Introduction
Extracellular vesicles (EVs), including exosomes and microvesicles, are now acknowledged to play important roles in a number of disease processes such as cancer metastasis and neurodegeneration [1,2]. The potential for EVs to transport cargo of microRNAs (miRNAs), lipids and proteins between cell populations suggests that these membrane-bound particles may be involved in crosstalk among different organ systems [3,4]. Such crosstalk is likely to underlie degenerative changes that can occur in multiple organ systems with aging and disease. For example, adipose tissue is recognized as a secretagogue for a number of inflammatory cytokines that can negatively impact multiple tissues [5], and muscle is a source of myokines that can positively impact various organs and tissues following exercise [6]. How EVs fit into this picture is not entirely clear, but EVs have been isolated from both adipocytes [7] and myotubes [8] suggesting that they may be able to function in a manner similar to adipokines and myokines, respectively. In support of this idea muscle-derived EVs have been detected in human serum, and their miRNA cargo is altered with physical activity [9].
Recently it has been discovered that cells undergoing senescence may secrete factors that can impact neighboring cells and tissues, factors collectively referred to as the senescence-associated secretory phenotype or SASP [10]. Most studies to date on the SASP have focused on inflammatory factors that are secreted by senescent cells such as IL-6 and IL-1β. It has, however, been proposed that EVs may be part of the SASP, and EVs have been identified that are secreted from senescent cells such as those cells in senescent lung [11]. The SASP is linked to age-related organ dysfunction since aging is associated with an accumulation of senescent cells. Yet the specific SASP-related factors carried by EVs with aging have not yet been defined, and their role in tissue crosstalk has not yet been explored. Aging is known to be accompanied by loss of muscle mass, a phenomenon referred to as sarcopenia [12,13]. Here we characterize EVs released from aged muscle in vivo, and the miRNAs secreted by muscle cells in response to age-associated stimuli in vitro. We show that muscle-derived EVs carrying senescence-associated miRNAs can induce cellular senescence in bone stem cells, revealing a potential mode of inter-organ crosstalk that may contribute directly to tissue dysfunction with aging.
Results
miR-34a expression is significantly increased with age in mouse skeletal muscle and in serum extracellular vesicles
Aging, inflammation, and oxidative stress are associated with increases in several miRNAs including miR-34a, miR-141, miR-155, and miR-183 which in turn target genes involved in cell survival such as Bcl2, Sirt1, FGF7, and Foxo1 among others [14–18]. We therefore analyzed expression of these miRNAs in skeletal muscle in serum EVs derived from young and aged male and female mice. Skeletal muscle from male and female aged mice showed a significant increase in miR-34a, whereas miR-141 was not significantly increased in male or female muscle and miR-155 and miR-183 were only increased in muscle from female mice (Figure 1A). EVs from serum of aged mice showed a significant increase in miR-34a in both males and females, whereas miR-155 was only increased in EVs from female mice (Figure 1B). EVs from male and female mice showed no marked increase in miR-183 or miR-141. Bivariate correlations indicate a significant (P<.001) association between muscle and serum EV miR-34a fold-change values (r=.66).
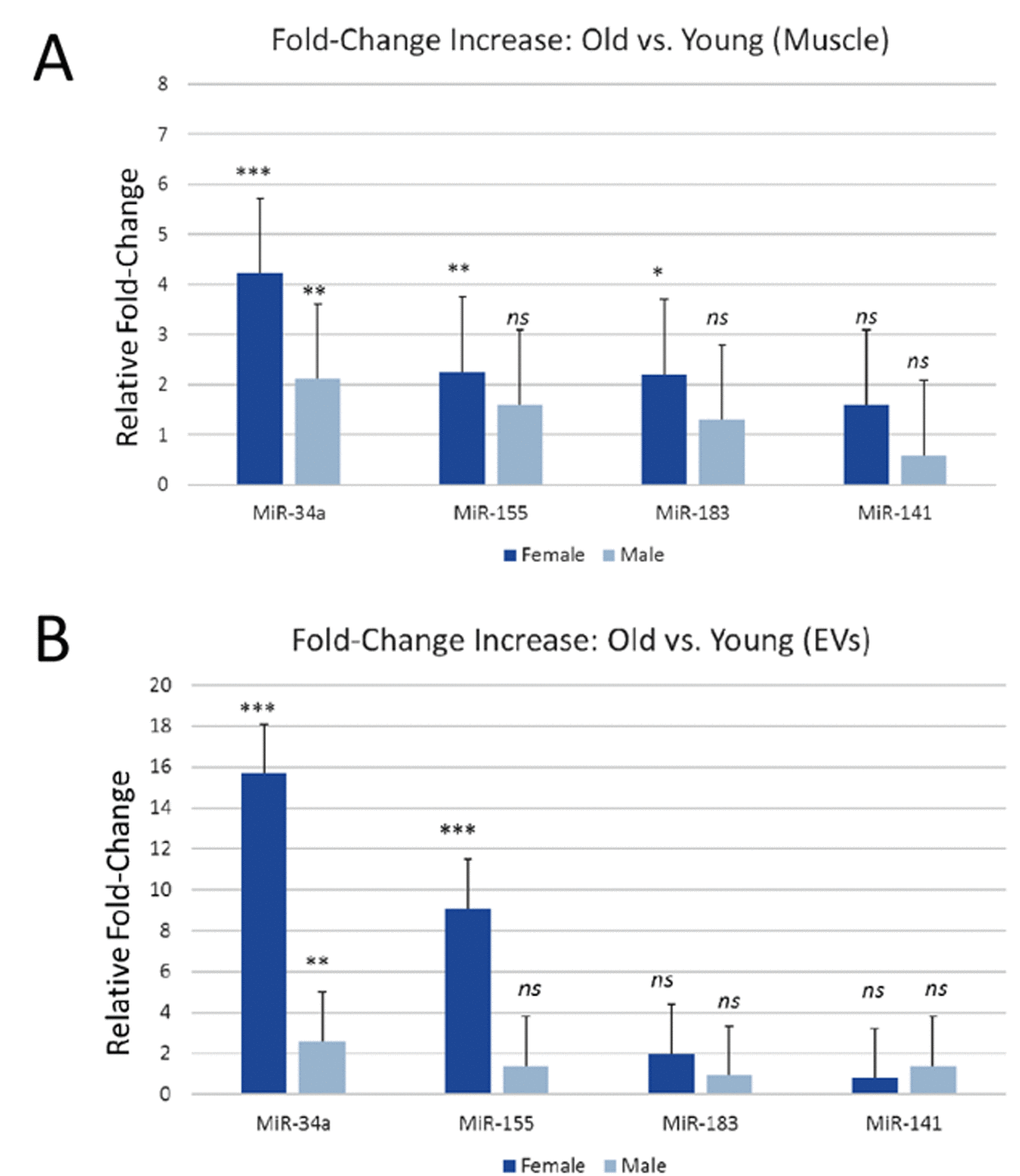
Figure 1. miR-34a is increased in mouse skeletal muscle and in serum EVs with age. (A) Expression levels of miR-34a, miR-155, miR-183, and miR-141 in skeletal muscle of young and aged male and female mice. (B) Expression levels of miR-34a, miR-155, miR-183, and miR-141 in serum EVs from young and aged male and female mice. *P<.05, **P<.01, ***P<.001.
Muscle-derived extracellular vesicles show increased miR-34a expression with age
We isolated EVs from serum in young and aged mice and then used immunoprecipitation for alpha-sarcoglycan (SGCA) to isolate muscle-derived EVs [9]. Representative particle distributions from nanoparticle tracking data show that particle sizes are similar between serum-derived EVs and SGCA+ EVs, and the particle diameters are less than <100 nm consistent with the known size of exosomes (Figure 2A). Nanoparticle tracking data indicate that SGCA+ EVs represent slightly less than 10% of EVs in serum (Figure 2B), consistent with previous findings using human serum [9]. SGCA+ EVs are significantly less abundant in bone marrow interstitial fluid compared to serum (Figure 2B). ANOVAs show no significant age effect on EV concentration, and there is a significant interaction between EV source and isolation approach, with SGCA+ EVs being significantly lower in marrow than in serum (Table 1). Real-time PCR data on SGCA+ EVs from serum indicate that miR-34a is significantly increased in SGCA+ EVs from aged mice compared to young mice (Figure 2C).
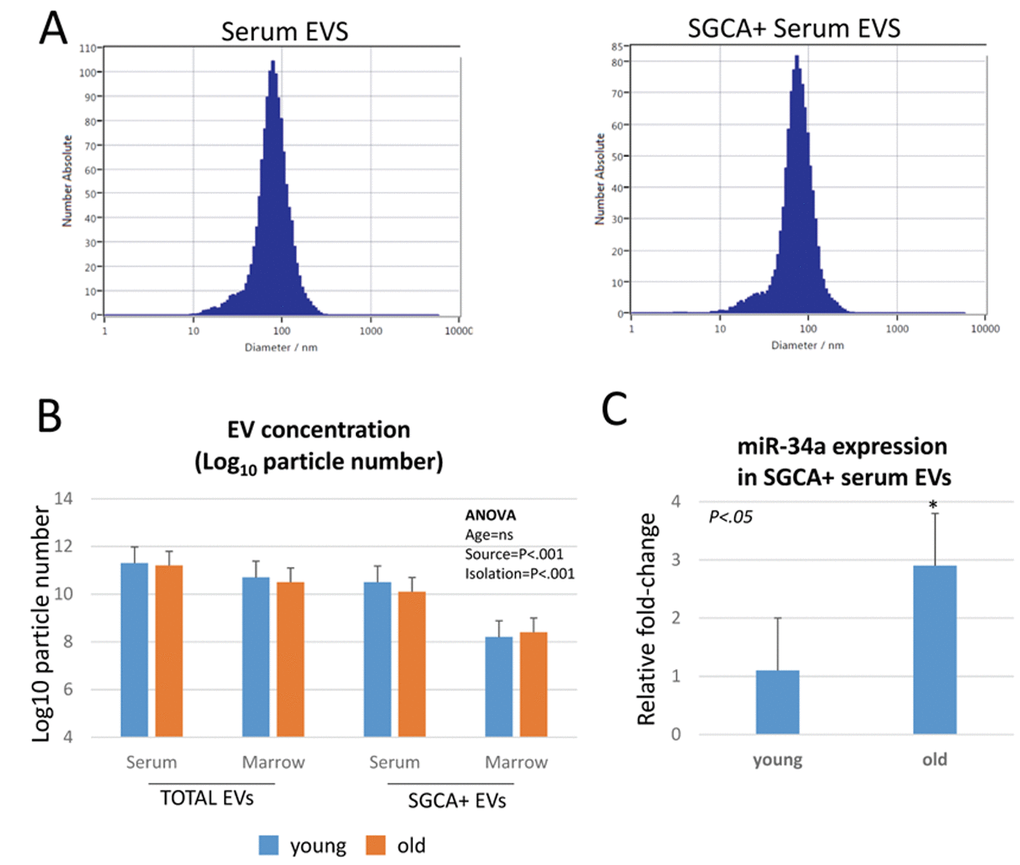
Figure 2. miR-34a is increased with age in muscle-derived, alpha-sarcoglycan positive (SGCA+) EVs. (A) Representative particle distribution histograms from nanoparticle tracking analysis showing similar particle sizes between serum EVs and SGCA+ serum EVs. (B) Particle concentrations of SGCA+ EVs are significantly lower than total serum EVs, and SGCA+ EVs in bone marrow are significantly less abundant than SGCA+ EVs in serum. (C) SGCA+ EVs show a significant increase in miR-34a expression with age.
Table 1. ANOVA results from nanoparticle tracking analysis of EV concentration (log-transformed values. Note significant interaction between source (serum or marrow) and isolation technique (normal or SGCA+).
| Source | Type III Sum of Squares | df | Mean Square | F | Sig. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Corrected Model | 172.153a | 7 | 24.593 | 10.496 | .000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 189.328 | 1 | 189.328 | 80.799 | .000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AGE | 2.532 | 1 | 2.532 | 1.080 | .306 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SOURCE | 45.066 | 1 | 45.066 | 19.233 | .000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ISOLATION | 92.315 | 1 | 92.315 | 39.397 | .000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AGE * SOURCE | 2.403 | 1 | 2.403 | 1.026 | .318 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AGE * ISOLATION | 1.974 | 1 | 1.974 | .843 | .365 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SOURCE * ISOLATION | 28.930 | 1 | 28.930 | 12.346 | .001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AGE * SOURCE * ISOLATION | .202 | 1 | .202 | .086 | .771 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Error | 82.012 | 35 | 2.343 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total | 390.383 | 43 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Corrected Total | 254.165 | 42 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| a. R Squared = .677 (Adjusted R Squared = .613) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Oxidative stress increases miR-34a in myoblast- and myotube-derived EVs, and these EVs can induce cellular senescence in bone stem cells
Reactive oxygen species are increased in skeletal muscle with aging, and as shown above muscles from aged mice show elevated levels of miR-34a and muscle-derived EVs from aged mice also have increased levels of miR-34a. We tested the hypothesis that oxidative stress might increase the production of EVs enriched in miR-34a in vitro by treating mouse C2C12 myoblasts and primary human myotubes with hydrogen peroxide. Hydrogen peroxide significantly increased levels of miR-34a in EVs isolated from the conditioned medium (Figure 3A, B). We then analyzed the bioactivity of these EVs by treating bone marrow mesenchymal (stromal) cells (BMSCs) from young adult mice. We utilized BMSCs for this system because aging is characterized by significant reduction in the population of BMSCs [19]. EVs from C2C12 cells exposed to hydrogen peroxide significantly decreased cell viability (Figure 3C) and increased cellular senescence (Figure 3D) compared to EVs from C2C12 cells not exposed to hydrogen peroxide.
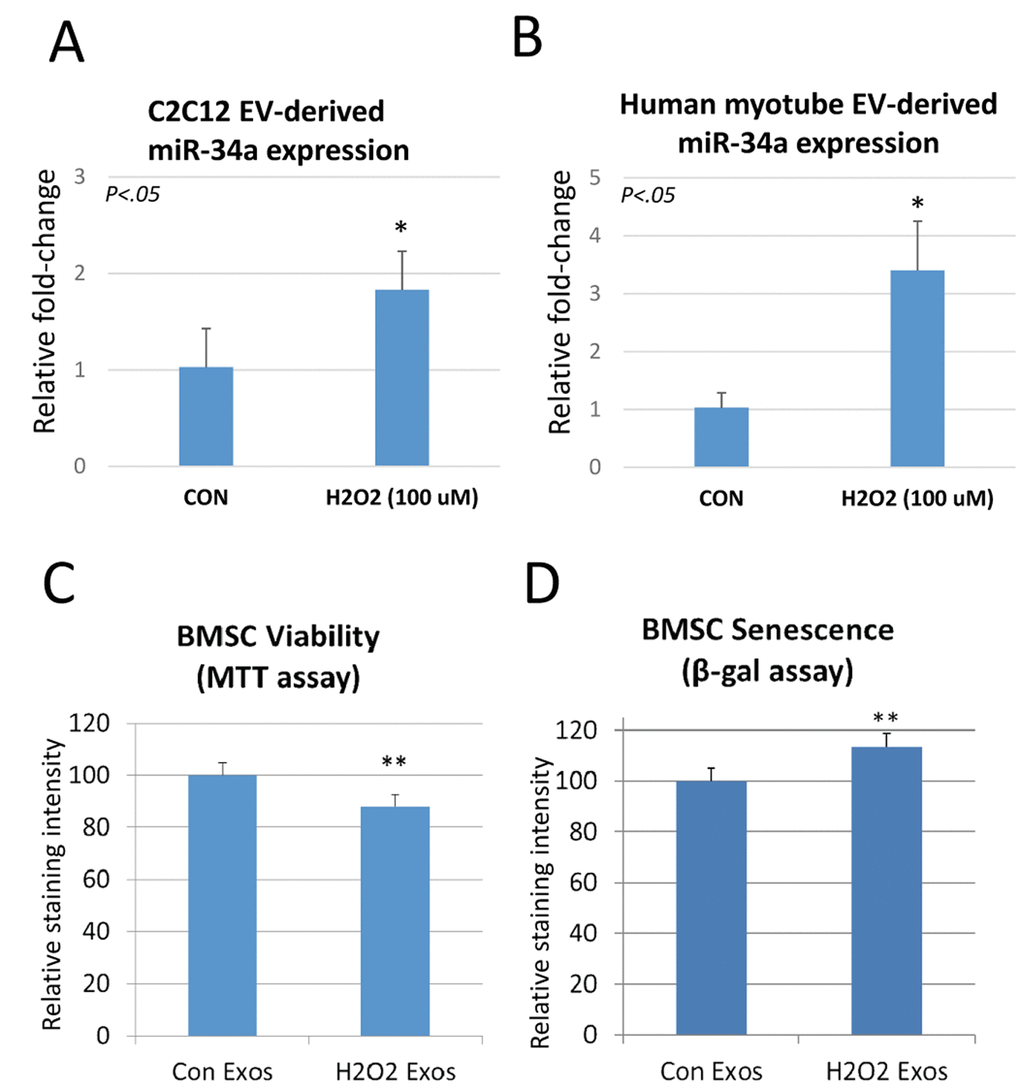
Figure 3. Hydrogen peroxide increases miR-34a in EVs secreted by C2C12 myoblasts and human myotubes, and these EVs can reduce bone stem cell (BMSC) viability and increase senescence. (A) EVs isolated from C2C12 cells treated with hydrogen peroxide show a significant increase in miR-34a. (B) EVs isolated from human myotubes treated with hydrogen peroxide show a significant increase in miR-34a. (C) BMSC viability indicated by MTT assay is significantly reduced after treatment with EVs isolated from these C2C12 cells exposed to hydrogen peroxide. (D) BMSC senescence measured by beta-galactosidase (β-gal) assay is significantly increased after treatment with EVs isolated from these C2C12 cells exposed to hydrogen peroxide. *P<.05, **P<.01.
EVs from cells overexpressing miR-34a can decrease BMSC viability and increase cellular senescence
We utilized a lentiviral vector system to overexpress miR-34a in C2C12 cells tagged to green-fluorescent protein. Validation of the system using confocal imaging shows that miR-34a is highly expressed in cells transfected with the lentivirus (Figure 4A). EVs isolated from conditioned medium of C2C12 cells overexpressing miR-34a show a three-fold increase in miR-34a compared to EVs from untransfected cells (Figure 4B). Confocal imaging of BMSCs treated with PKH67-labeled EVs from C2C12 cells overexpressing miR-34a show that BMSCs readily take up these labeled vesicles (Figure 5A). Quantitative analysis of BMSC viability and senescence shows that EVs from miR-34a overexpressing C2C12 cells significantly decrease cell viability and increase cellular senescence (Figure 5B, C).
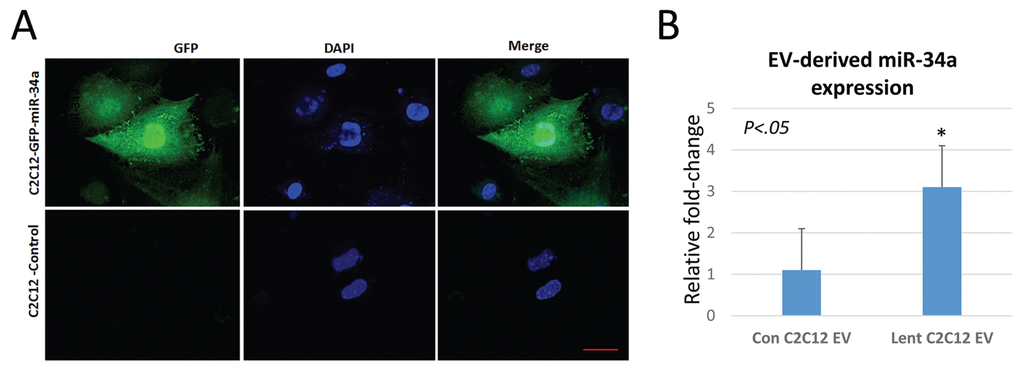
Figure 4. C2C12 cells overexpressing miR-34a secrete EVs with elevated levels of miR34a. (A) Confocal images of C2C12 cells transfected with a lentivirus overexpressing miR-34a. The virus contains a GFP reporter under a constitutive CMV promoter. Images show GFP expression in transfected cells. Blue staining represents nuclear DAPI staining. Scale bar = 20 µm. (B) Analysis of miR-34a expression in EVs from transfected and non-transfected cells shows a three-fold increase in miR-34a in EVs isolated from conditioned medium of transfected cells.
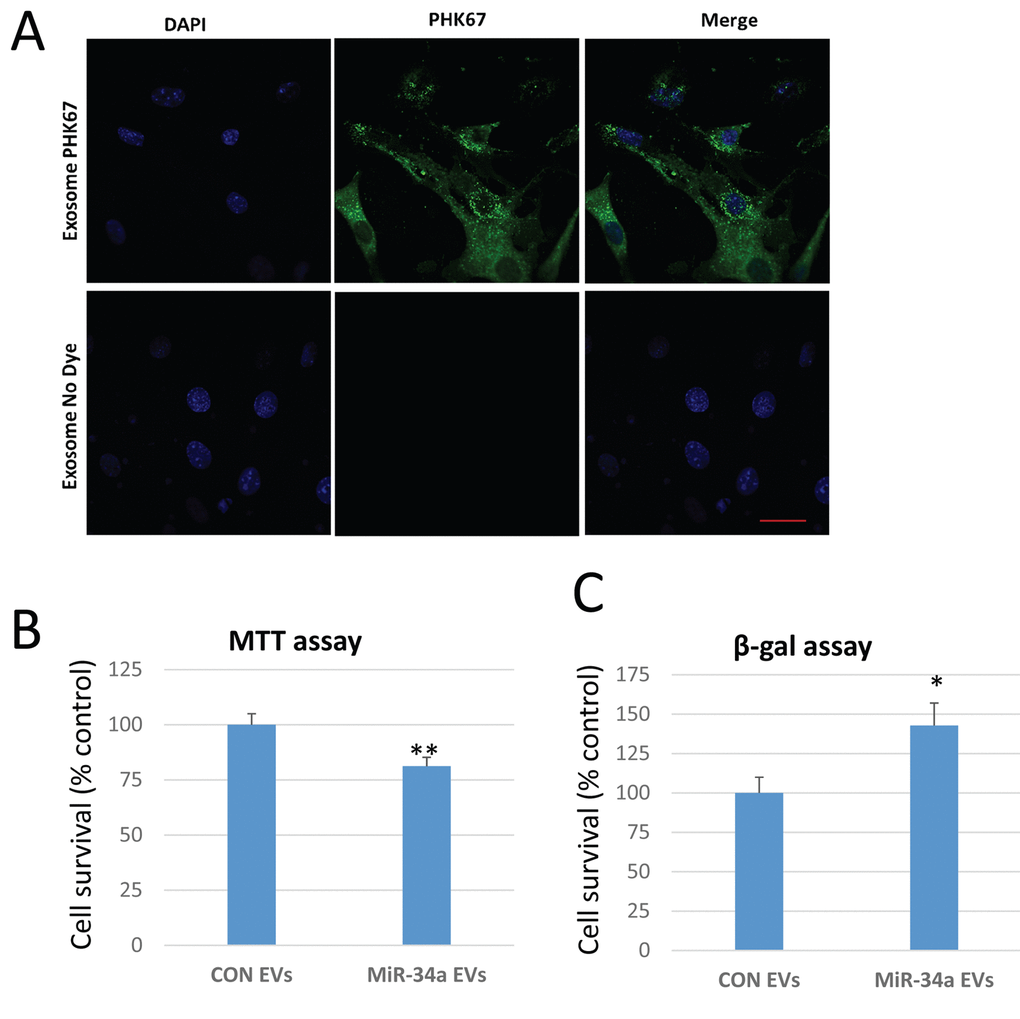
Figure 5. EVs from C2C12 cells overexpressing miR-34a reduce BMSC viability and increase senescence. (A) Confocal images of BMSCs treated with EVs isolated from conditioned medium of C2C12 cells overexpressing miR-34a. EVs are unlabeled (control, bottom row) or labeled with the membrane dye PKH67 (top row). Images show abundant EVs in cytoplasm of BMSCs. Blue staining represents nuclear DAPI staining. Scale bar = 20 µm. (B) BMSC viability indicated by MTT assay is significantly reduced after treatment with EVs isolated from C2C12 cells overexpressing miR-34a. (C) BMSC senescence measured by beta-galactosidase (β-gal) assay is significantly increased after treatment with EVs isolated from these C2C12 cells overexpressing miR-34a. *P<.05, **P<.01.
EVs from cells overexpressing miR-34a home to bone marrow in vivo and decrease Sirt1 expression ex vivo
Previous work suggests that EVs derived from C2C12 cells can travel to a variety of organs in vivo, including the limb [20]. We tested the hypothesis that EVs enriched in miR-34a may reach the bone marrow microenvironment in vivo by labeling these EVs with the infrared dye DiR and then imaging mice 24 hours after tail vein injection. Images show that DiR-labeled vesicles are detected in both the fore- and hindlimb (Figure 6A). We flushed primary bone marrow cells from the limb bones of untreated mice and cultured these cells ex vivo in the presence of EVs derived from miR-34a overexpressing cells or with EVs from untransfected C2C12 cells. We then analyzed expression for Sirt1, since miR-34a is known to target Sirt1 in BMSCs [18] and Sirt1 plays a key role in cell survival [21]. Analysis of mRNA levels and protein show that EVs from miR-34a overexpressing C2C12 significantly reduced Sirt1 mRNA and protein in primary bone marrow cells (Figure 6B, C).
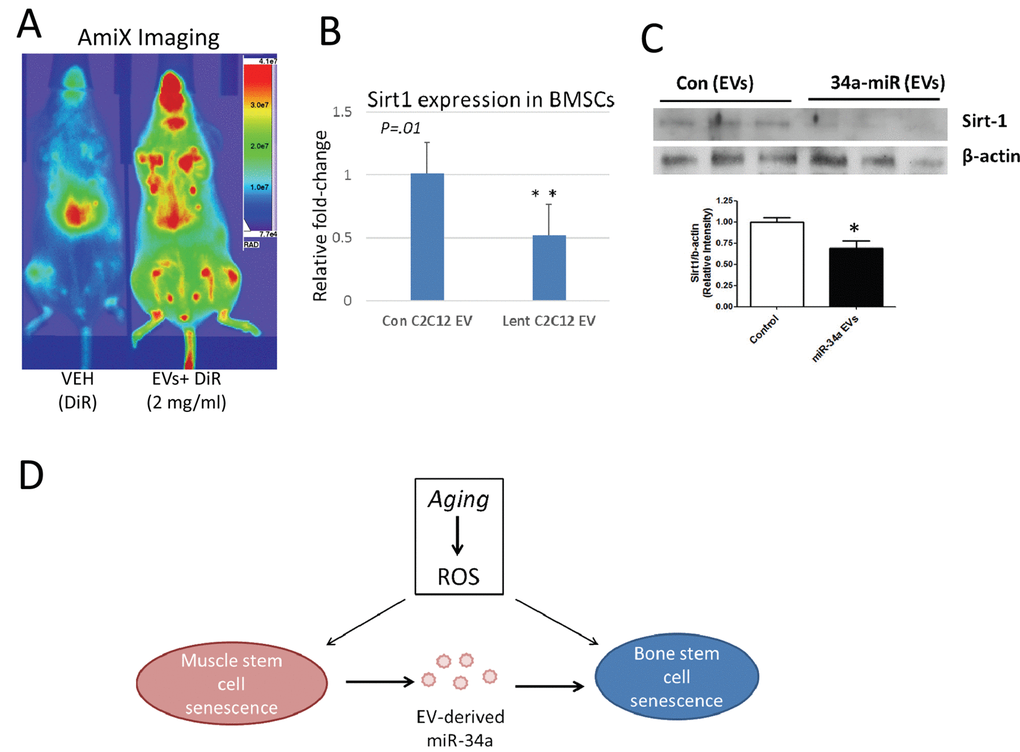
Figure 6. EVs from C2C12 cells overexpressing miR-34a home to bone marrow in vivo and reduce Sirt1 expression ex vivo. (A) Mice were injected via tail vein with DiR dye alone (VEH) or EVs from C2C12 cells overexpressing miR-34a labeled with DiR (EVs + DiR) and imaged with AmiX imaging. Mice receiving labeled EVs show high image intensity in the metaphyseal regions of long bones. (B) Bone marrow cells flushed from untreated mice and cultured in the presence of EVs from miR-34a overexpression cells show reduced Sirt1 expression compared to cells cultured with EVs from control C2C12 cells. (C) Bone marrow cells flushed from untreated mice and cultured in the presence of EVs from miR-34a overexpression cells show reduced Sirt1 protein compared to cells cultured with EVs from control C2C12 cells. Top image is from protein of adherent cells, graph includes pooled data from both adherent and non-adherent cells. *P<.05, **P<.01. (D) Working model summarizing changes in muscle and bone with age, and the role of EV-derived miR-34a in muscle and bone senescence. ROS = reactive oxygen species.
Discussion
Previous work suggests that extracellular vesicles may play important roles in the aging process. Changes in circulating EV cargo, such as miRNAs, proteins, and lipids characterize older versus younger populations, and many of these alterations in EV cargo are thought to reflect cell- and tissue-level changes in response to age-associated stimuli such as inflammation and oxidative stress [22–25]. Our work provides additional support for this concept, and indicates that elevated oxidative stress in skeletal muscle with age is associated with changes in the miRNA cargo of muscle-derived EVs. The in vivo data are corroborated by in vitro experiments showing the exposure of muscle cells to oxidative stress significantly alters the miRNA profile of secreted EVs. These findings are consistent with previous work demonstrating that myoblasts and myotubes actively secrete EVs in vitro [8,26–29], that muscle-derived EVs can be detected in the circulation in vivo [9], and that the cargo of muscle-derived EVs is altered by changes in the systemic environment [4,9].
We evaluated age-associated changes in several miRNAs with aging, and found that miR-34a-5p is significantly increased in muscle and in muscle-derived EVs with aging and with myoblast exposure to oxidative stress. These results support previous findings that miR-34a is increased with aging in the circulation [30] and in skeletal muscle [31], and that its elevated expression is associated with muscle atrophy and myopathy [31,32]. miR-34a is regulated at least in part by p53 [18] and its increased expression with aging is consistent with a role in for miR-34a in cell death. This role for miR-34a is further indicated by the capacity of miR-34a to promote DNA damage and inhibit DNA repair [33]. miR-34a is known to induce senescence in vascular smooth muscle cells and cardiomyocytes and to increase cardiac fibrosis [34,35]. miR-34a knockout attenuates cognitive deficits in APP/PS1 mice [36] and calorie restriction promotes cell survival in the mouse brain by downregulating miR-34a [37]. Together, these studies combined with our new data support the notion that increased miR-34a expression with aging in skeletal muscle is likely associated with elevated muscle damage. It is thought that a primary mechanism by which miR-34a mediates senescence and cell survival is by suppressing the pro-survival deacetylase Sirt1. Sirt1 is downregulated and miR-34a increased in various cell types with aging [38], Sirt1 is a well-established target of miR-34a [18], and decreased Sirt1 activity in muscle with aging is associated with impaired muscle performance [39]. Interestingly we also observed sex differences in miR-34a expression, with females showing a greater increase in miR-34a expression with age than males. A similar difference has been noted in mouse cardiac tissue, with females showing greater miR-34a expression than males [40]. There are well-documented sex differences in microRNA expression in a variety of cells and tissues [41], and in mice females show a more marked decline in grip strength and muscle absolute force with aging than male mice [42,43]. These sex-specific differences may explain the greater miR-34a expression with aging that we observed in female mouse muscle and in EVs.
It is well-known that crosstalk exists between muscle and bone, and that muscle can secrete a number of factors that can promote bone formation and suppress bone resorption [6,44,45]. Sirt1 has positive effects on bone mass through its activation of the osteoblast transcription factor Runx2 and its inhibition of NF-κB signaling [46,47]. miR-34a has detrimental effects on bone by suppressing bone formation by osteoblasts [48] and by decreasing survival of BMSCs [21,49] which are precursors of osteoblasts. Our data are consistent with these studies and indicate that muscle-derived EVs carrying miR-34a can reduce Sirt1 expression in BMSCs and also induce BMSC senescence. These results may represent a novel pathway by which muscle can influence bone physiology (Figure 6D). The majority of studies to date on muscle-bone crosstalk have revealed positive effects of muscle-derived factors on bone [44,45]. The data presented here suggest that the accumulation of reactive oxygen species in skeletal muscle with aging can potentially have negative effects on bone due to circulating, muscle-derived EVs carrying miR-34a (Figure 6D). Future studies may be directed at identifying other factors carried by muscle-derived EVs and determining their effects on various organs and tissues such as bone, liver, and adipose tissue. miR34a can target other factors important for muscle and bone repair and regeneration such as Notch, Jagged1 and Numb [18], and it is possible that the negative, age-associated effects of EV-derived miR-34a may be mediated by suppression of these factors in addition to Sirt1.
The in vivo and in vitro experiments performed here indicate that hydrogen peroxide stimulates and increase in EV-derived miR-34a. Previous studies have shown that, in general, overexpression of a particular miRNA in a cell is linked with greater abundance of that miRNA in secreted EVs [50]. A limitation of our study is that we have not yet performed mechanistic analyses linking ROS exposure to EV packaging and secretion. We hypothesize that the release of specific miRNAs within EVs is a mechanism for cell survival. Previous authors have proposed that EV release is a process that removes harmful materials from the cell cytoplasm such as accumulated nuclear DNA or lipids such as ceramides [51,52]. We expect that cellular stressors that occur with aging, such as exposure to inflammatory factors and/or reactive oxygen species, increase miR-34a expression as a by-product of p53 activation. This is known to occur in the setting of sepsis, injury and inflammation [53]. Removal of stress- and senescence-associated miRNAs such as miR-34a via extracellular vesicle secretion would be a mechanism to promote cell survival; however, it is likely that increasing the circulating concentration of such “inflammiRs” will ultimately have detrimental effects on neighboring cells and tissues by delivering these inflammiRs via EV membrane fusion and endocytosis. Interventions such as physical activity and dietary restriction may ultimately reduce the systemic burden of these stress- and senescence-associated miRNAs carried within circulating EVs and in turn reduce cell- and tissue-level damage with aging.
Materials and Methods
Sample collection and preparation from young and old mice
C57BL/6 mice were obtained from the National Institute on Aging at 6 and 24 months of age, approximately 5-6 males and 5-6 females per age group. Mice were euthanized by CO2 overdose followed by thoracotomy as approved by the Augusta University IACUC. Tibialis anterior muscles were dissected free and snap frozen in liquid nitrogen for miRNA isolation using the miRNeasy kit (Qiagen) following manufacturer specifications. Blood was collected via cardiac puncture and serum isolated by clotting for 1 hr and then centrifugation for 15 min at 5,000 rpm.
EV isolation and nanoparticle tracking analysis
EVs were isolated from serum and flushed bone marrow interstitial fluid from the femur [19] using 8% PEG as described by Rider and colleagues [54]. Particle size and concentration were quantified using the Zetaview instrument from Particle Matrix [55,56]. Immunoprecipitation for muscle-derived EVs was performed using an antibody specific for muscle alpha-sarcoglycan (SGCA) following ref [9]. In brief, we used the Expedeon Lightning-Link® Rapid Biotin – Type B kit to label the SGCA monoclonal antibody (IVD3(1)A9) from the Developmental Studies Hybridoma Bank. Immunoselection was performed using the Pierce™ Streptavidin Plus UltraLink™ Resin kit following manufacturer specifications. The EVs were initially mixed with prepared streptavidin resin for an hour to remove any nonspecific binding elements. EVs were removed and mixed with pretreated biotinylated SGCA-streptavidin resin for overnight incubation. After incubation the sample was spun down and supernatant removed. The resin was then treated with 0.1M glycine-HCl (pH 2.5-3.0) and supernatant collected after spin down and acidic conditions neutralized with Tris-HCL (pH 8.0). miRNAs were isolated from serum EVs as well as from SGCA+ serum EVs. Western blot for the exosomal markers CD63 (Santa Cruz #sc-15363) and TSG101 (Santa Cruz #sc-7964) was performed following standard procedures (Figure 7A). We have submitted all relevant data of our experiments to the EV-TRACK knowledge base (EV-TRACK ID: EV180077; http://evtrack.org/).
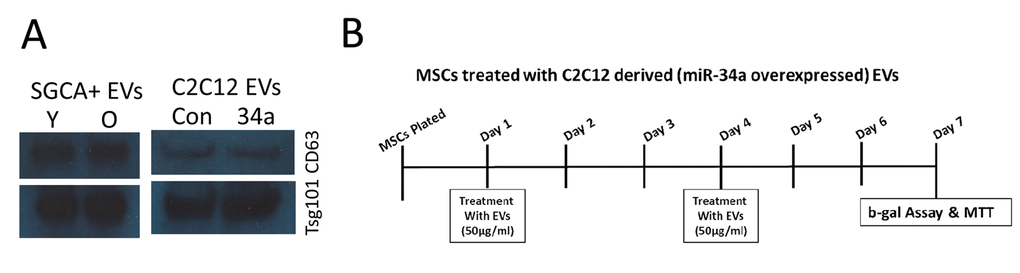
Figure 7. Western blot for exosome markers and experimental design for in vivo treatment. (A) SGCA+ EVs isolated from serum of young (Y) and old (O) mice as well as from conditioned medium of normal C2C12 cells and cells overexpressing miR-34a are positive for the exosome markers CD63 and TSG101. (B) Timeline, EV doses, and outcome measures for in vitro BMSC treatments.
Analysis of miRNA expression in skeletal muscle and EVs
The concentration of miRNA from skeletal muscle samples and EVs was determined using a NanoDrop spectrophotometer (Thermo Scientific). Specifically, we analyzed expression of miR-34a-5p, miR-155-5p, miR-183-5p, and miR-141-3p using primers from Qiagen. Two hundred nanograms of enriched miRNAs were converted into cDNA using miScript II RT Kit (from Qiagen). Fifty pictograms of cDNA were amplified in each qRT-PCR using SYBR Green I and miR-specific primers (Qiagen, Valencia, CA, USA). The real-time qRT-PCR was performed on a Bio-Rad MyiQ machine with following cycling parameters: 95 °C for 10 mins, then 40 cycles of 95 °C for 15 s, 60 °C for 30 s and 72 °C for 30 s. The average of RNU6 and SNORD was used as normalization reference genes for miRs. Relative expression of miRNA was evaluated by using the comparative CT method (ΔΔCt).
In vitro studies utilizing C2C12 myoblasts overexpressing miR-34a
We used a lentiviral system to overexpress miR-34a in mouse C2C12 myoblasts as a mechanism to produce exosomes enriched in miR-34a. The lentiviral particles were purchased from Genecopoeia (PP-MmiR3342-MR03), Polybrene® (sc-134220) and puromycin (sc-108071) was purchased from Santa Cruz Biotechnology, Inc. USA. In brief, C2C12 cells were plated at 30–50% confluence and transfected with appropriate dilutions of lentivirus particles and polybrene. Forty-eight hours after transfection, the cells were cultured in growth medium containing puromycin (2 μg/ml) to obtain the stable, transfected C2C12 cells. The efficiency of overexpression of miR-34a in C2C12 cells EVs was analyzed by fluorescence imaging real-time PCR (Figure 4). Primary mouse BMSCs were isolated from young adult mice as previously described [19,57]. BMSCs were treated with EVs (50 µg/ml) isolated from normal C2C12 cells or cells overexpressing miR-34a at day 1 and day 4 of culture (Figure 7B). At day 7 cells were analyzed using MTT assay (Fisher) to assess cell viability and β-galactosidase assay (Enzo Life Sciences) to measure cellular senescence following manufacturer specifications. Uptake of EVs was verified using PKH67 labeling of EVs followed by confocal imaging.
In vitro studies utilizing EVs from C2C12 cells and human myotubes exposed to hydrogen peroxide
C2C12 cells were prepared as described above, treated with H2O2 (100µM) for 24hrs, and EVs isolated as described for cells overexpressing miR-34a. C2C12 (ATCC® CRL-1772™) and Human Skeletal Myoblast (HSkM) cells (Gibco, catalog#A1140) were grown to 80% confluence in growth media for 2 days followed by myotube differentiation. Myotube differentiation is initiated upon reaching confluence by switching the cells to medium containing 2% horse serum for 5-6 days. Then, cells were treated with hydrogen peroxide (H2O2) in 1% exo-free media for 24hrs. Exosomes were isolated from cell culture supernatants as per published method. BMSCs cells were plated separately for MTT assay and β-galactosidase assay at an initial density of 3000/cm2 in 96-well plates using 5% FBS to support overnight attachment. The following day, cells were treated with control and H2O2 derived EVs (50μg/ml) separately with 1% FBS (exosome free) media for 48 hrs. MTT and β-galactosidase assays were performed as described above.
In vivo tracking of C2C12-derived EVs and ex vivo treatment of bone marrow cells
EVs isolated from conditioned medium of C2C12 cells were labeled labeled with the membrane dye DiR (Xenolight, Perkin Elmer) following manufacturers recommendations. DiR is a near-infrared lipophilic dye that binds the lipid bilayer surrounding EVs [20]. Its emission spectra is only visible using infrared CCD imaging. We injected 100 µl of DiR dye alone or DiR-labeled EVs at 1.0 mg/ml and then imaged (Ex =710, Em=790) 24 hrs later using an Ami X spectral imaging instrument. Bone marrow cells were flushed from long bones of young adult mice and adherent and non-adherent cells cultured and treated (50µg/ml EVs) with normal C2C12 EVs and miR-34a overexpressed EVs for 18 hrs. The cell lysate was prepared for western blot analysis as per our published method [57]. In brief, protein was extracted from cell culture lysate, subjected to SDS-PAGE, and transferred to nitrocellulose membranes. Membranes were incubated with a polyclonal antibody against Sirt1 (Millipore Anti-Sirt1 Cat # 07-131), and beta-actin (Santa Cruz Biotechnology, Santa Cruz, CA) overnight at 4 ºC, followed by incubation with appropriate secondary antibody. Proteins were visualized with an ECL Western blot detection system (Thermo Scientific, Waltham, MA).
Statistical analysis
ANOVA was used to compare fold changes in miRNAs between and among different age groups (young vs old), genders (male vs female), and source (muscle vs EVs). Students t-tests were performed for in vitro experiments using treated vs control designs.
Author Contributions
SF, CMI, and MH conceived of the experimental design and prepared the manuscript. BM performed MTT and β-gal staining, and maintained C2C12 cells in culture. AK performed EV immunoprecipitation and HK performed Amplex Red assays. YL performed nanoparticle tracking studies and MJ analyzed nanoparticle tracking and PCR data.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
Funding for this research was provided by the National Institute on Aging, US National Institutes of Health (AG 036675).
References
- 1. Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, Molina H, Kohsaka S, Di Giannatale A, Ceder S, Singh S, Williams C, Soplop N, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015; 527:329–35. https://doi.org/10.1038/nature15756 [PubMed]
- 2. Dinkins MB, Wang G, Bieberich E. Sphingolipid-enriched extracellular vesicles and Alzheimer’s disease: a decade of research. J Alzheimers Dis. 2017; 60:757–68. https://doi.org/10.3233/JAD-160567 [PubMed]
- 3. Hubal MJ, Nadler EP, Ferrante SC, Barberio MD, Suh JH, Wang J, Dohm GL, Pories WJ, Mietus-Snyder M, Freishtat RJ. Circulating adipocyte-derived exosomal MicroRNAs associated with decreased insulin resistance after gastric bypass. Obesity (Silver Spring). 2017; 25:102–10. https://doi.org/10.1002/oby.21709 [PubMed]
- 4. Whitham M, Parker BL, Friedrichsen M, Hingst JR, Hjorth M, Hughes WE, Egan CL, Cron L, Watt KI, Kuchel RP, Jayasooriah N, Estevez E, Petzold T, et al. Extracellular vesicles provide a means for tissue crosstalk during exercise. Cell Metab. 2018; 27:237–251.e4. https://doi.org/10.1016/j.cmet.2017.12.001 [PubMed]
- 5. Choi CH, Cohen P. Adipose crosstalk with other cell types in health and disease. Exp Cell Res. 2017; 360:6–11. https://doi.org/10.1016/j.yexcr.2017.04.022 [PubMed]
- 6. Hamrick MW. A role for myokines in muscle-bone interactions. Exerc Sport Sci Rev. 2011; 39:43–47. https://doi.org/10.1097/JES.0b013e318201f601 [PubMed]
- 7. Lazar I, Clement E, Dauvillier S, Milhas D, Ducoux-Petit M, LeGonidec S, Moro C, Soldan V, Dalle S, Balor S, Golzio M, Burlet-Schiltz O, Valet P, et al. Adipocyte exosomes promote melanoma aggressiveness through fatty acid oxidation: a novel mechanism linking obesity and cancer. Cancer Res. 2016; 76:4051–57. https://doi.org/10.1158/0008-5472.CAN-16-0651 [PubMed]
- 8. Romancino DP, Paterniti G, Campos Y, De Luca A, Di Felice V, d’Azzo A, Bongiovanni A. Identification and characterization of the nano-sized vesicles released by muscle cells. FEBS Lett. 2013; 587:1379–84. https://doi.org/10.1016/j.febslet.2013.03.012 [PubMed]
- 9. Guescini M, Canonico B, Lucertini F, Maggio S, Annibalini G, Barbieri E, Luchetti F, Papa S, Stocchi V. Muscle releases alpha-sarcoglycan positive extracellular vesicles carrying miRNAs in the bloodstream. PLoS One. 2015; 10:e0125094. https://doi.org/10.1371/journal.pone.0125094 [PubMed]
- 10. Tchkonia T, Zhu Y, van Deursen J, Campisi J, Kirkland JL. Cellular senescence and the senescent secretory phenotype: therapeutic opportunities. J Clin Invest. 2013; 123:966–72. https://doi.org/10.1172/JCI64098 [PubMed]
- 11. Kadota T, Fujita Y, Yoshioka Y, Araya J, Kuwano K, Ochiya T. Emerging role of extracellular vesicles as a senescence-associated secretory phenotype: insights into the pathophysiology of lung diseases. Mol Aspects Med. 2018; 60:92–103. https://doi.org/10.1016/j.mam.2017.11.005 [PubMed]
- 12. Bettis T, Kim BJ, Hamrick MW. Impact of muscle atrophy on bone metabolism and bone strength: implications for muscle-bone crosstalk with aging and disuse. Osteoporos Int. 2018; 29:1713–20. https://doi.org/10.1007/s00198-018-4570-1 [PubMed]
- 13. Novotny SA, Warren GL, Hamrick MW. Aging and the muscle-bone relationship. Physiology (Bethesda). 2015; 30:8–16. https://doi.org/10.1152/physiol.00033.2014 [PubMed]
- 14. Davis C, Dukes A, Drewry M, Helwa I, Johnson MH, Isales CM, Hill WD, Liu Y, Shi X, Fulzele S, Hamrick MW. MicroRNA-183-5p increases with age in bone-derived extracellular vesicles, suppresses bone marrow stromal (stem) cell proliferation, and induces stem cell senescence. Tissue Eng Part A. 2017; 23:1231–40. https://doi.org/10.1089/ten.tea.2016.0525 [PubMed]
- 15. Withrow J, Murphy C, Liu Y, Hunter M, Fulzele S, Hamrick MW. Extracellular vesicles in the pathogenesis of rheumatoid arthritis and osteoarthritis. Arthritis Res Ther. 2016; 18:286. https://doi.org/10.1186/s13075-016-1178-8 [PubMed]
- 16. Onodera Y, Teramura T, Takehara T, Obora K, Mori T, Fukuda K. miR-155 induces ROS generation through downregulation of antioxidation-related genes in mesenchymal stem cells. Aging Cell. 2017; 16:1369–80. https://doi.org/10.1111/acel.12680 [PubMed]
- 17. Fariyike B, Singleton Q, Hunter M, Hill WD, Isales CM, Hamrick MW, Fulzele S. Role of microRNA-141 in the aging musculoskeletal system: a current overview. Mech Ageing Dev. 2019; 178:9–15. https://doi.org/10.1016/j.mad.2018.12.001 [PubMed]
- 18. Rokavec M, Li H, Jiang L, Hermeking H. The p53/miR-34 axis in development and disease. J Mol Cell Biol. 2014; 6:214–30. https://doi.org/10.1093/jmcb/mju003 [PubMed]
- 19. Zhang W, Ou G, Hamrick M, Hill W, Borke J, Wenger K, Chutkan N, Yu J, Mi QS, Isales CM, Shi XM. Age-related changes in the osteogenic differentiation potential of mouse bone marrow stromal cells. J Bone Miner Res. 2008; 23:1118–28. https://doi.org/10.1359/jbmr.080304 [PubMed]
- 20. Wiklander OP, Nordin JZ, O’Loughlin A, Gustafsson Y, Corso G, Mäger I, Vader P, Lee Y, Sork H, Seow Y, Heldring N, Alvarez-Erviti L, Smith CI, et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J Extracell Vesicles. 2015; 4:26316. https://doi.org/10.3402/jev.v4.26316 [PubMed]
- 21. Zhang F, Cui J, Liu X, Lv B, Liu X, Xie Z, Yu B. Roles of microRNA-34a targeting SIRT1 in mesenchymal stem cells. Stem Cell Res Ther. 2015; 6:195. https://doi.org/10.1186/s13287-015-0187-x [PubMed]
- 22. Bertoldi K, Cechinel LR, Schallenberger B, Corssac GB, Davies S, Guerreiro IC, Belló-Klein A, Araujo AS, Siqueira IR. Circulating extracellular vesicles in the aging process: impact of aerobic exercise. Mol Cell Biochem. 2018; 440:115–25. https://doi.org/10.1007/s11010-017-3160-4 [PubMed]
- 23. Kangas R, Törmäkangas T, Fey V, Pursiheimo J, Miinalainen I, Alen M, Kaprio J, Sipilä S, Säämänen AM, Kovanen V, Laakkonen EK. Aging and serum exomiR content in women-effects of estrogenic hormone replacement therapy. Sci Rep. 2017; 7:42702. https://doi.org/10.1038/srep42702 [PubMed]
- 24. Terlecki-Zaniewicz L, Lämmermann I, Latreille J, Bobbili MR, Pils V, Schosserer M, Weinmüllner R, Dellago H, Skalicky S, Pum D, Almaraz JC, Scheideler M, Morizot F, et al. Small extracellular vesicles and their miRNA cargo are anti-apoptotic members of the senescence-associated secretory phenotype. Aging (Albany NY). 2018; 10:1103–32. https://doi.org/10.18632/aging.101452 [PubMed]
- 25. Xie Y, Gao Y, Zhang L, Chen Y, Ge W, Tang P. Involvement of serum-derived exosomes of elderly patients with bone loss in failure of bone remodeling via alteration of exosomal bone-related proteins. Aging Cell. 2018; 17:e12758. https://doi.org/10.1111/acel.12758 [PubMed]
- 26. Wang H, Wang B. Extracellular vesicle microRNAs mediate skeletal muscle myogenesis and disease. Biomed Rep. 2016; 5:296–300. https://doi.org/10.3892/br.2016.725 [PubMed]
- 27. Le Bihan MC, Bigot A, Jensen SS, Dennis JL, Rogowska-Wrzesinska A, Lainé J, Gache V, Furling D, Jensen ON, Voit T, Mouly V, Coulton GR, Butler-Browne G. In-depth analysis of the secretome identifies three major independent secretory pathways in differentiating human myoblasts. J Proteomics. 2012; 77:344–56. https://doi.org/10.1016/j.jprot.2012.09.008 [PubMed]
- 28. Choi JS, Yoon HI, Lee KS, Choi YC, Yang SH, Kim IS, Cho YW. Exosomes from differentiating human skeletal muscle cells trigger myogenesis of stem cells and provide biochemical cues for skeletal muscle regeneration. J Control Release. 2016; 222:107–15. https://doi.org/10.1016/j.jconrel.2015.12.018 [PubMed]
- 29. Forterre A, Jalabert A, Berger E, Baudet M, Chikh K, Errazuriz E, De Larichaudy J, Chanon S, Weiss-Gayet M, Hesse AM, Record M, Geloen A, Lefai E, et al. Proteomic analysis of C2C12 myoblast and myotube exosome-like vesicles: a new paradigm for myoblast-myotube cross talk? PLoS One. 2014; 9:e84153. https://doi.org/10.1371/journal.pone.0084153 [PubMed]
- 30. Li X, Khanna A, Li N, Wang E. Circulatory miR34a as an RNAbased, noninvasive biomarker for brain aging. Aging (Albany NY). 2011; 3:985–1002. https://doi.org/10.18632/aging.100371 [PubMed]
- 31. Zheng Y, Kong J, Li Q, Wang Y, Li J. Role of miRNAs in skeletal muscle aging. Clin Interv Aging. 2018; 13:2407–19. https://doi.org/10.2147/CIA.S169202 [PubMed]
- 32. Greco S, Perfetti A, Fasanaro P, Cardani R, Capogrossi MC, Meola G, Martelli F. Deregulated microRNAs in myotonic dystrophy type 2. PLoS One. 2012; 7:e39732. https://doi.org/10.1371/journal.pone.0039732 [PubMed]
- 33. Kofman AV, Kim J, Park SY, Dupart E, Letson C, Bao Y, Ding K, Chen Q, Schiff D, Larner J, Abounader R. microRNA-34a promotes DNA damage and mitotic catastrophe. Cell Cycle. 2013; 12:3500–11. https://doi.org/10.4161/cc.26459 [PubMed]
- 34. Badi I, Burba I, Ruggeri C, Zeni F, Bertolotti M, Scopece A, Pompilio G, Raucci A. MicroRNA-34a induces vascular smooth muscle cells senescence by SIRT1 downregulation and promotes the expression of age-associated pro-inflammatory secretory factors. J Gerontol A Biol Sci Med Sci. 2015; 70:1304–11. https://doi.org/10.1093/gerona/glu180 [PubMed]
- 35. Boon RA, Iekushi K, Lechner S, Seeger T, Fischer A, Heydt S, Kaluza D, Tréguer K, Carmona G, Bonauer A, Horrevoets AJ, Didier N, Girmatsion Z, et al. MicroRNA-34a regulates cardiac ageing and function. Nature. 2013; 495:107–10. https://doi.org/10.1038/nature11919 [PubMed]
- 36. Jian C, Lu M, Zhang Z, Liu L, Li X, Huang F, Xu N, Qin L, Zhang Q, Zou D. miR-34a knockout attenuates cognitive deficits in APP/PS1 mice through inhibition of the amyloidogenic processing of APP. Life Sci. 2017; 182:104–11. https://doi.org/10.1016/j.lfs.2017.05.023 [PubMed]
- 37. Khanna A, Muthusamy S, Liang R, Sarojini H, Wang E. Gain of survival signaling by down-regulation of three key miRNAs in brain of calorie-restricted mice. Aging (Albany NY). 2011; 3:223–36. https://doi.org/10.18632/aging.100276 [PubMed]
- 38. Owczarz M, Budzinska M, Domaszewska-Szostek A, Borkowska J, Polosak J, Gewartowska M, Slusarczyk P, Puzianowska-Kuznicka M. miR-34a and miR-9 are overexpressed and SIRT genes are downregulated in peripheral blood mononuclear cells of aging humans. Exp Biol Med (Maywood). 2017; 242:1453–61. https://doi.org/10.1177/1535370217720884 [PubMed]
- 39. Mohamed JS, Wilson JC, Myers MJ, Sisson KJ, Alway SE. Dysregulation of SIRT-1 in aging mice increases skeletal muscle fatigue by a PARP-1-dependent mechanism. Aging (Albany NY). 2014; 6:820–34. https://doi.org/10.18632/aging.100696 [PubMed]
- 40. Bernardo BC, Ooi JY, Matsumoto A, Tham YK, Singla S, Kiriazis H, Patterson NL, Sadoshima J, Obad S, Lin RC, McMullen JR. Sex differences in response to miRNA-34a therapy in mouse models of cardiac disease: identification of sex-, disease- and treatment-regulated miRNAs. J Physiol. 2016; 594:5959–74. https://doi.org/10.1113/JP272512 [PubMed]
- 41. Sharma S, Eghbali M. Influence of sex differences on microRNA gene regulation in disease. Biol Sex Differ. 2014; 5:3. https://doi.org/10.1186/2042-6410-5-3 [PubMed]
- 42. Sheth KA, Iyer CC, Wier CG, Crum AE, Bratasz A, Kolb SJ, Clark BC, Burghes AH, Arnold WD. Muscle strength and size are associated with motor unit connectivity in aged mice. Neurobiol Aging. 2018; 67:128–36. https://doi.org/10.1016/j.neurobiolaging.2018.03.016 [PubMed]
- 43. Chan S, Head SI. Age- and gender-related changes in contractile properties of non-atrophied EDL muscle. PLoS One. 2010; 5:e12345. https://doi.org/10.1371/journal.pone.0012345 [PubMed]
- 44. Kim H, Wrann CD, Jedrychowski M, Vidoni S, Kitase Y, Nagano K, Zhou C, Chou J, Parkman VA, Novick SJ, Strutzenberg TS, Pascal BD, Le PT, et al. Irisin mediates effects on bone and fat via αV integrin receptors. Cell. 2018; 175:1756–1768.e17. https://doi.org/10.1016/j.cell.2018.10.025 [PubMed]
- 45. Kitase Y, Vallejo JA, Gutheil W, Vemula H, Jähn K, Yi J, Zhou J, Brotto M, Bonewald LF. β-aminoisobutyric Acid, l-BAIBA, is a muscle-derived osteocyte survival factor. Cell Reports. 2018; 22:1531–44. https://doi.org/10.1016/j.celrep.2018.01.041 [PubMed]
- 46. Zainabadi K, Liu CJ, Guarente L. SIRT1 is a positive regulator of the master osteoblast transcription factor, RUNX2. PLoS One. 2017; 12:e0178520. https://doi.org/10.1371/journal.pone.0178520 [PubMed]
- 47. Edwards JR, Perrien DS, Fleming N, Nyman JS, Ono K, Connelly L, Moore MM, Lwin ST, Yull FE, Mundy GR, Elefteriou F. Silent information regulator (Sir)T1 inhibits NF-κB signaling to maintain normal skeletal remodeling. J Bone Miner Res. 2013; 28:960–69. https://doi.org/10.1002/jbmr.1824 [PubMed]
- 48. Chen L, Holmstrøm K, Qiu W, Ditzel N, Shi K, Hokland L, Kassem M. MicroRNA-34a inhibits osteoblast differentiation and in vivo bone formation of human stromal stem cells. Stem Cells. 2014; 32:902–12. https://doi.org/10.1002/stem.1615 [PubMed]
- 49. Liu Y, Zhang X, Chen J, Li T. Inhibition of microRNA-34a enhances survival of human bone marrow mesenchymal/stromal stem cells under oxidative stress. Med Sci Monit. 2018; 24:264–71. https://doi.org/10.12659/MSM.904618 [PubMed]
- 50. Kosaka N, Iguchi H, Yoshioka Y, Takeshita F, Matsuki Y, Ochiya T. Secretory mechanisms and intercellular transfer of microRNAs in living cells. J Biol Chem. 2010; 285:17442–52. https://doi.org/10.1074/jbc.M110.107821 [PubMed]
- 51. Takahashi A, Okada R, Nagao K, Kawamata Y, Hanyu A, Yoshimoto S, Takasugi M, Watanabe S, Kanemaki MT, Obuse C, Hara E. Exosomes maintain cellular homeostasis by excreting harmful DNA from cells. Nat Commun. 2017; 8:15287. https://doi.org/10.1038/ncomms15287 [PubMed]
- 52. Obata Y, Kita S, Koyama Y, Fukuda S, Takeda H, Takahashi M, Fujishima Y, Nagao H, Masuda S, Tanaka Y, Nakamura Y, Nishizawa H, Funahashi T, et al. Adiponectin/T-cadherin system enhances exosome biogenesis and decreases cellular ceramides by exosomal release. JCI Insight. 2018; 3:99680. https://doi.org/10.1172/jci.insight.99680 [PubMed]
- 53. Shetty SK, Tiwari N, Marudamuthu AS, Puthusseri B, Bhandary YP, Fu J, Levin J, Idell S, Shetty S. p53 and miR-34a feedback promotes lung epithelial injury and pulmonary fibrosis. Am J Pathol. 2017; 187:1016–34. https://doi.org/10.1016/j.ajpath.2016.12.020 [PubMed]
- 54. Rider MA, Hurwitz SN, Meckes DG
Jr . ExtraPEG: A polyethylene glycol-based method for enrichment of extracellular vesicles. Sci Rep. 2016; 6:23978. https://doi.org/10.1038/srep23978 [PubMed] - 55. Helwa I, Cai J, Drewry MD, Zimmerman A, Dinkins MB, Khaled ML, Seremwe M, Dismuke WM, Bieberich E, Stamer WD, Hamrick MW, Liu Y. A comparative study of serum exosome isolation using differential ultracentrifugation and three commercial reagents. PLoS One. 2017; 12:e0170628. https://doi.org/10.1371/journal.pone.0170628 [PubMed]
- 56. Kolhe R, Hunter M, Liu S, Jadeja RN, Pundkar C, Mondal AK, Mendhe B, Drewry M, Rojiani MV, Liu Y, Isales CM, Guldberg RE, Hamrick MW, Fulzele S. Gender-specific differential expression of exosomal miRNA in synovial fluid of patients with osteoarthritis. Sci Rep. 2017; 7:2029. https://doi.org/10.1038/s41598-017-01905-y [PubMed]
- 57. Sangani R, Naime M, Zakhary I, Ahmad S, Chutkan N, Zhu A, Ha Y, Hamrick M, Isales C, Elsalanty M, Smith S, Liou GI, Fulzele S. Regulation of vitamin C transporter in the type 1 diabetic mouse bone and bone marrow. Exp Mol Pathol. 2013; 95:298–306. https://doi.org/10.1016/j.yexmp.2013.08.007 [PubMed]



