Introduction
A recent WHO report predicts that, by 2030, one in six people globally will be over 60 years of age [1]. Aging profoundly impairs immune competence, a phenomenon termed immunosenescence, rendering older adults (≥60 years) highly vulnerable to infectious diseases such as tuberculosis (TB) [2]. Mycobacterium tuberculosis (Mtb), the causative pathogen of TB, induces hypermethylation and transcriptional reprogramming of host immune cells, further exacerbating age-related immune dysfunction [3]. Clinically, older adults exhibit reduced vaccine effectiveness alongside heightened susceptibility to TB, with epidemiological data indicating 2-3 times higher TB incidence and up to four times higher mortality than younger patients. In several East Asian regions, including Japan, Korea, and Hong Kong, older adults accounted for 45-69% of TB cases in 2020-21 [4]. Similarly, projections from Taiwan predict 78% of new TB cases by 2035 will arise in the geriatric population, which currently contributes to 82.1% of TB-associated mortality [5, 6]. These trends underscore the urgent need to elucidate age-associated immune defects driving TB vulnerability in an aging world.
Despite this growing burden, current TB research predominantly employs young adult mouse models (6-8 weeks old, equivalent to ~18-year-old humans), which do not adequately capture the immune landscape of older hosts [7]. Evidence from studies suggests that old (>18 months; M) mice exhibit higher bacterial burden, delayed CD4+ T cell responses, and altered immune activation compared to younger counterparts [8, 9, 10]. However, the impact of immunosenescence on bacterial clearance dynamics, immune cell phenotypes, and host responses during TB treatment remain largely unexplored.
To address this knowledge gap, we investigated age-dependent effects on Mtb infection and treatment in C57BL/6 mice spanning young (2-4M), mid-aged (9-12M; 38-44 human years), old (17-19M; ~54-60 human years), and aged (31M; >80 human years) stages. Following low-dose aerosol infection with Mtb H37Rv and treatment with rifampicin and isoniazid (RIF-INH), we monitored bacterial clearance kinetics across tissues, profiled systemic and tissue-specific immune responses, including circulating cytokines, Mtb-specific antibodies, liver micronutrients, and the proteome of splenic CD4+CD44+ T cells. Through this approach, we aimed to identify age-associated processes that compromise treatment efficacy, which can inform strategies to reduce the risk of relapse in geriatric TB patients.
Results
Lung Mtb burden in C57BL/6 mice remains unaffected by age or sex through 6 weeks post-infection
The influence of host age and sex on susceptibility to Mtb infection remains an area of interest, particularly at early time points. Thus, female and male C57BL/6 mice of wider age groups were aerosol infected with a low dose of H37Rv (Figure 1A). Mice were categorized as young (2-4M), middle-aged (9M females, 12M males) and old (17-19M males). Prior to Mtb infection, body weight was higher in middle-aged and older mice than in younger controls (Supplementary Figure 1). During the study period, old mice lost ~3% weight, whereas young mice gained ~8% weight (Figure 1B). Young female mice gained more body weight (~20%) than males (~10%) (Figure 1B). At 6 weeks post infection (w.p.i.), middle-aged male mice showed significant body weight loss (Figure 1B). Despite age-related variations in body weight, lung Mtb burden progressively increased up to 4 w.p.i. before plateauing by 6 w.p.i., with no significant differences attributable to age or sex (Figure 1C). Thus, immunosenescence manifests primarily during therapeutic clearance rather than initial containment phase, rationalizing our focus on age-associated differences in treatment response.
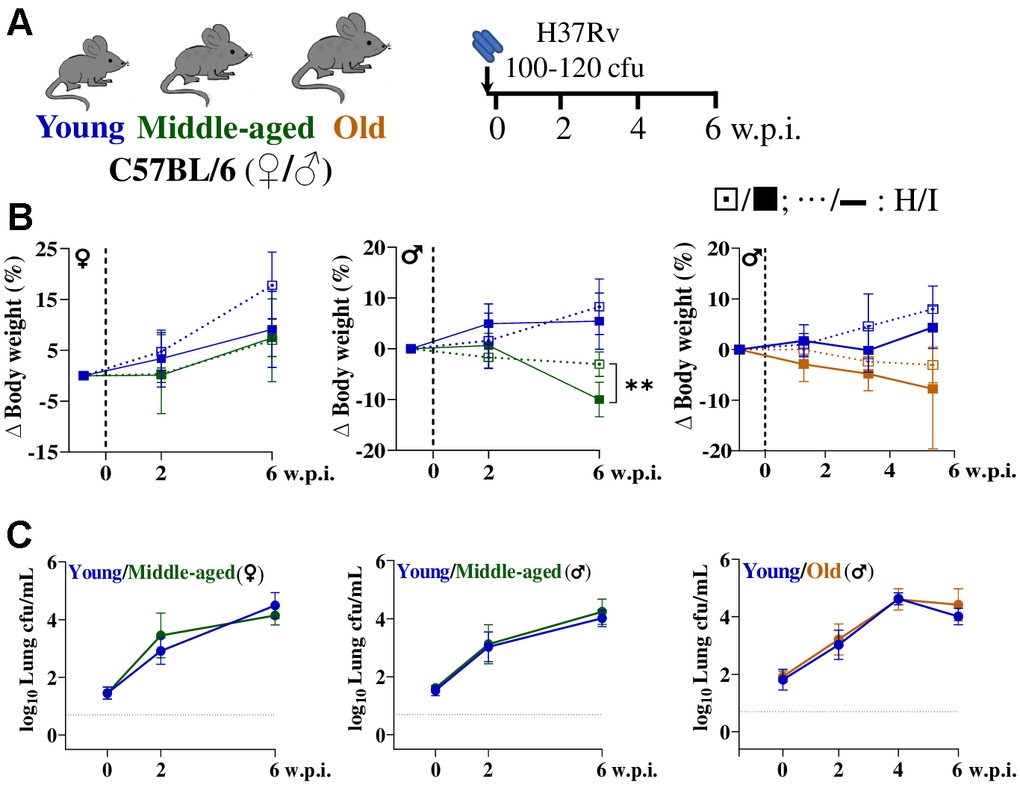
Figure 1. Lung Mtb burden remains similar till 6 weeks post-infection across age groups and sexes in C57BL/6 mice. (A) Schematic of the experimental design used for aerosol Mtb H37Rv infection of C57BL/6 mice [female (♀) and male (♂)] across age groups. (B) Changes in body weight of mice groups (healthy: H, ….; infected: I, ___) are presented, n=5-10/age group/condition; dashed vertical line represents day of infection. (C) Lung bacterial burden (in log10cfu/mL) in mice post Mtb infection; n=5-10/timepoint/age group/condition; dashed horizontal line represents limit of detection (LOD). Young (2-4 months) in blue, middle-aged (9-12 months) in green and old (17-19 months) mice in brown; cfu= colony forming unit; w.p.i.= weeks post infection; p-value: ** <0.005 at 95% confidence interval by Mann-Whitney test. Data shown as mean ± SD. See also Supplementary Figures 1, 2.
Inflammaging and immune defects drive delayed TB clearance in aging despite hepatic resilience
To evaluate the impact of age on treatment response, a subset of mice received RIF-INH following Mtb infection. Remarkably, older mice retained a significantly higher ~3 log10 cfu lung Mtb load at 2 weeks post-treatment (w.p.t.) compared to the young group (~1.5 log10 cfu). And by 4 weeks, both mouse age groups achieved complete bacterial clearance, i.e. resulting below the limit of detection (LOD) (Figure 2A, 2B). Histologically, old mice developed granulomatous lung lesions with prominent foamy cells later (6 w.p.i.) compared to young mice (4 w.p.i.) (Supplementary Figure 1). Lung inflammation was resolved in young mice by 4 w.p.t. but persisted in old mice (Supplementary Figure 1). Additionally, middle-aged mice were able to resolve the inflammation by 4 w.p.t., irrespective of sex (Supplementary Figure 2). In extrapulmonary sites, such as spleen and liver, mycobacterial clearance was observed by 2 w.p.t. in both age groups (Figure 2B).
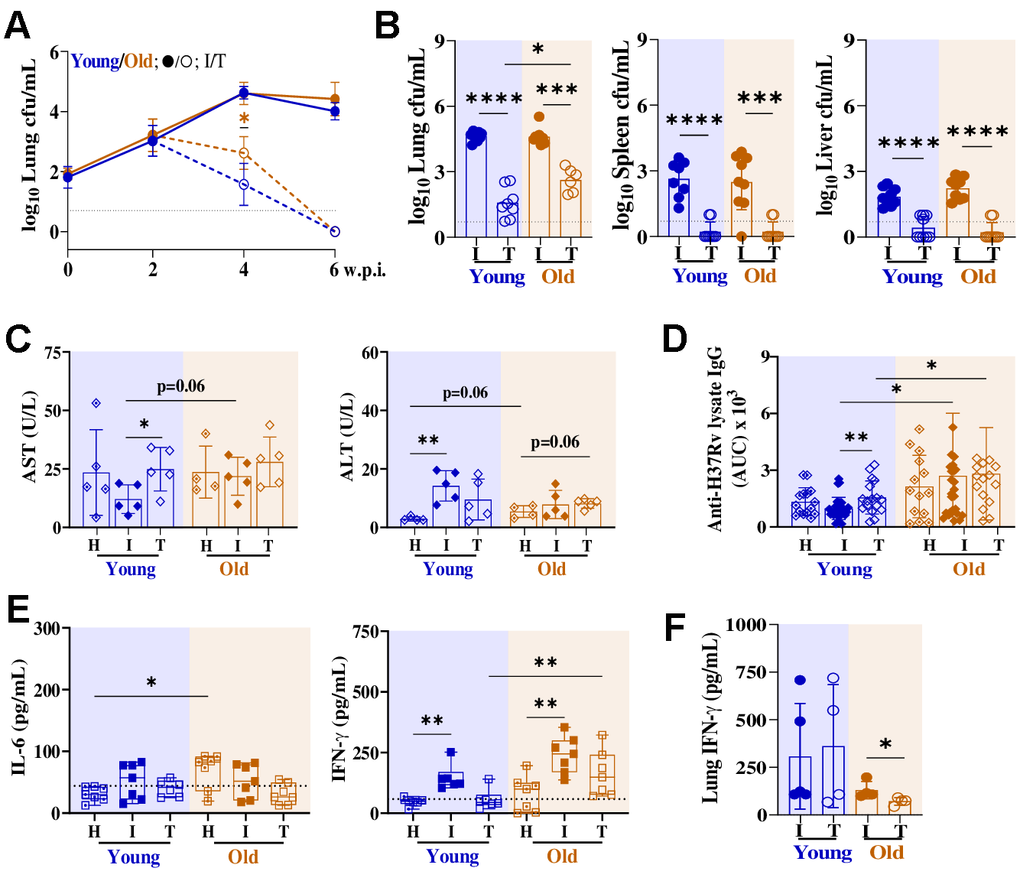
Figure 2. Old C57BL/6 mice exhibit delayed lung Mtb clearance following two weeks of RIF-INH treatment. (A) Lung bacterial burden (in log10cfu/mL) in male C57BL/6 mice from infected (I; at 2, 4 and 6 w.p.i.) and treated (T; at 2 and 4 w.p.t.; treatment started at 2 w.p.i.) groups; n=10/timepoint/age group/condition; dashed horizontal line represents limit of detection (LOD). A set of two independent experiments is depicted. (B–F) showing data of infected groups at 4 w.p.i. and treated groups at 2 w.p.t.. (B) Bacterial load (in log10cfu/mL) in lung, spleen and liver; individual data points at 4 w.p.i./2 w.p.t. presented in histograms (n=10/age group/condition). (C) Serum alanine transaminase (ALT) and aspartate aminotransferase (AST) activities in mice of different groups (n=5/age group/condition) at 4 w.p.i./2 w.p.t.. (D) Area under the curve (AUC) for Mtb H37Rv lysate-specific serum IgG in healthy (H; n=15/age group), infected (I; n=27-28/age group) and treated (T; n= 16-17/age group) mice at 4 w.p.i./2 w.p.t.. (E) Serum cytokines (in picogram per milliliter): IL-6, IFN-γ in different mice groups (n=6-7/age group/condition) at 4 w.p.i./2 w.p.t.. (F) Lung IFN-γ in the different mice groups (n=4-5/age group/condition) at 4 w.p.i./2 w.p.t.. Young (2-4 months) in blue and old (17-19 months) mice in brown; cfu= colony forming unit; w.p.i.= weeks post infection; w.p.t.= weeks post treatment; p-values: * ≤0.05, ** <0.005, *** <0.0005 and **** <0.0001 at 95% confidence interval by Mann-Whitney test. Data shown as mean ± SD. See also Supplementary Figures 1, 4.
Liver enzymes are pivotal for drug metabolism, with metal ions exerting substantial influence over their activity. Age-related changes may influence this variability in liver function and micronutrient status [11]. Thus, we monitored the liver micronutrient levels of these mouse groups using inductively coupled plasma mass spectrometry (ICP-MS) (Supplementary Figure 3). Old mice had higher hepatic Cu (4.28×), low Zn (0.14×) levels with similar Fe and Se levels compared to controls (Supplementary Figure 3). Additionally, old mice receiving treatment showed higher hepatic Cu levels (Supplementary Figure 3). Liver micronutrient status impacts activities of hepatic enzymes like alanine aminotransferase (ALT) and aspartate aminotransferase (AST), commonly used markers of liver function and injury. Old mice exhibited comparable serum ALT and AST activities to the young mice (Figure 2C). However, ALT activity showed a non-significant upward (p=0.06) trend in old mice (Figure 2C). Mtb infection did not impact serum AST or ALT activity in old mice, but ALT activity was higher in Mtb-infected younger mice (Figure 2C). Strikingly, young mice receiving treatment had higher serum AST activity reflecting drug-induced liver injury, while old mice remained unaffected (Figure 2C).
To assess age-related differences in humoral immunity against Mtb, if any, serum anti-H37Rv specific IgG titers were measured. The overall IgG response was significantly higher in old mice compared to young mice (Figure 2D). Importantly, old mice receiving treatment mounted higher IgG responses at a later time point i.e. 4 w.p.t., while young mice showed it at 2 w.p.t. compared to their respective infected controls (Figure 2D and Supplementary Figure 4). While robust humoral responses are beneficial for pathogen control, the delayed yet heightened antibody response in old mice could indicate persistent antigenic stimulation and likely reflect altered immune regulation with aging. Circulatory IL-6 levels were higher in old mice, consistent with a state of chronic low-grade inflammation or inflammaging (Figure 2E). Higher circulatory IFN-γ levels were observed in Mtb-infected young and old mice, which resolved partially in treated groups (Figure 2E). The cytokine response in old mice appears skewed toward a TH1-type response rather than a broad systemic inflammatory state, which may explain selective upregulation of IFN-γ without a parallel increase in IL-6. However, at the site of infection, IFN-γ levels trended higher in young mice compared to old mice (Figure 2F). Critically, despite RIF-INH treatment selectively reducing lung IFN-γ levels in old mice, yet bacterial clearance remained impaired (Figure 2F). This reveals a central paradox showing TH1 contraction coupled with effector dysfunction, where diminished IFN-γ, rather than unresolved inflammation, impairs bacterial killing during early therapy.
Splenic T cell proteome reveals age-specific mitochondrial collapse
To further elucidate the molecular drivers of delayed Mtb clearance, we conducted global proteomic profiling of flow-sorted CD4+CD44+ T cells (Figure 3D and Supplementary Table 2). In set-I, 109 proteins were identified, and Mtb-infected young and old mice showed 23 and 30 deregulated (p ≤ 0.1, log2fold change (log2fc): I/H > |0.58|) proteins, respectively (Figure 3E, 3F and Supplementary Figure 10 and Supplementary Table 3). In set-II, out of 41 identified proteins, ~20 were deregulated in the mouse groups receiving RIF-INH treatment (Figure 3G, 3H and Supplementary Table 4). Proteins involved in immune response (e.g., complement, serotransferrin) were suppressed in old mice, potentially reflecting age-related immune decline. Young mice exhibited few changes, with upregulation of stress response proteins like galectin-1 and superoxide dismutase, possibly reflecting a more robust acute response (Supplementary Table 3). Both mouse age groups showed downregulation of 4-hydroxy-2-oxoglutarate aldolase (HOGA), suggesting disrupted hydroxyproline degradation [14] (Supplementary Table 3). Upon Mtb infection, old mice showed an upregulation of prostaglandin E synthase 3 (PTGES3, log2fc: I/H = 1.15), indicating an age-biased induction of prostaglandin synthase E2 (PGE2) machinery (Supplementary Table 3). PGE2 disrupts alveolar macrophage metabolic fitness through mitochondrial dysfunction, suppressing electron transport chain gene expression [15].
Additionally, old mice showed elevated levels of mitochondrial AST after treatment (log2fc: T/I = 3.15), indicative of unresolved mitochondrial stress, consistent with increased serum AST and altered hepatic micronutrient status observed earlier (Supplementary Table 4). Old mice showed lower HOGA abundance after treatment, implying prolonged mitochondrial stress in T cells of old mice (Supplementary Table 4). Together, these proteomic signatures reveal age-specific mitochondrial defects, providing a molecular basis for T cell dysfunction, inflammaging, and impaired mycobacterial clearance in older hosts, linking cellular metabolic stress in T cells during Mtb infection and therapy.
Discussion
As the global demography is shifting towards the older (≥60 years) age groups, it is expected that immunosenescence and lack of an effective TB vaccine for adults will be key drivers for the elevated numbers of geriatric TB patients. Paradoxically, in countries where TB was contained successfully, the number of new TB cases is increasing, for example, in Kansas City and North Carolina in the United States. The majority of published reports focused on understanding host-pathogen interactions in Mtb-infected mice of 6-8 weeks (~2M), representing a human age equivalence of 18-20 years [7]. Thus, understanding how the older host responds to Mtb infection is crucial for developing appropriate and effective interventions.
TB demonstrates a marked gender disparity, with males showing increased susceptibility, with male to female ratio of 1.7 in TB prevalence, which is associated with reduced B cell follicle formation in the lung [16–18]. On the contrary, females exhibit increased T cell activity, production of TH1-type cytokines, particularly IFN-γ, and robust antibody responses [19]. Above 65 years of age, genomic disparities between the sexes become more pronounced, with males exhibiting higher innate and pro-inflammatory activity [20]. Thus, we focused on using old male mice to study the outcomes upon Mtb infection and post-treatment.
A low aerosol dose of Mtb infection to young (2-4M), middle-aged (9, 12M), old (17-19M) and aged (31M) C57BL/6 mice yielded similar lung mycobacterial burden up to 6 w.p.i.. Whereas earlier reports showed better Mtb control in older mice at earlier time points [8, 9, 21, 22]. Turner et al. reported that 18M C57BL/6 mice aerosol-infected with Erdman strain had a lower lung load at 14 d.p.i., compared to 3M controls [9]. 24M B6D2F1 hybrid mice infected with H37Rv showed lower load at 20 d.p.i. than young 2M controls [8]. Variations in reported tissue Mtb loads between earlier reports and this study may stem from differences in mouse strain and sex, Mtb strain and dose, infection method, and observation timing. Important to highlight that, to the best of our knowledge, this report demonstrates for the first time, that old mice showed a compromised Mtb clearance after 2 weeks of RIF-INH treatment compared to younger controls. These results corroborate the reported delayed viral clearance in 17M BALB/c mice infected with PR8 influenza virus [23].
Aging also impacts hepatic function, affecting the metabolism of exogenous molecules, thereby leading to hepatoxicity [24]. Old mice displayed a higher hepatic Cu/Zn ratio irrespective of infection status, consistent with reports that an elevated Cu/Zn ratio in aged hosts is a biomarker of chronic inflammation and oxidative stress. The accumulation of Cu and Zn in phagosomes increases bactericidal activity; however, Cu overload is detrimental to the host [25]. Low Zn levels in older mice may impair Cu/Zn superoxide-dismutase function, leading to pro-inflammatory effects and weakened hepatic function [26]. Mtb pathogenesis depletes the available Fe pool and is a typical host response in counteracting Mtb replication in TB patients [27]. The liver Fe level was reduced in young mice post-treatment; however, treated old mice retained a higher hepatic Fe pool, providing a niche that could support Mtb growth and persistence. Dysregulated iron homeostasis could influence bacterial burden partly through effects on ferroptosis, which is an important avenue for future mechanistic work. Hepatic micronutrient imbalances in old mice did not translate to significant serum ALT/AST elevations or drug-induced liver injury, unlike in young treated mice. This hepatic resilience underscores that age-related treatment delays stem from immune defects rather than metabolic or toxic limitations. Young mice maintained high lung IFN-γ to drive rapid bacterial killing while old mice exhibited premature TH1 contraction, creating a confluence of impairments during early-phase treatment.
Immune cells, such as CD4+ T cells play a critical role in controlling Mtb, and their function might be partially impaired in older hosts [28]. A higher lung mycobacterial load was predicted in 18M mice at 6 w.p.i. compared to 3M mice, due to a delayed CD4+ T cell response [28, 29]. We observed that Mtb-infected old C57BL/6 mice had similar TFH-like cells in the lungs and a higher frequency in the spleen at 4 w.p.i. compared to younger controls. However, a higher proportion of TFC-like cells was observed in the lungs and spleens in Mtb-infected old mice. The contribution of TH1 cells in TB is well established. However, our understanding of TFH cells, which link humoral and adaptive immunity, during Mtb infection is limited [30]. TFC cells suppress TFH cell helper function and antibody responses through multiple mechanisms, thereby inhibiting antibody production to maintain self-tolerance and regulate immune responses [31, 32]. Age is reported to negatively influence TFH cell function and differentiation, thereby compromising host responses [33]. Localization of TFH-like cells has been demonstrated within protective granuloma-associated lymphoid tissue in lungs of 2M C57BL/6 mice, which mediates Mtb control [34]. In effective responses, TFH cells dominate the follicular niche, provide IL-21 and CD40L to B cells, and help structure organized germinal centres or tertiary lymphoid-like granulomas. Immunosenescence drives this pathological skew: aging-associated TFH cells enable TFC expansion, disrupting germinal centre formation and granuloma organization.
Understanding tissue-level T cell distribution in Mtb-infected old mice has been attempted earlier; however, limited reports captured the changes during TB treatment [8, 9, 21, 28, 30]. At 2 w.p.t., young mice showed increased splenic TFH-like cells, which remained similar in old mice, suggesting the inability of the older host to elicit a proper TFH response. Compromised Mtb clearance in old mice might be due to the impaired TFH cell functionality, which was reported earlier in >20M old C57BL/6 mice [12, 33]. Future work should phenotypically characterize TFC-like cells to test their active suppression of protective TFH responses, establishing causality in age-dependent humoral dysregulation.
Proteome analyses of splenic cells from Mtb-infected old mice revealed altered abundance of mitochondrial enzymes involved in the catabolism of hydroxyproline (Hyp), a product of protein degradation, notably higher AST and lower HOGA [14] (Figure 4). AST catalyzes 4-hydroxy glutamate (4-OH-Glu) to 4-hydroxy-2-oxo-glutarate (HOG), which is converted into pyruvate and glyoxylate using HOGA. This imbalance likely causes HOG accumulation, inhibiting glyoxylate reductase and promoting excessive oxalate production, known to disrupt mitochondrial function independent of the lactate dehydrogenase pathway [35]. PTGES3 upregulation in Mtb-infected old mice further implicates prostaglandin E2 signaling, which prior work shows hampers alveolar macrophage proliferation and mitochondrial function in 18-22M mice [15]. These proteomic changes represent immunometabolic signatures of T cell dysfunction in Mtb-infected old mice rather than as direct causal drivers. The observed HOGA downregulation and PTGES3 signaling likely underlies defective CD4+ T cell responses and impaired Mtb clearance. This aligns with evidence that mitochondrial rescue strategies enhance TB immunity in older adults, positioning mitochondrial axis as a therapeutically actionable target [36, 37].
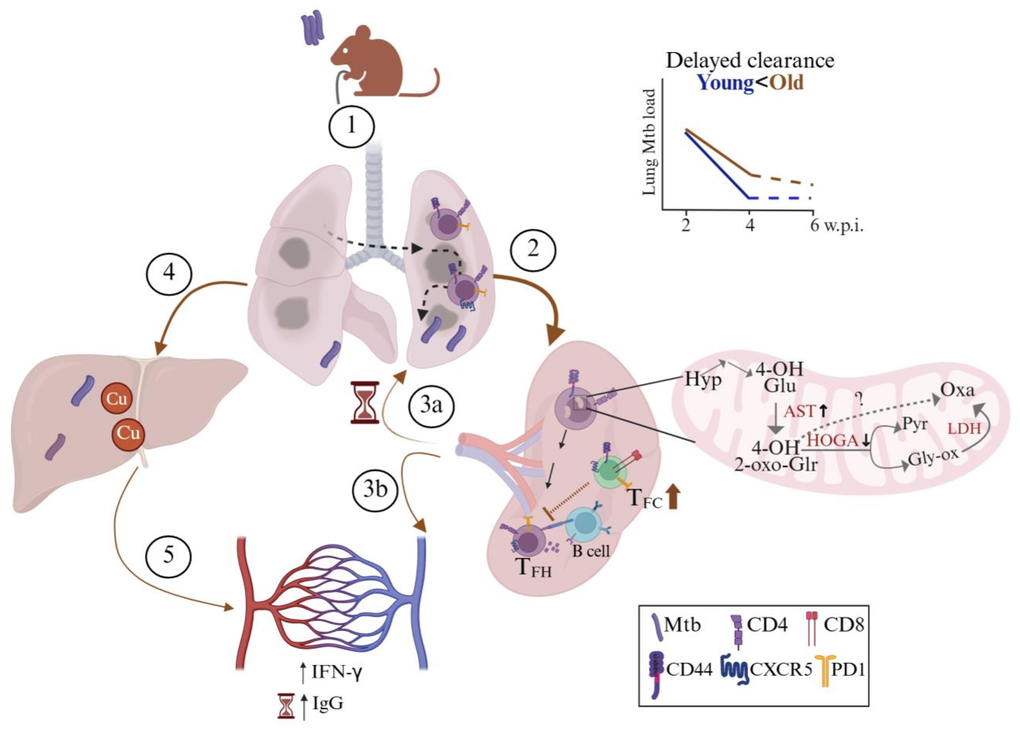
Figure 4. Age-associated mechanisms of delayed Mycobacterium tuberculosis lung clearance in old C57BL/6 mice during early rifampicin-isoniazid treatment. Illustrative summary of the results observed in the study. (1) Following Mtb infection via aerosol challenge, (2) antigen presentation takes place in the spleen. (3a) T cells reach the inflamed lung of old mice. (3b) Increased levels of proinflammatory cytokines: interferon-gamma (IFN-γ) and increased Mtb-specific IgG levels observed at later timepoint in Mtb-infected old mice. (4) Mtb disseminates to the liver via hepatic artery where copper (Cu) was observed to be accumulated (5) alongside increased systemic proinflammatory mediators. Old mice exhibit dysfunctional splenic T follicular helper (TFH : CD4+CD44+CXCR5+PD1+) cells as a virtue of high T follicular cytotoxic cells (TFC : CD8+CD44+CXCR5+PD1+), and leads to delayed lung Mtb clearance at 2 weeks post-treatment. Proteome analysis revealed splenic CD4+CD44+ T cells of old mice showed alterations in mitochondrial proteins (decreased HOGA: 4-hydroxy-2-oxoglutarate aldolase and increased AST), disrupting hydroxyproline (Hyp) degradation and promoting 4-hydroxy-2-oxo-glutarate (4-OH-2-oxo-Glr) accumulation, directing the reaction to oxalate (Oxa) via a pathway independent of lactate dehydrogenase (LDH), pyruvate (Pyr) and glyoxylate (Gly-ox). Created with BioRender.
This study has a few limitations. Technical constraints prevented direct proteomic profiling of lung TFH cells, necessitating analysis of splenic CD4+CD44+ T cells as a functional proxy. Mouse models in TB research have limitations for extrapolating findings to humans due to differences in disease pathology, notably the absence of key features such as granuloma liquefaction, cavitation, and fibrosis. Also, mice exhibit different metabolic and aging patterns compared to humans, with distinct changes in organ metabolism, immune responses, and biomarkers of aging. Thus, age-associated metabolic alterations identified should be viewed as hypotheses for mechanisms that may operate in older humans, rather than as directly translatable biomarkers. Functional validation of mitochondrial defects including mitophagy flux assay, Seahorse bioenergetics or electron microscopy, remains a key next step. In vitro modulation of HOGA/AST (via inhibitors or overexpression) in T cells of lungs would test causality in rescuing anti-Mtb function. Future directions extend beyond T cell-intrinsic changes to dendritic cell antigen presentation and alterations in myeloid cell populations, and using clinical Mtb isolates in aging models for enhanced translational relevance.
In summary, this study demonstrated impaired pulmonary Mtb clearance in old C57BL/6 mice during RIF-INH treatment, driven by a confluence of immunosenescence defects: inflammaging, delayed TH1 responses, higher TFC cell frequency and mitochondrial proteome collapse. These findings illuminate age-specific mechanisms contributing to suboptimal TB treatment responses in geriatric patients and identifying novel mitochondrial targets for adjunct therapy.
Materials and Methods
Experimental groups, Mtb infection and treatment
The procedures adopted in this study were performed following the recommended Guidelines for Care and Use of Laboratory Animals and approved by the Animal Ethics Committee of ICGEB- New Delhi (Ref No. ICGEB/IAEC/07032020/TH-10). C57BL/6 mice of different age groups (2-, 4-, 9-, 12-, 17-, 19- and 31 months, M) were procured from the ICGEB bio-experimentation facility. The mice age equivalents to humans, used in this study, is as follows: young (2-4M), mid-aged (9-12M; 38-44 human years), old (17-19M; ~54-60 human years), and aged (31M; >80 human years) [7]. These mice were transferred to the BSL-III Tuberculosis Aerosol Challenge Facility (TACF) for experiments involving Mtb infection. Mice were fed a chow diet, water ad libitum and maintained at 20-22° C; 45-60% humidity with a 12h light/dark cycle. Mice were weight-adjusted, randomly grouped and after 1 week of acclimatization in TACF, infected with a low dose (100-120 cfu) of Mtb H37Rv strain in the Madison aerosol exposure chamber. After 2 w.p.i., a subset of mice received RIF and INH (100 mg/L each) in drinking water and fresh drug solutions were replaced every 48 hours. Two 17M mice (one bearing an abscess, the other died before the completion of 4 w.p.t.) were excluded from the study.
Cfu assay
Tissues (lung, spleen and liver) were homogenized in PBS (1 mL) and plated on 7H11 agar supplemented with Middlebrook 10% OADC. These plates were incubated at 37° C, and bacterial colonies were enumerated 3 weeks later.
Histopathology
Formalin-fixed lungs were paraffin-embedded, sectioned and stained with hematoxylin and eosin, followed by capturing images using Nikon Eclipse TS2. The slides were scored based on the severity of inflammation, granuloma formation and necrosis.
Liver micronutrient profiling
Liver (~50 mg) was subjected to microwave digestion post addition of H2O2 and HNO3 (1:4). These were diluted and introduced to iCAP™-TQ ICP-MS using argon as carrier. Data acquisition was performed using Qtegra (Thermo Fisher Scientific, version 2.10.4345.39) and involved running different concentrations of a multielement standard mix (Supelco, #92091).
ALT and AST assay
Serum ALT and AST activities were measured using kits (Cayman Chemical, #700260 and #701640) according to the manufacturer’s instructions.
Immune cell isolation and flow cytometry
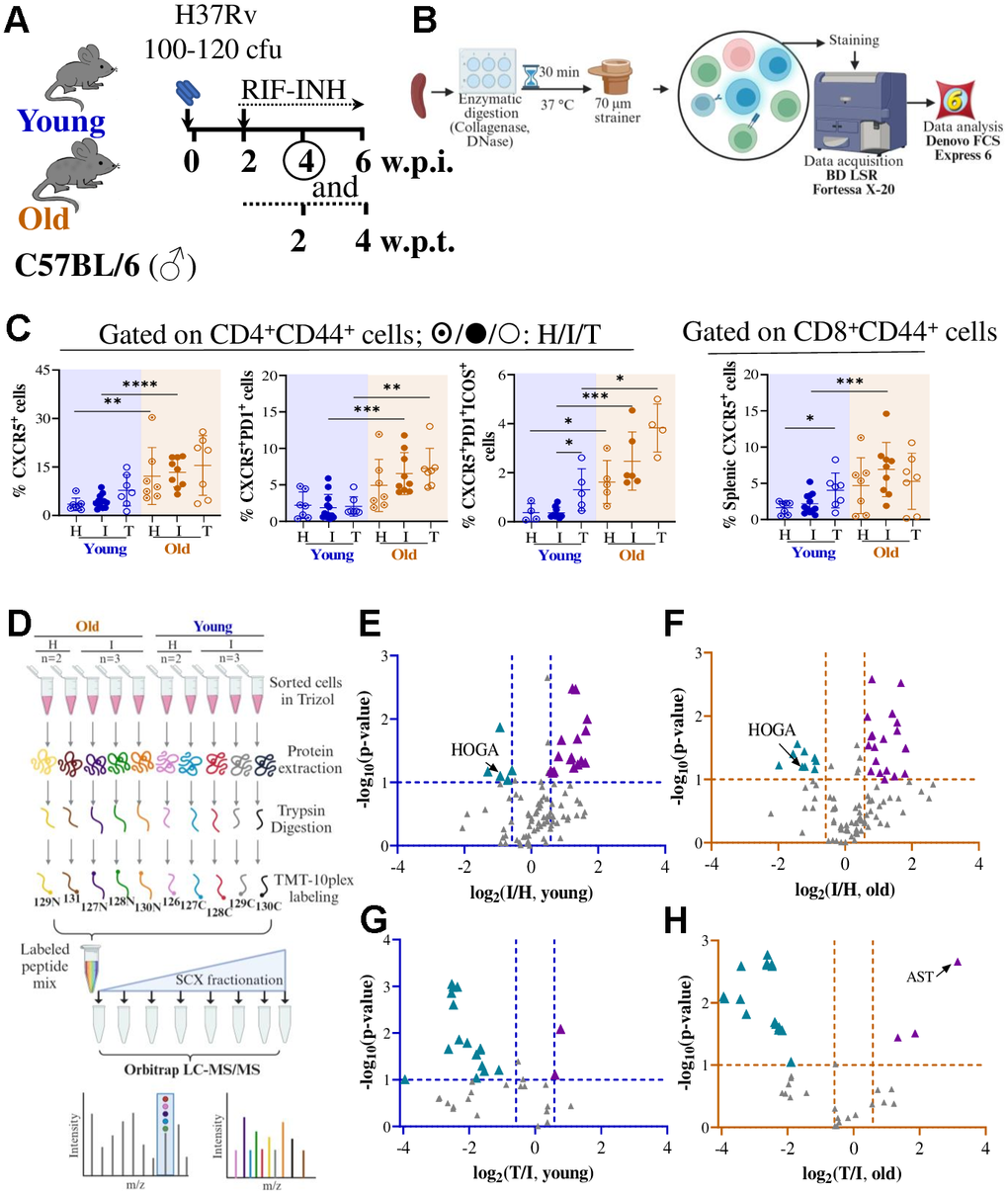
Harvested tissues (lung and spleen) were incubated with collagenase D (1 μg/mL) and DNase I (0.5 mg/mL). Peripheral blood mononuclear cells (PBMCs) were isolated using Histopaque-1077 following standard procedures. Isolated cells were stained with recommended dilutions by the manufacturer: CD3-BV510 (BioLegend, #100234), CD4-BV785 (BioLegend, #100552), CD8a-AF700 (BioLegend, #100730), CD44-BV605 (BioLegend, #103047), CXCR5-PE/Dazzle 594 BioLegend, #145522), PD1-PE-Cy7 (BioLegend, #109110), KLRG1-BV421 (BioLegend, #138414), CD153-PE (BioLegend, #106405), CD25-AF488 (BioLegend, #102017) and CD127-BV650 (BioLegend, #135043) and LIVE/DEAD™ Fixable Near IR stain (Invitrogen, #L34994) on ice for 45 min. After washing with buffer (0.2% FBS in 1× PBS), cells were treated with fixation (Invitrogen, #00-5123) and permeabilization buffer (Invitrogen, #00-8333) and kept on ice for 30 min. Intracellular staining was done with FoxP3-AF647 (BioLegend, #126408) and Tbet-BV711 (BioLegend, #644820) and incubated at RT for 45 min, followed by washing. Data (~1 million events/sample) was acquired using BD LSRFortessa X-20 and analyzed using FCS Express (DeNovo software, version 6).
Another set of immune cells were stained with: CD3-FITC (Invitrogen, #11-0032-82), CD4-SB600 (Invitrogen, #63-0042-82), CD8a-BV421 (BioLegend, #100753), CD44-BV510 (BioLegend, #103044), CXCR5-PE, ICOS-PerCP/Cy5.5 (BioLegend, #145504), PD1-PE-Cy7 (BioLegend, #109110) and LIVE/DEAD™ Fixable Near IR stain (Invitrogen, #L34994) on ice for 45 min. After washing with buffer (3% FBS in 1× PBS), splenic CD4+CD44+ T cells were sorted using BD FACS Aria III Fusion. Data was analyzed using FlowJo (BD Biosciences, version 10.8.1).
Lung IFN-γ estimation: IFN-γ was estimated in lung homogenates by using the ELISA MAX™ Standard Set Mouse IFN-γ kit (BioLegend, #430801) following the manufacturer’s instructions.
Serum cytokine estimation
Two times diluted serum samples were used to estimate cytokines using capture bead-based assay LEGENDplex (BioLegend) along with the standard (BioLegend, #740371, lot no. B330381). The samples were acquired on BD LSRFortressa X-20 using PE and APC as the reporter and bead classification channel, respectively. Data was analyzed using Qognit software.
Serum IgG antibody quantification
ELISA plate was coated with Mtb H37Rv whole cell lysate (10 μg/mL) and incubated overnight at 4° C. Then, wash buffer (0.05% Tween-20 in 1× PBS) was added, followed by blocking in 3% BSA in 1× PBS containing 0.05% Tween-20 and incubated at 37° C for 2h at 180 rpm. After blotting the plate dry, sera were serially diluted fourfold in 0.1% BSA in 1× PBS containing 0.05% Tween-20, and 100 μL was added per well. The plate was then incubated overnight at 4° C. The plate was washed three times, followed by incubation with anti-mouse IgG (Southern Biotech, #1031-05) at 37° C for 2h at 180 rpm. The plate was washed six times and developed by adding o-phenylenediamine dihydrochloride, incubated at RT for 5 min. The reaction was terminated by adding 1N HCl, and the absorbance was measured at 490 nm.
Splenic CD4+CD44+ cell proteome analysis
Proteins were isolated using TRIzol reagent from sorted splenic CD4+CD44+ cells and quantified using BCA Protein Assay. An equal amount (~10 μg) of protein from each sample in a set (n = 10) was used for TMT10plex (Thermo Fisher Scientific, #90113). TMT-labelled peptides from all samples were pooled, dried and subjected to strong cation exchange chromatography and eluted using ammonium formate. Later, these fractions (n = 5) were cleaned up using C18 spin columns and dried, resuspended in formic acid (0.1%, 20 μL) and injected into an Orbitrap Fusion Lumos Tribrid Mass Spectrometer connected to a nano-LC 1200. Data was analysed with Proteome Discoverer version 2.3 using Mus musculus (ID: UP000000589; 55,087 protein count). Proteins qualifying the parameters [log2 fold change ≥ ± 0.58; log10p-value ≥1; false discovery rate<0.05] were selected as important deregulated molecules.
Statistical analysis
Data are presented as mean ± standard deviation. Details on the sample size and statistical analysis are provided in the figure legends.
Supplementary Materials
Author Contributions
FP and RKN conceptualized the project. FP and SC carried out the mouse experiments and cytokine profiling. FP performed the flow cytometry data acquisition and prepared the liver micronutrient and proteomics samples. AG carried out antibody titers, ELISA experiments, and helped with proteomic data analysis. ShiC calculated histopathological scores. FP and RKN analyzed the data, wrote the original draft and revised it with the suggestions received from all the co-authors. RKN acquired funding, shared resources and administered the project. All co-authors reviewed and approved the manuscript.
Acknowledgements
Haripriya Priyadarsini, Adyasha Sarangi, Nidhi Yadav and Mothe Sravya are acknowledged for their help and support during the mouse experiments. Help from Shilpa Sharma and Jaya Baranwal, and the staff of the bio-experimentation facility and the BSL-III Tuberculosis Aerosol Challenge Facility (TACF) at ICGEB-New Delhi, is acknowledged. DBT, Government of India supports the TACF. We thank Dr. Anmol Chandele for her insightful comments and suggestions for revising the manuscript. Part of these research findings were presented in Keystone Symposia on Molecular Basis of Healthy Aging in 2023; World Conference on Lung Health 2022, organized by The Union, and the 2nd biennial meeting on Sex Differences in the Immune Systems Conference in 2022, organized by Trinity College Dublin and Whitehead Institute. Some figures were created using BioRender.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Ethical Statement
The animal procedures adopted in this study were performed following the recommended Guidelines for Care and Use of Laboratory Animals and approved by the Animal Ethics Committee of ICGEB- New Delhi (Protocol No. ICGEB/IAEC/07032020/TH-10).
Funding
FP was supported with a Junior and Senior Research fellowship from the Department of Biotechnology (DBT), Government of India. SC was a Shyama Prasad Mukherjee Fellow supported by the Council of Scientific and Industrial Research, Government of India. A Senior Research Fellowship from the DBT supports AG. The core support from the International Centre for Genetic Engineering and Biotechnology (ICGEB), New Delhi, to RKN is highly acknowledged.
References
- 1. Word Health Organization data website: https://www.who.int/news-room/fact-heets/detail/ageing-and-health (Accessed 10/02/2024).
- 2. Yew WW, Yoshiyama T, Leung CC, Chan DP. Epidemiological, clinical and mechanistic perspectives of tuberculosis in older people. Respirology. 2018; 23:567–75. https://doi.org/10.1111/resp.13303 [PubMed]
- 3. Bobak CA, Abhimanyu, Natarajan H, Gandhi T, Grimm SL, Nishiguchi T, Koster K, Longlax SC, Dlamini Q, Kahari J, Mtetwa G, Cirillo JD, O'Malley J, et al. Increased DNA methylation, cellular senescence and premature epigenetic aging in guinea pigs and humans with tuberculosis. Aging (Albany NY). 2022; 14:2174–93. https://doi.org/10.18632/aging.203936 [PubMed]
- 4. Teo AK, Morishita F, Islam T, Viney K, Ong CW, Kato S, Kim H, Liu Y, Oh KH, Yoshiyama T, Ohkado A, Rahevar K, Kawatsu L, et al. Tuberculosis in older adults: challenges and best practices in the Western Pacific Region. Lancet Reg Health West Pac. 2023; 36:100770. https://doi.org/10.1016/j.lanwpc.2023.100770 [PubMed]
- 5. Ku CC, Dodd PJ. Forecasting the impact of population ageing on tuberculosis incidence. PLoS One. 2019; 14:e0222937. https://doi.org/10.1371/journal.pone.0222937 [PubMed]
- 6. Yen YF, Feng JY, Pan SW, Chuang PH, Su VY, Su WJ. Determinants of mortality in elderly patients with tuberculosis: a population-based follow-up study. Epidemiol Infect. 2017; 145:1374–81. https://doi.org/10.1017/S0950268817000152 [PubMed]
- 7. Flurkey K, Currer JM, Harrison DE, Fox JG. The mouse in biomedical research. American College of Laboratory Animal Medicine series. Elsevier, AP: Amsterdam. 2007; 637–72. https://primo.qatar-weill.cornell.edu/permalink/974WCMCIQ_INST/lflmnj/alma991000123789706691.
- 8. Cooper AM, Callahan JE, Griffin JP, Roberts AD, Orme IM. Old mice are able to control low-dose aerogenic infections with Mycobacterium tuberculosis. Infect Immun. 1995; 63:3259–65. https://doi.org/10.1128/iai.63.9.3259-3265.1995 [PubMed]
- 9. Turner J, Frank AA, Orme IM. Old mice express a transient early resistance to pulmonary tuberculosis that is mediated by CD8 T cells. Infect Immun. 2002; 70:4628–37. https://doi.org/10.1128/IAI.70.8.4628-4637.2002 [PubMed]
- 10. Vesosky B, Rottinghaus EK, Davis C, Turner J. CD8 T Cells in old mice contribute to the innate immune response to Mycobacterium tuberculosis via interleukin-12p70-dependent and antigen-independent production of gamma interferon. Infect Immun. 2009; 77:3355–63. https://doi.org/10.1128/IAI.00295-09 [PubMed]
- 11. Jeyakumar SM. Micronutrient Deficiency in Pulmonary Tuberculosis - Perspective on Hepatic Drug Metabolism and Pharmacokinetic Variability of First-line Anti- Tuberculosis Drugs: Special Reference to Fat-soluble Vitamins A, D, & E and Nutri-epigenetics. Drug Metab Lett. 2021; 14:166–76. https://doi.org/10.2174/1872312814999211130093625 [PubMed]
- 12. Silva-Cayetano A, Fra-Bido S, Robert PA, Innocentin S, Burton AR, Watson EM, Lee JL, Webb LM, Foster WS, McKenzie RC, Bignon A, Vanderleyden I, Alterauge D, et al. Spatial dysregulation of T follicular helper cells impairs vaccine responses in aging. Nat Immunol. 2023; 24:1124–37. https://doi.org/10.1038/s41590-023-01519-9 [PubMed]
- 13. Slight SR, Rangel-Moreno J, Gopal R, Lin Y, Fallert Junecko BA, Mehra S, Selman M, Becerril-Villanueva E, Baquera-Heredia J, Pavon L, Kaushal D, Reinhart TA, Randall TD et al. CXCR5+ T helper cells mediate protective immunity against tuberculosis. J Clin Invest. 2013; 123:712–26. https://doi.org/10.1172/JCI65728 [PubMed]
- 14. Buchalski B, Wood KD, Challa A, Fargue S, Holmes RP, Lowther WT, Knight J. The effects of the inactivation of Hydroxyproline dehydrogenase on urinary oxalate and glycolate excretion in mouse models of primary hyperoxaluria. Biochim Biophys Acta Mol Basis Dis. 2020; 1866:165633. https://doi.org/10.1016/j.bbadis.2019.165633 [PubMed]
- 15. Chen J, Deng JC, Zemans RL, Bahmed K, Kosmider B, Zhang M, Peters-Golden M, Goldstein DR. Age-induced prostaglandin E2 impairs mitochondrial fitness and increases mortality to influenza infection. Nat Commun. 2022; 13:6759. https://doi.org/10.1038/s41467-022-34593-y [PubMed]
- 16. Dibbern J, Eggers L, Schneider BE. Sex differences in the C57BL/6 model of Mycobacterium tuberculosis infection. Sci Rep. 2017; 7:10957. https://doi.org/10.1038/s41598-017-11438-z Erratum in: Sci Rep. 2018; 8:6354. DOI: 10.1038/s41598-018-24598-3 PMID: 28887521.
- 17. Gupta M, Srikrishna G, Klein SL, Bishai WR. Genetic and hormonal mechanisms underlying sex-specific immune responses in tuberculosis. Trends Immunol. 2022; 43:640–56. https://doi.org/10.1016/j.it.2022.06.004 [PubMed]
- 18. Hertz D, Dibbern J, Eggers L, von Borstel L, Schneider BE. Increased male susceptibility to Mycobacterium tuberculosis infection is associated with smaller B cell follicles in the lungs. Sci Rep. 2020; 10:5142. https://doi.org/10.1038/s41598-020-61503-3 [PubMed]
- 19. Klein SL, Flanagan KL. Sex differences in immune responses. Nat Rev Immunol. 2016; 16:626–38. https://doi.org/10.1038/nri.2016.90 [PubMed]
- 20. Márquez EJ, Chung CH, Marches R, Rossi RJ, Nehar-Belaid D, Eroglu A, Mellert DJ, Kuchel GA, Banchereau J, Ucar D. Sexual-dimorphism in human immune system aging. Nat Commun. 2020; 11:751. https://doi.org/10.1038/s41467-020-14396-9 [PubMed]
- 21. Orme IM. Aging and immunity to tuberculosis: increased susceptibility of old mice reflects a decreased capacity to generate mediator T lymphocytes. J Immunol. 1987; 138:4414–8. [PubMed]
- 22. Lafuse WP, Wu Q, Kumar N, Saljoughian N, Sunkum S, Ahumada OS, Turner J, Rajaram MVS. Psychological stress creates an immune suppressive environment in the lung that increases susceptibility of aged mice to Mycobacterium tuberculosis infection. Front Cell Infect Microbiol. 2022; 12:990402. https://doi.org/10.3389/fcimb.2022.990402 [PubMed]
- 23. Toapanta FR, Ross TM. Impaired immune responses in the lungs of aged mice following influenza infection. Respir Res. 2009; 10:112. https://doi.org/10.1186/1465-9921-10-112 [PubMed]
- 24. Lossow K, Kopp JF, Schwarz M, Finke H, Winkelbeiner N, Renko K, Meçi X, Ott C, Alker W, Hackler J, Grune T, Schomburg L, Haase H, et al. Aging affects sex- and organ-specific trace element profiles in mice. Aging (Albany NY). 2020; 12:13762–90. https://doi.org/10.18632/aging.103572 [PubMed]
- 25. Rowland JL, Niederweis M. Resistance mechanisms of Mycobacterium tuberculosis against phagosomal copper overload. Tuberculosis (Edinb). 2012; 92:202–10. https://doi.org/10.1016/j.tube.2011.12.006 [PubMed]
- 26. Sakiyama H, Fujiwara N, Yoneoka Y, Yoshihara D, Eguchi H, Suzuki K. Cu, Zn-SOD deficiency induces the accumulation of hepatic collagen. Free Radic Res. 2016; 50:666–77. https://doi.org/10.3109/10715762.2016.1164856 [PubMed]
- 27. Cercamondi CI, Stoffel NU, Moretti D, Zoller T, Swinkels DW, Zeder C, Mhimibra F, Hella J, Fenner L, Zimmermann MB. Iron homeostasis during anemia of inflammation: a prospective study of patients with tuberculosis. Blood. 2021; 138:1293–303. https://doi.org/10.1182/blood.2020010562 [PubMed]
- 28. Orme IM, Griffin JP, Roberts AD, Ernst DN. Evidence for a defective accumulation of protective T cells in old mice infected with Mycobacterium tuberculosis. Cell Immunol. 1993; 147:222–9. https://doi.org/10.1006/cimm.1993.1062 [PubMed]
- 29. Friedman A, Turner J, Szomolay B. A model on the influence of age on immunity to infection with Mycobacterium tuberculosis. Exp Gerontol. 2008; 43:275–85. https://doi.org/10.1016/j.exger.2007.12.004 [PubMed]
- 30. Crotty S. T Follicular Helper Cell Biology: A Decade of Discovery and Diseases. Immunity. 2019; 50:1132–48. https://doi.org/10.1016/j.immuni.2019.04.011 [PubMed]
- 31. Kim HJ, Verbinnen B, Tang X, Lu L, Cantor H. Inhibition of follicular T-helper cells by CD8(+) regulatory T cells is essential for self tolerance. Nature. 2010; 467:328–32. https://doi.org/10.1038/nature09370 [PubMed]
- 32. Yu D, Ye L. A Portrait of CXCR5+ Follicular Cytotoxic CD8+ T cells. Trends Immunol. 2018; 39:965–79. https://doi.org/10.1016/j.it.2018.10.002 [PubMed]
- 33. Sage PT, Tan CL, Freeman GJ, Haigis M, Sharpe AH. Defective TFH Cell Function and Increased TFR Cells Contribute to Defective Antibody Production in Aging. Cell Rep. 2015; 12:163–71. https://doi.org/10.1016/j.celrep.2015.06.015 [PubMed]
- 34. Swanson RV, Gupta A, Foreman TW, Lu L, Choreno-Parra JA, Mbandi SK, Rosa BA, Akter S, Das S, Ahmed M, Garcia-Hernandez ML, Singh DK, Esaulova E, et al. Antigen-specific B cells direct T follicular-like helper cells into lymphoid follicles to mediate Mycobacterium tuberculosis control. Nat Immunol. 2023; 24:855–68. https://doi.org/10.1038/s41590-023-01476-3 [PubMed]
- 35. Patel M, Yarlagadda V, Adedoyin O, Saini V, Assimos DG, Holmes RP, Mitchell T. Oxalate induces mitochondrial dysfunction and disrupts redox homeostasis in a human monocyte derived cell line. Redox Biol. 2018; 15:207–15. https://doi.org/10.1016/j.redox.2017.12.003 [PubMed]
- 36. Headley CA, Gautam S, Olmo-Fontanez A, Garcia-Vilanova A, Dwivedi V, Akhter A, Schami A, Chiem K, Ault R, Zhang H, Cai H, Whigham A, Delgado J, et al. Extracellular Delivery of Functional Mitochondria Rescues the Dysfunction of CD4+ T Cells in Aging. Adv Sci (Weinh). 2024; 11:e2303664. https://doi.org/10.1002/advs.202303664 [PubMed]
- 37. Headley CA, Gautam S, Olmo-Fontanez A, Garcia-Vilanova A, Dwivedi V, Schami A, Weintraub S, Tsao PS, Torrelles JB, Turner J. Mitochondrial Transplantation promotes protective effector and memory CD4+ T cell response during Mycobacterium tuberculosis infection and diminishes exhaustion and senescence in elderly CD4+ T cells. Adv Sci (Weinh). 2024; 11:e2401077. https://doi.org/10.1002/advs.202401077 [PubMed]