Introduction
Alzheimer’s disease (AD) and vascular dementia (VD) are common neurocognitive disorders [1–3]. The cerebrospinal fluid (CSF) concentrations of phosphorylated Tau 181 (Tau-181) and amyloid-beta 42 (Aß-42) are considered biomarkers for AD [4–6]. There are no diagnostic or therapeutic biomarkers for VD [7]. Mild cognitive impairment (MCI) is a transitional and reversible stage that can diverge to normal aging and neurocognitive disorder [8, 9]. MCI increases the risk of developing neurocognitive disorders [9], but the trajectory of individuals varies. Identifying biomarkers of neurocognitive disorders in the MCI stage is critical for early diagnosis and intervention [10].
With the advances in biochemistry and sequencing techniques, over 150 RNA modifications have been identified in the past decade [11, 12]. N6-methyladenosine (m6A) is the most common RNA modification in eukaryotic cells [13–17]. The abundance of m6A in the brain gradually increases with age and peaks in adulthood [18]. M6A is highly enriched in adult brain tissue [19, 20] and plays a critical role in neurogenesis, neurodevelopment, and neurological disorders [18, 20–23]. M6A modification on messenger RNA (mRNA) affects the proliferation and differentiation of neural progenitor cells [24–26], and elucidating dysregulations and alterations of m6A perturbations facilitates a comprehensive understanding of RNA methylation-based stem cell or gene-targeted diagnosis and therapy [17, 27].
M6A modification is dynamically regulated by methyltransferases (also known as “writers”), demethylases (“erasers”) and binding proteins (“readers”) (Figure 1A, 1B) [15, 28]. This methylation installed by the “writers” can be reversed by “erasers” [29]. Dysregulations of m6A have been associated with the perturbations of cell proliferation and cell death in different diseases [11, 30, 31].
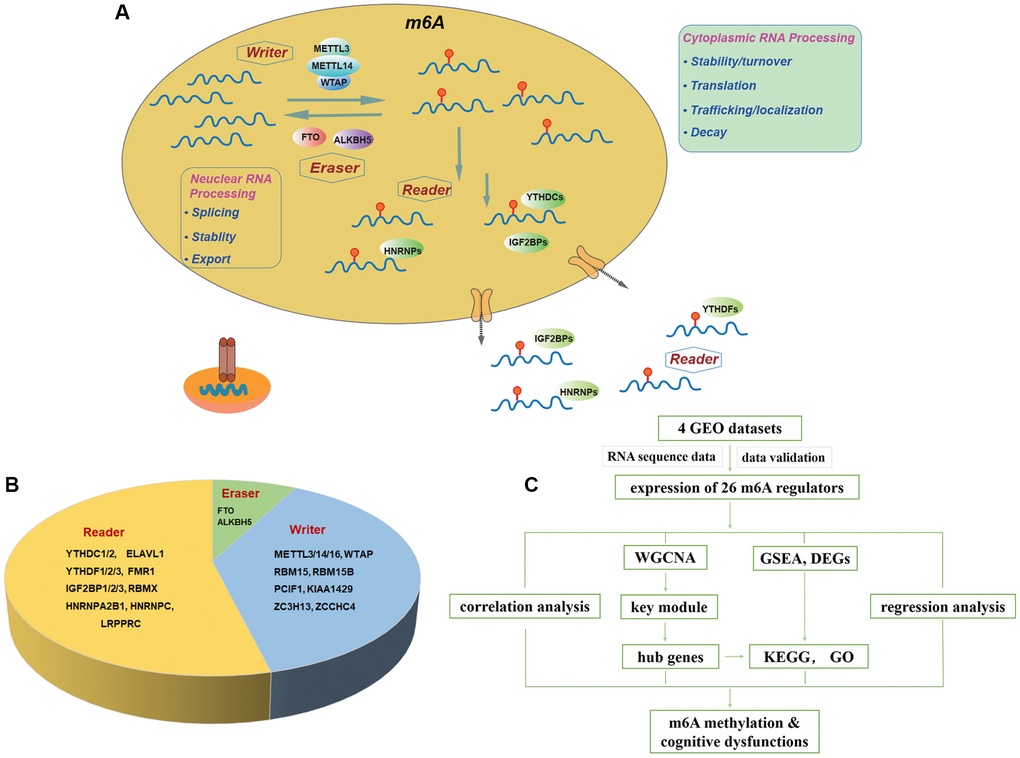
Figure 1. Landscape of included m6A regulators. (A) Overview of dynamic biological processes of m6A RNA methylation mediated by “writers”, “erasers” and “readers” in nucleus and cytoplasm. (B) Distribution of “writers”, “erasers” and “readers” among included 26 m6A regulators. (C) Workflow of the study design. Abbreviations: GEO: Gene Expression Omnibus; WGCNA: Weighted Gene Co-expression Network Analysis; GSEA: Gene Set Enrichment Analysis; DEGs: differentially expressed genes; KEGG: Kyoto Encyclopedia of Genes and Genomes; GO: Gene Ontology.
Alternations of RNA methylation modified genes in the central nervous system (CNS). Little evidence has elucidated the relationships between m6A regulators and neurodegeneration, such as dementia [32]. A recent study by Han et al. using APP/PS1 transgenic mice indicated that m6A abnormality (such as METTL3 and FTO genes) is closely related to AD [33]. To gain a thorough understanding regarding cognitive malfunction from a new perspective, we systematically investigated the molecular alterations of m6A regulators and their associations with AD, VD, and MCI using the database search.
Materials and Methods
Collection of m6A regulators
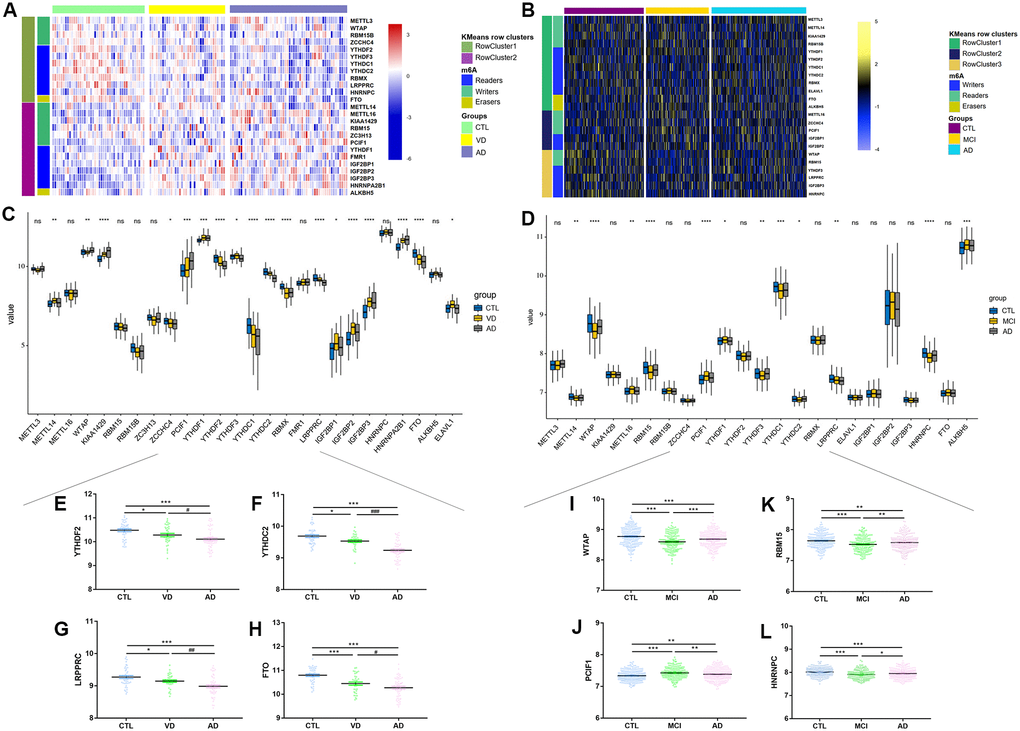
Our study was designed and conducted according to the flow chart (Figure 1C). Briefly, twenty-six m6A regulators were selected accordingly to recent publications [15, 34–37], including ten writers (METTL3, METTL14, METTL16, RBM15, RBM15B, WTAP, KIAA1429, PCIF1, ZCCHC4, ZC3H13), two erasers (FTO, ALKBH5) and fourteen readers (YTHDC1, YTHDC2, YTHDF1, YTHDF2, YTHDF3, IGF2BP1, IGF2BP2, IGF2BP3, HNRNPA2, HNRNPC, FMR1, RBMX, LRPPRC, ELAVL1) (Figure 1B). Figure 1A summarized the landscape of related regulators, including classification, biological functions, and molecular mechanisms.
Acquisition of microarray datasets and preprocessing
The gene-expression dataset with full clinical annotation was obtained from Gene Expression Omnibus (GEO), a publicly sponsored genomic database operated by the National Institutes of Health (NIH). GEO provides open access to many gene expression data from biological and statically comparable samples [38]. In total, four eligible datasets regarding AD, VD, or MCI, including GSE122063, GSE63060, GSE63061, and GSE84422, were selected. There are 711 blood samples from GSE63060 (n = 382) and GSE63061 (n = 329), including 238 control, 189 MCI and 284 AD. GSE122063 consists of 136 brain samples from either frontal or temporal lobe, and patients are divided into control (n = 11), VD (n = 8), and AD (n = 12) groups. GSE84422 collected 1053 post-mortem brain samples from 125 subjects with a full spectrum of AD.
The microarray platforms provided by Illumina and Agilent were downloaded in the format of normalized matrix files. The dataset retrieved from Affymetrix was downloaded in raw “CEL” form. The R/Bioconductor algorithm “RMA” and package “SVA” were used to preprocess gene chips normalization among datasets and to remove batch effects and other latent variations [39]. The overall workflow was presented in Figure 1C.
Analysis of unsupervised clustering for m6A regulators
Unsupervised clustering analysis is an effective machine learning tool for exploring the patterns of datasets in a complex system, which has been applied to AD studies [40] and single-cell RNA sequencing applications [41]. In the current study, the m6A-related regulators were classified into several distinct endotypes by employing unsupervised clustering methods [42, 43] and m6A modification patterns based on the mRNA sequencing of 21 m6A regulators were hereafter determined for further research. Consensus Cluster Plus R package was conducted to perform 1000 times repetitions to guarantee the stability of classification [44]. The number of clusters was determined by the consensus clustering cumulative distribution function (CDF) result (Supplementary Figure 1). The purpose of the CDF plot is to find the k at which the distribution reaches an approximate maximum, which indicates maximum stability, and after which divisions are equivalent to random picks rather than the true cluster structure [44]. Besides, patients were classified into different groups for deeper analysis by adopting an unsupervised clustering method for analyzing the significant difference in different clusters by consensus clustering.
Identification of differential gene expression
After data normalization, differentially expressed genes (DEGs) in datasets of GSE122063, GSE63060, and GSE63061 were identified using the “Limma” package from R/Bioconductor software [45]. The significance of DEGs was set as the adjusted P-value <0.05 and threshold of |log2FC|≥1. Different expression levels of 26 m6A regulators among groups were further verified by unsupervised clustering analysis.
Exploring KEGG pathway enrichment
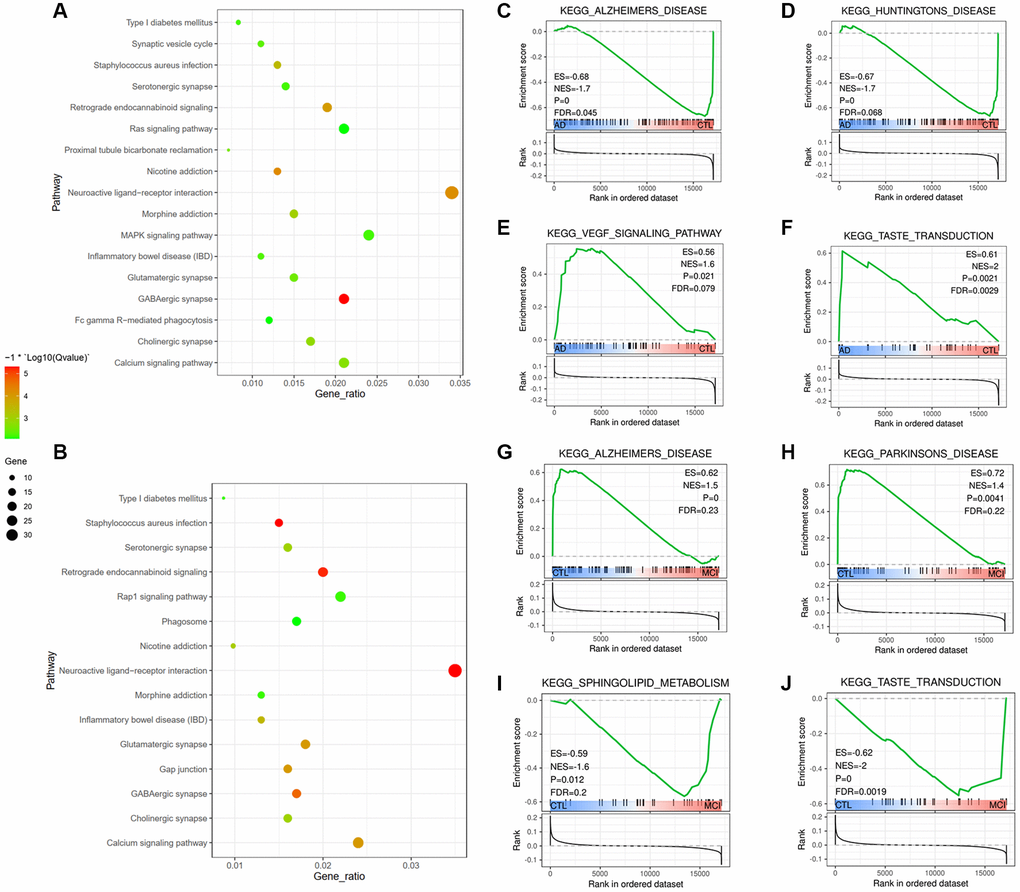
After identifying DEGs in frontal and temporal cortices, Database for Annotation, Visualization and Integrated Discovery (DAVID, https://david.ncifcrf.gov/, ver. 6.8) was further used to identify Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment of DEGs in the above two brain regions. The cutoff criteria were set as P values of 0.05. The results of top10 KEGG pathways in both cortices were picked up and constructed in a bubble plot via R Studio.
Gene set enrichment analysis and functional annotation
Gene set enrichment analysis (GSEA) has successfully been applied to interpret the molecular pathway activated in different biological states [46]. In this study, software “GSEA” (https://www.gsea-msigdb.org/) was utilized to identify the gene up- or down-regulation after filtering for gene set size (min = 5, max = 500) and ranked by t-score [47]. The gene sets of “c2.cp.kegg.v7.1.symbols” (MSigDB database) were used for GSEA analysis. The FDR-corrected q-value <0.25 and P-value <0.05 were set for significance.
Weighted gene co-expression network analysis
Weighted gene co-expression network analysis (WGCNA) was used to extract highly correlated clinical traits and calculate module membership measures from the data sets [48]. “WGCNA” R package was applied to determine hub genes and clinical traits-related modules among microarrays [48]. Genes with variations in the top 25% were extracted from DEGs analysis. Biweight miscorrelation (corType = “Pearson”) was set to detect the outliers. The topological overlap matrix (TOM) was transformed to find the connectivity in the adjacent matrix. Genes were after that divided into multiple sensitive modules according to the TOM-based dissimilarity measurement. Other analysis setting regarding the identification of key modules included soft-threshold power = 7, scale free R2 = 0.9, height = 33, cut height = 0.2, and minimal module size = 10. Subsequently, genes from the highest correlated module were picked up to perform Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analyses. Hub genes were determined by defying gene significance (GS) >0.3 and module membership (MM) >0.8.
Comparing the expression levels according to genotypes of apolipoprotein E gene
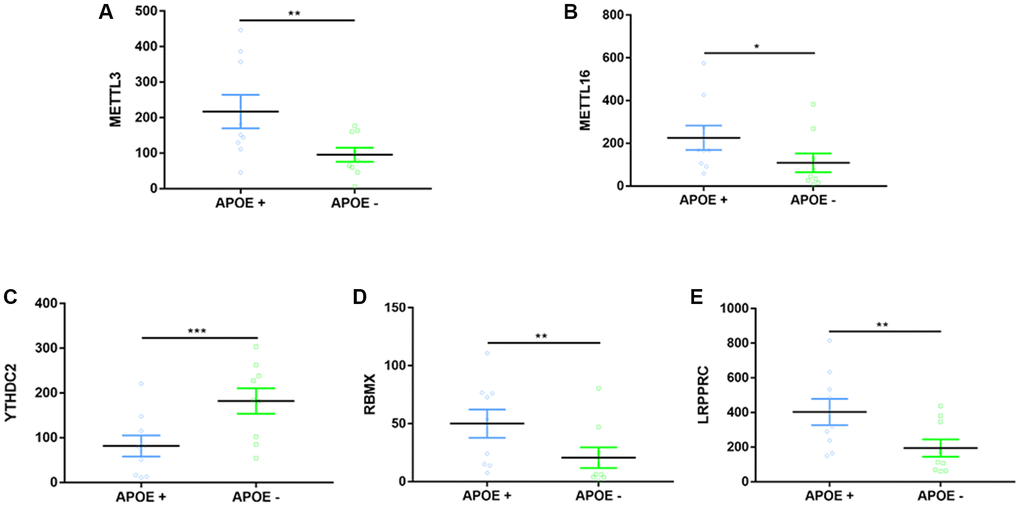
The association between apolipoprotein E (APOE) gene ɛ4 allele and m6A methylation regulators in AD patients was examined by extracting data from GSE29652. Eighteen post-mortem brain samples of AD were categorized into APOE ɛ4+ or APOE ɛ4− subtype. The expression levels of m6A regulators were compared between APOE ɛ4 genotype groups after data normalization and DEGs extraction.
Statistical analysis
The Statistical Package for the Social Sciences (SPSS) version 24.0 was used for statistical analysis. Patients in GSE63060 and GSE63061 were sub-grouped by age according to their cognitive functions. Normal distributed continuous variables were described using mean ± SD; categorical variables were presented as percentages (%). Differences between groups were compared by t-test, one-way ANOVA, or Kruskal-Wallis test for continuous variables, and chi-square for categorical variables.
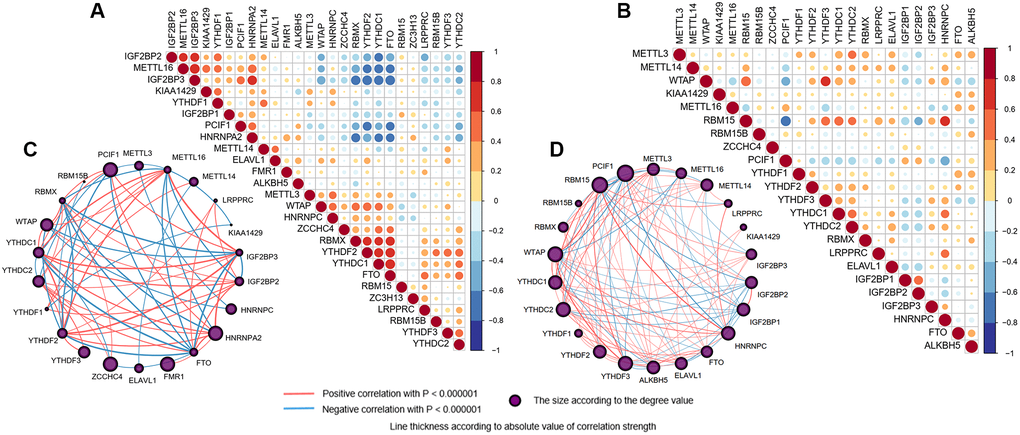
Correlation analyses were carried out to compute the strength of interrelationships between clinical traits and gene expression traits. Correlations between m6A regulators were computed by Spearman correlation analyses and visualized by the “corrplot” package in the R program. Univariate analysis examinations, filtering the meaningful independent variables, followed by multivariate logistic regression analysis, were conducted to estimate the association between m6A methylation levels and MCI and AD.
All statistical P values were two-tail, and p < 0.05 was regarded as statistically significant.
Data sharing statement
All relevant data supporting the key findings are available from the corresponding author upon reasonable request.
Results
Overview of included datasets and m6A regulators
Seven hundred and seventy-one blood samples from GSE63060 and GSE63061 were stratified into three age categories (≤70, 71–79, ≥80). There are significant differences in age distribution among CTL, MCI, and AD groups (χ2 = 26.2, P < 0.001) (Supplementary Table 1). 50% of patients over 80 years had AD, which is significantly higher than in younger age groups, supporting that AD is age-related.
Estimation of differentially regulated molecular pathways
We used the Gene Set Enrichment Analysis (GSEA) algorithm to identify molecular pathways differentially regulated according to cognitive dysfunctions. Of the 65 pathways in the AD group compared to the CTL group, 41 pathways were activated, while 24 pathways were down-regulated (Additional file 1). Generally, GSEA-based analysis highlighted a broad dysregulation of genes related to neurodegenerative disorders. Among the latter are genesets associated with AD, Parkinson’s Disease and Huntington’s Disease, taste transduction, sphingolipid metabolism, and calcium signaling pathway (Figure 4C–4J). It is interesting to find that taste transduction was the only pathway mediated between AD and MCI, and it was up-regulated in AD vs. CTL. However, down-regulated both in MCI vs. CTL and AD vs. MCI (Figure 4J, 4F, Additional file 1), indicating that taste transduction malfunction might play a role in cognitive impairment progress. Besides, Pathways such as the VEGF signaling pathway, complement and coagulation cascades, JAK-STAT signaling pathway, and MAPK signaling pathways were activated in the AD group compared to the CTL group (Additional file 1). The identified dysregulated pathways and related genes, e.g., VEGF, JAK, MAPK, and complements, could be therapeutic targets of AD.
Construction of gene co-expression network and module of interest identification
In this study, weighted gene co-expression network analysis (WGCNA) [50] was performed to identify the key modules most associated with AD and MCI clinical traits. Ages were stratified into three subgroups: ≤70, 71–79, and ≥80 years old. After setting the soft threshold = 7, 6 outliers were removed (height = 33), and the left 705 genes were, after that, theoretically classified according to the expression pattern (Supplementary Figure 5). Firstly, we identified 11 modules of highly co-expressed genes by considering clinical traits, including age/age stratification, gender, cognitive status (Figure 5A). Eigengene adjacency heatmap was further conducted and revealed four main branches among the genes, which verified the above interconnections (Figure 5B). Unique color identifiers were assigned to each module, with gray represented the remaining poorly connected genes. Then, the co-expressed gene network was constructed, and the topological overlap matrix (TOM) heatmap plot was employed to show the network landscape (Figure 5C). The rows and columns in the TOM plot corresponded to various genes. The color intensity represented values of Pearson correlation coefficients, which meant that the higher color intensity indicates higher co-expression similarity between genes included in the network. Herein, genes appeared highly interconnected between module green with modules turquoise and blue, module purple with module brown by visual inspection (Figure 5C).
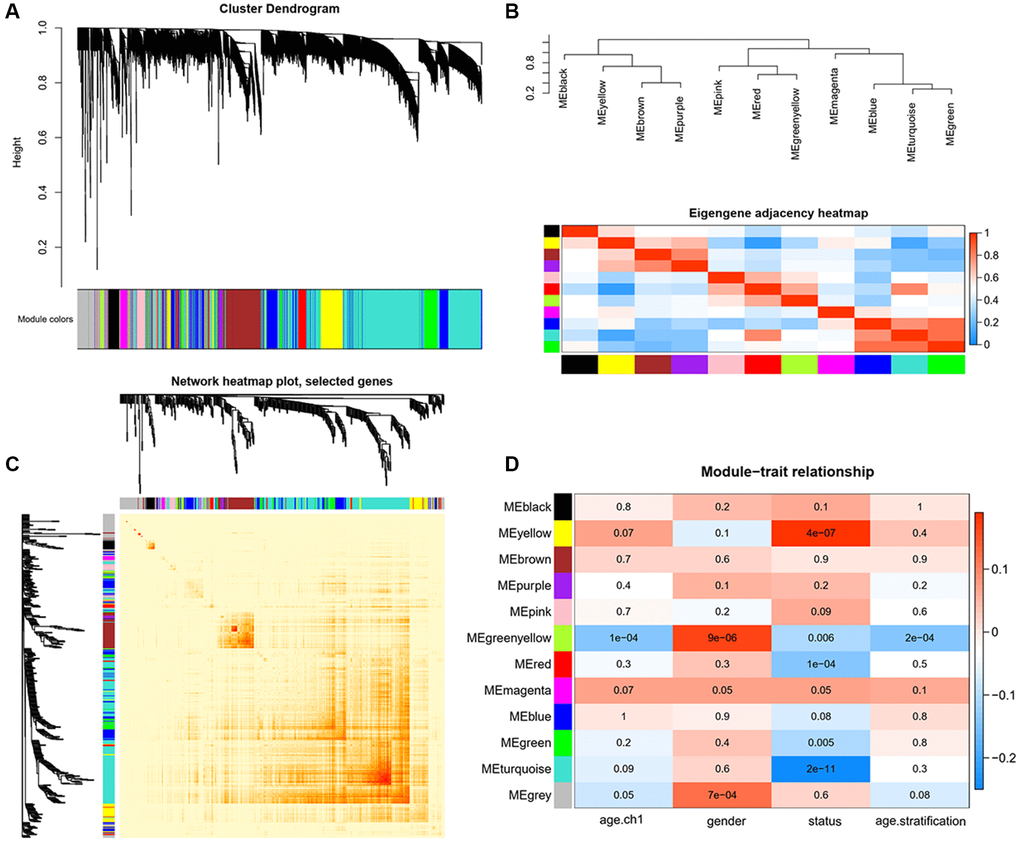
Figure 5. Identification of key modules correlated with clinical traits by WGCNA. (A) Cluster dendrograms of all genes, with dissimilarity based on topological overlap, and then various module colors were assigned. (B) The upper panel displays the hierarchical clustering dendrogram of hub genes that summarize the analyzed modules and branches of the dendrogram group with eigengenes are closely correlated. The lower panel shows the eigengene adjacency heatmap, with the trait weight included. The darker red color represents higher adjacency, while darker blue color represents low adjacency. (C) Heatmap plot of Topological Overlap Matrix (TOM) among selected genes. Each module corresponds to a branch in the hierarchical clustering dendrogram. Modules demonstrate more saturated yellow or even red colors indicate higher co-expression interconnection. Genes locate at the tip of each branch indicate highest interconnection with the rest of the genes in the module. (D) Heatmap of the associations between module eigengenes and clinical traits. Each row and column correspond to a module eigengene or a clinical trait. The plot is colored by corresponding correlation according to the legend, and each cell contains the corresponding P-value. The red color represents positive correlation, while blue color represents negative correlation.
To further strengthen the study of crucial module identification, we defined a measurement to discern the statistical significance between modules with clinical traits. As depicted in the Heatmap, the associations between module eigengenes and clinical traits were colored by corresponding plot, with a darker color (red or blue) indicating strong correlations. Therefore, modules turquoise and yellow were closely intimated with cognitive statues (Figure 5D), indicating that genes in these two modules had high relationships with different cognitive performance levels. The investigation of gene significance (GS) was further performed to find the modules most biologically connected to clinical traits. (Figure 6A–6C). What is more, the existence of significant correlations between GS and module membership (MM) implied that genes within the module turquoise tended to strongly interrelate to cognition (correlation coefficient = 0.35, P = 3.4e-49; Figure 6B).
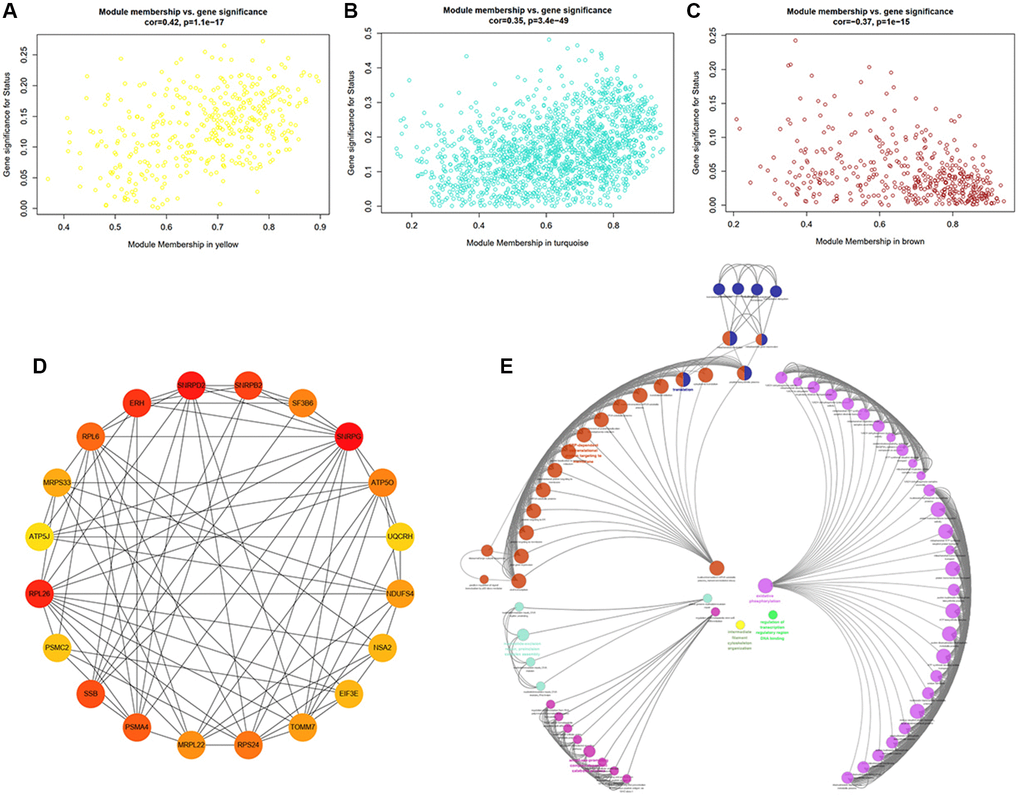
Figure 6. Identification of hub genes and functional annotation of the WGCNA module highly correlated with clinical traits. (A–C) Scatter plots of eigengenes in the representative modules yellow (A), turquoise (B), and brown (C), which have highly significant correlation between Gene Significance (GS) and Module Membership (MM). (D) PPI network of the top20 hub genes in the module turquoise. The circles with darker red represent higher gene rank. (E) Biological functional annotation of the top20 hub genes in the module turquoise by Geno Ontology (GO) enrichment analysis.
A total of 1666 genes in module turquoise and the top20 hub genes, including SNRPG, SNRPD2, RPL26, ERH, SNRPB2, and SSB, were selected, setting GS >0.3 and module membership (MM) >0.8. We can tell that these genes were inseparably associated with each other (Figure 6D). Herein, GO enrichment analysis was performed to identify potential biological functions of module turquoise-related genes (Figure 6E). The result revealed that genes within the module turquoise were most significantly enriched in translation, peptide biosynthetic process, nuclear-transcribed mRNA catabolic process, mRNA/RNA catabolic process.
Discussion
The discovery of m6A mRNA methylation has extended a new dimension in post-transcriptional gene expression [29]. Various animal experiments have previously suggested the modulation roles of m6A on neuronal functions [19, 20, 54–57]. However, the relevance of m6A RNA methylation in cognitive dysfunction remains mostly unexplored. The current study has identified certain m6A-related regulators and related modification patterns, which might serve as novel biomarkers and therapeutic strategies for cognitive dysfunction.
Firstly, we found that m6A-related regulators’ expression differs in different tissues and different cognition levels. A little evidence has confirmed that m6A regulators were closely related to hippocampal-dependent learning and memory [22, 56] and further proved the inner regulating mechanisms [58, 59]. We found that FTO, YTHDC2, and YTHDF2 were the most divergently expressed regulators in the brain between different cognitive groups. However, different from the above findings, we found that expression of METTL3 had no significant difference between CTL, MCI, and AD, while METTL14 had the highest expression in CTL but lowest in MCI. This may partly explain that the above two studies were based on animal research and tissue from the hippocampus, while we analyzed samples of blood from the elderly [22, 56]. Besides, recently reported studies have proved that oligodendrocyte (OL) lineage progression was accompanied by changes along with m6A-related regulators, such as METTL14, YTHDF2, or even a novel reader PRRC2A [60, 61]. Considering our previous findings that OLs and myelin were closely associated with animal cognition [62, 63], we may uncover a m6A-specific cognition modification. Conditional inactivation of m6A components might result in decreased OLs numbers, and CNS hypomyelination and the latter have been implicated in the development of cognitive impairment. Together, we hypothesize that it is possible to regulate OLs proliferation and differentiation by modulating RNA methylation and improving hippocampus-dependent cognitive function. Further in vivo and in vitro experiments are needed to validate the conjecture.
KEGG analysis revealed that DEGs in the brain were significantly enriched in neuroactive ligand-receptor interaction, consistent with a previous study. They reported that the regulation of neuroactive ligand-receptor interaction associated with AD was not preserved in healthy and MCI networks [64]. Another multinational study confirmed that genes regarding neuroactive ligand-receptor interaction were closely related to memory-modulation [65]. As for DEGs in blood, GSEA results surprisingly revealed enrichment of taste transduction. The alterations of taste perception were commonly found in aging and neurodegenerative disorders [66–68]. For example, frontotemporal dementia (FTD) is characterized by alterations in gustation, eating behaviors [69, 70], and appetite alteration are also significantly found in AD [70, 71]. The type 1 taste receptor member 3 (T1R3) is closely involved in taste perception and highly abundant in cognition-related brain areas, such as the hippocampus and cortex [72]. Besides, bioinformatics tools confirmed that the T1R3 receptor processes a strong structural similarity with metabotropic glutamate receptors, and the latter is crucial for learning, memory, and behavior [73, 74]. The loss of the T1R3 subunit is thereby demonstrated to cause learning and memory impairment [68]. Taken together with our and others' data suggest that taste transduction plays a crucial role in cognition procession, and alterations of taste might implicate as an indicator of cognitive dysfunction.
The present study has dug out critical proteins associated with MCI and AD through WGCNA. Belonging to the small nuclear ribonucleoprotein peptide family, SNRPG has been identified as one of the bridge regulators in the module network closely connected to MCI and AD [75, 76]. The decreased expression level of SNRPG might participate in the progression from MCI to AD [76]. Meanwhile, SNRPD2 interacts with nuclear retention elements, and a decrease of SNRPD2 also correlates with pathogenesis from MCI to AD [76, 77]. Moreover, several genes show overlaps in the potential pathogenesis of cancers. It is consistent with a previous bioinformatic study regarding VD by our group [accepted but unpublished] and another PD-associated research [78], which collectively indicated that genes associated with neurodegenerative diseases were always abnormally dysregulated in cancers [76]. Therefore, we may conclude that our study contributed to a better understanding of the pathological mechanisms from MCI to AD. Proteins like SNRPG, SNRPD2, or even cancer-related are expected to be novel biomarkers to predict for patients with MCI who are more likely to progress to AD.
For the first time, we demonstrated in the present study that APOE ɛ4 is closely correlated to five RNA methylation regulators (METTL3, METTL16, YTHDC2, RBMX, LRPPRC) in the AD brain. The ε4 allele of APOE is the most common and influential genetic risk factor for developing AD [79]. Lee et al. once reported that expression of all APOE RNA species was significantly higher in the AD brain than those in the control brain [80]. Similarly, we found a significant increase in most m6A-related regulators within the AD APOE ɛ4+ group, suggesting a complex regulation of epigenetic alterations between the ɛ4 allele and AD. A prospective cohort study by Keller et al. once reported an interaction between FTO and APOE. It proved that those carrying genes of both FTO and AOPE ɛ4 had an increased risk for dementia [81]. They further figured out that FTO's effect on dementia or AD risk mainly was through interaction with the APOE ɛ4 allele [81]. We did not find the difference of FTO expression between APOE ɛ4+/− groups, suggesting that we adopted different samples (brain vs. blood). Consistent with Han's study reporting an elevated level of METTL3 in AD mice, we found that the AD APOE ɛ4 + group has a higher expression of METTL3 [33]. Ectopic expression of RBMX was reported to decrease the APOE receptor’s splicing and was critical to cholesterol homeostasis and, possibly, AD development [82]. Loss or mutation of LRPPRC may contribute to manifestations of neurofibromatosis type 1, which has characteristics of cognitive dysfunction [83]. No previous studies have ever reported relationships between METTL16, YTHDC2 with AOPE ɛ4, or cognition.
Conclusions
The current study has demonstrated the prevalent genetic and expression alterations of RNA methylation regulators according to cognitive impairment. These differently modified patterns of m6A regulators deserve to be highlighted because they are tightly correlated with cognitive malfunctions. The systematic evaluation of m6A regulators-related molecular alterations might lay a critical foundation for understanding the characteristics of cognition. It will also contribute to guiding more therapeutic strategies for dementia.
Author Contributions
YJ and XB conceived and studies and provided supervision. BD, YJ, ML, and ZD collected the data and performed the bioinformatics analysis. BD and YZ wrote the original manuscript, and HL provided the edited version. BD and YJ completed the tables and figures, CF and HZ interpreted the results from clinical perspectives. All the authors read and approved the final manuscript.
Acknowledgments
We thank the National Natural Science Foundation of China and Health System Talent Training Program of Shanghai Municipal Health and Family Planning Commission for their financial support. The funders had no role in study design, data collection, data analysis, interpretation or writing of the report. We thank Ph.D. Qilin Huang from Department of Neurosurgery, Central Theater General Hospital, Wuhan, P.R. China for his technical support.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This work was funded by the National Natural Science Foundation of China (81871040) and the Shanghai Health System Talent Training Program (2018BR29).
References
- 1. Kivipelto M, Mangialasche F, Ngandu T. Lifestyle interventions to prevent cognitive impairment, dementia and Alzheimer disease. Nat Rev Neurol. 2018; 14:653–66. https://doi.org/10.1038/s41582-018-0070-3 [PubMed]
- 2. Shigemizu D, Akiyama S, Asanomi Y, Boroevich KA, Sharma A, Tsunoda T, Matsukuma K, Ichikawa M, Sudo H, Takizawa S, Sakurai T, Ozaki K, Ochiya T, Niida S. Risk prediction models for dementia constructed by supervised principal component analysis using miRNA expression data. Commun Biol. 2019; 2:77. https://doi.org/10.1038/s42003-019-0324-7 [PubMed]
- 3. Robinson L, Tang E, Taylor JP. Dementia: timely diagnosis and early intervention. BMJ. 2015; 350:h3029. https://doi.org/10.1136/bmj.h3029 [PubMed]
- 4. Olsson B, Lautner R, Andreasson U, Öhrfelt A, Portelius E, Bjerke M, Hölttä M, Rosén C, Olsson C, Strobel G, Wu E, Dakin K, Petzold M, et al. CSF and blood biomarkers for the diagnosis of Alzheimer's disease: a systematic review and meta-analysis. Lancet Neurol. 2016; 15:673–84. https://doi.org/10.1016/S1474-4422(16)00070-3 [PubMed]
- 5. Fagan AM, Shaw LM, Xiong C, Vanderstichele H, Mintun MA, Trojanowski JQ, Coart E, Morris JC, Holtzman DM. Comparison of analytical platforms for cerebrospinal fluid measures of β-amyloid 1–42, total tau, and p-tau181 for identifying Alzheimer disease amyloid plaque pathology. Arch Neurol. 2011; 68:1137–44. https://doi.org/10.1001/archneurol.2011.105 [PubMed]
- 6. Kweon OJ, Youn YC, Lim YK, Lee MK, Kim HR. Clinical utility of serum hepcidin and iron profile measurements in Alzheimer's disease. J Neurol Sci. 2019; 403:85–91. https://doi.org/10.1016/j.jns.2019.06.008 [PubMed]
- 7. Petersen RC. Conversion. Neurology. 2006 (Suppl 3); 67:S12–13. https://doi.org/10.1212/wnl.67.9_suppl_3.s12 [PubMed]
- 8. Albert MS, DeKosky ST, Dickson D, Dubois B, Feldman HH, Fox NC, Gamst A, Holtzman DM, Jagust WJ, Petersen RC, Snyder PJ, Carrillo MC, Thies B, Phelps CH. The diagnosis of mild cognitive impairment due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011; 7:270–79. https://doi.org/10.1016/j.jalz.2011.03.008 [PubMed]
- 9. Roberts R, Knopman DS. Classification and epidemiology of MCI. Clin Geriatr Med. 2013; 29:753–72. https://doi.org/10.1016/j.cger.2013.07.003 [PubMed]
- 10. Petersen RC, Stevens JC, Ganguli M, Tangalos EG, Cummings JL, DeKosky ST. Practice parameter: early detection of dementia: mild cognitive impairment (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2001; 56:1133–42. https://doi.org/10.1212/wnl.56.9.1133 [PubMed]
- 11. Roundtree IA, Evans ME, Pan T, He C. Dynamic RNA Modifications in Gene Expression Regulation. Cell. 2017; 169:1187–200. https://doi.org/10.1016/j.cell.2017.05.045 [PubMed]
- 12. Boccaletto P, Machnicka MA, Purta E, Piatkowski P, Baginski B, Wirecki TK, de Crécy-Lagard V, Ross R, Limbach PA, Kotter A, Helm M, Bujnicki JM. MODOMICS: a database of RNA modification pathways. 2017 update. Nucleic Acids Res. 2018; 46:D303–07. https://doi.org/10.1093/nar/gkx1030 [PubMed]
- 13. Alarcón CR, Lee H, Goodarzi H, Halberg N, Tavazoie SF. N6-methyladenosine marks primary microRNAs for processing. Nature. 2015; 519:482–85. https://doi.org/10.1038/nature14281 [PubMed]
- 14. Patil DP, Chen CK, Pickering BF, Chow A, Jackson C, Guttman M, Jaffrey SR. m(6)A RNA methylation promotes XIST-mediated transcriptional repression. Nature. 2016; 537:369–73. https://doi.org/10.1038/nature19342 [PubMed]
- 15. Yang Y, Hsu PJ, Chen YS, Yang YG. Dynamic transcriptomic m6A decoration: writers, erasers, readers and functions in RNA metabolism. Cell Res. 2018; 28:616–24. https://doi.org/10.1038/s41422-018-0040-8 [PubMed]
- 16. Wang S, Chai P, Jia R, Jia R. Novel insights on m6A RNA methylation in tumorigenesis: a double-edged sword. Mol Cancer. 2018; 17:101. https://doi.org/10.1186/s12943-018-0847-4 [PubMed]
- 17. Li Y, Xiao J, Bai J, Tian Y, Qu Y, Chen X, Wang Q, Li X, Zhang Y, Xu J. Molecular characterization and clinical relevance of m6A regulators across 33 cancer types. Mol Cancer. 2019; 18:137. https://doi.org/10.1186/s12943-019-1066-3 [PubMed]
- 18. Livneh I, Moshitch-Moshkovitz S, Amariglio N, Rechavi G, Dominissini D. The m6A epitranscriptome: transcriptome plasticity in brain development and function. Nat Rev Neurosci. 2020; 21:36–51. https://doi.org/10.1038/s41583-019-0244-z [PubMed]
- 19. Weng YL, Wang X, An R, Cassin J, Vissers C, Liu Y, Liu Y, Xu T, Wang X, Wong SZH, Joseph J, Dore LC, Dong Q, et al. Epitranscriptomic m6A Regulation of Axon Regeneration in the Adult Mammalian Nervous System. Neuron. 2018; 97:313–25.e6. https://doi.org/10.1016/j.neuron.2017.12.036 [PubMed]
- 20. Meyer KD, Saletore Y, Zumbo P, Elemento O, Mason CE, Jaffrey SR. Comprehensive analysis of mRNA methylation reveals enrichment in 3' UTRs and near stop codons. Cell. 2012; 149:1635–46. https://doi.org/10.1016/j.cell.2012.05.003 [PubMed]
- 21. Yoon KJ, Ringeling FR, Vissers C, Jacob F, Pokrass M, Jimenez-Cyrus D, Su Y, Kim NS, Zhu Y, Zheng L, Kim S, Wang X, Doré LC, et al. Temporal Control of Mammalian Cortical Neurogenesis by m6A Methylation. Cell. 2017; 171:877–89.e17. https://doi.org/10.1016/j.cell.2017.09.003 [PubMed]
- 22. Shi H, Zhang X, Weng YL, Lu Z, Liu Y, Lu Z, Li J, Hao P, Zhang Y, Zhang F, Wu Y, Delgado JY, Su Y, et al. m6A facilitates hippocampus-dependent learning and memory through YTHDF1. Nature. 2018; 563:249–53. https://doi.org/10.1038/s41586-018-0666-1 [PubMed]
- 23. Widagdo J, Anggono V. The m6A-epitranscriptomic signature in neurobiology: from neurodevelopment to brain plasticity. J Neurochem. 2018; 147:137–52. https://doi.org/10.1111/jnc.14481 [PubMed]
- 24. Zhou H, Wang B, Sun H, Xu X, Wang Y. Epigenetic Regulations in Neural Stem Cells and Neurological Diseases. Stem Cells Int. 2018; 2018:6087143. https://doi.org/10.1155/2018/6087143 [PubMed]
- 25. Yao B, Christian KM, He C, Jin P, Ming GL, Song H. Epigenetic mechanisms in neurogenesis. Nat Rev Neurosci. 2016; 17:537–49. https://doi.org/10.1038/nrn.2016.70 [PubMed]
- 26. Boles NC, Temple S. Epimetronomics: m6A Marks the Tempo of Corticogenesis. Neuron. 2017; 96:718–20. https://doi.org/10.1016/j.neuron.2017.11.002 [PubMed]
- 27. Li J, Yang X, Qi Z, Sang Y, Liu Y, Xu B, Liu W, Xu Z, Deng Y. The role of mRNA m6A methylation in the nervous system. Cell Biosci. 2019; 9:66. https://doi.org/10.1186/s13578-019-0330-y [PubMed]
- 28. Lee M, Kim B, Kim VN. Emerging roles of RNA modification: m(6)A and U-tail. Cell. 2014; 158:980–87. https://doi.org/10.1016/j.cell.2014.08.005 [PubMed]
- 29. Yue Y, Liu J, He C. RNA N6-methyladenosine methylation in post-transcriptional gene expression regulation. Genes Dev. 2015; 29:1343–55. https://doi.org/10.1101/gad.262766.115 [PubMed]
- 30. Fu Y, Dominissini D, Rechavi G, He C. Gene expression regulation mediated through reversible m6A RNA methylation. Nat Rev Genet. 2014; 15:293–306. https://doi.org/10.1038/nrg3724 [PubMed]
- 31. Tong J, Cao G, Zhang T, Sefik E, Amezcua Vesely MC, Broughton JP, Zhu S, Li H, Li B, Chen L, Chang HY, Su B, Flavell RA, Li HB. m6A mRNA methylation sustains Treg suppressive functions. Cell Res. 2018; 28:253–56. https://doi.org/10.1038/cr.2018.7 [PubMed]
- 32. Qin L, Min S, Shu L, Pan H, Zhong J, Guo J, Sun Q, Yan X, Chen C, Tang B, Xu Q. Genetic analysis of N6-methyladenosine modification genes in Parkinson's disease. Neurobiol Aging. 2020; 93:143.e9–143.e13. https://doi.org/10.1016/j.neurobiolaging.2020.03.018 [PubMed]
- 33. Han M, Liu Z, Xu Y, Liu X, Wang D, Li F, Wang Y, Bi J. Abnormality of m6A mRNA Methylation Is Involved in Alzheimer's Disease. Front Neurosci. 2020; 14:98. https://doi.org/10.3389/fnins.2020.00098 [PubMed]
- 34. Arguello AE, DeLiberto AN, Kleiner RE. RNA Chemical Proteomics Reveals the N6-Methyladenosine (m6A)-Regulated Protein-RNA Interactome. J Am Chem Soc. 2017; 139:17249–52. https://doi.org/10.1021/jacs.7b09213 [PubMed]
- 35. Panneerdoss S, Eedunuri VK, Yadav P, Timilsina S, Rajamanickam S, Viswanadhapalli S, Abdelfattah N, Onyeagucha BC, Cui X, Lai Z, Mohammad TA, Gupta YK, Huang TH, et al. Cross-talk among writers, readers, and erasers of m6A regulates cancer growth and progression. Sci Adv. 2018; 4:eaar8263. https://doi.org/10.1126/sciadv.aar8263 [PubMed]
- 36. Akichika S, Hirano S, Shichino Y, Suzuki T, Nishimasu H, Ishitani R, Sugita A, Hirose Y, Iwasaki S, Nureki O, Suzuki T. Cap-specific terminal N6-methylation of RNA by an RNA polymerase II-associated methyltransferase. Science. 2019; 363:eaav0080. https://doi.org/10.1126/science.aav0080 [PubMed]
- 37. Ma H, Wang X, Cai J, Dai Q, Natchiar SK, Lv R, Chen K, Lu Z, Chen H, Shi YG, Lan F, Fan J, Klaholz BP, et al. N6−Methyladenosine methyltransferase ZCCHC4 mediates ribosomal RNA methylation. Nat Chem Biol. 2019; 15:88–94. https://doi.org/10.1038/s41589-018-0184-3 [PubMed]
- 38. Edgar R, Domrachev M, Lash AE. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002; 30:207–10. https://doi.org/10.1093/nar/30.1.207 [PubMed]
- 39. Leek JT, Johnson WE, Parker HS, Jaffe AE, Storey JD. The sva package for removing batch effects and other unwanted variation in high-throughput experiments. Bioinformatics. 2012; 28:882–83. https://doi.org/10.1093/bioinformatics/bts034 [PubMed]
- 40. Alashwal H, El Halaby M, Crouse JJ, Abdalla A, Moustafa AA. The Application of Unsupervised Clustering Methods to Alzheimer's Disease. Front Comput Neurosci. 2019; 13:31. https://doi.org/10.3389/fncom.2019.00031 [PubMed]
- 41. Kiselev VY, Andrews TS, Hemberg M. Challenges in unsupervised clustering of single-cell RNA-seq data. Nat Rev Genet. 2019; 20:273–82. https://doi.org/10.1038/s41576-018-0088-9 [PubMed]
- 42. Zhuang J, Lin C, Ye J. m6 A RNA methylation regulators contribute to malignant progression in rectal cancer. J Cell Physiol. 2020; 235:6300–06. https://doi.org/10.1002/jcp.29626 [PubMed]
- 43. Hartigan J, Wong M. Algorithm AS 136: a K-means clustering algorithm. Appl Stat. 1979; 28:9. https://doi.org/10.2307/2346830
- 44. Wilkerson MD, Hayes DN. ConsensusClusterPlus: a class discovery tool with confidence assessments and item tracking. Bioinformatics. 2010; 26:1572–73. https://doi.org/10.1093/bioinformatics/btq170 [PubMed]
- 45. Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015; 43:e47. https://doi.org/10.1093/nar/gkv007 [PubMed]
- 46. Hung JH, Yang TH, Hu Z, Weng Z, DeLisi C. Gene set enrichment analysis: performance evaluation and usage guidelines. Brief Bioinform. 2012; 13:281–91. https://doi.org/10.1093/bib/bbr049 [PubMed]
- 47. Song ZY, Chao F, Zhuo Z, Ma Z, Li W, Chen G. Identification of hub genes in prostate cancer using robust rank aggregation and weighted gene co-expression network analysis. Aging (Albany NY). 2019; 11:4736–56. https://doi.org/10.18632/aging.102087 [PubMed]
- 48. Langfelder P, Horvath S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics. 2008; 9:559. https://doi.org/10.1186/1471-2105-9-559 [PubMed]
- 49. Zhang B, Wu Q, Li B, Wang D, Wang L, Zhou YL. m6A regulator-mediated methylation modification patterns and tumor microenvironment infiltration characterization in gastric cancer. Mol Cancer. 2020; 19:53. https://doi.org/10.1186/s12943-020-01170-0 [PubMed]
- 50. Horvath S, Zhang B, Carlson M, Lu KV, Zhu S, Felciano RM, Laurance MF, Zhao W, Qi S, Chen Z, Lee Y, Scheck AC, Liau LM, et al. Analysis of oncogenic signaling networks in glioblastoma identifies ASPM as a molecular target. Proc Natl Acad Sci U S A. 2006; 103:17402–07. https://doi.org/10.1073/pnas.0608396103 [PubMed]
- 51. Corder EH, Saunders AM, Strittmatter WJ, Schmechel DE, Gaskell PC, Small GW, Roses AD, Haines JL, Pericak-Vance MA. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer's disease in late onset families. Science. 1993; 261:921–23. https://doi.org/10.1126/science.8346443 [PubMed]
- 52. Foraker J, Millard SP, Leong L, Thomson Z, Chen S, Keene CD, Bekris LM, Yu CE. The APOE Gene is Differentially Methylated in Alzheimer's Disease. J Alzheimers Dis. 2015; 48:745–55. https://doi.org/10.3233/JAD-143060 [PubMed]
- 53. Tannorella P, Stoccoro A, Tognoni G, Petrozzi L, Salluzzo MG, Ragalmuto A, Siciliano G, Haslberger A, Bosco P, Bonuccelli U, Migliore L, Coppedè F. Methylation analysis of multiple genes in blood DNA of Alzheimer's disease and healthy individuals. Neurosci Lett. 2015; 600:143–47. https://doi.org/10.1016/j.neulet.2015.06.009 [PubMed]
- 54. Li L, Zang L, Zhang F, Chen J, Shen H, Shu L, Liang F, Feng C, Chen D, Tao H, Xu T, Li Z, Kang Y, et al. Fat mass and obesity-associated (FTO) protein regulates adult neurogenesis. Hum Mol Genet. 2017; 26:2398–411. https://doi.org/10.1093/hmg/ddx128 [PubMed]
- 55. Hess ME, Hess S, Meyer KD, Verhagen LA, Koch L, Brönneke HS, Dietrich MO, Jordan SD, Saletore Y, Elemento O, Belgardt BF, Franz T, Horvath TL, et al. The fat mass and obesity associated gene (Fto) regulates activity of the dopaminergic midbrain circuitry. Nat Neurosci. 2013; 16:1042–48. https://doi.org/10.1038/nn.3449 [PubMed]
- 56. Widagdo J, Zhao QY, Kempen MJ, Tan MC, Ratnu VS, Wei W, Leighton L, Spadaro PA, Edson J, Anggono V, Bredy TW. Experience-Dependent Accumulation of N6-Methyladenosine in the Prefrontal Cortex Is Associated with Memory Processes in Mice. J Neurosci. 2016; 36:6771–77. https://doi.org/10.1523/JNEUROSCI.4053-15.2016 [PubMed]
- 57. Walters BJ, Mercaldo V, Gillon CJ, Yip M, Neve RL, Boyce FM, Frankland PW, Josselyn SA. The Role of The RNA Demethylase FTO (Fat Mass and Obesity-Associated) and mRNA Methylation in Hippocampal Memory Formation. Neuropsychopharmacology. 2017; 42:1502–10. https://doi.org/10.1038/npp.2017.31 [PubMed]
- 58. Zhang Z, Wang M, Xie D, Huang Z, Zhang L, Yang Y, Ma D, Li W, Zhou Q, Yang YG, Wang XJ. METTL3-mediated N6-methyladenosine mRNA modification enhances long-term memory consolidation. Cell Res. 2018; 28:1050–61. https://doi.org/10.1038/s41422-018-0092-9 [PubMed]
- 59. Koranda JL, Dore L, Shi H, Patel MJ, Vaasjo LO, Rao MN, Chen K, Lu Z, Yi Y, Chi W, He C, Zhuang X. Mettl14 Is Essential for Epitranscriptomic Regulation of Striatal Function and Learning. Neuron. 2018; 99:283–92.e5. https://doi.org/10.1016/j.neuron.2018.06.007 [PubMed]
- 60. Xu H, Dzhashiashvili Y, Shah A, Kunjamma RB, Weng YL, Elbaz B, Fei Q, Jones JS, Li YI, Zhuang X, Ming GL, He C, Popko B. m6A mRNA Methylation Is Essential for Oligodendrocyte Maturation and CNS Myelination. Neuron. 2020; 105:293–309.e5. https://doi.org/10.1016/j.neuron.2019.12.013 [PubMed]
- 61. Wu R, Li A, Sun B, Sun JG, Zhang J, Zhang T, Chen Y, Xiao Y, Gao Y, Zhang Q, Ma J, Yang X, Liao Y, et al. A novel m6A reader Prrc2a controls oligodendroglial specification and myelination. Cell Res. 2019; 29:23–41. https://doi.org/10.1038/s41422-018-0113-8 [PubMed]
- 62. Du B, Li H, Zheng H, Fan C, Liang M, Lian Y, Wei Z, Zhang Y, Bi X. Minocycline Ameliorates Depressive-Like Behavior and Demyelination Induced by Transient Global Cerebral Ischemia by Inhibiting Microglial Activation. Front Pharmacol. 2019; 10:1247. https://doi.org/10.3389/fphar.2019.01247 [PubMed]
- 63. Du B, Liang M, Zheng H, Fan C, Zhang H, Lu X, Du Z, Lian Y, Zhang Y, Bi X. Anti-mouse CX3CR1 Antibody Alleviates Cognitive Impairment, Neuronal Loss and Myelin Deficits in an Animal Model of Brain Ischemia. Neuroscience. 2020; 438:169–81. https://doi.org/10.1016/j.neuroscience.2020.05.011 [PubMed]
- 64. Kelly J, Moyeed R, Carroll C, Luo S, Li X. Genetic networks in Parkinson's and Alzheimer's disease. Aging (Albany NY). 2020; 12:5221–43. https://doi.org/10.18632/aging.102943 [PubMed]
- 65. Papassotiropoulos A, Gerhards C, Heck A, Ackermann S, Aerni A, Schicktanz N, Auschra B, Demougin P, Mumme E, Elbert T, Ertl V, Gschwind L, Hanser E, et al. Human genome-guided identification of memory-modulating drugs. Proc Natl Acad Sci U S A. 2013; 110:E4369–74. https://doi.org/10.1073/pnas.1314478110 [PubMed]
- 66. Shin YK, Cong WN, Cai H, Kim W, Maudsley S, Egan JM, Martin B. Age-related changes in mouse taste bud morphology, hormone expression, and taste responsivity. J Gerontol A Biol Sci Med Sci. 2012; 67:336–44. https://doi.org/10.1093/gerona/glr192 [PubMed]
- 67. Martin B, Maudsley S, White CM, Egan JM. Hormones in the naso-oropharynx: endocrine modulation of taste and smell. Trends Endocrinol Metab. 2009; 20:163–70. https://doi.org/10.1016/j.tem.2009.01.006 [PubMed]
- 68. Martin B, Wang R, Cong WN, Daimon CM, Wu WW, Ni B, Becker KG, Lehrmann E, Wood WH
3rd , Zhang Y, Etienne H, van Gastel J, Azmi A, et al. Altered learning, memory, and social behavior in type 1 taste receptor subunit 3 knock-out mice are associated with neuronal dysfunction. J Biol Chem. 2017; 292:11508–30. https://doi.org/10.1074/jbc.M116.773820 [PubMed] - 69. Woolley JD, Gorno-Tempini ML, Seeley WW, Rankin K, Lee SS, Matthews BR, Miller BL. Binge eating is associated with right orbitofrontal-insular-striatal atrophy in frontotemporal dementia. Neurology. 2007; 69:1424–33. https://doi.org/10.1212/01.wnl.0000277461.06713.23 [PubMed]
- 70. Ahmed RM, Irish M, Kam J, van Keizerswaard J, Bartley L, Samaras K, Hodges JR, Piguet O. Quantifying the eating abnormalities in frontotemporal dementia. JAMA Neurol. 2014; 71:1540–46. https://doi.org/10.1001/jamaneurol.2014.1931 [PubMed]
- 71. Mendez MF, Licht EA, Shapira JS. Changes in dietary or eating behavior in frontotemporal dementia versus Alzheimer's disease. Am J Alzheimers Dis Other Demen. 2008; 23:280–85. https://doi.org/10.1177/1533317507313140 [PubMed]
- 72. Ren X, Zhou L, Terwilliger R, Newton SS, de Araujo IE. Sweet taste signaling functions as a hypothalamic glucose sensor. Front Integr Neurosci. 2009; 3:12. https://doi.org/10.3389/neuro.07.012.2009 [PubMed]
- 73. Talevi A, Enrique AV, Bruno-Blanch LE. Anticonvulsant activity of artificial sweeteners: a structural link between sweet-taste receptor T1R3 and brain glutamate receptors. Bioorg Med Chem Lett. 2012; 22:4072–74. https://doi.org/10.1016/j.bmcl.2012.04.076 [PubMed]
- 74. Ménard C, Quirion R. Group 1 metabotropic glutamate receptor function and its regulation of learning and memory in the aging brain. Front Pharmacol. 2012; 3:182. https://doi.org/10.3389/fphar.2012.00182 [PubMed]
- 75. Heinrichs V, Hackl W, Lührmann R. Direct binding of small nuclear ribonucleoprotein G to the Sm site of small nuclear RNA. Ultraviolet light cross-linking of protein G to the AAU stretch within the Sm site (AAUUUGUGG) of U1 small nuclear ribonucleoprotein reconstituted in vitro. J Mol Biol. 1992; 227:15–28. https://doi.org/10.1016/0022-2836(92)90678-d [PubMed]
- 76. Tao Y, Han Y, Yu L, Wang Q, Leng SX, Zhang H. The Predicted Key Molecules, Functions, and Pathways That Bridge Mild Cognitive Impairment (MCI) and Alzheimer's Disease (AD). Front Neurol. 2020; 11:233. https://doi.org/10.3389/fneur.2020.00233 [PubMed]
- 77. Azam S, Hou S, Zhu B, Wang W, Hao T, Bu X, Khan M, Lei H. Nuclear retention element recruits U1 snRNP components to restrain spliced lncRNAs in the nucleus. RNA Biol. 2019; 16:1001–09. https://doi.org/10.1080/15476286.2019.1620061 [PubMed]
- 78. Feng Y, Wang X. Systematic analysis of microarray datasets to identify Parkinson's disease-associated pathways and genes. Mol Med Rep. 2017; 15:1252–62. https://doi.org/10.3892/mmr.2017.6124 [PubMed]
- 79. Liu CC, Liu CC, Kanekiyo T, Xu H, Bu G. Apolipoprotein E and Alzheimer disease: risk, mechanisms and therapy. Nat Rev Neurol. 2013; 9:106–18. https://doi.org/10.1038/nrneurol.2012.263 [PubMed]
- 80. Lee EG, Tulloch J, Chen S, Leong L, Saxton AD, Kraemer B, Darvas M, Keene CD, Shutes-David A, Todd K, Millard S, Yu CE. Redefining transcriptional regulation of the APOE gene and its association with Alzheimer's disease. PLoS One. 2020; 15:e0227667. https://doi.org/10.1371/journal.pone.0227667 [PubMed]
- 81. Keller L, Xu W, Wang HX, Winblad B, Fratiglioni L, Graff C. The obesity related gene, FTO, interacts with APOE, and is associated with Alzheimer's disease risk: a prospective cohort study. J Alzheimers Dis. 2011; 23:461–69. https://doi.org/10.3233/JAD-2010-101068 [PubMed]
- 82. Ling IF, Estus S. Role of SFRS13A in low-density lipoprotein receptor splicing. Hum Mutat. 2010; 31:702–09. https://doi.org/10.1002/humu.21244 [PubMed]
- 83. Arun V, Wiley JC, Kaur H, Kaplan DR, Guha A. A novel neurofibromin (NF1) interaction with the leucine-rich pentatricopeptide repeat motif-containing protein links neurofibromatosis type 1 and the French Canadian variant of Leigh's syndrome in a common molecular complex. J Neurosci Res. 2013; 91:494–505. https://doi.org/10.1002/jnr.23189 [PubMed]