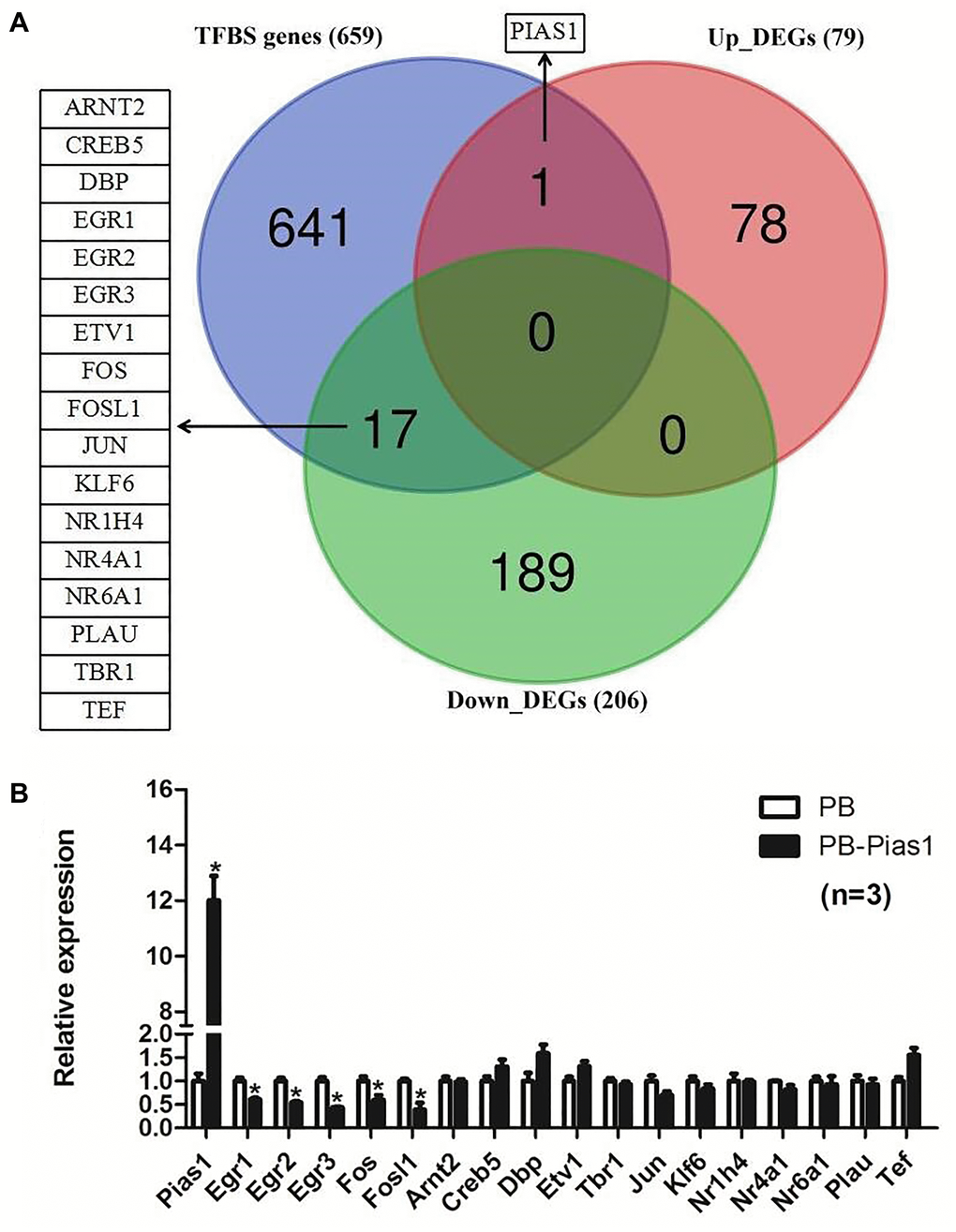
In conclusion, the regulatory mechanism of the Pias1 gene in MHNC was elucidated in our functional genomics study by using RNA-seq. The overexpression of Pias1 was determined to repress several AD markers expressions and to accelerate the proliferation of HT-22 cells. There were 79 upregulated genes and 206 downregulated genes, significantly identified in Pias1/+ cells compared to WT cells through the DEGs analysis. Functional enrichment analysis and TFBS prediction results showed that basic region leucine zipper domain-containing transcription factors including zinc finger C2H2 (zf-C2H2), homeobox and basic/helix-loop-helix (bHLH) in several signaling pathways were significantly involved in PIAS1 regulation in MHNCs. We also reconstructed the regulatory network under PIAS1 overexpression with 43 coding proteins, especially NR3C2, which directly interacted with PIAS1.
Materials and Methods
PIAS1 gene expression in AD brains
The expression profiles of the PIAS1 gene in four brain regions in humans, including the frontal pole (including 111 AD patients and 76 controls), the inferior frontal gyrus (including 90 AD patients and 64 controls), the parahippocampal gyrus (including 90 AD patients and 68 controls), and the superior temporal gyrus (including 102 AD patients and 65 controls), were sourced from the database Synapse [42, 43]. Next, we tested the differences of PIAS1 gene expressions between AD patients and controls in each brain region. The Wilcoxon signed-rank test was used to analyze the significance.
Cell culture and gene overexpression
For the in vitro experiments, we employed one subclone of the mouse hippocampal HT-22 cells, which were purchased from CHI SCIENTIFIC, Shanghai, China. HT-22 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM, Sigma) supplemented with 10% fetal bovine serum (FBS, Gibco BRL, USA), MEM nonessential amino acids (Invitrogen), L-glutamine (Invitrogen), penicillin and streptomycin (Sigma), 0.01 mM β-mercaptoethanol (Invitrogen) and 1000 units/ml LIF (Millipore). Based on the database of GenBank, the reference sequence of Mus musculus Pias1 (NM_019663.3) was obtained. The gene insertion and transfection were performed by using a vector of PiggyBac (PB) in addition with 2 μg transposase using LTX (Invitrogen). The cell selection was continued for seven days by using 2 μg/ml puromycin.
Cell proliferation assay
The experiments of Cell Counting Kit-8 (CCK-8) was performed to assess the proliferation of HT-22 cells based on the manufacturer’s instructions. Cell culture in vitro was conducted in 96-well plates with 100 μl DMEM supplemented with 10% FBS. The cell density was set as 1000 cells per well. Three time points (24 hours, 36 hours, 48 hours and 60 hours after transfection) were selected for observation. The cell viability was determined based on the absorbance at 450 nm by using a microplate reader (Spectramax i3 platform, MD, Austria). All of the experiments were performed in three biological repeats.
Experimental grouping and RNA sequencing
Two groups of the control (named as PB-1 and PB-2) and two groups of the treatment (named as Pias1-1 and Pias1-2) were selected in the following experiments. Total RNA in each group was extracted by using TRIzol, the cDNA was synthesized from 1 μg of total RNA by using reverse transcriptase (Takara). The preparation of the cDNA library and RNA sequencing (RNA-Seq) utilizing the platform of Illumina HiSeq 3000 were performed by Personal Biotechnology (Shanghai, China). On average, 6 G reads per sequencing sample were obtained. Based on the general process of data analysis, our sequencing data was analyzed [44]. The data preprocessing and quality control were performed by using the softwares of Trim Galore and FastQC. Using STAR software, the preprocessing sequence alignment was performed based on the mouse reference genome sequences (Mus_musculus.GRCm.38.83) downloaded from the database of Ensembl. Cufflinks software was used to perform transcript assembly and Cuffcompare software was employed to parsimoniously merge the assembled transfrags. The DEGs analysis was performed by using the package of DESeq2 under R software [45]. The significance threshold is defined as p < 0.05 and |log2 FC| > 1. The functional enrichment analysis was performed by GSEA to identify the significantly enriched KEGG pathways and GO terms, including biological process (BP), molecular function (MF) and cellular component (CC) [46]. The significance cutoff was chosen as FDR < 0.05. The web server SMART version 8.0 was employed to identify and annotate the specific domains of the target proteins [47]. The protein-protein interaction network was reconstructed by using the tool of STRING version 11.0 [48].
Transcription factor binding site (TFBS) prediction
The promoter sequences, including 3000 bases upstream and 50 bases downstream of STAT1, were downloaded from the UCSC Genome Browser [49]. The animal transcription factor database AnimalTFDB 3.0 was employed to predict the transcription factor binding sites (TFBS), as well as predicting the associated TFs and their family information [50]. Next, we compared the above TFBS genes with the DEGs identified by RNA-seq data analysis to explore the overlapping genes that were affected by overexpressing PIAS1.
Quantitative polymerase chain reaction (qPCR)
The primers of target genes were designed by using Primer 5.0. The experiments of qPCR were performed using TransStart Tip Green qPCR SuperMix (Takara). The normalization of each transcript expression was performed by using the 2−ΔΔCt method with the reference gene ribosomal protein L19 (Rpl19). The sequences of above primers are shown in Table 4.
Table 4. The primers of target genes for qPCR.
| Genes | Forward | Reverse |
| Arnt2 | GTTCCAGGACATGCTACCCAT | ATGCCCAGGTCAGCAAAGTC |
| Creb5 | GTTGAAAAACGAGGTGGCCC | AGGGCTGCTCTCTGGACTTA |
| Dbp | CAAGCCCAAAGAACCGGC | AGCGGCGCAAAAAGACTCG |
| Etv1 | TGCAAAAGGTGGCTGGAGAA | ATGTCCGTCTTCAGCAGTGG |
| Fosl1 | GTTCCACCTTGTGCCAAGCA | GGACTGTACTGGGGATAGGC |
| Jun | CAAAACTCCGAGCTGGCATC | TGCGTTAGCATGAGTTGGCA |
| Klf6 | CTTGAAAGCACATCAGCGCA | TCTTGCAAAACGCCACTCAC |
| Nr1h4 | GAGATGGGGATGTTGGCTGA | CAGCGTGCTGCTTCACATTT |
| Nr4a1 | CCGGTGACGTGCAACAATTT | TCCCCTCACCGGGTTTAGAT |
| Nr6a1 | CATCCAGTAGGTCTGTGGAACT | TGGTGAGTGGCCAGAATAGC |
| Plau | TAAAATGCTGTGTGCTGCGG | CCGGGCTTGTTTTTCTCTGC |
| Tef | GAAGCTGATGGAGAACCCCC | GAAGGACTCGCCATCGTAGG |
| Egr1 | AGTGATGAACGCAAGAGGCA | TAGCCACTGGGGATGGGTAA |
| Egr2 | ATCGAAAGCCGTTTCCCTGT | GCGGATTATAAGGGGTGGCA |
| Egr3 | GATGGCTACAGAGAATGTGATGG | TTGGAAGGAGAGTCGAAAGCG |
| Fos | TCGCATCAAGGCCATCATTG | TACGCACGTAGACCAGGATC |
| Tbr1 | GGATTTACGAGCAGGCCAAG | CTATGTCCTTGGCGCAGTTC |
| Fabp4 (p15) | GGGATGGAAAGTCGACCACA | CTTGTGGAAGTCACGCCTTT |
| Stmn1 (p18) | TTCTCAGCCCTCGGTCAAAA | CTGCTTCAAGACTTCCGCCT |
| Cdkn1a (p21) | AGTACTTCCTCTGCCCTGCT | GAATCTTCAGGCCGCTCAGA |
| Cdkn1b (p27) | TCGCAAAACAAAAGGGCCAA | TTACGTCTGGCGTCGAAGG |
| Trp53 (p53) | GCCCATGCTACAGAGGAGTC | TCAGGCCCCACTTTCTTGAC |
| Coro1a (p57) | CTACCTCTGTGGCAAGGGTG | CCATACCACGCTGAGACTCC |
| Rpl19 | GACGGAAGGGCAGGCATATG | TGTGGATGTGCTCCATGAGG |
Immunofluorescence staining
Cells were fixed in 4% paraformaldehyde (PFA fixative) for 20 mins at room temperature, washed in PBS three times, permeabilized for 1 h at 37°C in PBS-T solution(PBS containing 5% BSA and 0.2% Triton X-100), rinsed with PBS three times and incubated overnight at 4°C with the following primary antibodies: APP (25524-1-AP, Proteintech, 1:500). In dark, Alexa Fluor 594 (Invitrogen, 1:1000)-conjugated secondary antibody was used at 1:1000 and nuclei were stained with Hoechst (Invitrogen, 1:5000).
Western blotting
Cells were lysed in pre-cooled RIPA cell buffer (P0013B, Beyotime Biotechnology, China) containing protease inhibitor (DI111-02, TRANSGEN BIOTECH, China, 1: 100). Proteins were separated by a 10% SDS-PAGE gel at RT and electrotransferred to a PVDF membrane in ice-bath. Probing was performed with specific primary antibodies and HRP-conjugated secondary antibodies. The primary antibodies used were PIAS1 (ab2474-1, Abcam, 1:1000) and GAPDH (SC-8035, Santa Cruz, 1:2000). Proteins were detected in super ECL detection reagent (No.180-501, Tanon, China).
Author Contributions
DL and KH conceived and designed the study. JZ, JL, LGL, YC, SY, QB and RP performed the experiments and data analysis. DL and KH wrote the paper. DL, JZ, JL and KH reviewed and edited the manuscript. All authors read and approved the manuscript.
Acknowledgments
We acknowledge financial supports by the National Natural Science Foundation of China (#81570376 and #81870307), the University Special Innovative Research Program of Department of Education of Guangdong Province (#2017KTSCX189), Natural Science Foundation Project of Anhui Province (#1908085MC87) and the Scientific Research Foundation and Academic & Technology Leaders Introduction Project, and 211 Project of Anhui University (#10117700023).
Conflicts of Interest
The authors declare that they have no competing interests.
References
- 1. Liu B, Mink S, Wong KA, Stein N, Getman C, Dempsey PW, Wu H, Shuai K. PIAS1 selectively inhibits interferon-inducible genes and is important in innate immunity. Nat Immunol. 2004; 5:891–98. https://doi.org/10.1038/ni1104 [PubMed]
- 2. Kitamura Y, Shimohama S, Ota T, Matsuoka Y, Nomura Y, Taniguchi T. Alteration of transcription factors NF-kappaB and STAT1 in Alzheimer’s disease brains. Neurosci Lett. 1997; 237:17–20. https://doi.org/10.1016/S0304-3940(97)00797-0 [PubMed]
- 3. Rezai-Zadeh K, Ehrhart J, Bai Y, Sanberg PR, Bickford P, Tan J, Shytle RD. Apigenin and luteolin modulate microglial activation via inhibition of STAT1-induced CD40 expression. J Neuroinflammation. 2008; 5:41. https://doi.org/10.1186/1742-2094-5-41 [PubMed]
- 4. Cho HJ, Kim SK, Jin SM, Hwang EM, Kim YS, Huh K, Mook-Jung I. IFN-gamma-induced BACE1 expression is mediated by activation of JAK2 and ERK1/2 signaling pathways and direct binding of STAT1 to BACE1 promoter in astrocytes. Glia. 2007; 55:253–62. https://doi.org/10.1002/glia.20451 [PubMed]
- 5. Li XG, Hong XY, Wang YL, Zhang SJ, Zhang JF, Li XC, Liu YC, Sun DS, Feng Q, Ye JW, Gao Y, Ke D, Wang Q, et al. Tau accumulation triggers STAT1-dependent memory deficits by suppressing NMDA receptor expression. EMBO Rep. 2019; 20:e47202. https://doi.org/10.15252/embr.201847202 [PubMed]
- 6. Ohshima T, Koga H, Shimotohno K. Transcriptional activity of peroxisome proliferator-activated receptor gamma is modulated by SUMO-1 modification. J Biol Chem. 2004; 279:29551–57. https://doi.org/10.1074/jbc.M403866200 [PubMed]
- 7. Liu B, Yang R, Wong KA, Getman C, Stein N, Teitell MA, Cheng G, Wu H, Shuai K. Negative regulation of NF-kappaB signaling by PIAS1. Mol Cell Biol. 2005; 25:1113–23. https://doi.org/10.1128/MCB.25.3.1113-1123.2005 [PubMed]
- 8. Pascual G, Fong AL, Ogawa S, Gamliel A, Li AC, Perissi V, Rose DW, Willson TM, Rosenfeld MG, Glass CK. A SUMOylation-dependent pathway mediates transrepression of inflammatory response genes by PPAR-gamma. Nature. 2005; 437:759–63. https://doi.org/10.1038/nature03988 [PubMed]
- 9. Tahk S, Liu B, Chernishof V, Wong KA, Wu H, Shuai K. Control of specificity and magnitude of NF-kappa B and STAT1-mediated gene activation through PIASy and PIAS1 cooperation. Proc Natl Acad Sci USA. 2007; 104:11643–48. https://doi.org/10.1073/pnas.0701877104 [PubMed]
- 10. Zhao X, Zheng B, Huang Y, Yang D, Katzman S, Chang C, Fowell D, Zeng WP. Interaction between GATA-3 and the transcriptional coregulator Pias1 is important for the regulation of Th2 immune responses. J Immunol. 2007; 179:8297–304. https://doi.org/10.4049/jimmunol.179.12.8297 [PubMed]
- 11. Prigge JR, Schmidt EE. Interaction of protein inhibitor of activated STAT (PIAS) proteins with the TATA-binding protein, TBP. J Biol Chem. 2006; 281:12260–69. https://doi.org/10.1074/jbc.M510835200 [PubMed]
- 12. Dadke S, Cotteret S, Yip SC, Jaffer ZM, Haj F, Ivanov A, Rauscher F
3rd , Shuai K, Ng T, Neel BG, Chernoff J. Regulation of protein tyrosine phosphatase 1B by sumoylation. Nat Cell Biol. 2007; 9:80–85. https://doi.org/10.1038/ncb1522 [PubMed] - 13. Du JX, Yun CC, Bialkowska A, Yang VW. Protein inhibitor of activated STAT1 interacts with and up-regulates activities of the pro-proliferative transcription factor Krüppel-like factor 5. J Biol Chem. 2007; 282:4782–93. https://doi.org/10.1074/jbc.M603413200 [PubMed]
- 14. Lee H, Quinn JC, Prasanth KV, Swiss VA, Economides KD, Camacho MM, Spector DL, Abate-Shen C. PIAS1 confers DNA-binding specificity on the Msx1 homeoprotein. Genes Dev. 2006; 20:784–94. https://doi.org/10.1101/gad.1392006 [PubMed]
- 15. Liu B, Tahk S, Yee KM, Fan G, Shuai K. The ligase PIAS1 restricts natural regulatory T cell differentiation by epigenetic repression. Science. 2010; 330:521–25. https://doi.org/10.1126/science.1193787 [PubMed]
- 16. Rabellino A, Carter B, Konstantinidou G, Wu SY, Rimessi A, Byers LA, Heymach JV, Girard L, Chiang CM, Teruya-Feldstein J, Scaglioni PP. The SUMO E3-ligase PIAS1 regulates the tumor suppressor PML and its oncogenic counterpart PML-RARA. Cancer Res. 2012; 72:2275–84. https://doi.org/10.1158/0008-5472.CAN-11-3159 [PubMed]
- 17. Liu Y, Zhang YD, Guo L, Huang HY, Zhu H, Huang JX, Liu Y, Zhou SR, Dang YJ, Li X, Tang QQ. Protein inhibitor of activated STAT 1 (PIAS1) is identified as the SUMO E3 ligase of CCAAT/enhancer-binding protein β (C/EBPβ) during adipogenesis. Mol Cell Biol. 2013; 33:4606–17. https://doi.org/10.1128/MCB.00723-13 [PubMed]
- 18. Liu B, Yee KM, Tahk S, Mackie R, Hsu C, Shuai K. PIAS1 SUMO ligase regulates the self-renewal and differentiation of hematopoietic stem cells. EMBO J. 2014; 33:101–13. https://doi.org/10.1002/embj.201283326 [PubMed]
- 19. Liu Y, Ge X, Dou X, Guo L, Liu Y, Zhou SR, Wei XB, Qian SW, Huang HY, Xu CJ, Jia WP, Dang YJ, Li X, Tang QQ. Protein Inhibitor of Activated STAT 1 (PIAS1) Protects Against Obesity-Induced Insulin Resistance by Inhibiting Inflammation Cascade in Adipose Tissue. Diabetes. 2015; 64:4061–74. https://doi.org/10.2337/db15-0278 [PubMed]
- 20. Liu SY, Ma YL, Hsu WL, Chiou HY, Lee EH. Protein inhibitor of activated STAT1 Ser503 phosphorylation-mediated Elk-1 SUMOylation promotes neuronal survival in APP/PS1 mice. Br J Pharmacol. 2019; 176:1793–1810. https://doi.org/10.1111/bph.14656 [PubMed]
- 21. Ochaba J, Monteys AM, O’Rourke JG, Reidling JC, Steffan JS, Davidson BL, Thompson LM. PIAS1 Regulates Mutant Huntingtin Accumulation and Huntington’s Disease-Associated Phenotypes In Vivo. Neuron. 2016; 90:507–20. https://doi.org/10.1016/j.neuron.2016.03.016 [PubMed]
- 22. O’Rourke JG, Gareau JR, Ochaba J, Song W, Raskó T, Reverter D, Lee J, Monteys AM, Pallos J, Mee L, Vashishtha M, Apostol BL, Nicholson TP, et al. SUMO-2 and PIAS1 modulate insoluble mutant huntingtin protein accumulation. Cell Rep. 2013; 4:362–75. https://doi.org/10.1016/j.celrep.2013.06.034 [PubMed]
- 23. Hoefer J, Schäfer G, Klocker H, Erb HH, Mills IG, Hengst L, Puhr M, Culig Z. PIAS1 is increased in human prostate cancer and enhances proliferation through inhibition of p21. Am J Pathol. 2012; 180:2097–107. https://doi.org/10.1016/j.ajpath.2012.01.026 [PubMed]
- 24. Yang N, Zhao B, Rasul A, Qin H, Li J, Li X. PIAS1-modulated Smad2/4 complex activation is involved in zinc-induced cancer cell apoptosis. Cell Death Dis. 2013; 4:e811. https://doi.org/10.1038/cddis.2013.333 [PubMed]
- 25. Karasawa F, Shiota A, Goso Y, Kobayashi M, Sato Y, Masumoto J, Fujiwara M, Yokosawa S, Muraki T, Miyagawa S, Ueda M, Fukuda MN, Fukuda M, et al. Essential role of gastric gland mucin in preventing gastric cancer in mice. J Clin Invest. 2012; 122:923–34. https://doi.org/10.1172/JCI59087 [PubMed]
- 26. Anderson WJ, Zhou Q, Alcalde V, Kaneko OF, Blank LJ, Sherwood RI, Guseh JS, Rajagopal J, Melton DA. Genetic targeting of the endoderm with claudin-6CreER. Dev Dyn. 2008; 237:504–12. https://doi.org/10.1002/dvdy.21437 [PubMed]
- 27. Jones MW, Errington ML, French PJ, Fine A, Bliss TV, Garel S, Charnay P, Bozon B, Laroche S, Davis S. A requirement for the immediate early gene Zif268 in the expression of late LTP and long-term memories. Nat Neurosci. 2001; 4:289–96. https://doi.org/10.1038/85138 [PubMed]
- 28. Qin X, Wang Y, Paudel HK. Early Growth Response 1 (Egr-1) Is a Transcriptional Activator of β-Secretase 1 (BACE-1) in the Brain. J Biol Chem. 2016; 291:22276–87. https://doi.org/10.1074/jbc.M116.738849 [PubMed]
- 29. Qin X, Wang Y, Paudel HK. Inhibition of Early Growth Response 1 in the Hippocampus Alleviates Neuropathology and Improves Cognition in an Alzheimer Model with Plaques and Tangles. Am J Pathol. 2017; 187:1828–47. https://doi.org/10.1016/j.ajpath.2017.04.018 [PubMed]
- 30. Heroux NA, Osborne BF, Miller LA, Kawan M, Buban KN, Rosen JB, Stanton ME. Differential expression of the immediate early genes c-Fos, Arc, Egr-1, and Npas4 during long-term memory formation in the context preexposure facilitation effect (CPFE). Neurobiol Learn Mem. 2018; 147:128–38. https://doi.org/10.1016/j.nlm.2017.11.016 [PubMed]
- 31. Nafez S, Oikawa K, Odero GL, Sproule M, Ge N, Schapansky J, Abrenica B, Hatherell A, Cadonic C, Zhang S, Song X, Kauppinen T, Glazner GW, et al. Early growth response 2 (Egr-2) expression is triggered by NF-κB activation. Mol Cell Neurosci. 2015; 64:95–103. https://doi.org/10.1016/j.mcn.2014.12.008 [PubMed]
- 32. Desmazières A, Decker L, Vallat JM, Charnay P, Gilardi-Hebenstreit P. Disruption of Krox20-Nab interaction in the mouse leads to peripheral neuropathy with biphasic evolution. J Neurosci. 2008; 28:5891–900. https://doi.org/10.1523/JNEUROSCI.5187-07.2008 [PubMed]
- 33. Li L, Yun SH, Keblesh J, Trommer BL, Xiong H, Radulovic J, Tourtellotte WG. Egr3, a synaptic activity regulated transcription factor that is essential for learning and memory. Mol Cell Neurosci. 2007; 35:76–88. https://doi.org/10.1016/j.mcn.2007.02.004 [PubMed]
- 34. Abe K, Takeichi M. NMDA-receptor activation induces calpain-mediated beta-catenin cleavages for triggering gene expression. Neuron. 2007; 53:387–97. https://doi.org/10.1016/j.neuron.2007.01.016 [PubMed]
- 35. Wei B, Li L, He A, Zhang Y, Sun M, Xu Z. Hippocampal NMDAR-Wnt-Catenin signaling disrupted with cognitive deficits in adolescent offspring exposed to prenatal hypoxia. Brain Res. 2016; 1631:157–64. https://doi.org/10.1016/j.brainres.2015.11.041 [PubMed]
- 36. Fan YS, Eddy RL, Byers MG, Haley LL, Henry WM, Nowak NJ, Shows TB. The human mineralocorticoid receptor gene (MLR) is located on chromosome 4 at q31.2. Cytogenet Cell Genet. 1989; 52:83–84. https://doi.org/10.1159/000132846 [PubMed]
- 37. Arp JM, ter Horst JP, Kanatsou S, Fernández G, Joëls M, Krugers HJ, Oitzl MS. Mineralocorticoid receptors guide spatial and stimulus-response learning in mice. PLoS One. 2014; 9:e86236. https://doi.org/10.1371/journal.pone.0086236 [PubMed]
- 38. Tallec LP, Kirsh O, Lecomte MC, Viengchareun S, Zennaro MC, Dejean A, Lombès M. Protein inhibitor of activated signal transducer and activator of transcription 1 interacts with the N-terminal domain of mineralocorticoid receptor and represses its transcriptional activity: implication of small ubiquitin-related modifier 1 modification. Mol Endocrinol. 2003; 17:2529–42. https://doi.org/10.1210/me.2003-0299 [PubMed]
- 39. Cuffe JS, Walton SL, Steane SE, Singh RR, Simmons DG, Moritz KM. The effects of gestational age and maternal hypoxia on the placental renin angiotensin system in the mouse. Placenta. 2014; 35:953–61. https://doi.org/10.1016/j.placenta.2014.09.004 [PubMed]
- 40. Speth RC, Carrera EJ, Bretón C, Linares A, Gonzalez-Reiley L, Swindle JD, Santos KL, Schadock I, Bader M, Karamyan VT. Distribution of non-AT1, non-AT2 binding of 125I-sarcosine1, isoleucine8 angiotensin II in neurolysin knockout mouse brains. PLoS One. 2014; 9:e105762. https://doi.org/10.1371/journal.pone.0105762 [PubMed]
- 41. Zheng J, Li G, Chen S, Bihl J, Buck J, Zhu Y, Xia H, Lazartigues E, Chen Y, Olson JE. Activation of the ACE2/Ang-(1–7)/Mas pathway reduces oxygen-glucose deprivation-induced tissue swelling, ROS production, and cell death in mouse brain with angiotensin II overproduction. Neuroscience. 2014; 273:39–51. https://doi.org/10.1016/j.neuroscience.2014.04.060 [PubMed]
- 42. Wang M, Beckmann ND, Roussos P, Wang E, Zhou X, Wang Q, Ming C, Neff R, Ma W, Fullard JF, Hauberg ME, Bendl J, Peters MA, et al. The Mount Sinai cohort of large-scale genomic, transcriptomic and proteomic data in Alzheimer’s disease. Sci Data. 2018; 5:180185. https://doi.org/10.1038/sdata.2018.185 [PubMed]
- 43. Wang M, Roussos P, McKenzie A, Zhou X, Kajiwara Y, Brennand KJ, De Luca GC, Crary JF, Casaccia P, Buxbaum JD, Ehrlich M, Gandy S, Goate A, et al. Integrative network analysis of nineteen brain regions identifies molecular signatures and networks underlying selective regional vulnerability to Alzheimer’s disease. Genome Med. 2016; 8:104. https://doi.org/10.1186/s13073-016-0355-3 [PubMed]
- 44. Trapnell C, Roberts A, Goff L, Pertea G, Kim D, Kelley DR, Pimentel H, Salzberg SL, Rinn JL, Pachter L. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc. 2012; 7:562–78. https://doi.org/10.1038/nprot.2012.016 [PubMed]
- 45. Wang L, Feng Z, Wang X, Wang X, Zhang X. DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics. 2010; 26:136–38. https://doi.org/10.1093/bioinformatics/btp612 [PubMed]
- 46. Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005; 102:15545–50. https://doi.org/10.1073/pnas.0506580102 [PubMed]
- 47. Letunic I, Doerks T, Bork P. SMART: recent updates, new developments and status in 2015. Nucleic Acids Res. 2015; 43:D257–60. https://doi.org/10.1093/nar/gku949 [PubMed]
- 48. Szklarczyk D, Gable AL, Lyon D, Junge A, Wyder S, Huerta-Cepas J, Simonovic M, Doncheva NT, Morris JH, Bork P, Jensen LJ, Mering CV. STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019; 47:D607–13. https://doi.org/10.1093/nar/gky1131 [PubMed]
- 49. Kent WJ, Sugnet CW, Furey TS, Roskin KM, Pringle TH, Zahler AM, Haussler D. The human genome browser at UCSC. Genome Res. 2002; 12:996–1006. https://doi.org/10.1101/gr.229102 [PubMed]
- 50. Hu H, Miao YR, Jia LH, Yu QY, Zhang Q, Guo AY. AnimalTFDB 3.0: a comprehensive resource for annotation and prediction of animal transcription factors. Nucleic Acids Res. 2019; 47:D33–38. https://doi.org/10.1093/nar/gky822 [PubMed]