Introduction
Autophagy is a process that disassembles unnecessary or dysfunctional cellular components for degradation and recycling [1]. The formation of autophagosome, a double-membraned vesicle that eventually fuses with lysosomes, is a critical step in macroautophagy. Autophagy-related genes (ATGs) family and microtubule-associated protein 1A/1B-light chain 3 (LC3) are crucial regulators in autophagosome formation [2]. The cytosolic form of LC3 (LC3-I) is conjugated to phosphatidylethanolamine to form LC3-II, which is recruited to autophagosomal membranes. After fusion of autophagosome with lysosomes, LC3-II on the cytosolic face of autophagosomes is delipidated by Atg4B to form LC3-I for recycling, and LC3-II on the lumenal face of autophagosomes is degraded by lysosomal hydrolases [3]. Therefore, LC3-II is used as a promising autophagosomal marker to reflect autophagic activity. Autophagy is a double-eged sword in cell function [4]. It may promote cell survival through inhibition of apoptosis [5] or induce cell death through caspase and apoptosis-independent mechanisms [6]. Oxidative stress has been shown to cause the accumulation of autophagosomes in different types of somatic cells [7]. Various conditions that induce oxidative stress, such as starvation [8], infection [9], and environmental stress [10], can activate autophagy. Excessive light exposure has been shown to induce autophagy in different types of cells. Constant UV light exposure for 24 h activates autophagy in epidermal cells [11]. Near-infrared photothermal therapy increases autophagic cell death in breast cancer cells [12]. Light emitting diodes (LEDs), with superior efficiency, lower cost and high variability of wavelengths ranging from the ultraviolet to the near-infrared region of the spectrum, have been developed to replace traditional light bulbs. LED light exposure has been reported to cause cell damage through activation of autophagy in colon cancer cells [13], retinal pigment epithelium cells [14], retinal photoreceptor cells [15] and lymphoid cells [16]. We reported previously that white LED light exposure induces apoptosis and cell cycle arrest in hippocampal neuron cells [17]. However, it remains unknown whether LED light may activate autophagy in hippocampal neuron cells.
The activation of autophagy is regulated by complex intracellular signaling networks. Glucocorticoid receptor (GR) is highly expressed in hippocampus to mediate the central response to peripheral glucocorticoids under basal and stressful situations [18]. Glucocorticoids are reported to induce the autophagic processes in N1511 chondrocyte cells [19] and rat bone marrow mesenchymal stem cells [20]. Also, a ligand-independent activation of GR is involved in the transcriptional regulation of autophagy-related genes in HT-22 hippocampal neuron cells [21]. Autophagy flux is known to have circadian rhythms [22], while circadian clock genes, which are regulated by transcription factors such as retinoid-related orphan receptor alpha (RORα) [23, 24], are reported to regulate autophagy [25]. Moreover, the serine/threonine kinase glycogen synthase kinase-3 (GSK-3) plays a paradoxical role in autophagy. Inhibition of GSK-3β suppresses autophagic cell death in adult hippocampal neural stem cells [26], whereas inhibition of GSK-3 activates autophagy in human pancreatic cancer cells [27], breast tumor cell line MCF7 [28], and prostate cancer cells [29]. Although the role of GSK-3, GR and RORα in the regulation of autophagy has been reported respectively, it remains unknown whether and how they are involved in mediating the effects of white LED light on hippocampal neural cell autophagy.
In this study, we first show that LED light exposure significantly activated autophagy and disrupted the circadian rhythms of clock related genes in hippocampal neuron cells, which was associated with significantly increased GSK-3, phospho-GR and RORα protein expression. To further delineate the signaling pathway of LED light-induced autophagy in hippocampal neuron cells, we used inhibitors or shRNAs to specifically knockdown RORα, GR and GSK-3, respectively. The results indicate that GSK-3-mediated GR and RORα pathways participate in the regulation of the autophagy activation in white LED light-exposed hippocampal neuron cells.
Results
White LED light exposure induces autophagy in hippocampal neuron cells
White LED light-induced autophagy in hippocampal neuron cells was demonstrated by using different methods. Firstly, transmission electron microscopy (Figure 1A–1D) demonstrated that the number of autophagic vacuoles was significantly increased (p < 0.01) in Light group (Figure 1E). Secondly, autophagy-related genes, such as LC3B, Beclin and ATG7, were significantly (p < 0.01) up-regulated in Light group at both mRNA (Figure 1F) and protein (Figure 1G) levels. Thirdly, the GFP-LC3B immuofluorescence was significantly increased (Figure 1H–1I) in light-exposed cells transfected with the GFP-tagged MAP1LC3B expression plasmid (GFP-MAP1LC3B). Furthermore, blocking the autophagy with 3-MA, an early stage autophagy inhibitor, rescued light-induced decrease of cell viability, in a dose-dependent manner (Supplementary Figure 2).
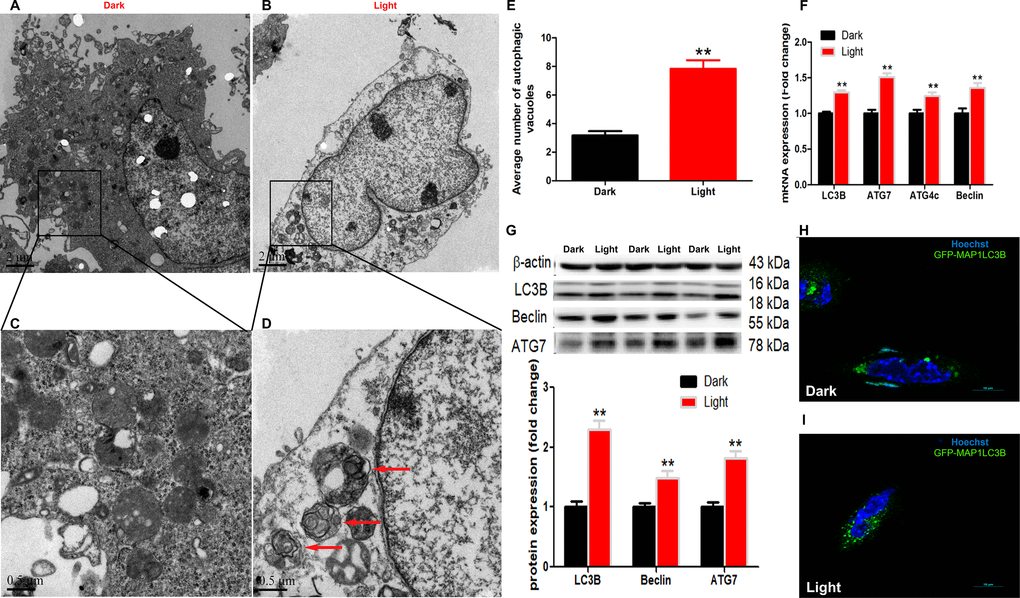
Figure 1. White LED light induces activation of autophagy process in HT-22 cells. (A–B) TEM images of autophagic vacuoles in Dark and Light groups, respectively. Scale bars, 2 μm, n = 3; (C–D) Higher magnification of indicated regions in A and B (squares), showing autophagosomes with double membrane (red arrows). Scale bars, 0.5 μm; (E) The number of autophagic vacuoles. Values are means ± SEM, ** p < 0.01 compared with Dark group, n = 3. (F) Quantitative Real-time PCR analysis of autophagy-related gene lc3b, atg7, atg4c and beclin. Values are means ± SEM, ** p < 0.01 compared with Dark group, n = 6; (G) Western blot analysis of autophagy-related protein LC3B, Beclin and ATG7. Values are means ± SEM, ** p < 0.01 compared with Dark group, n = 6; (H–I) Representative fluorescence images of HT-22 cells transfected with GFP-LC3 plasmid. Cells were counterstained with Hochest (nuclei in blue) and more autophagosomes (green) were seen in Light group. Scale bar, 10 μm.
White LED light exposure enhance autophagy flux in hippocampal neuron cells
To detected the effect of white LED light on autophagy flux, we added 50 μM chloroquine (CQ) in the last 2 h of white LED light exposure. CQ significantly increased LC3II and p62 protein levels (p < 0.01) both in Dark and Light group (Figure 2B and 2E). Meanwhile, white LED light exposure significantly increased (p < 0.01) LC3II flux both in net flux and relative flux (Figure 2C and 2D). Concurrently, white LED light exposure significantly increased (p < 0.01) the relative flux of p62 (Figure 2G), yet not influence net flux of p62 (Figure 2F).
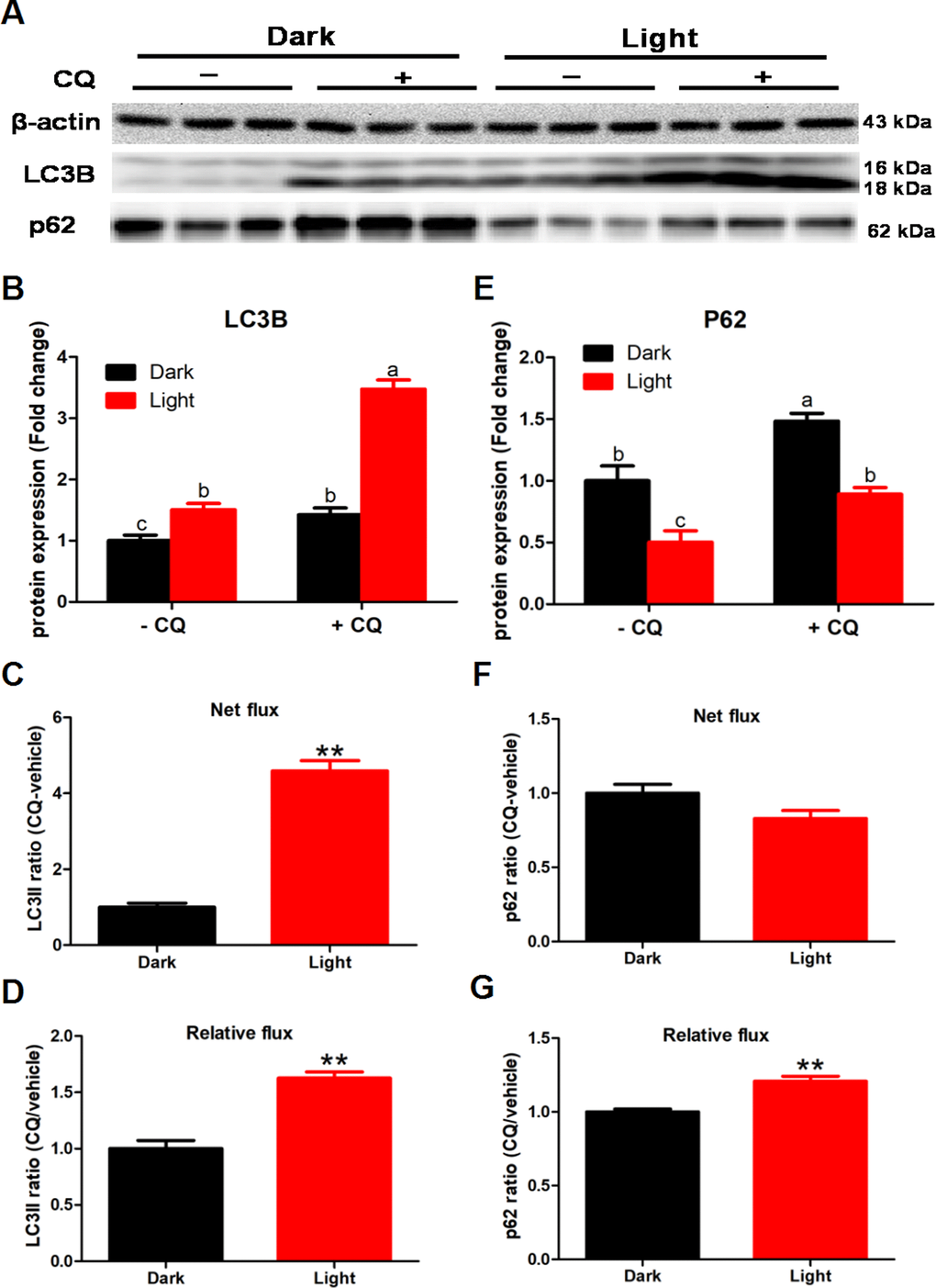
Figure 2. White LED light exposure enhance autophagy flux in hippocampal neuron cells. To detect the effect of white LED light on autophagy flux, we added 50 μM chloroquine (CQ) in the last 2 h of white LED light exposure. (A) Images of bands detected in Western blot analyses; (B) LC3B protein levels in Dark and Light group with or without CQ. Values are means ± SEM. Bars with different superscripts are significantly different from each other (p < 0.05, n = 3); (C) Net flux of LC3II protein. Values are means ± SEM, ** p < 0.01 compared with Dark group, n = 3; (D) Relative flux of LC3II protein. Values are means ± SEM, ** p < 0.01 compared with Dark group, n = 3; (E) p62 protein levels in Dark and Light group with or without CQ. Values are means ± SEM. Bars with different superscripts are significantly different from each other (p < 0.05, n = 3); (F) Net flux of p62 protein. Values are means ± SEM, n = 3; (G) Relative flux of p62 protein. Values are means ± SEM, ** p < 0.01 compared with Dark group, n = 3.
Surprisingly, we found that different CQ added mode lead to different autophagy flux results. LC3II and p62 protein levels were significantly increased (p < 0.01) both in Dark and Light group with added 50 μM CQ in 2 h after white LED light exposure (Supplementary Figure 3B and 3E). However, white LED light exposure significantly decreased LC3II flux both in the net autophagy flux (p < 0.01) and the relative autophagy flux (p < 0.05) (Supplementary Figure 3C and 3D). Concurrently, white LED light exposure significantly increased (p < 0.05) the relative flux of p62 (Supplementary Figure 3G), yet not influence the net flux of p62 (Supplementary Figure 3F).
White LED light activates GSK-3 and nuclear receptors GR and RORα in HT-22 cells
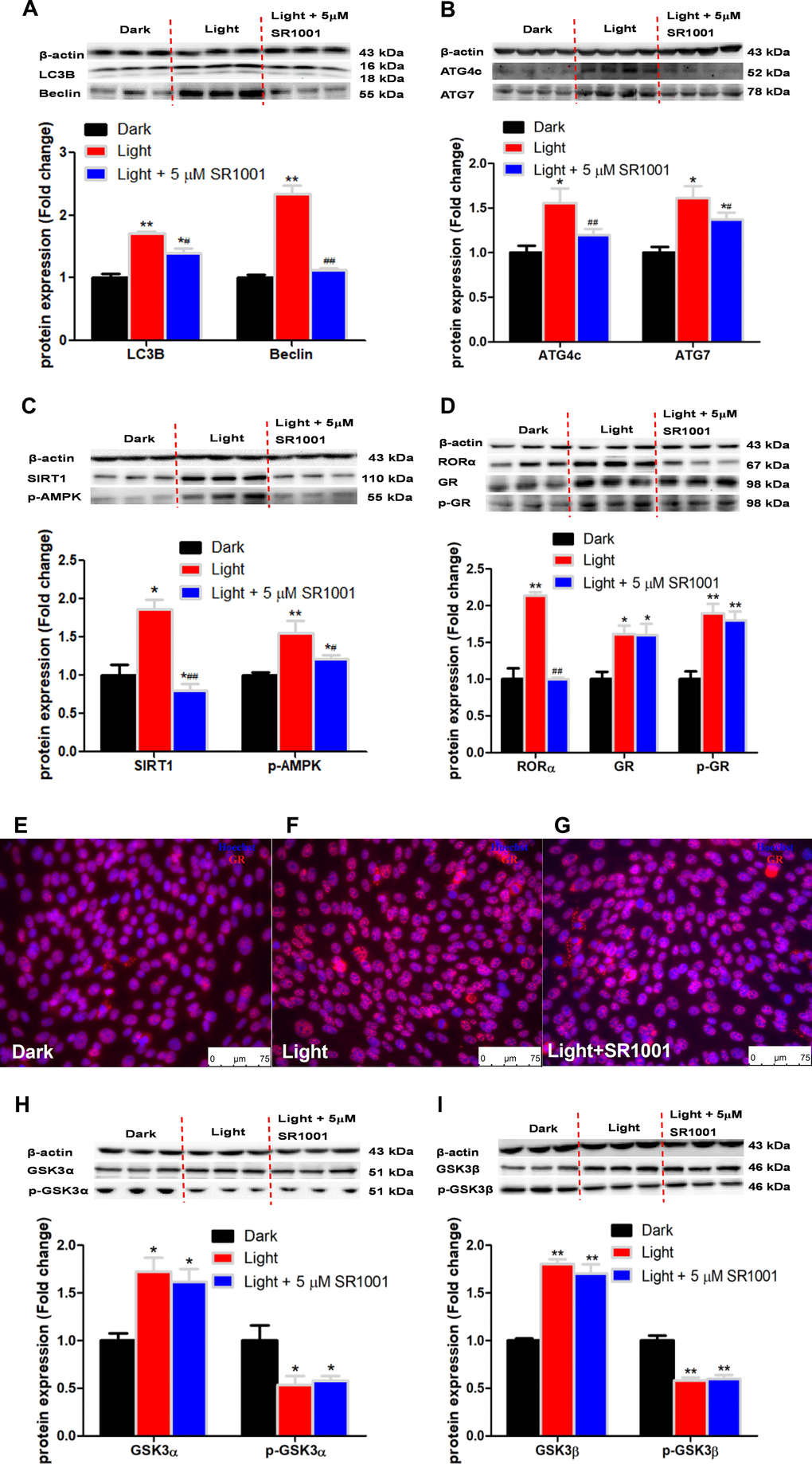
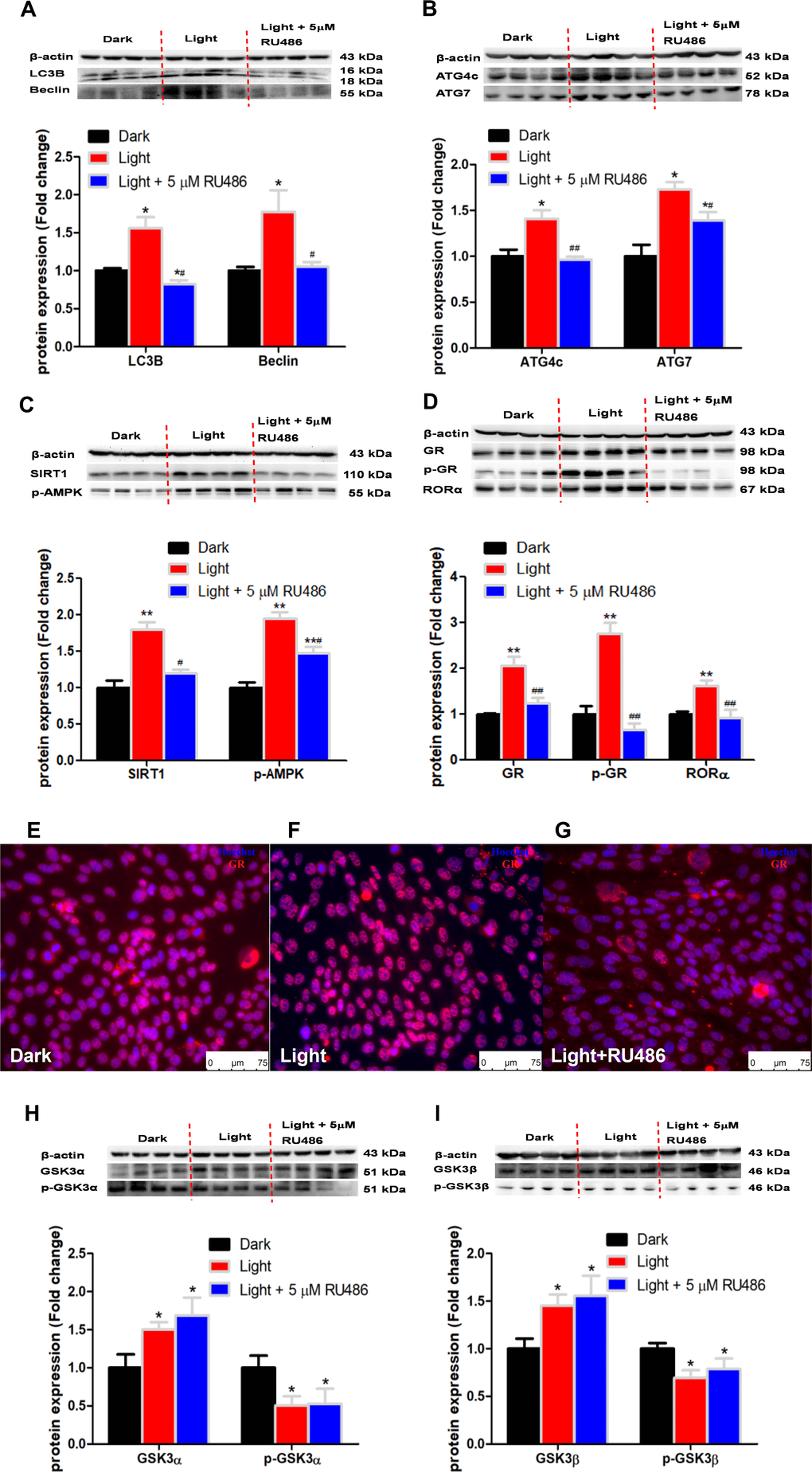
White LED light exposure significantly increased protein levels of GSK-3α and GSK-3β (p < 0.01), yet the serine-21 phosphorylated GSK-3α (Figure 3A) and serine-9 phosphorylated GSK-3β (Figure 3B) were significantly decreased (p < 0.01), indicating enhanced GSK-3 activity as phosphorylation at these particular serine residues inhibits the activity of GSK-3α and GSK-3β, respectively [30]. Concurrently, protein contents of GR and phospho-GR were also significant increased (p < 0.01). Moreover, RORα was significantly increased (p < 0.01), while REV-ERBα significantly decreased (p < 0.01) in light-exposed cells (Figure 3C), implicating possible disruption of clock genes and their circadian rhythms. Furthermore, SIRT1/AMPK pathway, which is known to regulate autophagy, is activated in response to white LED light exposure. The protein levels of SIRT1 and phospho-AMPK were significantly increased (p < 0.01) after LED light exposure (Figure 3D).
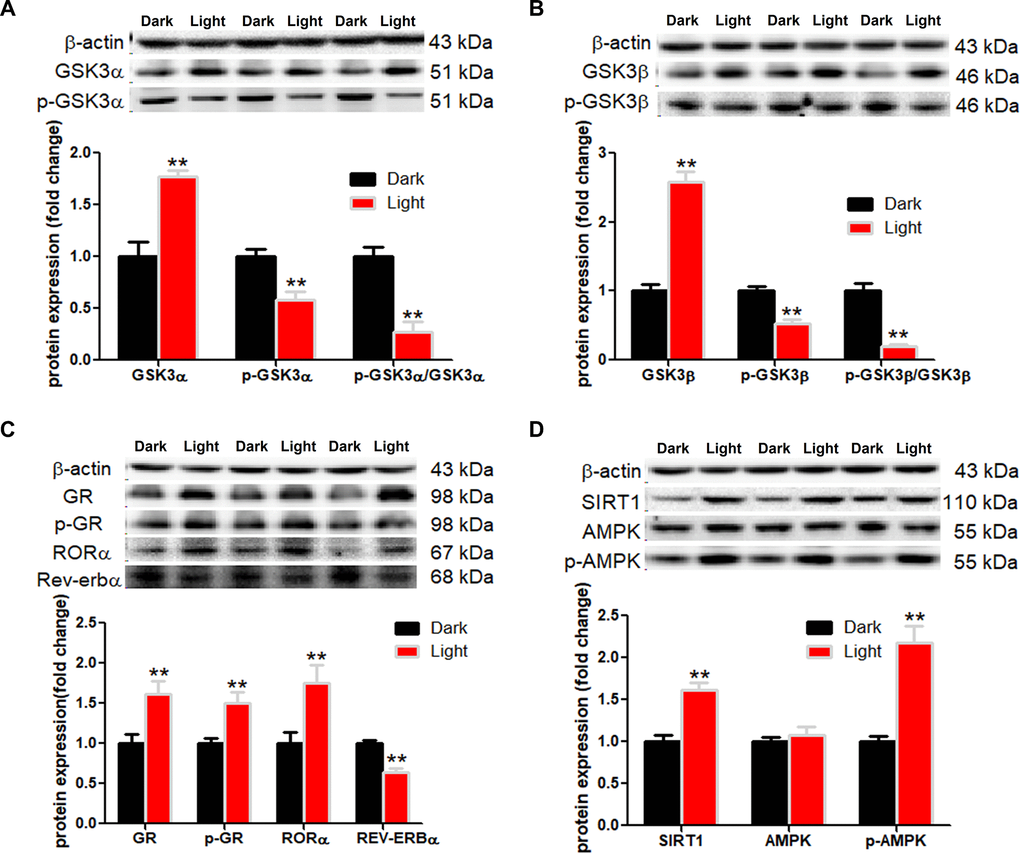
Figure 3. White LED light activates GSK3 and nuclear receptor GR, RORα in HT-22 cells. (A–B) Protein content of total and phospho-GSK-3α/β (Ser21/9). Significant decrease of p-GSK3/GSK3 ratio in Light group indicates activation of both GSK3α and GSK3β; (C) Protein content of GR, phospho-GR, RORα and REV-ERBα, showing up-regulation of both GR and RORα in Light group; (D) Protein content of SIRT1, AMPK and phospho-AMPK. Values are means ± SEM. **p < 0.01, compared with Dark group (n = 6).
White LED light disrupts the circadian rhythm of circadian clock genes in HT-22 cells
In order to elaborate the effect of white LED light on circadian rhythm of clock genes, we examined the mRNA expression profile of clock genes every 6 h for 48 h. In general, LED light exposure disrupted circadian rhythm of clock genes, in a gene-specific manner (Figure 4). Among 9 genes detected, clock (Figure 4A), bmal1 (Figure 4B), cry1 (Figure 4G) and cry2 (Figure 4H) genes displayed obvious time-phase shift and significantly suppressed (p < 0.01) oscillation amplitudes, while per1 (Figure 3D), per2 (Figure 4E) and per3 (Figure 4F) genes showed a delayed appearance of the 2nd peak, indicating reduced frequency of oscillation. Interestingly, rorα (Figure 4C) completely lost its circadian rhythm and kept increasing (p < 0.01) over the period of examination.
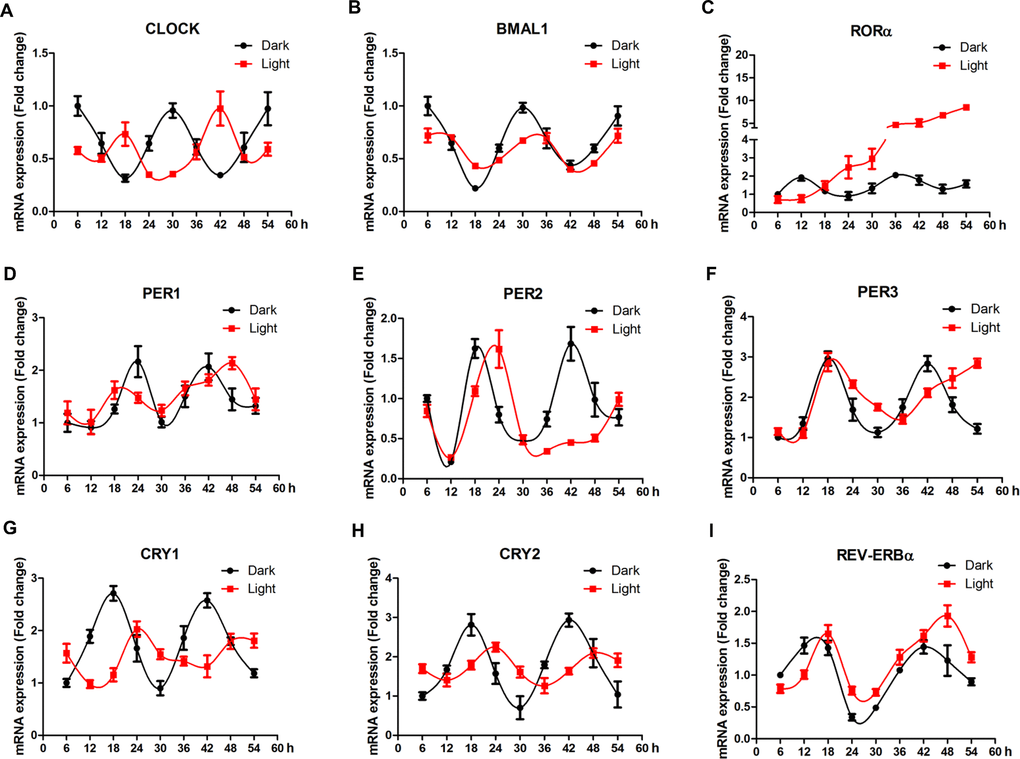
Figure 4. White LED light influences the circadian rhythm of clock-related genes in HT-22 cells. (A–B) Clock and bmal1 mRNA, showing time-phase shift and suppressed oscillation amplitudes in Light group; (C) Rorα mRNA, showing diminished circadian rhythm and a continous increase in mRNA abundance over the period of examination in Light group; (D–E) Per1, per2 and per3 mRNA expression, showing reduced oscillation frequencies in Light group; (G–H) Cry1 and cry2 mRNA, displaying obvious time-phase shift and significantly suppressed oscillation amplitudes in Light group; (I) Rev-erbα mRNA, showing an obvious time-phase shift in Light group.
White LED light disrupts the circadian rhythm of autophagy genes in HT-22 cells
Compared to clock genes, autophagy-related genes showed less clear pattern of circadian rhythms (Figure 5). No significant effect of LED light exposure was observed for LC3B (Figure 5A), yet the fluctuation of ATG3 (Figure 5B) and ATG5 (Figure 5C) was significantly blunted (p < 0.05) in light-exposed cells. Interestingly, ATG7 (Figure 5D) was significantly up-regulated (p < 0.01), reaching a plateau at 42 h after light exposure.
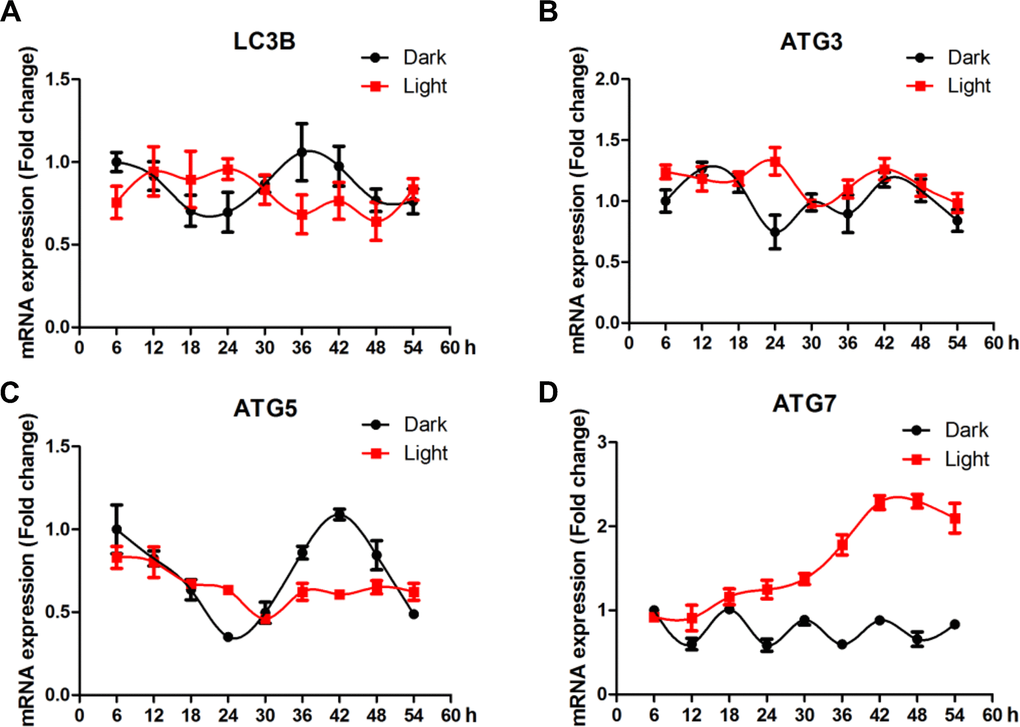
Figure 5. White LED light influences the circadian rhythm of autophagy-related genes expression in HT-22 cells. (A) Lc3b mRNA, with no clear pattern of circadian rhythm and no significant alterations in Light group; (B–C) Atg3 and atg5 mRNA, showing significantly blunted fluctuation in light-exposed cells; (D) Atg7 mRNA, showing a continuous increase in mRNA abundance until a plateau was reached 42 h after light exposure.
Discussion
A number of studies indicate that constant light exposure significantly suppresses the circadian rhythm of clock-related genes [31, 32], which is associated with impaired hippocampal neurogenesis and cognitive performance [33, 34] in mice. In this study, we found that white LED light exposure significantly disrupted the rhythmic expression of clock-related genes. Interestingly, different genes responded to light exposure differently. Genes such as clock, bmal1, cry1 and cry2, displayed time-phase shift and suppressed oscillation amplitudes, while others (per1, per2 and per3) demonstrated reduced frequency of oscillation. These results agree with previous reports that UVB exposure significantly altered the circadian rhythm of clock gene in human keratinocytes [35] and blue light irradiation disrupted the circadian rhythm of per1, per2, per3, cry1 and cry2 genes in Puffer Fish-derived Fugu eye cells [36]. In this study, we observed a striking light response in rorα gene that completely lost its circadian rhythm and kept increasing over the period of examination. Similar result was reported that prenatal exposure to continuous light significantly increased rorα mRNA expression in the adult rat offspring [37]. Concurrently, the autophagy-related gene atg7 showed a similar pattern, which is significantly up-regulated reaching a plateau 42 h after light exposure. These findings implicate a possible role of RORα in light-induced autophagy in HT-22 cells.
RORα has been reported to play an important role in the regulation of circadian rhythm [38]. In this study, we found that white LED light exposure significantly up-regulated RORα protein expression in HT-22 cells in association with activation of SIRT1/AMPK signaling, a classical pathway involved in autophagy [39]. RORα inhibitor SR1001 was able to normalize the light-induced autophagy and the expression of autophagy-related proteins. Our results agree with previous report that RORα deprivation results in autophagy dysfunction by interrupting autophagosome clearance in myocardial ischemia/reperfusion injury mice model [40]. Complex interactions exist between the circadian and glucocorticoid systems [41]. In the present study, white LED light significantly induced GR phosphorylation and nuclear translocation. GR antagonist RU486 significantly suppressed autophagy as well as RORα protein expression. Considering that RORα inhibitor SR1001 was not able to restore light-induced GR activation, we presume that GR is up-stream of RORα in the regulation of light-induced autophagy in HT-22 cells. Nevertheless, an in-depth molecular mechanism of GR-mediated RORα activation awaits further investigation. Given that GR activation in response to light exposure in HT-22 cells is ligand-independent, we speculate that there may be a kinase involved in the regulation of GR activation.
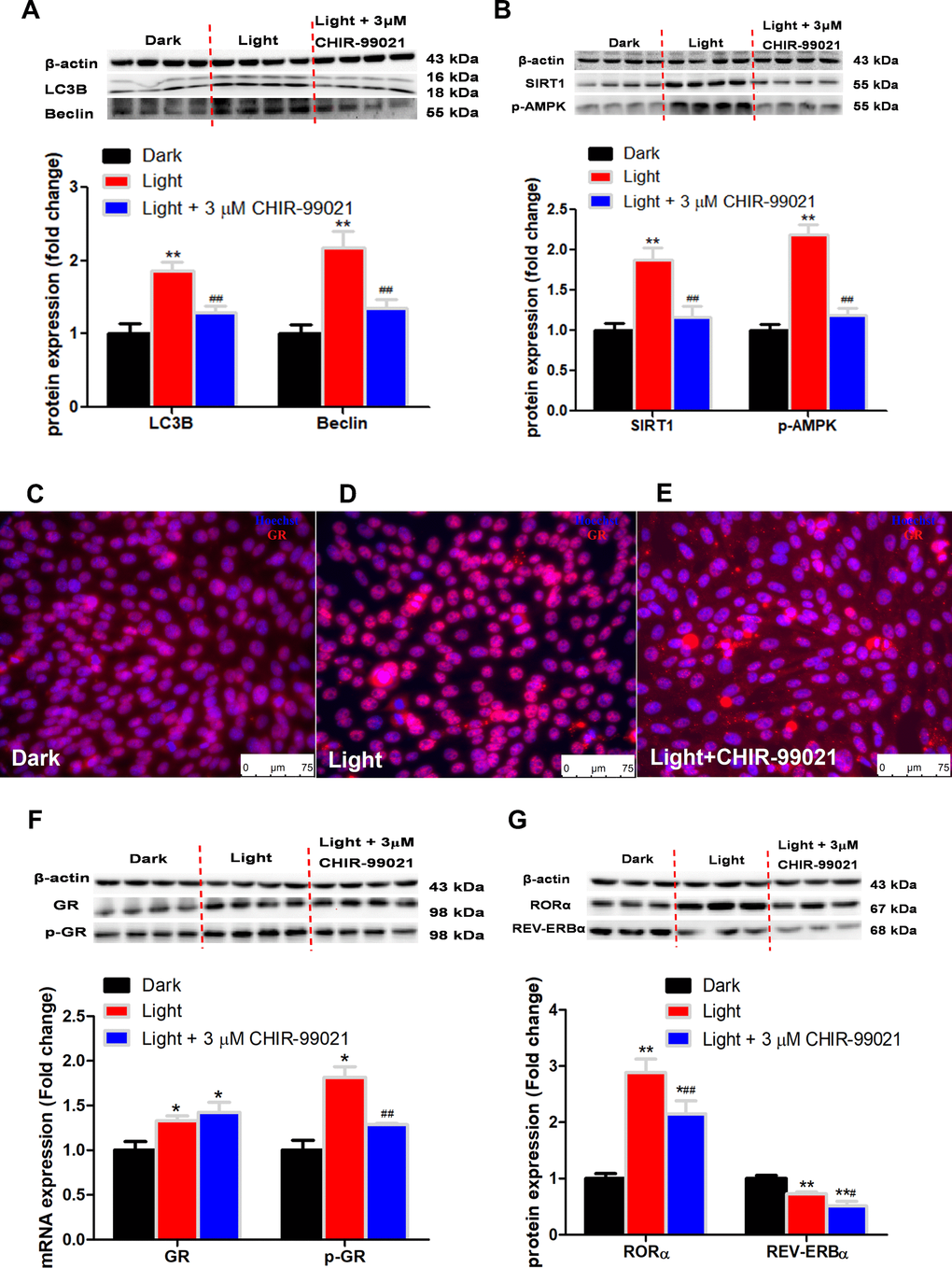
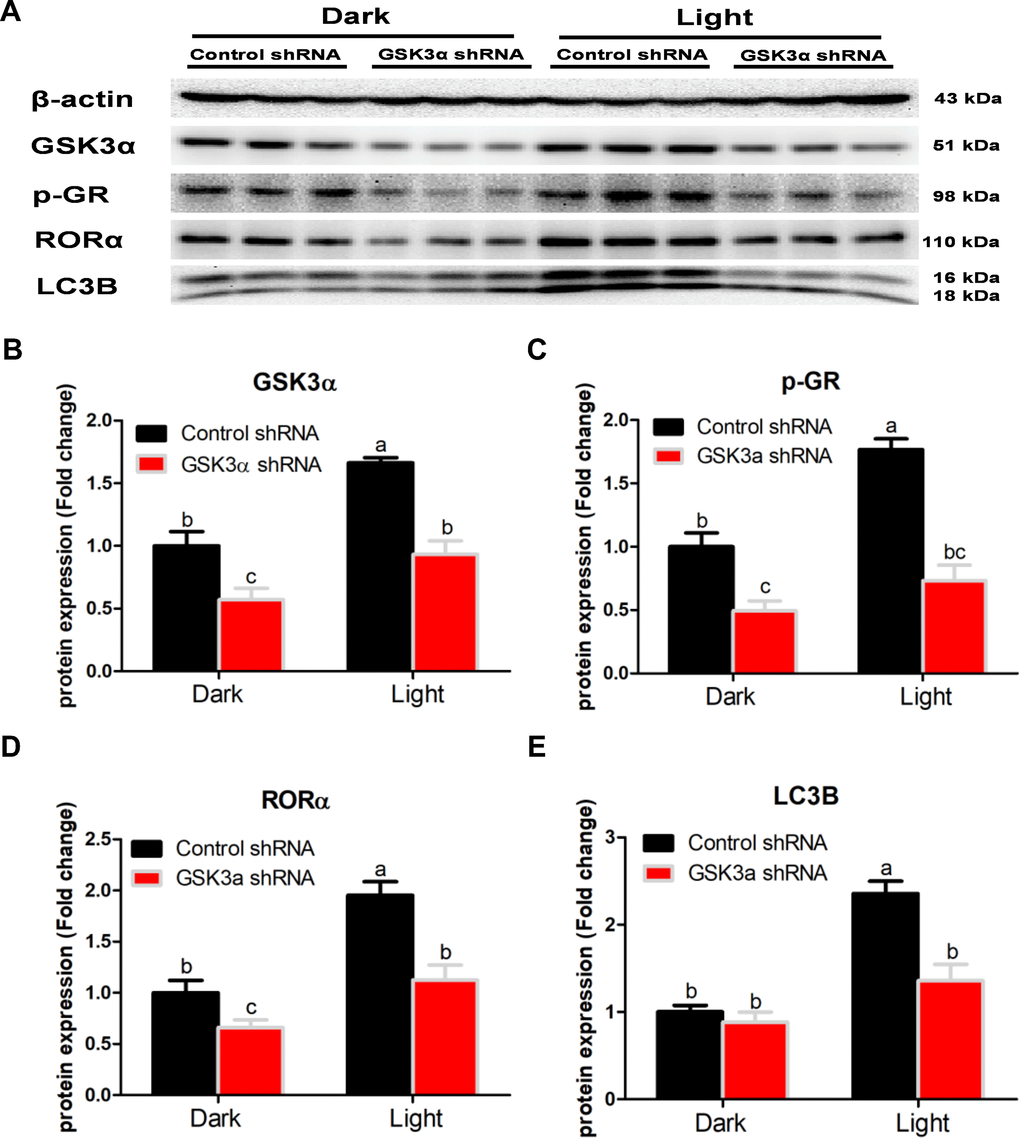
GSK-3 is a multifunctional kinase with a critical role in the regulation of autophagy. Previous studies reported that inhibition of GSK-3 activates autophagy in human pancreatic cancer cells [27], breast tumor cell line MCF7 [28], and prostate cancer cells [29]. Contradictory findings are also reported that inhibition of GSK-3β suppresses autophagic cell death in adult hippocampal neural stem cells [26]. The kinase activity of GSK-3 is inhibited through phosphorylation of a serine residue located at the N-terminus of the proteins, S21 and S9 of GSK-3α and GSK-3β, respectively [30]. In the present study, the protein expression of total GSK-3α and GSK-3β was significantly increased, while that of phosphor-GSK-3α (Ser9) and phosphor-GSK-3β (Ser9) was decreased in response to white LED light exposure, indicating enhanced GSK-3 activity. Inhibition of GSK-3 with CHIR-99021 or shRNA was able to rescue the LED light-induced cell autophagy associated with normalized protein expression of LC3B and Beclin. Interestingly, CHIR-99021 and GSK-3α shRNA also restored the protein expression of phospho-GR and RORα, while light-induced GSK activation was not normalized by RORα or GR inhibition. These results imply that GSK is up-stream of GR and RORα activation induced by white LED light exposure. Our findings are consistent with a previous study that GSK-3 phosphorylates GR on Ser404 and promotes GR activity [42].
In conclusion, the present study provides the first evidence that 24 h exposure under the white LED light induces cell autophagy with disrupted circadian rhythm of clock-related and autophagy-related genes in HT-22 cells. A signaling cascade of GSK-3-mediated GR and RORα pathways is delineated underlying such effects. Our findings indicate that the effects of light exposure in our daily life or in therapy can be detrimental. Caution should be taken to avoid light pollution that has significant impacts on hippocampal neuron survival and related health consequences.
Materials and Methods
Cell culture and LED exposure
Mouse hippocampal neuron cells (HT-22) purchased from Shanghai HuiYing Biological Technology Co. Ltd. (Shanghai, China) were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) (Lot AAH204831, SH30243.01, Hyclone) containing 10 % (v/v) FBS at 37°C under 5% CO2. Cells were divided into Dark and Light groups and cultured in constant dark or light (2500 lux white LED) condition for 24 h. The white LED light was purchased from Wangdeng Technology Co., LTD (ShenZhen, China). The CCT of the light source was 6500 K (Light spectrums in Supplementary Figure 1) and the surface power density was 15 mW/cm2. After cultured under respective condition for 24 h, cells were harvested and the culture media were collected.
Serum shock procedures
Approximately 5 × 105 cells/well were seeded in 6-well plates and cultured in DMEM containing 10 % (v/v) FBS at 37°C under 5% CO2 for 24 h before the medium was replaced with serum-rich medium (DMEM, supplemented with 50% horse serum, Hyclone) for serum shock. Two hours after the serum shock (time = 0), the medium was replaced with serum-free DMEM and the cells were divided into Dark and Light groups. Cells were collected every 6 h to 54 h after light treatment. At the indicated times, the cells were washed twice with ice-cold PBS, frozen on a layer of liquid nitrogen, and kept at -80°C until the extraction of whole cell RNA.
Total RNA isolation and real-time PCR
Total RNA was extracted from the HT-22 cells using 700 μL TRIzol reagents (Cat#3101-100, Invitrogen, USA). Two micrograms of total RNA were treated with RNase-Free DNase and reverse-transcribed to cDNA using the HiScript® Q RT SuperMix (Vazyme, China). Four microliters of diluted cDNA (1:25, vol/vol) were used for real-time PCR performed in Mx3000P (Stratagene, USA). Peptidylprolyl isomerase A (PPIA) was used as an internal control to normalize the technical variations. Data were analyzed using the method of 2–ΔΔCT and presented relative to the Dark group. Nucleotide sequences of specific primers are shown in Table 1.
Table 1. Nucleotide sequences of specific primers.
| Target genes | Primer sequences (5’ to 3’) | GenBank accession | |
| lc3b | F: TAACCAAGCCTTCTTCCTCC | R: GCTGTCCCGAATGTCTCC | NM_026160 |
| atg3 | F: AACAAGAACATACGACCTG | R: GCTCATCATAGCCAAAC | NM_001356366 |
| atg4c | F: CTGGCGATTGGTATGGA | R: CACGGAGTCAGTCTGCTTA | NM_175029 |
| atg5 | F: CAAGGATGCGGTTGAGGC | R: TGAGTTTCCGGTTGATGG | NM_053069 |
| atg7 | F: CCTCGCTGGGACTTGTG | R: TGAATCCTTCTCGCTCGT | NM_001253717 |
| beclin | F: GGCCAATAAGATGGGTCTGA | R: GCTGCACACAGTCCAGAAAA | NM_001359820 |
| clock | F: TCACTCAGGACAGACAGAT | R: TGGCGAAGGTAGGATAGG | NM_007715 |
| bmal1 | F: GCTGGACGAAGACAATGA | R: AAGTTCCTGTGGTAGATACG | NM_007489 |
| cry1 | F: ATCTTGATGCCAATCTACGA | R: AGTGATGTTCCATTCCTTGA | NM_007771 |
| cry2 | F: TGTCCCTTCCTGTGTGGAAGA | R: GCTCCCAGCTTGGCTTGA | NM_009963 |
| per1 | F: ACCTCTGGCTGTTCCTAC | R: CCTCTGCTTGTCATCATCA | NM_011065 |
| per2 | F: ATGCTCGCCATCCACAAGA | R: GCGGAATCGAATGGGAGAAT | NM_011066 |
| per3 | F: CTCTGGCTTCTGAACATACT | R: TCATACTGCGAGGCTCTT | NM_001289878 |
| nr3c1 | F: AGCAGTGGAAGGACAGCAC | R: GTAGGGGTAAGCTGTGGCAG | NM_008173 |
| rorα | F: ATGCACCTGACCGAAGACGAA | R: AGCTTTTCCGTATGTCGTCCAC | NM_013646 |
| rev-erbα | F: GAAGTGTCTCTCCGTTGGCA | R: CTGCTCAGTTGGTTGTTGGC | NM_007489 |
| gsk3α | F: GCTTCTCCCCTCACCACTTC | R: AGGGTAGCAGTTGTGGCATC | NM_001031667 |
| gsk3β | F: GAGCAGGACATTTCACCCCA | R: AAGAGTGCAGGTGTGTCTCG | NM_001347232 |
| ppia | F: GCAAGACCAGCAAGAAGA | R: CAGTGAGAGCAGAGATTACA | NM_008907 |
Total protein extraction and Western blotting
Whole cell lysates were extracted and the protein concentration was determined using Pierce BCA Protein Assay kit (Rockford, IL, USA) according to the product instruction. Whole cell proteins (30 µg) were loaded on a 10% SDS-PAGE gel for electrophoresis. Western blot analysis for LC3B (AP0762, Bioworld, USA, diluted 1:500), Beclin (AP0769, Bioworld, USA, diluted 1:500), ATG4c (5262, Cell Signaling Technology, USA, diluted 1:1000), ATG7 (2631, Cell Signaling Technology, USA, diluted 1:1000), GSK-3α (9338, Cell Signaling Technology, USA, diluted 1:1000), phospho-GSK-3α (9316, Cell Signaling Technology, USA, diluted 1:1000), GSK-3β (9315, Cell Signaling Technology, USA, diluted 1:500), phospho-GSK-3β (9336, Cell Signaling Technology, USA, diluted 1:500), GR (ab2768, Abcam, USA, diluted 1:500), phospho-GR (4161, Cell Signaling Technology, USA, diluted 1:1000), RORα (ab60134, Abcam, USA, diluted 1:500), REV-ERBα (ab174309, Abcam, USA, diluted 1:500), SIRT1 (ab12193, Abcam, USA, diluted 1:1000), AMPK (BS1009, Bioworld Technology USA, diluted 1:1000), phospho-AMPK (2531, Cell Signaling Technology, USA, diluted 1:1000), SQSTM1/p62 (bs-2951R, Bioss, USA, diluted 1:1000), was carried out according to the recommended protocols provided by the manufacturers, and β-actin (AP0060, Bioworld, USA, diluted 1:10,000) was used as loading control. Images were captured by VersaDoc 4000MP system (Bio-Rad, USA) and the band density was analyzed with Quantity One software (Bio-Rad, USA).
Measurement of autophagosome formation
HT-22 cells were seeded on coverslips in 12-well plates, and grown to 70% to 80% confluency before a Lipofectamine 2000-mediated transfection with the green fluorescent protein (GFP)-tagged microtubule-associated protein 1 light chain 3 beta (MAP1LC3B) expression plasmid (GFP-MAP1LC3B), which was a gift from Prof. Honglin Liu (Nanjing Agricultural University). Cells were cultured for 48 h before exposed to light treatments, and the cytoplasmic GFP-MAP1LC3B puncta were then observed under a laser-scanning confocal microscope (Carl Zeiss, Zeiss LSM 710 META). Experiments were performed in triplicate, and the punctate GFP-MAP1LC3B was counted in 3 randomly selected fields in each coverslip.
Determination of autophagy flux
To detected the effect of white LED light on autophagy flux, HT-22 cells were treated with 50 μM chloroquine (CQ, C6628, sigma, USA), a specific protease inhibitor, in the last 2 h of white LED light exposure. Then the levels of autophagy related protein LC3B and p62, as well as both the net and relative autophagy flux of LC3B and p62 were detected.
Transmission electron microscopy
HT-22 cells were treated with typsin for 5 min at 37 °C and centrifuged at 200 ×g for 5 min. After the supernatant was removed, the cell pellets were fixed in 2% glutaraldehyde in 0.1 M sodium cacodylate (NaCac) buffer, pH 7.4, postfixed in 2% osmium tetroxide in NaCac, stained en bloc with 2% uranyl acetate, dehydrated in a graded ethanol series, and embedded in Epon-Araldite resin. Thin sections were made using a diamond knife on a Leica EM UC6 ultramicrotome (Leica Microsystems, Wetzlar, Germany), collected on copper grids, and stained with uranyl acetate and lead citrate. Cells were observed under a JEM 1230 transmission electron microscope (JEOL USA, Peabody, MA) at 110 kV and imaged with an UltraScan 4000 CCD camera and First Light Digital Camera Controller (Gatan, Pleasanton, CA). Autophagic vacuoles were counted in individual cells from multiple fields and nonserial sections.
Determination of GR nuclear translocation
Sub-cellular localization of GR protein in HT-22 cells was determined through immunofluorescence staining analysis. After 24 h culture, HT-22 cells were fixed with 4% paraformaldehyde for 10 min and treated with 0.3% TritonX-100 for 30 min on coverslips. After washing with PBS for three times and blocking with 10% Fetal Bovine Serum for 10 min, HT-22 cells were incubated with GR antibodies (ab2768, Abcam, USA, diluted 1:500) at 4°C overnight. Signal of primary antibody was amplified by TRITC-labelled goat anti-rabbit IgG (ab97200, Abcam, USA, diluted 1:1000) and were visualized and imaged with a fluorescence microscope.
Knockdown of GSK-3α by shRNA transfection
To analyze the function of GSK-3α, HT-22 cells were transfected with a shRNA plasmid constructed to specifically knockdown GSK-3α (pPLK/GFP+Puro-Gsk3a shRNA-1). HT-22 cell line was maintained at 37°C in a humidified incubator containing 5% CO2. Cells were grown in medium supplemented with 10% FBS in 6-well plates 24 h prior to transfection and the medium was replaced by fresh DMEM immediately before transfection. The transfection was conducted following the protocols of Lipofectamine 2000 Transfection Reagent (11668-019, Invitrogen, US) and Opti-MEM (31985, Gibco, USA) provided by the manufacturers.
Statistical analysis
All data are presented as means ± SEM and the differences between groups were analyzed using independent-samples T-Test with SPSS 20.0 for Windows. The differences were considered statistically significant when P < 0.05.
Supplementary Materials
Acknowledgments
This work was supported by the National Key Research and Development Program of China (2016YFD0500502), the Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX18_0716), the National Basic Research Program of China (2012CB124703), the Fundamental Research Funds for the Central Universities (KYZ201212), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), and Jiangsu Collaborative Innovation Center of Meat Production and Processing, Quality and Safety Control.
Conflicts of Interest
There are no conflicts of interest.
References
- 1. Denton D, Xu TQ, Kumar S. Autophagy as a pro-death pathway. Immunol Cell Biol. 2015; 93:35–42. https://doi.org/10.1038/icb.2014.85 [PubMed]
- 2. Tanida I. Autophagosome formation and molecular mechanism of autophagy. Antioxid Redox Signal. 2011; 14:2201–2214. https://doi.org/10.1089/ars.2010.3482 [PubMed]
- 3. Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T, Kominami E, Ohsumi Y, Yoshimori T. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J. 2000; 19:5720–5728. https://doi.org/10.1093/emboj/19.21.5720 [PubMed]
- 4. Thorburn A. Autophagy and its effects: making sense of double-edged swords. PLoS Biol. 2014; 12:e1001967. https://doi.org/10.1371/journal.pbio.1001967 [PubMed]
- 5. Zhao DX, Yuan HP, Yi F, Meng CY, Zhu QS. Autophagy prevents doxorubicin-induced apoptosis in osteosarcoma. Mol Med Rep. 2014; 9:1975–1981. https://doi.org/10.3892/mmr.2014.2055 [PubMed]
- 6. Yu L, Wan F, Dutta S, Welsh S, Liu Z, Freundt E, Baehrecke EH, Lenardo M. Autophagic programmed cell death by selective catalase degradation. Proc Natl Acad Sci USA. 2006; 103:4952–4957. https://doi.org/10.1073/pnas.0511288103 [PubMed]
- 7. Szumiel I. Autophagy, reactive oxygen species and the fate of mammalian cells. Free Radic Res. 2011; 45:253–265. https://doi.org/10.3109/10715762.2010.525233 [PubMed]
- 8. Dice JF, Walker CD, Byrne B, Cardiel A. General characteristics of protein degradation in diabetes and starvation. Proc Natl Acad Sci USA. 1978; 75:2093–2097. https://doi.org/10.1073/pnas.75.5.2093 [PubMed]
- 9. Deretic V, Saitoh T, Akira S. Autophagy in infection, inflammation and immunity. Nat Rev Immunol. 2013; 13:722–737. https://doi.org/10.1038/nri3532 [PubMed]
- 10. Takahashi T, Shimizu K, Shimazaki K, Toda H, Nibuya M. Environmental enrichment enhances autophagy signaling in the rat hippocampus. Brain Res. 2014; 1592:113–123. https://doi.org/10.1016/j.brainres.2014.10.026 [PubMed]
- 11. Yang Y, Wang H, Wang S, Xu M, Liu M, Liao M, Frank JA, Adhikari S, Bower KA, Shi X, Ma C, Luo J. GSK3β signaling is involved in ultraviolet B-induced activation of autophagy in epidermal cells. Int J Oncol. 2012; 41:1782–1788. https://doi.org/10.3892/ijo.2012.1620 [PubMed]
- 12. Zhang M, Kim HS, Jin T, Moon WK. Near-infrared photothermal therapy using EGFR-targeted gold nanoparticles increases autophagic cell death in breast cancer. J Photochem Photobiol B. 2017; 170:58–64. https://doi.org/10.1016/j.jphotobiol.2017.03.025 [PubMed]
- 13. Matsumoto N, Yoshikawa K, Shimada M, Kurita N, Sato H, Iwata T, Higashijima J, Chikakiyo M, Nishi M, Kashihara H, Takasu C, Eto S, Takahashi A, et al. Effect of light irradiation by light emitting diode on colon cancer cells. Anticancer Res. 2014; 34:4709–4716. [PubMed]
- 14. Jaadane I, Villalpando Rodriguez GE, Boulenguez P, Chahory S, Carré S, Savoldelli M, Jonet L, Behar-Cohen F, Martinsons C, Torriglia A. Effects of white light-emitting diode (LED) exposure on retinal pigment epithelium in vivo. J Cell Mol Med. 2017; 21:3453–3466. https://doi.org/10.1111/jcmm.13255 [PubMed]
- 15. Ogawa K, Kuse Y, Tsuruma K, Kobayashi S, Shimazawa M, Hara H. Protective effects of bilberry and lingonberry extracts against blue light-emitting diode light-induced retinal photoreceptor cell damage in vitro. BMC Complement Altern Med. 2014; 14:120. https://doi.org/10.1186/1472-6882-14-120 [PubMed]
- 16. Oh PS, Hwang H, Jeong HS, Kwon J, Kim HS, Kim M, Lim S, Sohn MH, Jeong HJ. Blue light emitting diode induces apoptosis in lymphoid cells by stimulating autophagy. Int J Biochem Cell Biol. 2016; 70:13–22. https://doi.org/10.1016/j.biocel.2015.11.004 [PubMed]
- 17. Yang Y, Zong Y, Sun Q, Jia Y, Zhao R. White light emitting diode suppresses proliferation and induces apoptosis in hippocampal neuron cells through mitochondrial cytochrome c oxydase-mediated IGF-1 and TNF-α pathways. Free Radic Biol Med. 2017; 113:413–423. https://doi.org/10.1016/j.freeradbiomed.2017.10.382 [PubMed]
- 18. Oakley RH, Cidlowski JA. The biology of the glucocorticoid receptor: new signaling mechanisms in health and disease. J Allergy Clin Immunol. 2013; 132:1033–1044. https://doi.org/10.1016/j.jaci.2013.09.007 [PubMed]
- 19. Zhao Y, Zuo Y, Huo HJ, Xiao YL, Yang XJ, Xin DQ. Glucocorticoid induced autophagy in N1511 chondrocyte cells. Eur Rev Med Pharmacol Sci. 2014; 18:3573–3579. [PubMed]
- 20. Wang L, Fan J, Lin YS, Guo YS, Gao B, Shi QY, Wei BY, Chen L, Yang L, Liu J, Luo ZJ. Glucocorticoids induce autophagy in rat bone marrow mesenchymal stem cells. Mol Med Rep. 2015; 11:2711–2716. https://doi.org/10.3892/mmr.2014.3099 [PubMed]
- 21. Sun Q, Yang Y, Li X, He B, Jia Y, Zhang N, Zhao R. Folate deprivation modulates the expression of autophagy- and circadian-related genes in HT-22 hippocampal neuron cells through GR-mediated pathway. Steroids. 2016; 112:12–19. https://doi.org/10.1016/j.steroids.2016.04.010 [PubMed]
- 22. Ma D, Li S, Molusky MM, Lin JD. Circadian autophagy rhythm: a link between clock and metabolism? Trends Endocrinol Metab. 2012; 23:319–325. https://doi.org/10.1016/j.tem.2012.03.004 [PubMed]
- 23. Smarr BL, Jennings KJ, Driscoll JR, Kriegsfeld LJ. A time to remember: the role of circadian clocks in learning and memory. Behav Neurosci. 2014; 128:283–303. https://doi.org/10.1037/a0035963 [PubMed]
- 24. Nader N, Chrousos GP, Kino T. Interactions of the circadian CLOCK system and the HPA axis. Trends Endocrinol Metab. 2010; 21:277–286. https://doi.org/10.1016/j.tem.2009.12.011 [PubMed]
- 25. Bell-Pedersen D, Cassone VM, Earnest DJ, Golden SS, Hardin PE, Thomas TL, Zoran MJ. Circadian rhythms from multiple oscillators: lessons from diverse organisms. Nat Rev Genet. 2005; 6:544–556. https://doi.org/10.1038/nrg1633 [PubMed]
- 26. Ha S, Ryu HY, Chung KM, Baek SH, Kim EK, Yu SW. Regulation of autophagic cell death by glycogen synthase kinase-3β in adult hippocampal neural stem cells following insulin withdrawal. Mol Brain. 2015; 8:30. https://doi.org/10.1186/s13041-015-0119-9 [PubMed]
- 27. Marchand B, Arsenault D, Raymond-Fleury A, Boisvert FM, Boucher MJ. Glycogen synthase kinase-3 (GSK3) inhibition induces prosurvival autophagic signals in human pancreatic cancer cells. J Biol Chem. 2015; 290:5592–5605. https://doi.org/10.1074/jbc.M114.616714 [PubMed]
- 28. Gavilán E, Sánchez-Aguayo I, Daza P, Ruano D. GSK-3β signaling determines autophagy activation in the breast tumor cell line MCF7 and inclusion formation in the non-tumor cell line MCF10A in response to proteasome inhibition. Cell Death Dis. 2013; 4:e572. https://doi.org/10.1038/cddis.2013.95 [PubMed]
- 29. Sun A, Li C, Chen R, Huang Y, Chen Q, Cui X, Liu H, Thrasher JB, Li B. GSK-3β controls autophagy by modulating LKB1-AMPK pathway in prostate cancer cells. Prostate. 2016; 76:172–183. https://doi.org/10.1002/pros.23106 [PubMed]
- 30. Godemann R, Biernat J, Mandelkow E, Mandelkow EM. Phosphorylation of tau protein by recombinant GSK-3beta: pronounced phosphorylation at select Ser/Thr-Pro motifs but no phosphorylation at Ser262 in the repeat domain. FEBS Lett. 1999; 454:157–164. https://doi.org/10.1016/S0014-5793(99)00741-3 [PubMed]
- 31. Nováková M, Polidarová L, Sládek M, Sumová A. Restricted feeding regime affects clock gene expression profiles in the suprachiasmatic nucleus of rats exposed to constant light. Neuroscience. 2011; 197:65–71. https://doi.org/10.1016/j.neuroscience.2011.09.028 [PubMed]
- 32. Grone BP, Chang D, Bourgin P, Cao V, Fernald RD, Heller HC, Ruby NF. Acute light exposure suppresses circadian rhythms in clock gene expression. J Biol Rhythms. 2011; 26:78–81. https://doi.org/10.1177/0748730410388404 [PubMed]
- 33. Fujioka A, Fujioka T, Tsuruta R, Izumi T, Kasaoka S, Maekawa T. Effects of a constant light environment on hippocampal neurogenesis and memory in mice. Neurosci Lett. 2011; 488:41–44. https://doi.org/10.1016/j.neulet.2010.11.001 [PubMed]
- 34. Ma WP, Cao J, Tian M, Cui MH, Han HL, Yang YX, Xu L. Exposure to chronic constant light impairs spatial memory and influences long-term depression in rats. Neurosci Res. 2007; 59:224–230. https://doi.org/10.1016/j.neures.2007.06.1474 [PubMed]
- 35. Park S, Kim K, Bae IH, Lee SH, Jung J, Lee TR, Cho EG. TIMP3 is a CLOCK-dependent diurnal gene that inhibits the expression of UVB-induced inflammatory cytokines in human keratinocytes. FASEB J. 2018; 32:1510–1523. https://doi.org/10.1096/fj.201700693R [PubMed]
- 36. Okano K, Ozawa S, Sato H, Kodachi S, Ito M, Miyadai T, Takemura A, Okano T. Light- and circadian-controlled genes respond to a broad light spectrum in Puffer Fish-derived Fugu eye cells. Sci Rep. 2017; 7:46150. https://doi.org/10.1038/srep46150 [PubMed]
- 37. Voiculescu SE, Le Duc D, Roşca AE, Zeca V, Chiţimuş DM, Arsene AL, Drăgoi CM, Nicolae AC, Zăgrean L, Schöneberg T, Zăgrean AM. Behavioral and molecular effects of prenatal continuous light exposure in the adult rat. Brain Res. 2016; 1650:51–59. https://doi.org/10.1016/j.brainres.2016.08.031 [PubMed]
- 38. Jetten AM. Retinoid-related orphan receptors (RORs): critical roles in development, immunity, circadian rhythm, and cellular metabolism. Nucl Recept Signal. 2009; 7:e003. https://doi.org/10.1621/nrs.07003 [PubMed]
- 39. Park SK, Seong RK, Kim JA, Son SJ, Kim Y, Yokozawa T, Shin OS. Oligonol promotes anti-aging pathways via modulation of SIRT1-AMPK-Autophagy Pathway. Nutr Res Pract. 2016; 10:3–10. https://doi.org/10.4162/nrp.2016.10.1.3 [PubMed]
- 40. He B, Zhao Y, Xu L, Gao L, Su Y, Lin N, Pu J. The nuclear melatonin receptor RORα is a novel endogenous defender against myocardial ischemia/reperfusion injury. J Pineal Res. 2016; 60:313–326. https://doi.org/10.1111/jpi.12312 [PubMed]
- 41. Dickmeis T, Weger BD, Weger M. The circadian clock and glucocorticoids—interactions across many time scales. Mol Cell Endocrinol. 2013; 380:2–15. https://doi.org/10.1016/j.mce.2013.05.012 [PubMed]
- 42. Galliher-Beckley AJ, Williams JG, Collins JB, Cidlowski JA. Glycogen synthase kinase 3beta-mediated serine phosphorylation of the human glucocorticoid receptor redirects gene expression profiles. Mol Cell Biol. 2008; 28:7309–7322. https://doi.org/10.1128/MCB.00808-08 [PubMed]